Abstract
Background
We previously reported peptide candidates of disease biomarkers for pregnancy-induced hypertension syndrome using a novel peptidomic analytical method, BLOTCHIP®-MS. The aim of this study was to establish a sandwich enzyme-linked immunosorbent assay system for quantitation of such peptides and to validate their usefulness as disease biomarkers of pregnancy-induced hypertension syndrome including gestational hypertension/pre-eclampsia.
Methods
We focused on three peptide fragments, kininogen-1439–456 (PDA039), kininogen-1438–456 (PDA044) and cysteinyl α2-HS-glycoprotein341–367 (PDA071). Using polyclonal antibodies specific for each peptide, suitable conditions for the sandwich enzyme-linked immunosorbent assay system were investigated. The quantitative enzyme-linked immunosorbent assay values were confirmed by quantitative matrix assisted laser desorption/ionization time-of-flight MS analyses. Using the established enzyme-linked immunosorbent assay systems, serum samples from gestational hypertension/pre-eclampsia patients and paired serum samples from healthy pregnant females were analysed.
Results
The optimum sandwich enzyme-linked immunosorbent assay conditions for PDA039/044 quantitation were developed. Quantitation of PDA071 by enzyme-linked immunosorbent assay failed, presumably due to issues with polyclonal antibody specificity for the native peptide. Bland–Altman plots showed a satisfactory correlation between the serum PDA039/044 concentration by enzyme-linked immunosorbent assay and that by quantitative MS analysis. Although the PDA044 concentration showed no significant change during pregnancy, including gestational hypertension/pre-eclampsia patients, the serum PDA039 concentration was significantly increased (P < 0.0001) in the patients.
Conclusions
The simple quantitation technology for PDA039 by enzyme-linked immunosorbent assay was established for the first time. PDA039 confirmed its clinical utility as a disease biomarker for gestational hypertension/pre-eclampsia by the enzyme-linked immunosorbent assay system using clinical samples. The information provided from the present study would be a new valuable addition in the field of gestational hypertension/pre-eclampsia research.
Keywords
Introduction
Hypertensive disorders of pregnancy such as pregnancy-induced hypertension (PIH) syndrome is one of the most serious complications of pregnancy. 1 The continuous high blood pressure of these patients sometimes causes a convulsive state known as eclampsia, 2 a leading cause of substantial maternal/fetal morbidity and mortality. 3 Although the pathological relationship between gestational hypertension (GH) and pre-eclampsia (PE) is still controversial among the hypertensive disorders of pregnancy,1,4 the Japan Society for the Study of Hypertension in Pregnancy provides the clinical classification of PIH syndrome that includes both GH and PE. 5 At present, the most effective method of predicting the syndrome is monitoring of blood pressure despite the performance of numerous basic and clinical studies. However, no essential treatment method for GH/PE has been developed. Instead, patients must hope for spontaneous recovery after delivery. Thus, new disease biomarkers (DBMs) for GH/PE should be developed to improve clinical management and diagnostic prediction. Accurate monitoring using such DBMs may reduce fetal and maternal mortality due to GH/PE.
