Abstract
Background
Obesity is associated with a state of systemic inflammation, mediated by adipose tissue-derived cytokines that may also have metabolic effects, including an effect on insulin resistance. The aim of this study was to compare the serum profile of pro- and anti-inflammatory cytokines in obese and non-obese subjects.
Methods
A total of 242 subjects who were either overweight or obese (body mass index [BMI] ≥ 25 kg/m2) and non-obese subjects (body mass index <25 kg/m2), were recruited in Mashhad in northeastern Iran. The concentrations of serum interleukin-1α, -1β, -2, -4, -6, -8 and -10 (IL-1α, IL-1β, IL-2, IL-4, IL-6, IL-8 and IL-10), were measured in all subjects, together with serum vascular endothelial growth factor, interferon-γ, epidermal growth factor, monocyte chemoattractant protein-1 and tumour necrosis factor-α.
Results
The groups differed significantly with respect to measures of adiposity and fasted lipid profile. Serum pro-inflammatory cytokines interferon-γ and interleukin-1α, and anti-inflammatory cytokines, interleukin-10, and epidermal growth factor were significantly different between obese and non-obese individuals, as was serum high-sensitivity C-reactive protein. Multivariate regression showed that waist circumference was significantly and independently related to serum monocyte chemoattractant protein-1concentrations (P = 0.001).
Conclusion
Despite significant differences in several cytokines between the groups, only monocyte chemoattractant protein-1appeared to be independently related to a measure of adiposity in this population sample from Iran.
Introduction
Obesity is an important public health challenge globally. There are large differences in the prevalence of overweight and obesity regionally. 1 Obesity is a growing problem in both developed and low-income countries.2–5 The World Health Organization (WHO) has reported that there are 1 million excess deaths and 12 million life-years of ill health attributable to obesity every year. Sociocultural and environmental factors are determinants of diet and physical activity; consequently, there is a significant difference in the prevalence of obesity between different countries. 1 In Iran, there has been a rapid change in dietary habits and physical activity levels that started during the 1990s and has led to an increased obesity rate among the Iranian people.4,6
Obesity is a common condition that is defined by an excess accumulation of fat in the adipose tissue. It is associated with metabolic, haematological and musculoskeletal complications, including, dyslipidaemia, heart disease, diabetes and increased risk of some malignancies, leading to a shortened life span.2,7 Visceral obesity is defined by the presence of excess fat in the abdominal cavity, and is an independent risk factor for several other conditions. 7
Inflammation arising from within the adipose tissue has been identified as a major source of systemic inflammation and may also be associated with insulin resistance. Adipocytes and adipose tissue-associated macrophages from obese individuals are an important source of inflammatory mediators such as tumour necrosis factor-α (TNF-α), interleukin (IL)-6, IL-1, and monocyte chemoattractant protein-1 (MCP)-1. 8 It has been proposed that adipokines such as leptin can induce T-helper cells to secrete pro-inflammatory cytokines including TNF-α, and interferon-γ (IFN-γ).9,10 IL-1 suppresses adipocyte differentiation and the expression of lipoprotein lipase in adipose tissue. A member of this family, IL-1α, is involved in the regulation of adipogenesis and energy expenditure. 11 Indeed, activation of inflammatory pathways in adipose tissue promotes the release of free fatty acids from triglyceride (TG) stores and results in macrophage infiltration into tissue. 8 In contrast, decreased concentrations of the anti-inflammatory cytokine, IL-10, have been shown to be associated with obesity and metabolic syndrome. IL-10 inhibits the synthesis of pro-inflammatory cytokines via suppression of NF-kB in macrophages. 12 A negative relationship between serum IL-10 and body mass index (BMI) has been reported previously. 2
Because of the potential importance of cytokine activity in obesity and its related complications, we aimed to compare a panel of pro-inflammatory and anti-inflammatory cytokines in the serum of obese, overweight and non-obese people living in northeastern Iran.
