Abstract
Objective
The purpose of this study was to explore serum miR-135a-5p expression in colorectal cancer and examine the potential usefulness of this molecule as a biomarker for diagnosis in colorectal cancer.
Methods
Serum samples were collected from 60 patients with primary colorectal cancer, 40 patients with colorectal polyps and 50 healthy controls. Serum miR-135a-5p expression levels were detected by reverse transcription quantitative real-time quantitative polymerase chain reaction. Serum carcinoembryonic antigen and carbohydrate antigen 199 concentrations were detected by MODULAR ANALYTICS E170.
Results
The relative expression level of serum miR-135a-5p in colorectal cancer patients, colorectal polyps patients and healthy controls was 2.451 (1.107, 4.413), 0.946 (0.401, 1.942) and 0.949 (0.194, 1.415), respectively, indicating that it was significantly higher in colorectal cancer patients than that in the other two groups (U = 351.0, 313.0, both P < 0.001). Additionally, it was significantly correlated with different degrees of tumour differentiation (U = 215.0, P = 0.029) and different tumour stages (U = 202.0, P = 0.013). There was no significant correlation between the relative expression of serum miR-135a-5p and carcinoembryonic antigen (r2 = 0.023, P = 0.293) or carbohydrate antigen 199 (r2 = 0.067, P = 0.068) in colorectal cancer patients. Compared with colorectal polyps group, AUCROC of serum miR-135a-5p in colorectal cancer group was 0.832 with 95% CI 0.73–0.93; compared with healthy control group, AUCROC was 0.875 with 95% CI 0.80–0.95.
Conclusion
Serum miR-135a-5p expression in colorectal cancer patients was higher than that in patients with colorectal polyps and healthy controls, suggesting that serum miR-135a-5p may prove to be an important biomarker for auxiliary diagnosis of colorectal cancer.
Keywords
Introduction
Colorectal cancer (CRC) is one of the leading causes of cancer-related deaths, the incidence of which ranks the third of all malignant tumours worldwide. 1 There is an upward trend in both incidence and fatality of CRC, which is the fifth most common cause of cancer-related deaths in China. 2 Most CRC-related deaths can be prevented through early diagnosis, surgical removal of early-stage cancer and precancerous lesions. Despite development of screening strategies, such as colonoscopy, fecal-blood testing and stool DNA test, early diagnosis for CRC remains difficult. Carcinoembryonic antigen (CEA) and carbohydrate antigen 199 (CA199), the two most frequently used serum tumour markers, have been used in the clinical diagnosis and prognostic prediction of CRC, but their sensitivity and specificity are limited. Therefore, there is an urgent need for new non-invasive biomarkers to improve the detection of CRC in clinical practice.
MicroRNAs (miRNAs) are small, non-coding RNAs that regulate the translation of specific protein-coding genes, which also take part in the genesis and development of diseases. 3 The discovery that miRNA expression is frequently dysregulated in a cancer-specific manner provides an opportunity to develop these RNAs as biomarkers for cancer detection. 4 Some studies identified a number of miRNAs that were aberrantly expressed in CRC tissue.5,6 Recently, plasma or serum miRNAs have been shown to be stable, 7 and the change in plasma or serum miRNA expression is closely correlated with the development and progression of cancer. Some studies 8 suggest that it could be an excellent candidate for the early diagnosis of cancer.
In the present study, we examined expression of miR-135a-5p in serum samples from 60 CRC patients, 40 patients with colorectal polyps and 50 healthy controls. Furthermore, we investigated the feasibility of using miR-135a-5p as a serum marker or auxiliary tool for the clinical diagnosis of CRC.
Materials and methods
Study population
Included in this study were 60 CRC patients who were admitted in the affiliated hospital of Nantong University (Nantong, China) between January and December 2014. They included 32 males and 28 females ranging in age from 35 to 84 years with a mean of 63 years. Tumour staging and grading were in accordance with the 2010 Cancer Staging Manual of the American Joint Committee on Cancer. 9 Inclusion criteria were (1) patients with CRC confirmed by cytology or pathohistology; (2) CRC patients who had not received treatment previously; (3) CRC patients who had not received chemotherapy and antitumour medical treatment preoperatively; (4) CRC patients with complete data records and (5) CRC patients without significant abnormalities in liver/kidney function and routine blood test or other associated or co-existing diseases. Patients were excluded if they had any of the following issues: (1) patients without confirmed pathohistologic diagnosis; (2) non-first-visit CRC patients; (3) patients without complete data records and (4) patients without a clear clinical stage or a definite therapeutic effect. Additional 40 patients with colorectal polyps who were admitted in the Department of Gastroenterology of the affiliated hospital of Nantong University during the same period were used as the benign cancer control group. They included 21 males and19 females ranging in age from 37 to 78 years with a mean of 61 years. All these cases were diagnosed as colorectal polyps by clinical, imaging or pathologic evaluations. In addition, 50 healthy individuals who underwent physical examination in the physical examination center of the affiliated hospital of Nantong University were used as the healthy control group. They included 27 males and 23 females ranging in age from 38 to 82 years with a mean of 60 years. The study protocol was approved by the ethics committee of the hospital, and all participants provided written consent and indicated willingness to donate their blood samples for research.
Specimen collection and serum RNA extraction
Blood samples were collected using plastic gel vacuum blood collection tubes and centrifuged at 1000 rpm for 10 min. The separated serum were placed in RNase-free centrifuge tubes and stored at −80℃ for use. Total RNA was extracted using the serum RNA extraction kit (Life Technologies, US), and stored at −80℃ after confirmation of the concentration and the purity by ultraviolet spectrophotometry.
cDNA synthesis
The extracted RNA was reverse transcribed into cDNA using the reverse transcription kit (Thermo Fisher Scientific, US), miR-135a-5p and U6 stem-loop RT reverse primer (Shanghai Generay Biotech Co., Ltd., Shanghai, China). The reverse transcription (RT) system was as follows: RNA 300 ng, 5 × reaction buffer solution 4 μL, RT primer 1 μL, DNA 2 μL, RNA inhibitor 1 μL, RTase 1 μL and addition of nucleic acid-free pure water to 20 μL. The mix was incubated at 42℃ for 60 min and 70℃ for 5 min. The synthesized cDNA was kept at −20℃ until further use.