To identify novel DBMs for intractable ‘poor-prognosis’ diseases such as GH/PE, comprehensive proteomic analysis of humoral fluids is one of the most promising experimental approaches. 6 Because more than 20,000 peptide fragments derived from various precursor proteins circulate in peripheral blood, 7 we are currently focused on proteomic (peptidomic) analyses that have potential to facilitate the identification of novel DBM(s) in blood samples.8–10 However, DBM discovery using a proteomic approach has several drawbacks. The techniques generally used for blood proteomics (peptidomics) have technical limitation in terms of the analytical process, i.e. the protocol requires removal of high-abundance plasma/serum proteins prior to analysis for omitting undesirable disturbance in the detection of small bio-products such as peptide fragments. This process likely resulted in some important DBMs being overlooked in some cases.11,12 However, peptidomics analysis may be useful for DBM discovery, because peptide fragments in blood show greater variation in total number and structure compared with a genome/proteome. 13 Therefore, sequestered peptides not detected (or overlooked) to date by conventional peptidomic approaches may represent an important source of candidate DBMs for subsequent clinical validation. Additionally, these peptides will expect to provide novel pathophysiological information in terms of the molecular composition of the circulation.11,14
We previously developed a one-step direct transfer technology for matrix-assisted laser desorption/ionization (MALDI) MS (BLOTCHIP®-MS) 15 that does not require reduction of protein concentrations in test samples prior to analysis. This technique enabled detection of peptides in blood samples, including those that would otherwise be adsorbed to blood proteins and thus escape detection. Using this technology, we found 23 characteristic peptides as potential DBMs for PIH syndrome (including GH/PE as stated above) in the serum of pregnant females; 7 of the 23 peptides were identified as fragments of kininogen-1 (three peptides), α-2-HS-glycoprotein, fibrinogen-α, complement component C4-A/B and inter-α-trypsin inhibitor heavy chain H4 and thus to be candidate DBMs for PIH syndrome. 16
Because direct application of a method like peptidomic analysis is not suitable for clinical screening, simple, reliable and low-cost peptide quantitation in blood samples is necessary for the evaluation of the usefulness of peptides as DBMs. The aim of the present study was to establish a simple and reliable quantitation system, sandwich enzyme-linked immunosorbent assay (ELISA), for potential peptide DBMs for GH/PE and to validate its clinical utility. Among the candidate peptides, three (two peptides derived from kininogen-1 and one from α-2-HS-glycoprotein) were initially used as target molecules based on their relatively high performance for PIH syndrome diagnosis, as demonstrated in our preliminary study. 16