Methods
Study design and participants
A total of 242 subjects were enrolled into the study. The subjects with obesity were referred to the clinic of Ghaem Hospital, Mashhad for nutritional advice. Using the WHO (BMI) classification, 13 77 individuals were classified as non-obese (BMI = 20–24.9 kg/m2), 76 individuals were classified as overweight (BMI = ≥ 25–29.9 kg/m2) and 89 individuals were classified as obese (BMI = ≥30). The control subjects did not have a history of major systemic disease, including renal or infectious disease or lupus. The non-obese, control group, was recruited from the normal population of Mashhad, Iran. We determined the fasting blood glucose (mmol/L) concentrations of each individual in the non-obese group, and this was less than 5.2 mmol/L for each subject. Exclusion criteria included: a history of endocrinological abnormalities, congestive heart disease, liver and/or renal disease, pregnancy and alcohol consumption or treatment with medications that altered blood pressure, glucose or lipid metabolism.
Blood collection and routine biochemistry
Blood samples (10 mL) were obtained in the early morning after an overnight fast. Blood samples were collected into plain Vacutainer™ tubes for lipid profile measurements, and into Vacutainer™ tubes containing fluoride-oxalate for measurement of fasting blood glucose.
Blood samples were centrifuged and stored at −80℃. Low-density lipoprotein cholesterol, high-density lipoprotein, total cholesterol (TC), cholesterol and glucose were measured using routine techniques using a Cobas Auto Analyser system (ABX Diagnostics, Montpellier, France). Subjects with diabetes or with a serum high-sensitivity C-reactive protein (hsCRP) ≥ 95.24 mmol/L were excluded from the study.
The study was approved by the Ethics Committee of the Mashhad University of Medical Sciences, and informed consent was obtained from individuals who were included in the criteria for inclusion.
Measurement of cytokines
Cytokines measurements were performed using Biochip Array Technology on a Randox Evidence Investigator analyser (Randox Laboratories, Belfast, Northern Ireland). The Evidence Investigator Biochip Array Technology was used to perform simultaneous quantitative detection of multiple analytes using a single patient sample. Intra- and interassay CVs for cytokine markers were ≤10%. 14 The cytokine array biochip employs a sandwich chemiluminescent immunoassay for a high-throughput measurement of circulating cytokines. The light signal generated from each of the test regions on the biochip is detected using digital imaging technology and compared to that from a stored calibration curve. The serum concentrations of the cytokines were derived from the calibration curve. Serum IL-1α, IL-1β, IL-2, IL-4, IL-6, IL-8, IL-10, vascular endothelial growth factor (VEGF), IFN-γ, epidermal growth factor (EGF), MCP-1 and TNFα were determined.
Measurement of baseline data
Demographic data and anthropometric parameters were obtained by using a general questionnaire. Weight was measured using electronic scales when subject with wearing light clothing without shoes. Height was measured using a wall-mounted stadiometer. Blood pressure was measured using a mercury sphygmomanometer according to the standard procedures, and periodical validity and reliability of instrument were checked.
Statistical analyses
Data analyses were performed using the SPSS software (version 16). Comparisons between groups were performed using either t-test or Mann-Whitney U test. Normally distributed data were presented as means ± standard deviation (SD). Analysis of trends was performed using linear regression models to determine the effect of anthropometric characteristics on serum cytokine and growth factor level. A P < 0.05 was considered to be statistically significant.
Results
Participant characteristics associated with evaluation base line data
Base line demographic, anthropometric and biochemical data of subjects in each group.