Detection of miR-135a-5p expression level by reverse transcription quantitative real-time polymerase chain reaction (RT-qPCR)
Fluorescence reverse transcription quantitative real-time polymerase chain reaction (RT-qPCR) was performed using the PCR mixture FastStart Universal SYBR Green I Master (Roche, Germany) and 7500 qPCR amplifier (ABI, US). The miR-135a-5p primer sequences are as follows: forward primer: 5′-ACACTCCAGCTGGGGTGCATTGTAGTTGCA-3′, and backward primer: 5′-TGGTGTCGTGGAGTCG-3′; U6 primer sequences: forward primer: 5′-CTCGCTTCGGCAGCACA-3′, and backward primer: 5′-AACGCTTCACGAATTTGCGT-3′. Reaction system: SYBR Green I mixture 10 μL, cDNA 6 μL, forward primer 0.5 μL, backward primer 0.5 μL and RNA-free pure water 3 μL. The mix was incubated at 95℃ for 10 min, 95℃ for 15 s and 60℃ for 1 min, totaling 40 cycles. Each sample was run in triplicates, and the mean value was used for analysis. The relative expression level of miR-135a-5p was normalized to U6 and calculated with the 2−ΔΔCq: ΔΔCq = mean value of study group (CqmiRNA−Cq U6 ) − mean value of control group (CqmiRNA − Cq U6 ).
Detection of CEA and CA199 concentrations
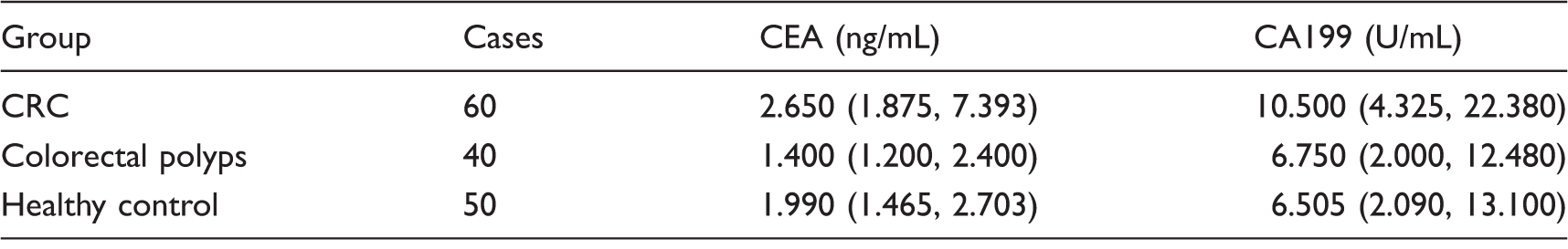
The concentrations of CEA and CA199 in the serum samples from CRC patients were detected in Laboratory Medicine Center of the Affiliated Hospital of Nantong University by MODULAR ANALYTICS E170 (Roche, Germany). The content of CEA > 5 ng/mL or CA199 > 37 U/mL was considered abnormal.
Statistical analysis
Statistical analyses were performed using the SPSS 17.0 statistical software (SPSS, Chicago, IL, USA) and GraphPad Prism 5 (GraphPad Software Inc., CA, USA). Data were expressed as medians (lower quartile and upper quartile). Differences of the relative expression level of serum miR-135a-5p between the CRC, polyps and healthy groups were calculated using Mann-Whitney U test. Paired comparison between the miR-135a-5p concentration and the clinicopathologic features in CRC group was evaluated through Mann-Whitney U test. Correlations between miR-135a-5p and CEA and CA199 concentrations in CRC patients were analysed by Spearman correlation analysis. Their diagnostic efficacy as a diagnostic marker was assessed by receiver-operating characteristics (ROC) and the area under the curve (AUC) (95% CI). Values of P < 0.05 were considered statistically significant.
Results
Range of linearity
The sample miR-135a-5p cDNA was diluted by multiple proportions of 1:10, 1:100, 1:1000 and 1:10000, respectively, and then tested with fluorescent RT-qPCR for Cq values which were used to draw standard curves. From the results, the standard curve equation of Y = −3.443X + 18.01 was obtained as shown Figure 1, with the curve correlation coefficient of r2 = 0.999. According to the formula of E = 10−1/slope − 1, the amplification efficiency of E = 0.95 was obtained by calculation, indicating that this method has good linearity and is suitable for testing serum miR-135a-5p.