Patients and methods
Blood sample collection
The Ethic Committee board members of Juntendo University and Yamagata Saisei Hospital approved the study protocol prior to the present study (registration #19-017 and #144, respectively).
Clinical profiles of serum samples isolated from pregnant patients with hypertensive disorders.
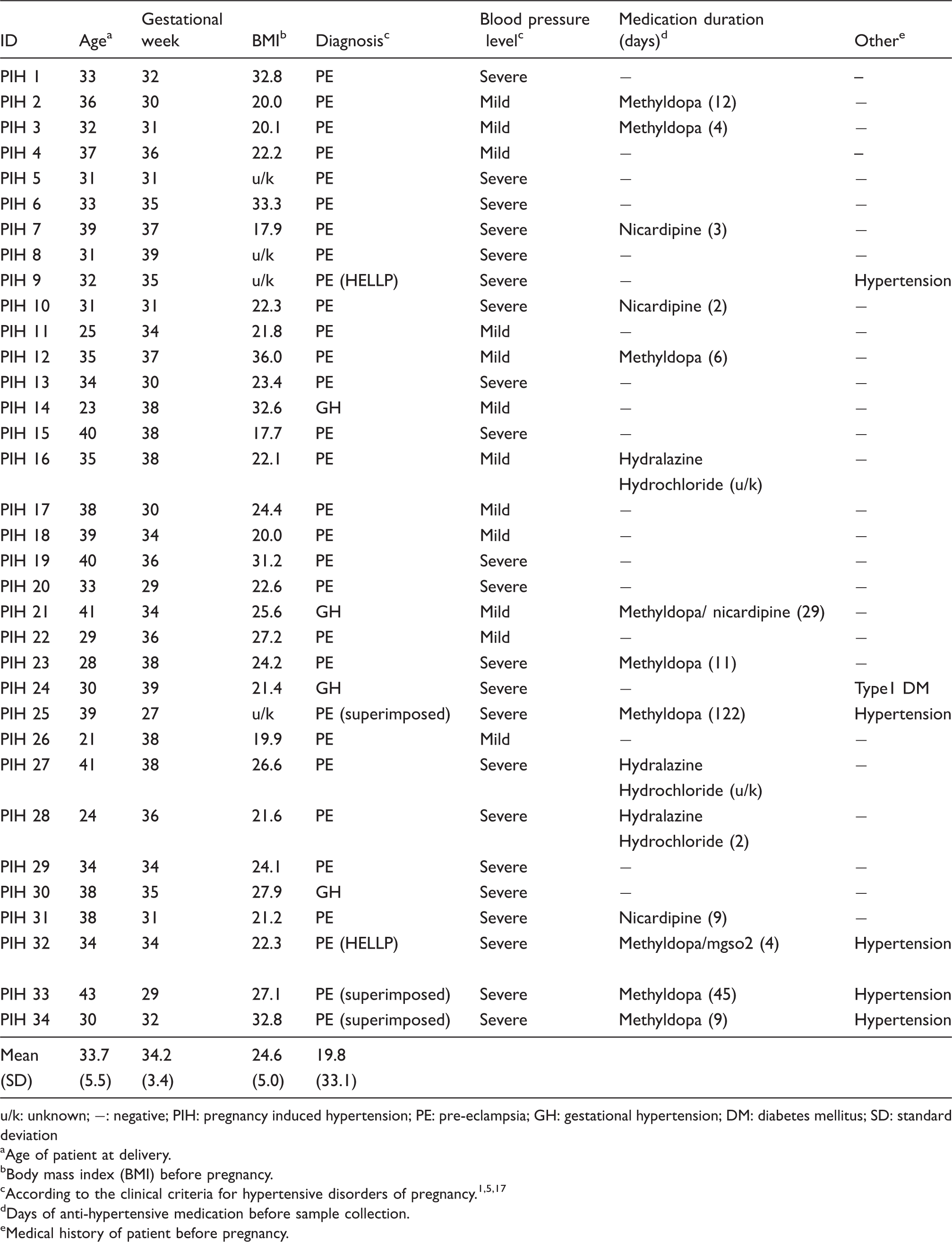
u/k: unknown; −: negative; PIH: pregnancy induced hypertension; PE: pre-eclampsia; GH: gestational hypertension; DM: diabetes mellitus; SD: standard deviation
Age of patient at delivery.
Body mass index (BMI) before pregnancy.
Days of anti-hypertensive medication before sample collection.
Medical history of patient before pregnancy.
Chemicals
BLOCK ACE® powder was purchased from DS Pharma Biomedical Co., Ltd (Osaka, Japan). High-sensitivity streptoavidine-horseradish peroxidase (HRP) was from Wako Pure Chemical Industries, Ltd, Osaka, Japan. Bovine serum albumin (BSA) (fraction V) and SIGMAFAST™ o-phenylenediamine dihydrochloride (OPD) tablets were purchased from Sigma-Aldrich Co. LLC., St. Louis, MO, USA. All other chemicals were obtained commercially and were of the highest purity available.
Polyclonal antibody production for peptides
Potential biomarker candidate peptides for GH/PE (kininogen-1439–456, m/z 2081.00 termed as PDA039; kininogen-1438–456, m/z 2209.12 [PDA044], and cysteinyl α2-HS-glycoprotein341–367, m/z 2858.61 [PDA071]) 16 were chemically synthesized. As immunogens, the amino acid residues of each N-/C-terminal peptide (Supplementary Table S1) conjugated with keyhole limpet haemocyanin (KLH) at their non-terminus sites were used.
For polyclonal antibodies (pAbs) production, rabbits were immunized subcutaneously with 0.15 mg of KLH-conjugated peptides emulsified in complete Freund's adjuvant. Two weeks after the first injection, three subcutaneous booster injections of 0.3 mg antigen emulsified in incomplete Freund's adjuvant were performed every two weeks. The rabbits were sacrificed 20 days after the last immunization and the serum samples were isolated from total bloods.
Anti-N-/C-terminal peptides pAbs were affinity purified from the isolated serum by each immunogen-peptide coupled with cyanogen bromide-activated Sepharose 4B (GE Healthcare Life Sciences, Uppsara, Sweden). A part of each pAb was biotinylated for use as a secondary antibody (Ab) in sandwich ELISA system for peptide quantitation (see below).
The production of pAbs for N-/C-terminal peptides described above was performed commercially by Ab production custom services in Scrum Inc., Tokyo, Japan.
