Abstract
Background
Thrombospondin-1 (TSP-1) is a circulating matricellular glycoprotein produced from many cell types including platelets. Currently TSP-1 is measured in either plasma or serum, using expensive commercial assays.
Aim
To develop and validate a cost effective in-house immunoassay for human TSP-1 suitable for quantitating levels from both plasma and serum.
Methods
An in-house enzyme-linked immunosorbent assay (ELISA) was developed for the measurement of human TSP-1. Sixteen healthy volunteers (8 male and 8 female), mean age 29 years (range 21–49), body mass index (BMI) mean 23.3 kg/m2 (range 17.3–26.7) had non-fasted venous blood sampled at 0800 h and 1600 h for both plasma and serum TSP-1.
Results
The assay limit of quantitation was 7.8 μg/L, inter assay CV was 17–31%, intra assay CV was 3–4% for plasma and <9% for serum. Plasma TSP-1 ranged from 133 to 478 μg/L (mean concentration 290 μg/L) in normal volunteers. Serum TSP-1 was approximately 100-fold higher, ranging from 13,700 to 44,400 μg/L (mean concentration 257,00 μg/L). There was no correlation between plasma and serum TSP-1.
Conclusions
TSP-1 can be readily measured in human plasma using ELISA. Serum concentrations are 100-fold higher, reflecting documented TSP-1 release by platelets, and does not provide a meaningful measure of circulating concentrations.
Introduction
Thrombospondin-1 (TSP-1) is a large matricellular glycoprotein originally identified as being released from platelets, characterized as a thrombin-sensitive factor important for platelet aggregation. 1 It has since been discovered that TSP-1 is produced and secreted from a number of other cells types, including endothelium, adipose and macrophages. 2 TSP-1 is readily detectable in the circulation, and can be sequestered to the extracellular matrix where it binds to a number of cell surface receptors including CD36, CD51, CD47, transforming growth factor-β (TGF-β) and members of the integrin family. Each of these receptors engages distinct downstream signalling pathways, allowing TSP-1 to modulate a variety of cellular processes in a context-dependent manner. 2
There have been a number of studies examining circulating TSP-1 concentrations in conditions such as peripheral vascular disease, 3 diabetes 4 and cancer.5,6 Thus, the importance of accurately assessing circulating TSP-1 is of considerable clinical interest. The aim of the study was to develop a cost-effective ELISA assay using readily available commercial antibodies for the measurement of circulating TSP-1, and obtain preliminary data concerning plasma and serum concentrations in healthy individuals.
Materials and methods
Healthy volunteers
Sixteen healthy volunteers, eight male and eight female, were recruited for the study. The mean age of participants was 29 years (range 21–49) and mean body mass index was 23.3 kg/m2 (range 17.3–26.7). Venous blood was sampled at 0800 h and 1600 h in a non-fasting state. Blood samples were collected at room temperature (RT) into EDTA tubes for plasma, or untreated tubes with gel separator for serum (VACCUETTE, Greiner Bio-one, Austria). Tubes were spun at 1500g for 10 min at 4℃, and serum and plasma stored at −80℃ for up to eight months prior to assay.
TSP-1 immunoassay
An indirect sandwich ELISA was developed and validated to assess the concentration of TSP-1 in plasma and serum. All washing steps used five cycles of 300 μL/well phosphate buffered saline with 0.05 % Tween 20 (PBST). Ninety-six-well plates were coated with 100 μL/well of monoclonal human anti-TSP-1 antibodies (1000 μg/L) (MAB3074, R&D Systems, Minneapolis, MN) in 0.05 M carbonate buffer (pH 9.6) and incubated overnight at 4℃. After washing, plates were blocked with 1% fraction V, IgG-free BSA buffer (blocking buffer) (Life Technologies, Victoria, Australia) for 1 h at RT. Plates were washed before the addition of standards and samples (100 μL) and all dilutions were performed in blocking buffer. Recombinant human TSP-1 (3074-TH-050, R&D Systems) was serially diluted from 1000 μg/L to 15.63 μg/L. Plasma and serum samples were diluted at 1:4 and 1:100, respectively, and tested in duplicate. Plates were incubated for 2 h at RT, washed, and biotinylated polyclonal human anti-TSP-1 antibodies (BAF3074, R&D Systems) (100 μg/L) were added (100 μL/well) and plates incubated for 2 h at RT. Following washing, Poly-horseradish peroxidase (Poly-HRP) Streptavidin conjugate (Thermo Scientific, Victoria, Australia) diluted 1:20,000 was added (100 μL/well) for 30 min at RT. Plates were washed, and incubated at RT for 20 min in peroxidase substrate 3,3′,5,5′-tetramethylbenzidine (TMB) solution (Cell-Signalling, Danvers, MA) (100 μL/well). The reaction was terminated by the addition of stop solution (Cell-Signalling) (100 μL/well) and the optical density (OD) was measured at 450 nm using a POLARstar Omega plate-reader (BMG Labtech, Offenburg, Germany). The calibration curve was obtained using a four parameter logistics (4-PL) curve fit. A blank of blocking buffer was included on every plate.