The standard curve of testing serum miR-135a-5p by RT-qPCR.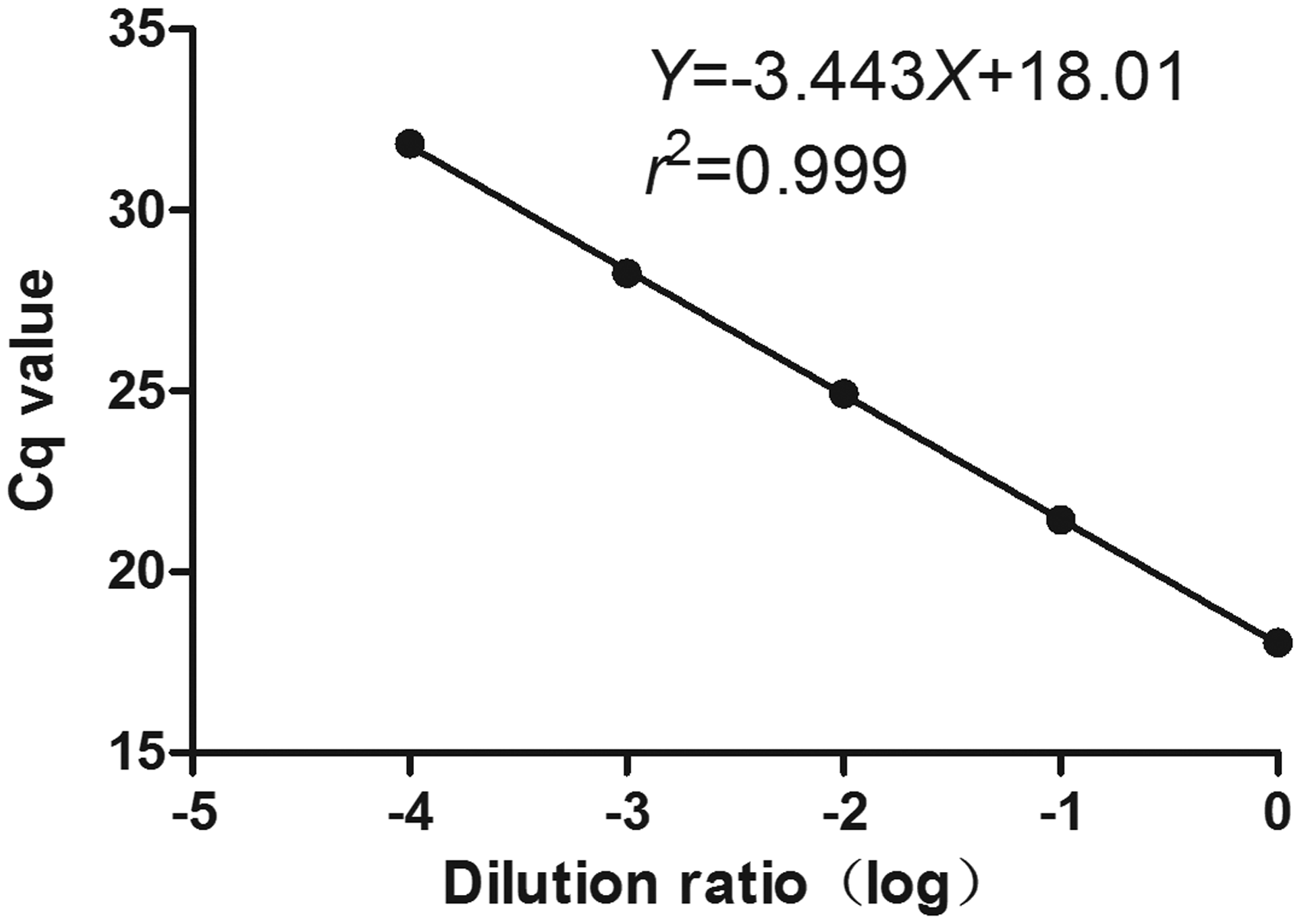
Reproducibility test
High- and low-concentration cDNA specimen of the CRC patients and U6 were taken. The Cq values of miR-135a-5p and U6 were measured by setting 20 parallel holes in a test of fluorescent RT-qPCR, and then the intra-assay coefficient of variation (CV) was calculated. The results showed that for high-concentration miR-135a-5p, the Cq value was 12.51 ± 0.019 and the intra-assay CV value was 0.679%; for low-concentration miR-135a-5p, the Cq value was 31.92 ± 0.059 and the intra-assay CV value was 0.827%, and for high-concentration U6, the Cq value was 7.38 ± 0.012 and the intra-assay CV value was 0.727%, for low-concentration U6, the Cq value was 20.16 ± 0.032 and the intra-assay CV value was 0.71%. Tests of fluorescent RT-qPCR were carried out for the same specimen for successive 20 days so as to calculate the inter-assay CV values. The results showed that the Cq and inter-assay CV values of high- and low-concentration miR-135a-5p were 13.06 ± 0.089, 3.048% and 33.08 ± 0.211, 2.853%, respectively, and those of U6 were 7.42 ± 0.048, 2.893% and 20.58 ± 0.153, 3.325%, respectively. Therefore, all intra-assay CV values of miR-135a-5p and U6 are less than 1% and their inter-assay CV values less than 4%, which indicate that this method has good reproducibility.
Specificity
As a specific unimodal curve, the melting curve of fluorescent RT-qPCR indicated that the melting temperature of miR-135a-5p was about 76℃, and that of U6 was about 78℃, which shows that the specificity of this amplification is high (Figure 2).
The melting curve of testing serum miR-135a-5p and U6 by RT-qPCR.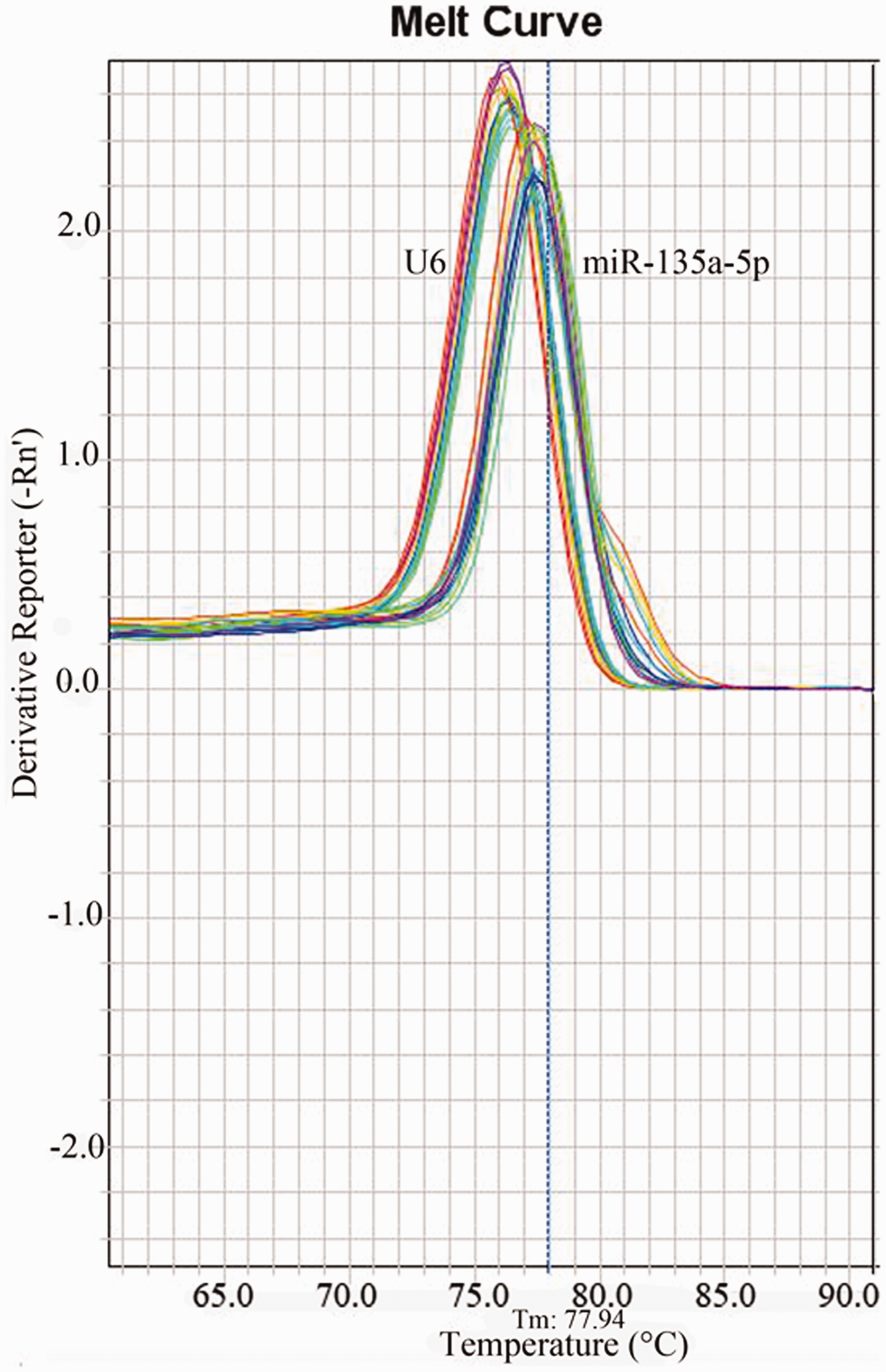
Serum miR-135a-5p expression in CRC, colorectal polyps and healthy control groups
miR-135a-5p in serum was detected by RT-qPCR. It was found that the relative expression levels of serum miR-135a-5p in CRC, colorectal polyps and healthy control groups were 2.451 (1.107, 4.413), 0.946 (0.401, 1.942) and 0.949 (0.194, 1.415), respectively. It was significantly higher in CRC group than that in the other two groups (U = 351.0, 313.0, both P < 0.001), and there was no significant difference between colorectal polyps and healthy control groups (U = 339.0, P = 0.172) (Figure 3). These results suggested that serum miR-135a-5p could be helpful in the auxiliary diagnosis of CRC.