Sandwich ELISA for quantitation of the peptides
Each of the purified pAbs was coated on a 96-well flat-bottom 2HB plate (cat# 3455, Thermo Fisher Scientific Inc., Waltham, MA, USA) in a volume of 100 µL of coating buffer (pH 8.5) containing 0.017 M Na2B4O710H2O, 0.12 M NaCl for 2 h at 37 ℃. After blocking with 1% (w/v) BLOCK ACE® in ultra pure water for 1 h at 37 ℃, 100 µL of sample solutions were added to each well and reacted for over 20 h at 4 ℃. For dilution of samples, 0.1% BSA in phosphate buffer saline (PBS) (pH 7.4) was used. After washing with 0.4% (w/v) BLOCK ACE® solution containing 0.05% (v/v) Tween-20 (washing solution) three times, the plate was treated with various concentrations of biotin-labelled secondary pAb (volume of each was 100 µL) in 0.4% (w/v) BLOCK ACE® solution for over 20 h at 4 ℃. At the end of reaction, each plate was washed at least three times with washing solution, and then each well was treated with 100 µL of 0.01% (v/v) high-sensitivity streptoavidine-HRP in PBS (pH 7.4) containing 0.25% (w/v) BSA and 0.05% (v/v) Tween-20 for 1 h at ambient temperature. The bound antibody was determined by reacting with OPD for 45 min at 37 ℃, and then stopped by the addition of 100 μL 1 M H2SO4. The amount of reaction product was read by absorbance at 492 nm using ELISA plate reader (Wallac 1420 ARVO MX, PerkinElmer Inc., Waltham, MA, USA).
Immunoaffinity-supported stable-isotope dilution MS quantitation
Preparation of anti-peptide pAb-conjugated beads
Each pAb against the N-terminal of PDA039, PDA044, and the C-terminal of the both peptides that shared the same C-terminal sequence (Supplementary Table S1) was covalently conjugated to N-hydroxysuccinimide-activated Sepharose 4B Fast Flow (GE Healthcare) according to the manufacturer's instruction. Each pAb (0.75 mg) was mixed in 2.0 mL solution of 0.2 M NaHCO3 (pH 8.3), 0.5 M NaCl containing 1.0 mL of the activated beads. The mixture was reacted for 2 h at ambient temperature with gentle swirling. After the incubation, complete immobilization of the pAbs was confirmed by measuring protein concentration of the reaction mixture with Coomassie Protein Assay reagent (Thermo Fisher). Three millilitres of 0.1 M tris(hydroxymethyl)aminomethane-HCl (Tris-HCl) buffer (pH 8.5) were added to block the residual active functional groups. The mixture was reacted for 30 min at ambient temperature, and then continuously incubated overnight at 4 ℃. The pAb-immobilized beads were washed for five times each with 0.1 M Tris-HCl (pH 8.5) and 0.1 M acetate (pH 4.5) containing 0.5 M NaCl. Finally, the beads were equilibrated with 5 mL PBS (pH 7.4) containing 0.1% (w/v) NaN3 and stored at 4 ℃ until use.