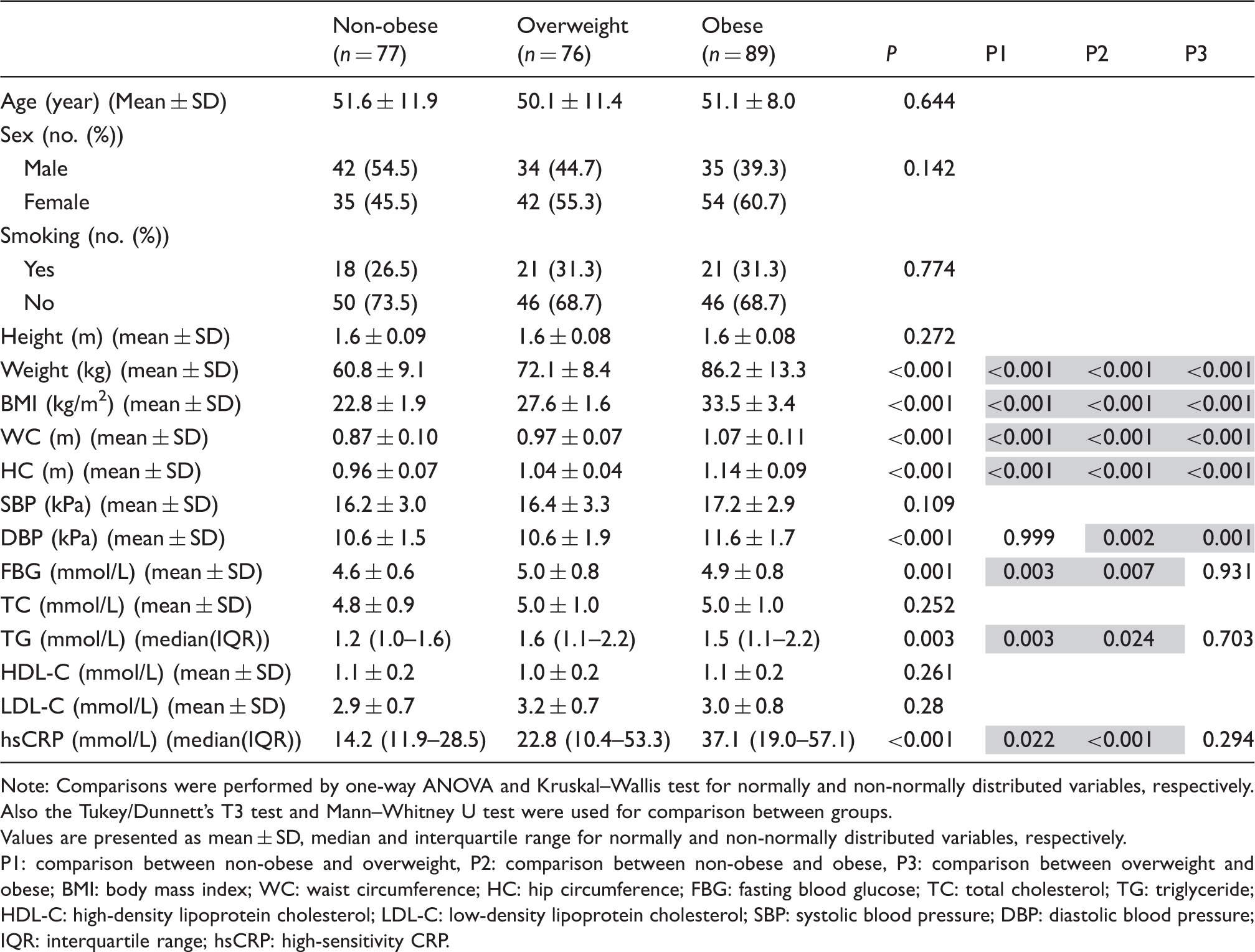
Note: Comparisons were performed by one-way ANOVA and Kruskal–Wallis test for normally and non-normally distributed variables, respectively. Also the Tukey/Dunnett’s T3 test and Mann–Whitney U test were used for comparison between groups.
Values are presented as mean ± SD, median and interquartile range for normally and non-normally distributed variables, respectively.
P1: comparison between non-obese and overweight, P2: comparison between non-obese and obese, P3: comparison between overweight and obese; BMI: body mass index; WC: waist circumference; HC: hip circumference; FBG: fasting blood glucose; TC: total cholesterol; TG: triglyceride; HDL-C: high-density lipoprotein cholesterol; LDL-C: low-density lipoprotein cholesterol; SBP: systolic blood pressure; DBP: diastolic blood pressure; IQR: interquartile range; hsCRP: high-sensitivity CRP.
Serum cytokines and growth factor levels
Comparison of serum cytokine and growth factor concentrations in groups of individuals with different degrees of adiposity.