The relative expression of serum miR-135a-5p in CRC, colorectal polyps and healthy control groups.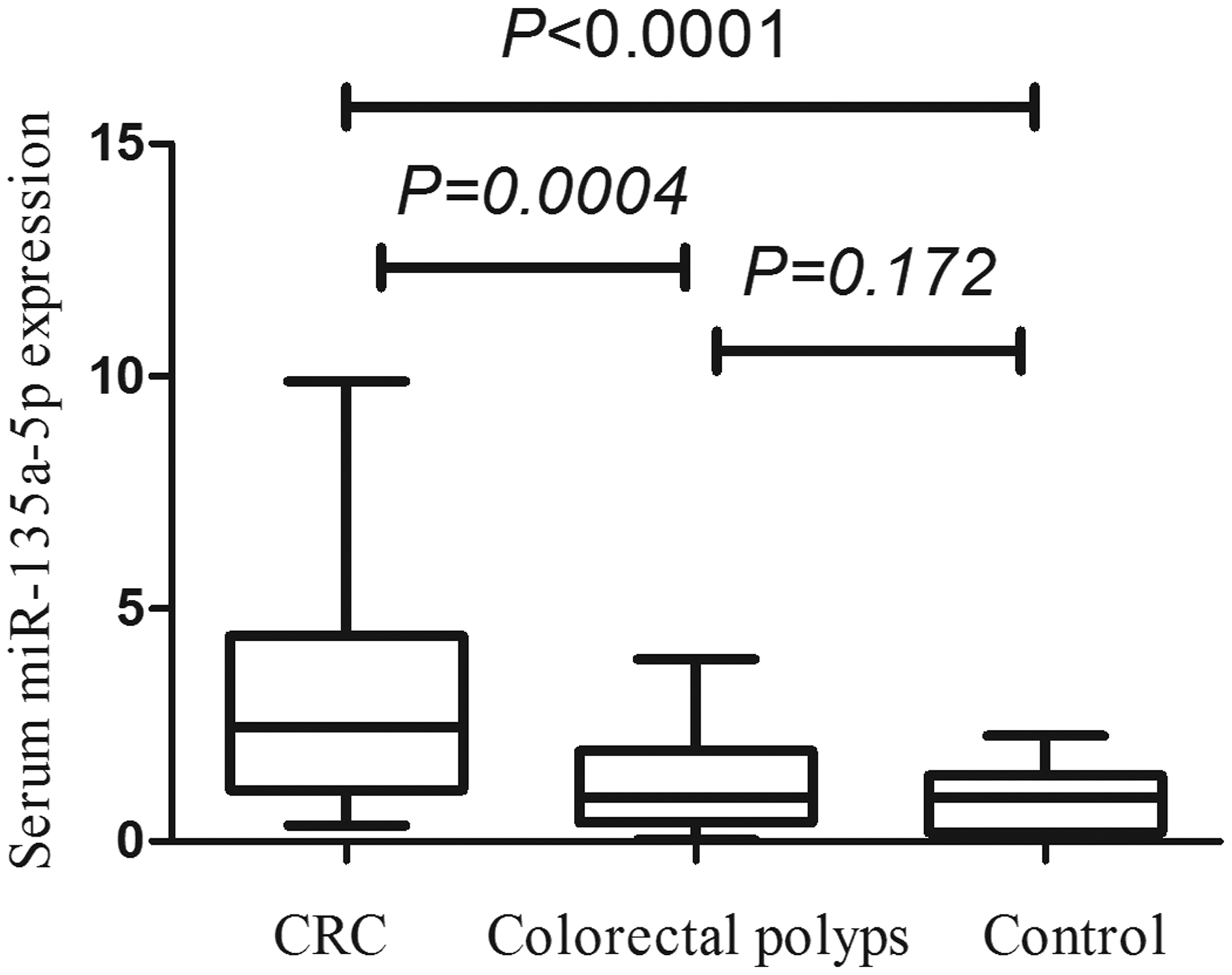
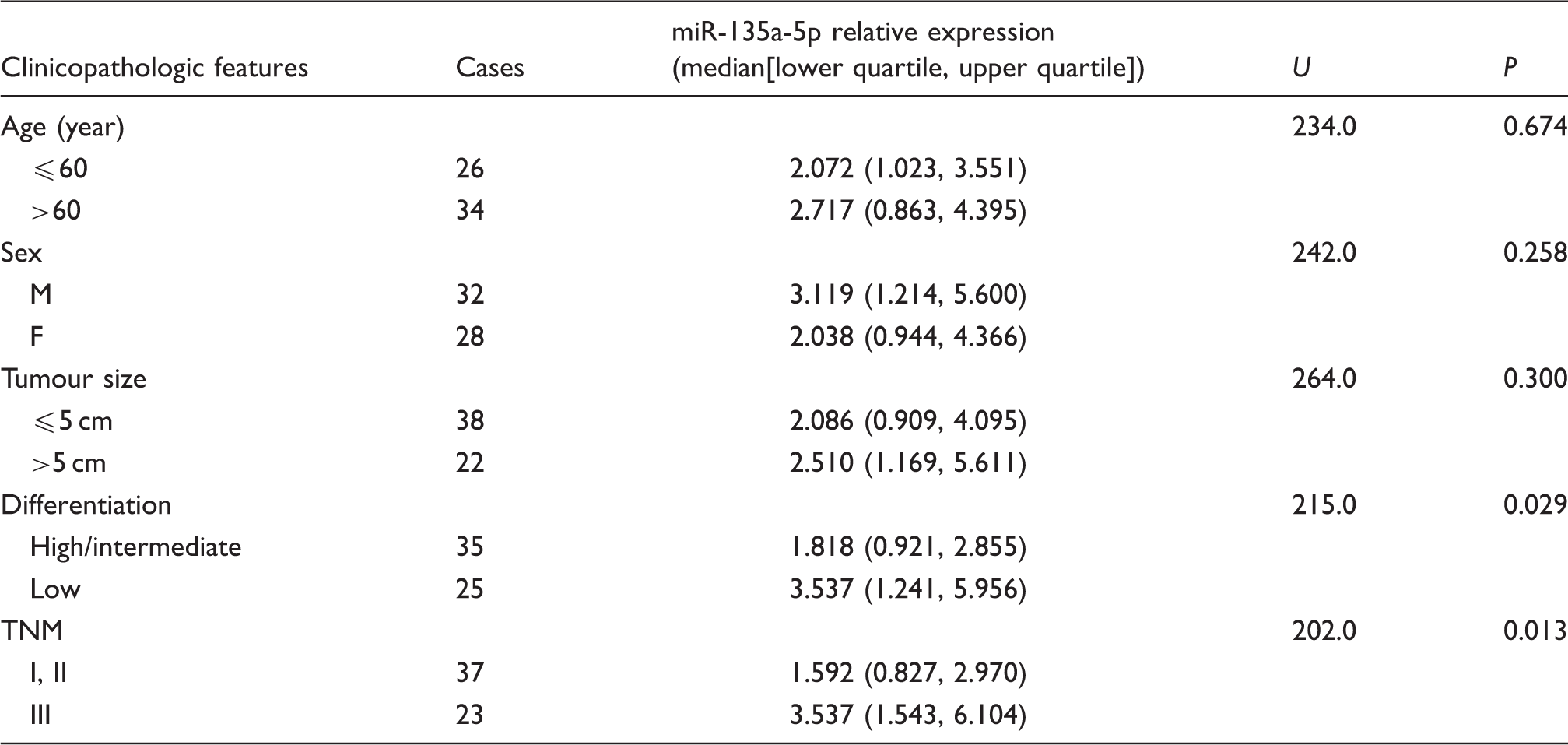
Correlations between the serum miR-135a-5p expression level and the clinicopathologic features in CRC patients
Correlations between the relative expression of serum miR-135a-5p and clinicopathologic features in 60 CRC patients.
Correlations between the relative expression of serum miR-135a-5p and CEA and CA199 in CRC patients
Correlations between the relative expression of miR-135a-5p and CEA and CA199 in CRC patients were analysed. The results are shown in Table 2, there was no significant correlation between the relative expression level of serum miR-135a-5p and CEA and CA199 in CRC patients (r2 = 0.023, P = 0.293; r2 = 0.067, P = 0.068) (Figure 4).
Correlations between the relative expression of serum miR-135a-5p and CEA (a), CA199 (b) in CRC patients. Expression level of serum CEA and CA199 in CRC, colorectal polyps and healthy control groups. CRC: colorectal cancer; CEA: carcinoembryonic antigen; CA199: carbohydrate antigen 199.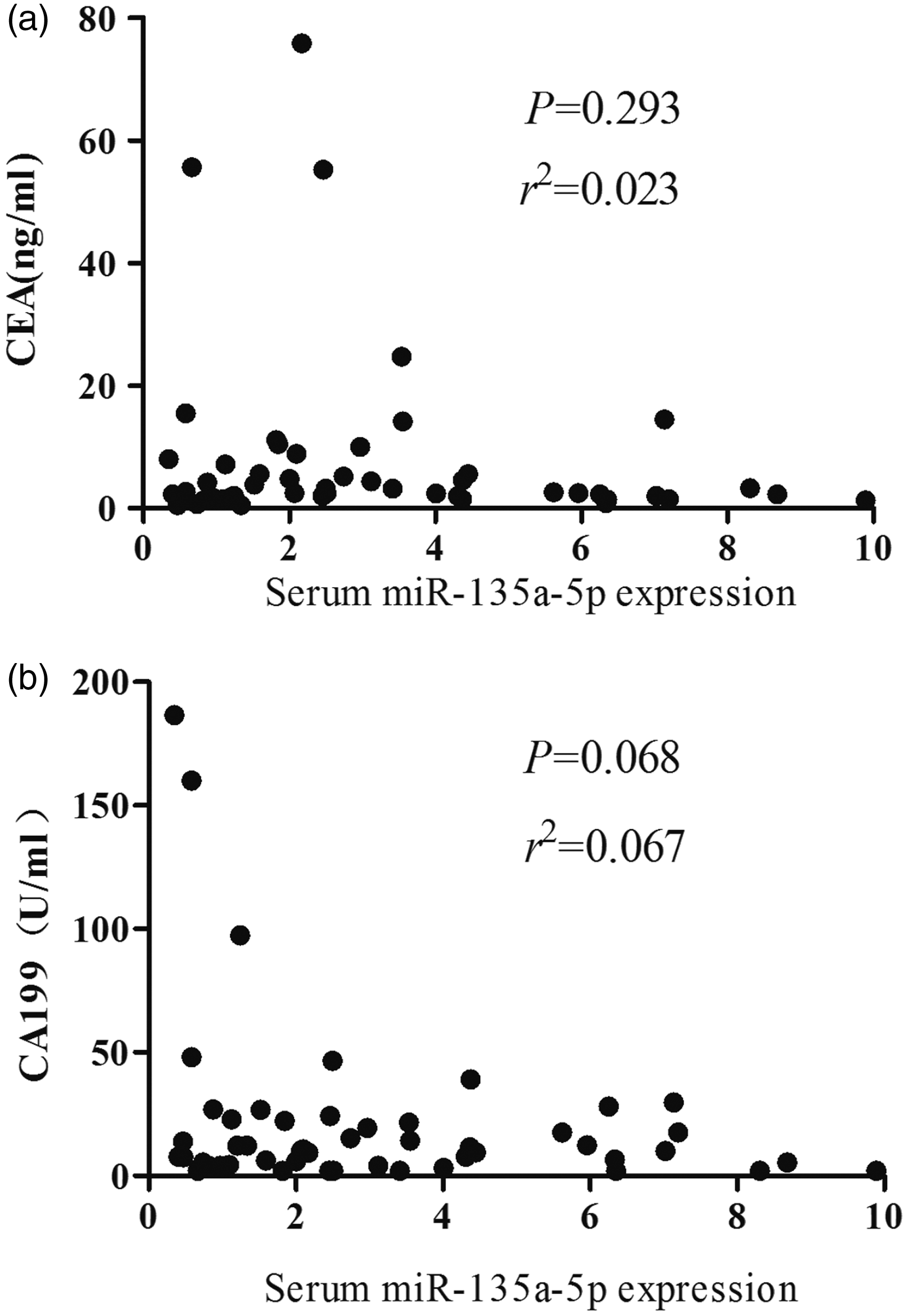
Analysis of the ROC curve
The relative expression levels of serum miR-135a-5p, CEA and CA199 in CRC, colorectal polyps and healthy control groups were mapped as ROC curves. In terms of comparison between CRC and colorectal polyps groups, at the cut-off value of 1.737 for miR-135a-5p, the related sensitivity, specificity and accuracy were 71.67%, 80% and 75%, respectively vs. 76.67%, 65% and 72% for CEA, and 36.67%, 90% and 58% for CA199. The AUC of miR-135a-5p was 0.832 with 95% CI 0.73–0.93 (Figure 3). In terms of comparison between CRC and healthy control groups, at the cut-off value of 1.760 for miR-135a-5p, the related sensitivity, specificity and accuracy were 76.67%, 88% and 81.82%, respectively, vs. 58.33%, 86% and 70.91% for CEA, and 58.33%, 68% and 62.73% for CA199. The AUC of miR-135a-5p was 0.875 with 95% CI 0.80–0.95 (Figure 5). Our results indicated that serum miR-135a-5p, as a potential biomarker, had a positive diagnosis value for CRC.