Quantitation of potential DBM peptides (PDA039/044) in serum samples with MS
Stable isotope-labelled PDA039 (PDA039H21: HNL(13C6,15N)GHG(13C2,15N)HK(13C6,15N2)HERDQG(13C2,15N)HGHQ) and PDA044 (PDA044H16: K(13C6,15N2)HNLGHGHK(13C6,15N2)HERDQGHGHQ) were chemically synthesized (Scrum). Absolute quantitation of PDA039/044 was conducted by MALDI-TOF-MS in combination with stable-isotope dilution method using PDA039H21/PDA044H16 and specific binding and elution of peptides with the Ab-immobilized gel. Since the peptide isolation step was essential for precise quantitation in this procedure, the gel immobilized with Ab against the C-terminal sequence of PDA039/044 was utilized to capture non-labelled and stable-isotope labelled peptides. Blood samples (totally 45 serum samples randomly selected from each group; 23/33-week healthy pregnant women and GH/PE patients; 15 samples each) were used for the measurement. A serum sample (20 μL; n = 2) was diluted with 960 μL PBS (pH 7.4) in a new 1.5 mL low-binding tube (Proteosave SS; Sumitomo Bakelite Co. Ltd, Tokyo, Japan). Stable-isotope labelled peptide (10 μL) (PDA039H21 or PDA044H16; 50 or 100 fmol/μL, respectively) was added as an internal standard. After mixing with inversion, the solution was immediately mixed with 5 μL of pAb-conjugated Sepharose 4B Fast Flow gel (50% slurry). The mixtures were gently stirred for 1 h and the gels were spun down with centrifugation for 12,000 g for 20 s. The solution was removed by vacuum aspiration. Subsequently, the gels were washed two times with 20 mM potassium phosphate, pH 7.2 containing 0.5 M KCl (for the purpose of quantitation) or with PBS (pH 7.4) containing 0.5 M NaCl (for the others). Washing the gels with potassium-containing buffer converted part of the peptide to its potassium-adduct (+43.964 u), which did not interfere with MS measurement of the stable-isotope labelled peptide (PDA039H21/PDA044H16; +21.000/+16.000 u from the unlabelled peptide, respectively) (Supplementary Figure S1). Finally, the gels were washed with ultra pure water. The antibody-bound peptides were eluted with 25 μL of 0.1% trifluoroacetic acid (TFA) containing 10% acetonitrile for 5 min at ambient temperature and then centrifuged. The supernatant was collected as the eluate.
All mass spectra were obtained on an UltraFlex II TOF/TOF-MS (Bruker Daltonics, Bremen, Germany) in the reflector mode (for quantitation) or in the linear mode (for the others). Each analyte solution was mixed with saturated α-cyano-4-hydroxycinnamic acid in 0.1% TFA/acetonitrile (50:50) in the ratio of 1:2 and spotted (2 μL per spot) in duplicate onto MTP 384 Ground Steel TF target plate (Bruker Daltonics).
Standard samples were prepared for optimization of measurement conditions with MS. According to the optimized conditions, the PDA039/044 concentration in the subject serum samples was calculated by considering the isotope purity. For more detailed descriptions of these matters, refer to Supplementary Methods, Figure S1, S2 and Table S2.
Statistical analysis
Statistical analyses were performed with R statistical environment software (R Core Team, http://www.R-project.org/). Receiver operation characteristic (ROC) analysis was conducted to determine the diagnostic performance of DBM candidate peptide concentration in distinguishing GH/PE patients and healthy pregnant women with package Epi (http://CRAN.R-project.org/package=Epi) within R software. Area under the curve (AUC), sensitivity (SN), specificity (SP) and threshold values (positive/negative predictive value; PPV/NPV) were calculated from the ROC curve as an indicator of the diagnostic value. The optimal cut-off thresholds for diagnosis were determined according to Youden's index. 18 Confidence interval (CI) of diagnostic values for AUC was calculated with the non-parametric bootstrapping method of sample data. Mann-Whitney U-test for non-parametric data was used for analysis between two groups of differences. A probability of P < 0.05 was considered statistically significant.
Results
Conditions for peptide quantitation by sandwich ELISA
As an initial step, we identified the optimum sandwich ELISA conditions for quantitation of the PDA039/044/071 synthetic peptides. Affinity-purified pAbs against N-/C-terminal peptides at several concentrations were assessed as the captured (primary) Ab. A biotinylated pAb against the opposite terminus of the peptide was used as the detection (secondary) Ab. Two primary Ab concentrations, 5 and 10 µg/mL diluted in PBS (pH 7.4) containing 0.1% (w/v) BSA, were used followed by 0.03 to 1 µg of each biotinylated secondary pAb.