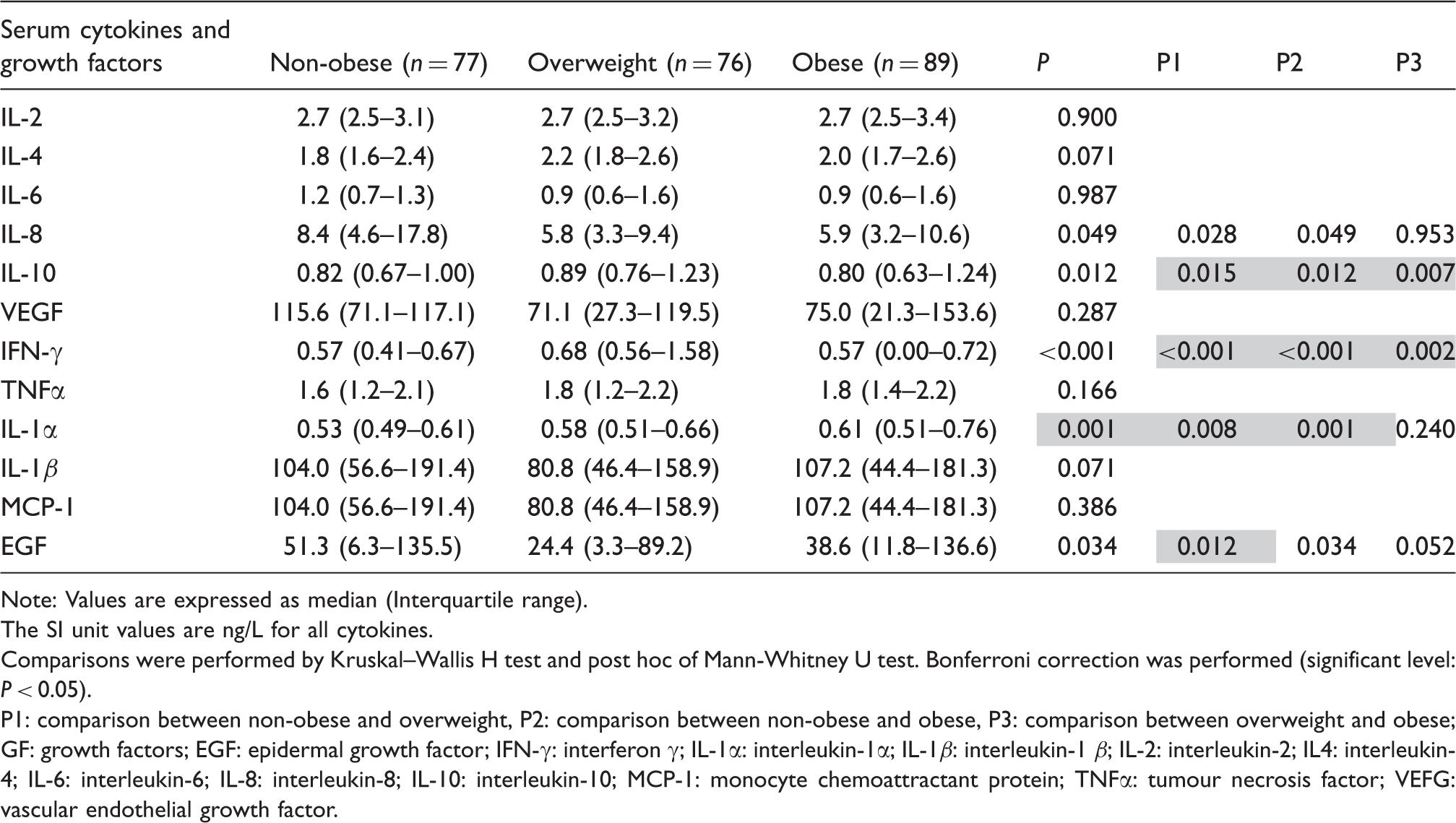
Note: Values are expressed as median (Interquartile range).
The SI unit values are ng/L for all cytokines.
Comparisons were performed by Kruskal–Wallis H test and post hoc of Mann-Whitney U test. Bonferroni correction was performed (significant level: P < 0.05).
P1: comparison between non-obese and overweight, P2: comparison between non-obese and obese, P3: comparison between overweight and obese; GF: growth factors; EGF: epidermal growth factor; IFN-γ: interferon γ; IL-1α: interleukin-1α; IL-1β: interleukin-1 β; IL-2: interleukin-2; IL4: interleukin-4; IL-6: interleukin-6; IL-8: interleukin-8; IL-10: interleukin-10; MCP-1: monocyte chemoattractant protein; TNFα: tumour necrosis factor; VEFG: vascular endothelial growth factor.
The effect of anthropometric characteristics on cytokine and growth factors
Association of anthropometrics variables and serum cytokines and growth factors using linear regression models.