ROC curve of miR-135a-5p, CEA and CA199 for differentiating CRC patients from colorectal polyps group (a) and healthy control group (b).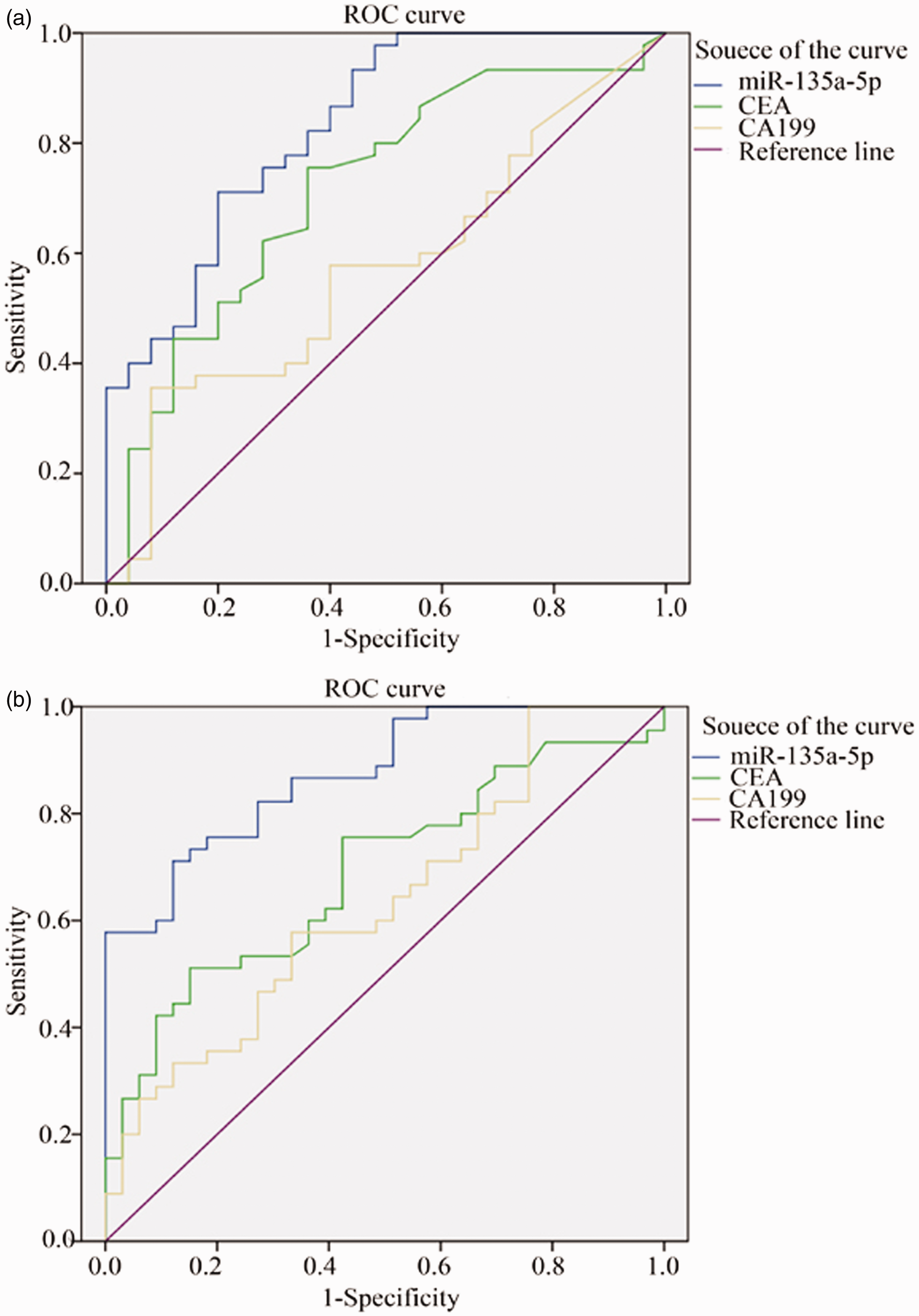
Combined detection of serum miR-135a-5p, CEA and CA199
Diagnostic efficacy of serum miR-135a-5p, CEA and CA199 in differentiating CRC patients from colorectal polyps group.