Data obtained in the above preliminary experiments suggested that the optimum conditions for PDA039/044 quantitation were as follows: primary Ab, anti-C-terminal pAb for PDA039/044 at the concentration of 5 µg/mL; detection Ab, anti-N-terminal biotinylated pAb for each peptide at 0.25 µg/mL. Using these conditions, typical reaction curves for synthetic PDA039/PDA044 were obtained (Figure 1). The standard curve was established within the range from 0.1 to 10 ng/mL for both PDA039 (Figure 1(a)) and PDA044 (Figure 1(b)) quantitation. Under these conditions, the linear dynamic range was 0.5–5 ng/mL. Although similar standard curves were obtained when synthetic peptides were spiked into standard serum diluted with PBS (pH 7.4) containing 0.01% BSA (Figure 1(c) and (d)), the optical densities at 492 nm were somewhat shifted in the PDA039 quantitation (Figure 1(c)).
Peptide quantitation of PDA039/044 peptides by sandwich ELISA system. Typical metage lines for standard synthetic peptides. Anti-C-terminal affinity purified PDA039/044 pAbs (5 µg/mL) were coated as primary Abs, respectively. Each anti-N-terminal peptide biotinyl pAb (PDA039 (a); PDA044 (b)) was used as a detection Ab. Chemically synthetic peptides were diluted in PBS (pH 7.4) containing 0.1% BSA and their absorbance values were monitored. Open circles, PDA039; Closed circle, PDA044. Identical sandwich ELISA system for the detection of peptides in 20-fold (c) or 200-fold (d) diluted serum with PBS (pH 7.4) containing 0.01% BSA.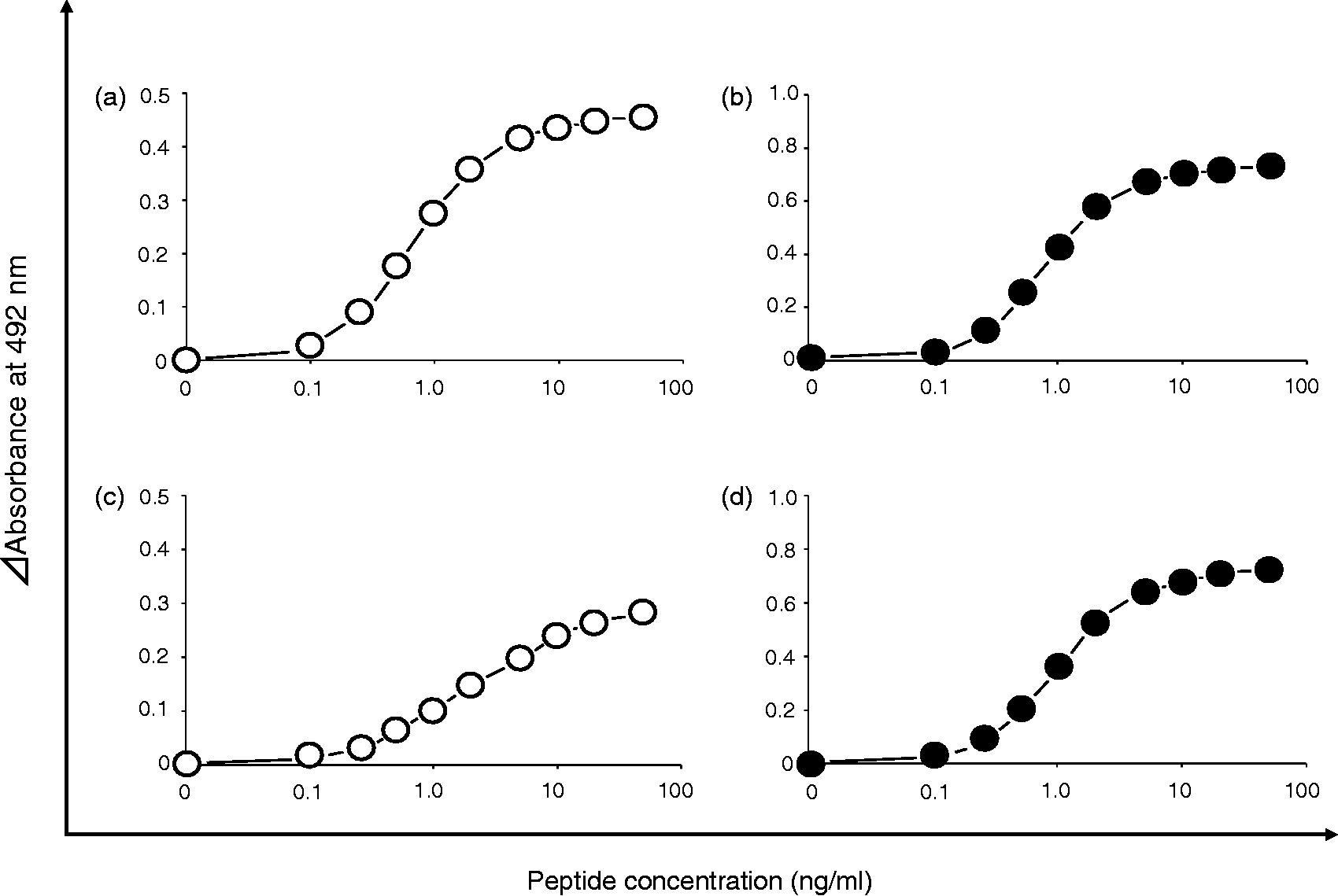
For PDA071 quantitation, however, the conditions above yielded no meaningful analytical data. Because a reasonable quantity of each pAb was obtained by affinity purification using synthesized N/C-terminal immunogen-peptide fragments (Supplementary Table S1) immobilized on Sepharose 4B beads, the pAbs to PDA071 terminal peptides were likely unsuitable for the sandwich ELISA system. Using the sandwich ELISA system with the conditions above, we examined the immunoreactivity of the PDA071 pAbs using various detergents, including deoxycholic acid or low concentration of sodium dodecyl sulphate; 19 however, no significant pAb reactivity with PDA071 was observed. Instead, the PDA071 anti-N-terminal pAb, but not the anti-C-terminal, showed immnoreactivity only when the peptide was directly coated on the ELISA plate (data not shown).