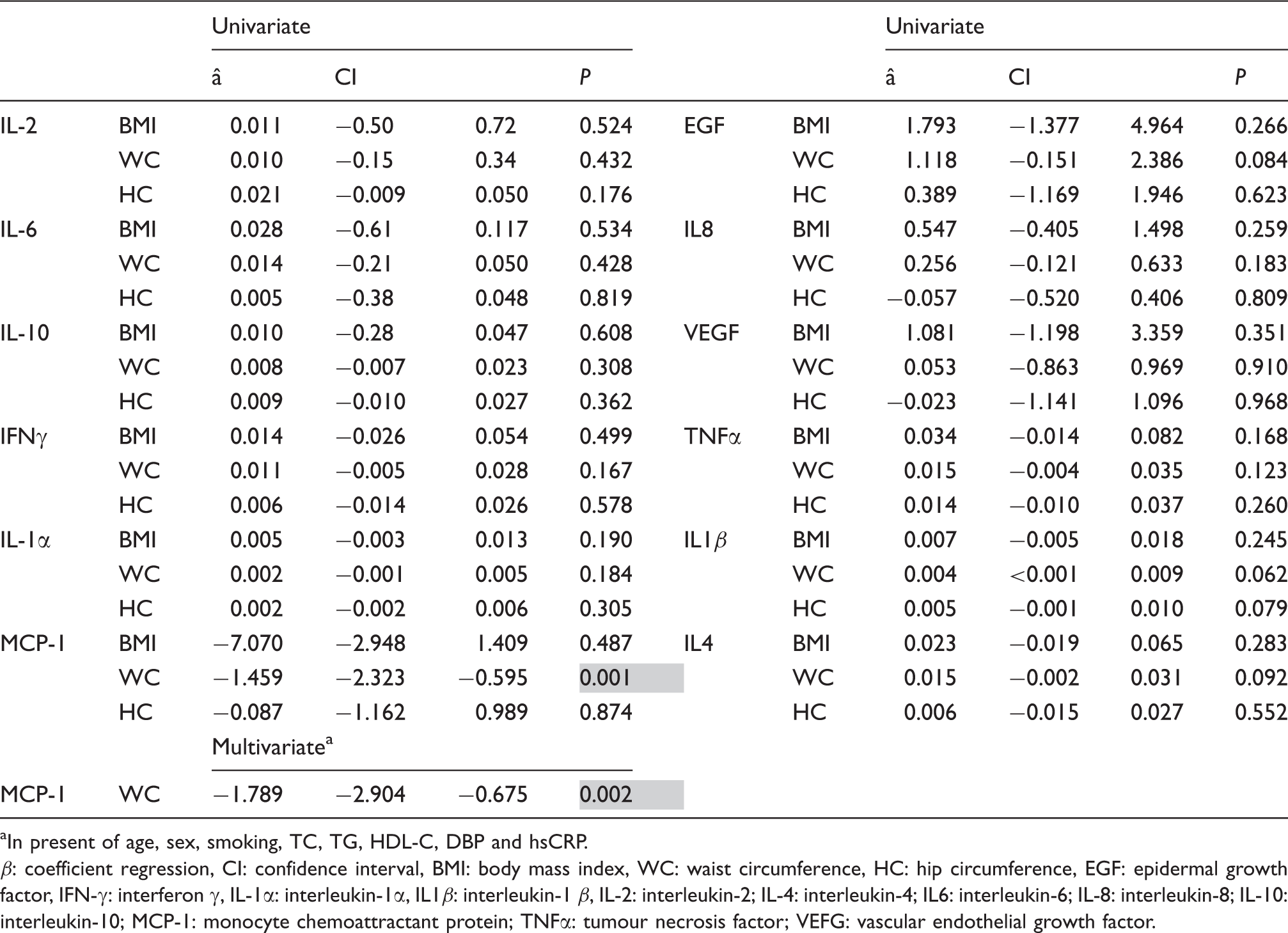
In present of age, sex, smoking, TC, TG, HDL-C, DBP and hsCRP.
β: coefficient regression, CI: confidence interval, BMI: body mass index, WC: waist circumference, HC: hip circumference, EGF: epidermal growth factor, IFN-γ: interferon γ, IL-1α: interleukin-1α, IL1β: interleukin-1 β, IL-2: interleukin-2; IL-4: interleukin-4; IL6: interleukin-6; IL-8: interleukin-8; IL-10: interleukin-10; MCP-1: monocyte chemoattractant protein; TNFα: tumour necrosis factor; VEFG: vascular endothelial growth factor.