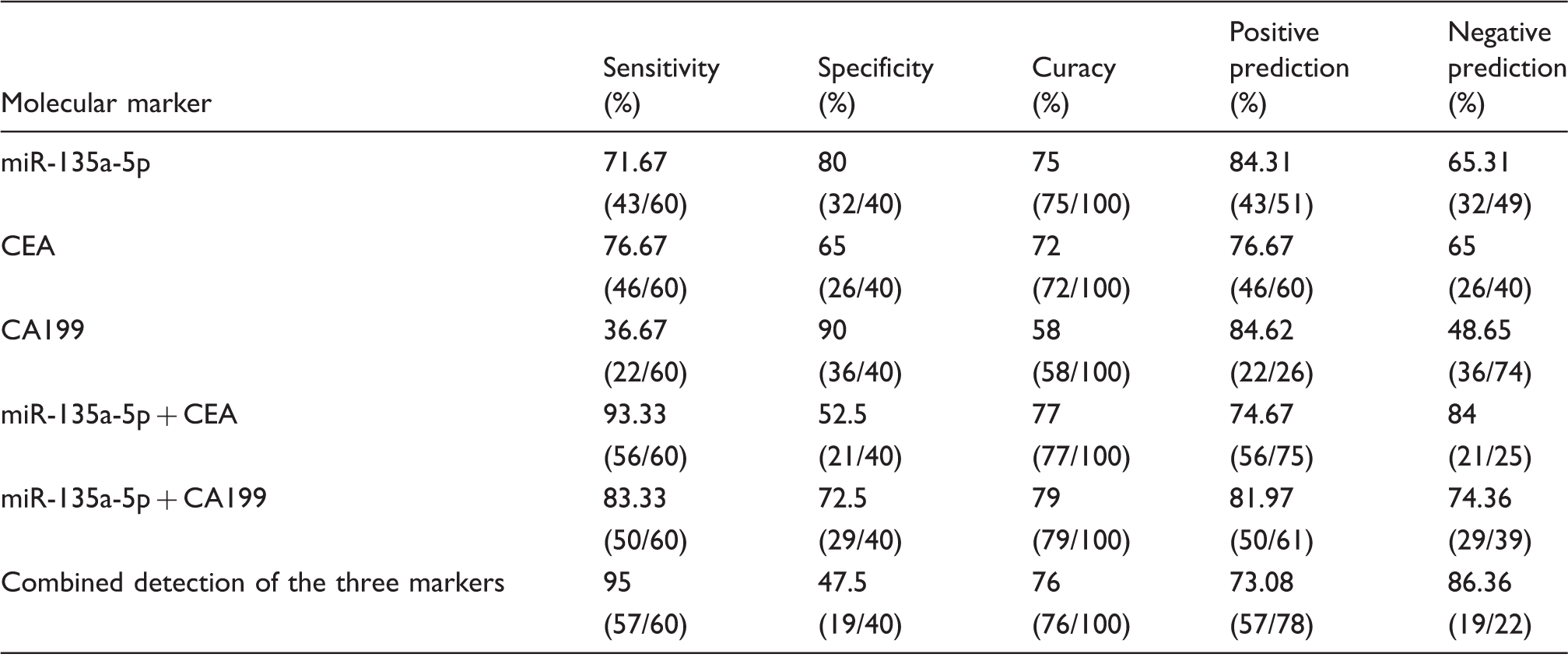
CEA: carcinoembryonic antigen; CA199: carbohydrate antigen 199.
Diagnostic efficacy of serum miR-135a-5p, CEA and CA199 in differentiating CRC patients from healthy control group.
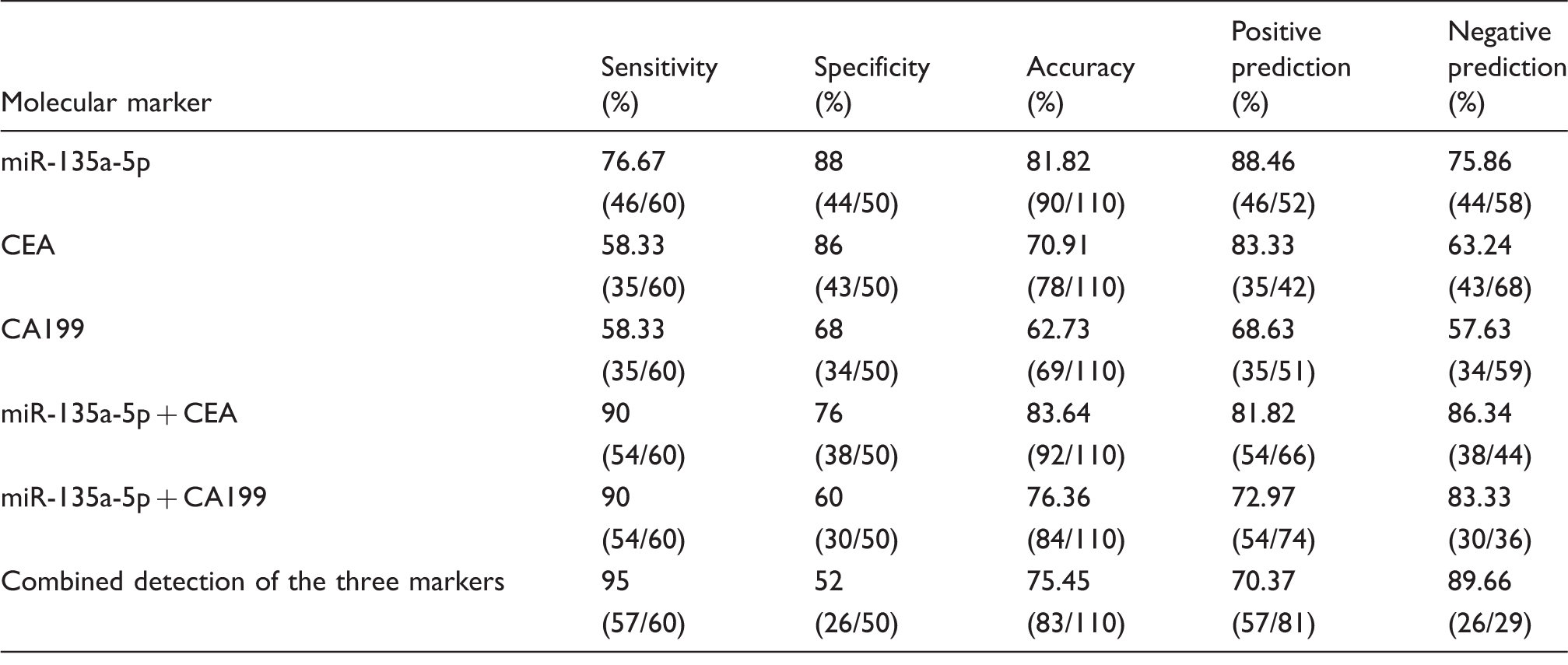
CEA: carcinoembryonic antigen; CA199: carbohydrate antigen 199.