These data suggest that the steric structure of the C-terminus of PDA071 in nature is somewhat different compared to that of the synthesized C-terminal peptide oligomer (Supplementary Table S1) that was used for the affinity purification of the pAb. Because the anti-N-terminal pAb of PDA071 enabled quantitation of the peptide, we are currently producing Abs against a more characteristic site of the candidate DBM, i.e. in the vicinity of the cysteinyl site at α2-HS-glycoprotein358 (Supplementary Table S1). We also aim to confirm the serum peptide concentration in females with PIH syndrome and healthy pregnant females using stable isotope-labelled PDA071 by quantitative MS analyses. These multidirectional approaches will enable clarification of the usefulness of PDA071 as a DBM for GH/PE.
Specificity of pAbs against N-terminal PDA039/044 peptides
Among the biomarker candidate peptides for GH/PE that we identified previously, both the PDA039 and 044 peptides originated from kininogen-1.
16
In addition, PDA044 possesses one additional amino acid residue at the N-terminus compared to PDA039 (Supplementary Table S1). Therefore, we assessed the antigenic specificity of both anti-N-terminal pAbs. Using conditions identical to those for quantitation of the PDA039/044 sandwich ELISA systems (Figure 1), we evaluated the specificity of each peptide detection system. As expected, the PDA039 detection system did not cross-react with that of PDA044 and vice versa (Supplementary Figure S3). The MS spectra of the eluates from anti-kininogen peptide antibody-immobilized beads indicate that each anti-N-terminal Ab recognizes its appropriate antigen peptide, and the anti-C-terminal Ab reacts specifically with the common terminal of PDA39/044 (Figure 2).