Commercial TSP-1 ELISA
A human TSP-1 Immunoassay (DTSP10, R&D Systems) was used as per the manufacturer’s instructions to measure plasma and serum TSP-1 concentrations from eight individuals.
Validation
The assay limit of quantitation was calculated as the lowest concentration which could be repeatedly measured (5 times) with a CV of no greater than 10%. The inter assay CV was calculated from six standard curves assayed on separate days, across two different batches of reagents (2 replicates from one batch, 4 replicates from another batch). Each data set was applied to a reference standard curve to generate TSP-1 concentrations. The intra assay CV was calculated from 14 separate plasma, and 14 separate serum samples assayed consecutively in duplicate. The linearity of the assay was determined by performing serial dilutions of four different plasma or serum samples. Sample recovery was evaluated by spiking plasma samples from four individuals with 400 μg/L or 800 μg/L, and serum samples from four individuals with 10,000 μg/L or 20,000 μg/L.
Cross-reactivity
Recombinant human TSP-2 (1635-T2, R&D Systems) was used to create a standard curve which was tested in our in-house assay. According to the manufacturer MAB3074 displays no cross-reactivity with TSP-2, and BAF3074 displays less than 0.2% cross-reactivity with TSP-2.
Statistical analysis
Comparisons were analysed by two-tailed unpaired t-tests. Correlation between parameters were analysed using the Pearson coefficient. All analysis was performed using GraphPad Prism version 6 (GraphPad, CA). Passing–Bablok regression analysis was used to compare the commercial ELISA kit and the in-house assay. 7 If the 95% confidence interval (CI) for intercept includes the value 0, it is concluded that there is no constant difference between two methods; if the 95% CI for the slope included the value 1, it is concluded that there is no proportional difference between the two methods.
Results
The assay limit of quantitation was 7.8 μg/L. The inter assay CV was 17–31% across all concentrations, the intra assay CV was 3–4% for plasma and <9% for serum. The linearity of the assay was determined for both plasma and serum samples. Linearity was maintained within the range of 1:4 to 1:64 in plasma (Figure 1a), and within 1:100 to 1:800 in serum (Figure 1b). In order to assess the ability of the assay to recover analyte, both plasma and serum samples (n = 4) were spiked with 400 and 800 μg/L or 10,000 and 20,000 μg/L recombinant TSP-1, respectively. The mean percent recovery was 101 for plasma (range 94–105), and 97 from serum (range 53–107) (Figure 1c). The manufacturers claim that the antibodies used in this assay demonstrate minimal cross-reactivity with the highly related TSP-2. To test this, recombinant human TSP-2 was used at a range of concentrations (Figure 1d). Even at high concentrations (1000 μg/L), signal from TSP-2 did not rise above background levels.