Discussion
Ideal biomarkers should be easy to measure and have a strong association with clinical outcome. miRNAs could match these proposed criteria. 10 Although tumour tissue miRNA have been demonstrated to be associated with the development and prognosis of tumours in many studies, the complex manoeuver and large trauma to patients during its determination make it difficult to be applied to clinical practice. On the contrary, the concentrations of tumour-associated miRNAs in the serum of the peripheral blood are much easier to determine, and are more feasible for clinical application. Previous studies have revealed that circulating miRNAs are not only derived from circulating tumour cells and blood cells but also originate from cancer tissues or other tissue cells affected by disease. 11 Many articles have reported the association between serum miRNA and tumours, which provides a solid foundation for the application of serum miRNA as a molecular marker in the early diagnosis and prognosis of tumours.12,13 Lawrie et al. 14 first reported that serum miR-155 and miR-210 expressions were elevated in B-cell lymphoma patients, and that the expression level of miR-21 was correlated with tumour-free survival of the patients. Many other researchers have demonstrated that the change of miRNA expression level in the circulating blood or body fluid of cancer patients is a direct reflection of the physiologic or pathologic process of the body. Therefore, miRNA may prove to be a valuable non-invasive tumour marker for the diagnosis and prognostic prediction of patients with malignant tumours. Ng et al. 10 first discovered that miR-17a-3p and miR-92a were highly expressed in the serum of CRC patients and decreased seven days after surgery. Yuan et al. 15 detected the expression level of plasma miR-183 in 118 CRC patients and 61 healthy controls using fluorescence RT-qPCR and found that plasma miR-183 expression was highly expressed in CRC patients and decreased significantly after surgery, suggesting that miR-183 could be used as a diagnostic marker of CRC. Studies in recent years have also shown that miRNA is predictive of the prognosis in CRC patients. For instance, high expression of serum miR-885-5p in CRC patients could predict the prognosis, lymph node metastasis and distant metastasis independently. 16 In a study of using epithelial to mesenchymal transition-related miRNA as a serum marker to predict the prognosis and metastasis of CRC patients, Toiyama et al. 17 found that high expression of serum miR-200c predicted the existence of lymph node metastasis, distal metastasis and poor prognosis. In a study of differential expression of serum miR-126, miR-141 and miR-21 in CRC patients with hepatic metastasis, Yin et al. 18 reported that increased serum expression of miR-141 and miR-21 might prove to be a marker for early detection of hepatic metastasis of CRC. miRNA can also be used to predict the effect of neoadjuvant chemotherapy. Chen et al. 19 detected the serum expression of miR-155, miR-200 and miR-210 in CRC patients receiving cetuximab-assisted oxaliplatin therapy and found that continuous elevation of miR-155 expression could be used as a marker of resistance to chemotherapy in postoperative CRC patients. Rasmussen et al. 20 analysed 94 metastatic CRC patients who received XELOX or FOLFOX chemotherapy postoperatively and found that the expression of miR-625-3p, miR-181b and miR-27b were correlated with the effect of chemotherapy. They also demonstrated that the high expression of miR-625-3p was positively correlated with poor prognosis. These findings suggested that miR-625-3p could be used as an independent therapeutic response marker.
The carcinogenesis of CRC involves multiple alterations of oncogenes and tumour suppressor genes, such as inactivation of adenomatous polyposis coli (APC) and tumour protein 53 and oncogenic activation of KRAS.21,22 miR-135 had been shown to suppress expression of the tumour suppressor gene APC, inducing Wnt pathway signaling, possibly contributing to the development of colorectal adenomas and carcinomas. 23 Further, a study exploring miRNAs expression in exfoliated colonocytes from feces had shown significantly higher miR-135 in patients with CRC compared with healthy volunteers. 24 Vickers et al. 25 presented further evidence that miR-135 was higher in primary colorectal tumours compared with normal adjacent tissues and higher in tumours that have the ability to metastasize. Additionally, it had been indicated that miR-135a-5p promoted the growth and invasion of CRC cells, through targeting metastasis suppressor 1 (MTSS 1). 26
However, the diagnostic value of miR-135a-5p in serum has not been fully evaluated to date. In the present study, we detected the serum miR-135a-5p expression in 60 CRC patients, 40 colorectal polyps patients and 50 healthy controls and found that the relative expression level of serum miR-135a-5p in CRC patients was significantly higher than that in the other two groups (both P < 0.001), and there was no significance difference between colorectal polyps and healthy control group (P > 0.05). Previous study demonstrated that miR-135a expression was upregulated in CRC tissue and cell lines. 26 The result of the present study showed that miR-135a-5p was highly expressed in the serum of CRC patients, which was consistent with the report of Zhou et al. 26
Additionally, we analysed the possible correlations between the relative expression level of serum miR-135a-5p and the clinicopathologic features of CRC patients and found that it was not correlated with sex, age and maximum tumour diameter (all P > 0.05) but significantly correlated with different degrees of tumour differentiation and stages (both P < 0.05), indicating that there was a tendency of increased serum miR-135a-5p expression in late-stage and poorly differentiated CRC patients. These results suggested that detection of serum miR-135a-5p level might have some clinical significant in monitoring the progression of the disease and prediction of prognosis in CRC patients.
We also detected serum miR-135a-5p, CEA and CA199 concentrations in all participants of the present study. The result of correlation analysis showed that the serum miR-135a-5p expression was not correlated with CEA and CA199 concentrations in CRC patients. ROC curve analysis showed that in terms of comparison between CRC and colorectal polyps groups, AUC of miR-135a-5p was 0.832 with 95% CI 0.73–0.93. At the cut-off value of 1.737, the accuracy of miR-135a-5p was superior to that of CEA and CA199. In terms of comparison between CRC and healthy control groups, AUC of miR-135a-5p was 0.875 with 95% CI 0.80–0.95. At the cut-off value of 1.760, the sensitivity, specificity and accuracy of miR-135a-5p were all superior to those of CEA and CA199. Our study demonstrated that combined detection of miR-135a-5p, CEA and CA199 could greatly improve the diagnostic efficacy of CRC.
In summary, miR-135a-5p expression was elevated in the serum of CRC patients. Fluorescence RT-qPCR detection of miR-135a-5p level in the serum of CRC patients could be used as a means of auxiliary diagnosis of CRC. Combined detection of miR-135a-5p, CEA and CA199 could improve the diagnostic efficacy of CRC. miR-135a-5p may prove to be an important biomarker for the diagnosis of CRC. As the sample size of the present study is relatively small, it is necessary to conduct further identification with clinical specimens to confirm our conclusion, and its expression in CRC tissue and action mechanism in CRC are need to be further explored.
Footnotes
Acknowledgements
We would like to thank Surgical Comprehensive Laboratory of Affiliated Hospital of Nantong University.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
This study was supported by the National Nature Science Foundation of China (No. 81301498, 81271920, 81201351), Jiangsu Provincial Medical Innovation Team and Leading Talents (LJ201133).
Ethical approval
The study was approved by the Ethics Committee of Nantong University affiliated Hospital with the permit number: 2014(126).
Guarantor
SJ.
Contributorship
QW researched the literature and conceived the study. QW, HZ and XS were involved in protocol development, gaining ethical approval, patient recruitment and data analysis. QW wrote the first draft of the manuscript. All authors reviewed and edited the manuscript and approved the final version of the manuscript.