Discussion
As for several previous studies,15,16 we found that in addition to age, gender and blood pressure, several biochemical parameters, including, blood glucose and TGs were associated with degrees of adiposity. Cytokines are mediators of pro- and anti-inflammatory responses. We found that among our panel of pro/anti-inflammatory cytokines and growth factors, IL-10, IFN-γ and IL-1α were significantly (P < 0.05) different between non-obese, overweight and obese people. We observed an incremental trend in the serum concentrations of these cytokines from non-obese to obese groups. We also showed that serum EGF concentrations were significantly lower in overweight people compared to the non-obese controls. Previous reports have also shown that people with obesity have a higher concentration of serum IL-10, IFN-γ, TNF-α, and IL-12, and that high concentrations of IL-4, IL-10 and IL-13 are related to low physical activity in obese people. 17 A further study has reported a high concentration of IL-10 in obese people, but that the metabolic syndrome was not associated with low concentrations of IL-10. 18 A study in young adolescents in Taiwan has reported low concentration of serum IL-10 and high concentrations of serum IL-1β in overweight and obese subjects. The authors also reported a negative correlation of IL-10 with IL-1β cytokines in obesity. 12 In a further study on the association of inflammatory cytokines in obese and non-obese women, it was observed that the concentration of serum TNF-α, IL-6, P-selectin, intercellular adhesion molecule 1 (ICAM-1) and vascular cell adhesion molecule 1 (VCAM-1) was higher in obese women, and that weight loss reduced the concentrations of these cytokines in the obese group. 19 For other cytokines and growth factors including IL-2, IL-4, IL-6, IL-8, VEGF, TNF-α, IL-1β and MCP-1, we did not observe a significant (P < 0.05) differences between our groups. We found a high variation in serum cytokine concentrations within each group, highlighting the dependency of the circulatory cytokine concentrations to various conditions.
Although there have been previous reports of high concentrations of IL-1, IL-18, IL-8 and IL-6 in obese people,20–22 we did not find any significant differences in serum IL-6 and IL-8 between our groups.
VEGF did not differ significantly between the groups. The overexpression of VEGF in white and brown adipose tissue has been shown to protect the animals against high-fat diet-induced hypoxia and obesity. 23 IL-4 is another important cytokine involved in adipogenesis inhibition and lipolysis enhancement. 24 An increased concentration of IL-4 has been reported in girls with central obesity. 25 In present study, we observed a statistically non-significant difference in IL-4 concentration from non-obese to obese group.
We performed a univariate analysis to evaluate the effects of three anthropometric variables (BMI, WC and HC) on serum cytokine concentrations. As data in Table 3 show, there was an independent negative relationship between WC and serum MCP-1 concentrations. WC which in contrast to BMI is independent of age 26 showed a significant independent association with serum MCP-1 when this was evaluated after correction for confounding factors. Several studies have identified a strong association between plasma MCP-1 concentrations and body weight.27–29 An increase in MCP-1 expression by adipose tissue may affect macrophage infiltration into this tissue, causing an enhancement of the inflammatory state in obesity.28,30
In summary, we have found a complex relationship between adiposity and serum cytokine and growth factor profile. However, there was a consistent and independent relationship between adiposity and serum MCP-1 concentrations. Serum MCP-1 may enhance the pro-inflammatory milieu by increasing macrophage infiltration of adipose tissue.
Footnotes
Acknowledgements
This work was conducted as a MSc degree thesis (thesis number: A450) with financial support provided by the Research Council at the Mashhad University of Medical Sciences, Mashhad, Iran.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
This work was supported by the Research Council at the Mashhad University of Medical Sciences, Mashhad, Iran.
Ethical approval (including reference number)
The study was approved by the Ethics Committee of the Mashhad University of Medical Sciences (REC Number: 938052) and informed consent was obtained from individuals who were included in the criteria for inclusion.
Guarantor
Mashhad University of Medical Sciences.
Contributorship
Majid Ghayour-Mobarhan was involved in protocol development, gaining ethical approval, patient recruitment and data analysis. Elahe Mahdipour was involved in developing and conducting the laboratory experiments and the manuscript revision. Maryam Azizian and Seyed Reza Mirhafez have done the experiments and the statistical analysis. Sara Shoeibi wrote the first draft of the manuscript. All authors reviewed and edited the manuscript and approved the final version of the manuscript.