In-house TSP-1 ELISA validation. TSP-1 concentrations determined from plasma (a) or serum (b) of four healthy individuals serially diluted to assess linearity; (c) recovery of recombinant TSP-1 spiked into plasma or serum samples from four individuals; (d) assessment of cross reactivity with recombinant TSP-2 at a range of concentrations; (e) comparison of in-house method with a commercially available kit using Passing–Bablok regression analysis. Values presented are for diluted samples Black line indicates regression analysis, red line indicates identity line (colour version available online).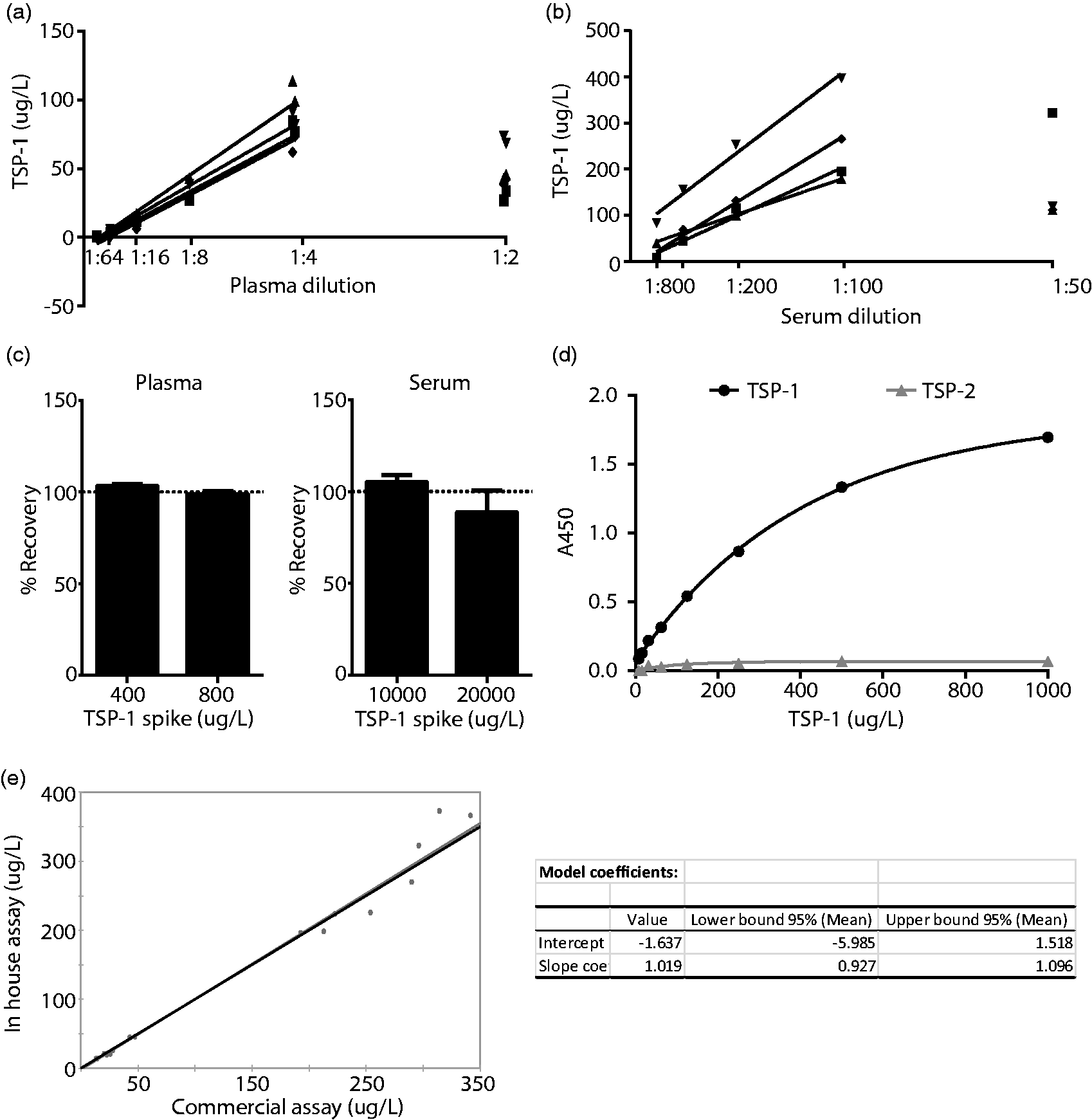
In order to compare our assay with a commercially available kit, we tested eight serum samples and eight plasma samples in both systems and used this data to perform a Passing–Bablok regression analysis (Figure 1e). The 95% CI for the intercept included 0, and the 95% CI for the slope included 1, allowing us to conclude that there is no proportional difference between the two methods.
The cost effectiveness of our assay was determined by calculating the cost per 96-well plate taking into account the plate, capture antibody, detection antibody, recombinant TSP-1 for standard curve, HRP, blocking buffer, TMB, stop solution and wash buffer. This was estimated to be AUD92 (USD67), compared to the commercial kit which cost AUD753 (USD548).
Based on this validation, plasma and serum TSP-1 were measured from eight males and eight females. Plasma TSP-1 ranged from 133 μg/L to 478 μg/L (mean concentration 290 μg/L) (Figure 2a). Serum TSP-1 was approximately 100-fold higher, ranging from 13,700 to 44,400 μg/L (mean concentration 25,700 µg/L) (Figure 2b). There was no correlation between plasma and serum TSP-1 concentrations (Figure 2c).
Discussion
TSP-1 is a multi-functional glycoprotein which is of considerable interest clinically across numerous specialities. It regulates multiple cellular events involved in tissue repair and is involved in the activation of TGF-β1. It results in attenuation of vasodilatation and maintenance of blood pressure, stimulates neuronal synaptogenesis, inhibits angiogenesis through its interaction with CD36 and is positively associated with invasive carcinomas.2,8
The quantification of TSP-1 from plasma or serum is routinely performed using commercially available kits, which are costly. Further, there is limited appreciation for the vast differences in TSP-1 concentrations measured from plasma and serum, which stems from platelet activation in serum. 1 To date, TSP-1 has been measured from either plasma or serum, or in some cases both, with minimal comment regarding the variation between these values. 5 In contrast, others have used the difference between plasma and serum TSP-1 concentration to make specific estimations of platelet TSP-1 stores. 6
This study details and evaluates an in house assay which is approximately eight-fold cheaper than a commercial kit. Spike experiments suggest that high concentrations of TSP-1 in serum can negatively impact on recovery, and assessment of linearity indicates that serum should be diluted to 1:100 or greater, and plasma diluted to 1:4 or greater. This appears to be the result of an apparent high dose hook effect, where high concentrations of analyte give rise to an underestimation of concentration. Thus samples should be tested at multiple dilutions, and when two separate dilutions give the same result it can be concluded that the sample has been diluted appropriately. This is an obvious limitation of the assay, however we feel that it is not prohibitive, given the cost of this system relative to commercial alternatives. The in-house assay does not cross react with TSP-2, and was comparable to a commercially available ELISA kit according to Passing–Bablok regression analysis. 7 Despite the preliminary nature of the presented precision data, we conclude that this in-house assay for human TSP-1 represents a cost effective method to quantitate circulating levels in both serum and plasma when appropriately diluted and compared against a simultaneously run standard curve.
Using this assay, we compared the circulating concentration of TSP-1 in both plasma and serum from healthy volunteers. We saw no time of day effects and no effect of gender. We have confirmed that TSP-1 is approximately 100-fold higher in serum compared to plasma. Mean plasma concentrations were 290 μg/L (95% CI 262–319 μg/L) whilst mean serum concentrations were 25,700 μg/L (95% CI 23,300–28,100 µg/L). Importantly, serum TSP-1 concentrations do not correlate with TSP-1 concentrations measured in plasma. This suggests that TSP-1 levels measured from serum largely reflect the reserve stored in platelets for injury response, whereas plasma TSP-1 reflects the secreted biologically active pool.1,6 Importantly, plasma TSP-1 concentrations detected using the in-house assay were comparable to published levels.4,5 It is now planned to collect plasma on 100 healthy community dwelling volunteers between the ages of 18 and 75 years to define a reference interval for plasma TSP-1 concentrations in adults.
In conclusion, we report a cost effective ELISA to quantify circulating TSP-1, which should be measured in plasma. Serum concentrations are 100-fold higher and mainly reflect TSP-1 stored and released during platelet activation.
Serum TSP-1 is elevated and does not correlate with plasma TSP-1. TSP-1 concentrations from 16 healthy individuals at 0800 h and 1600 h in plasma (a), serum (b), and the correlation between plasma and serum concentrations (c). Males are represented by closed circles; females are represented by open circles.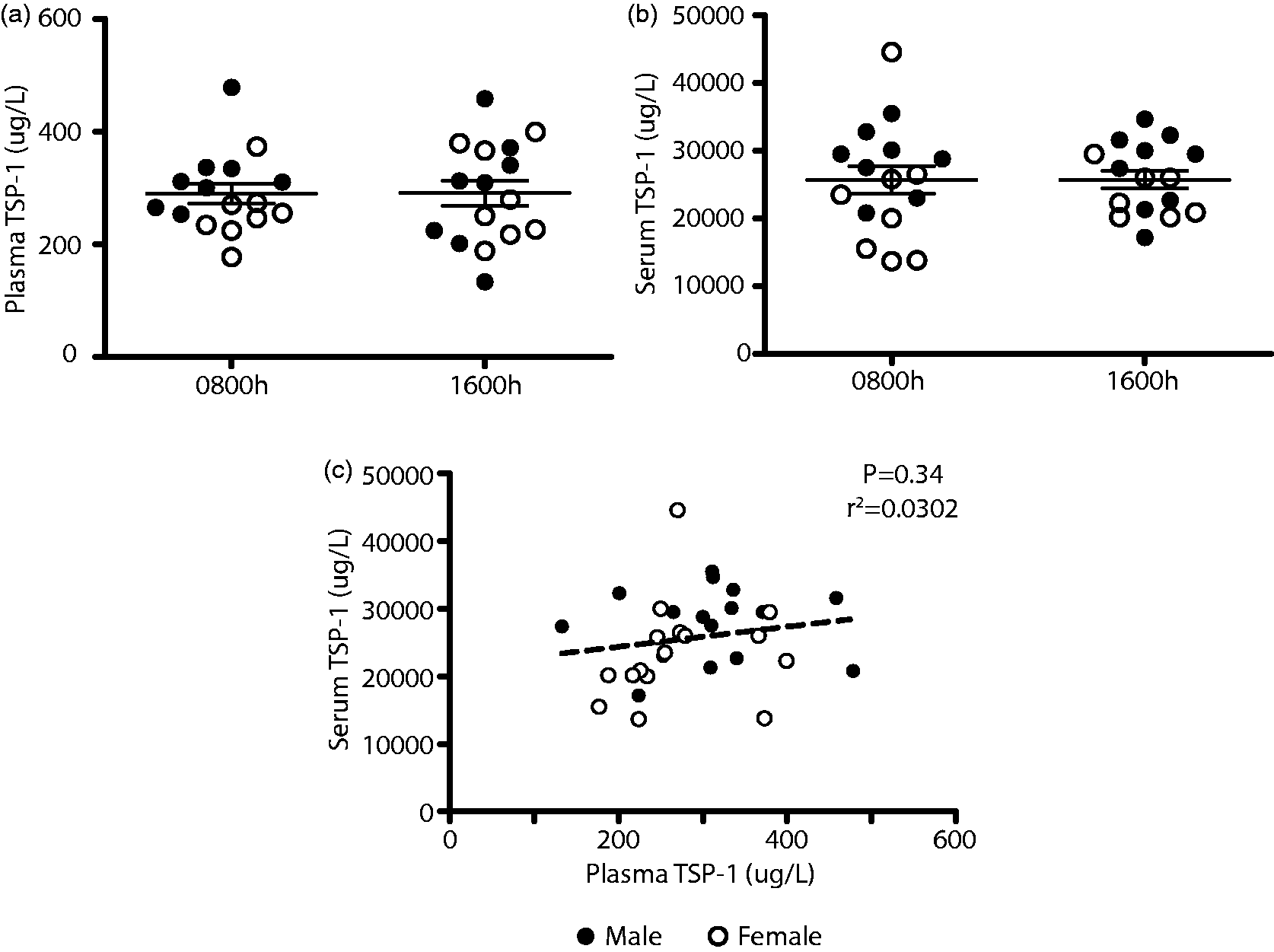
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: a new appointment grant to WJI from the Princess Alexandra Research Support Scheme. SK was supported by a University of Queensland Research Scholarship (UQRS) and a Mater Research Frank Clair Scholarship.
Ethical approval
The study was approved by the local Human Research Ethics Committee (Reference number HREC/14/QPAH/24) and written informed consent was obtained from each participant.
Guarantor
JLB.
Contributorship
WJI, JLB and JPW designed the study, WJI, JLB and SK performed the experiments, WJI, JLB and SK prepared the manuscript. WJI, JLB, JPW and SK edited the manuscript.