MS analysis of eluates from anti-kininogen peptide antibody immobilized beads. The beads were mixed with serum samples from GH/PE patients, washed and bound peptides were eluted. Two pairs of anti N-terminal and C-terminal kininogen peptide antibodies were found to bind respective antigen peptides. Bound kininogen peptides were eluted from immobilized anti-PDA044 N-terminal pAb (a), anti-PDA039 N-terminal pAb (b) and anti-PDA039/044 C-terminal Ab (c) on Sepharose 4B Fast Flow beads. Closed circles indicate peaks that also found when control beads (no-Ab immobilized beads) were used instead. Open circles indicate mono-(+22u), di-(+44u) or tri-(+66u) sodiated peptides.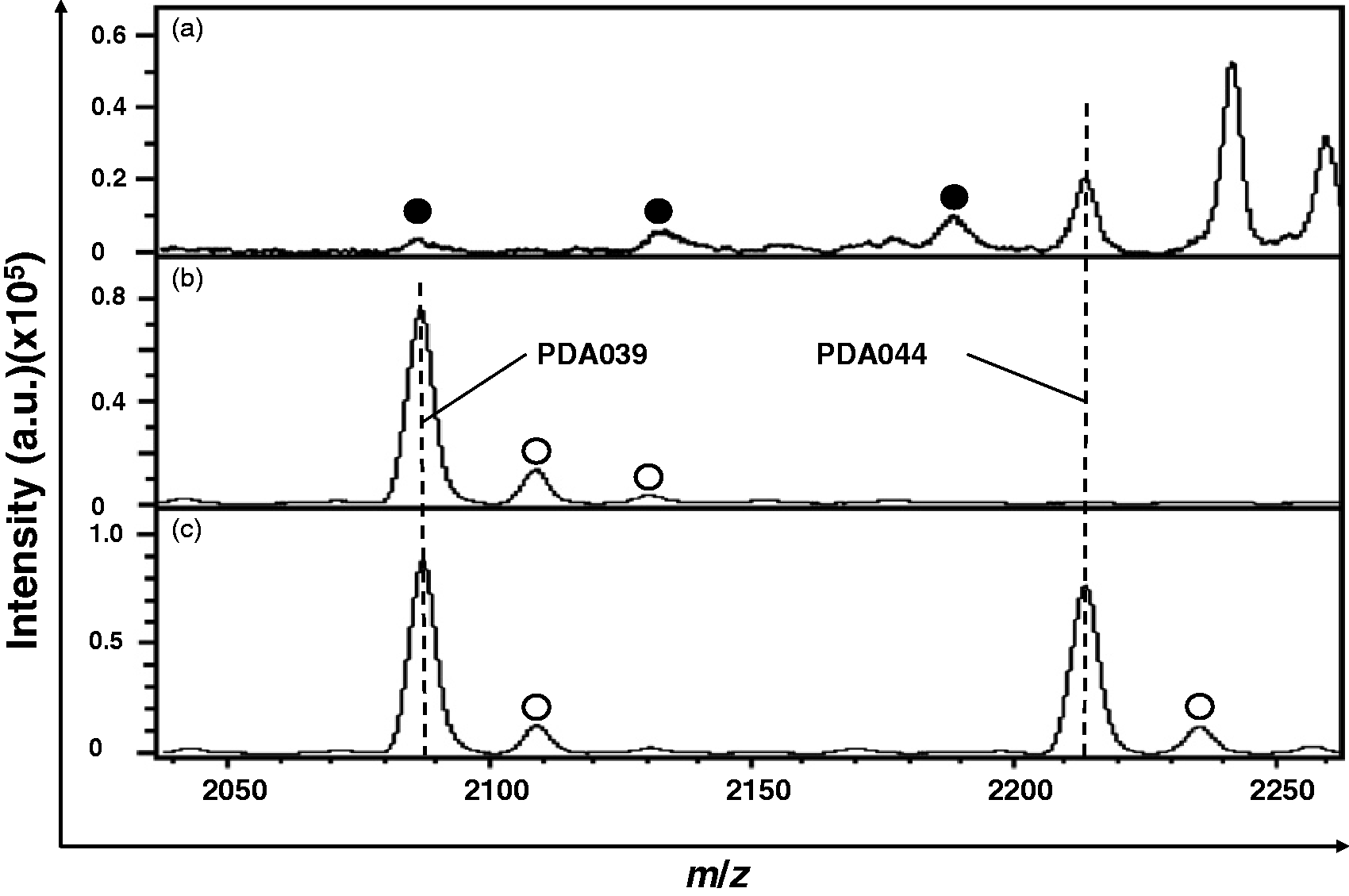
Taken together, these results indicate that the sandwich ELISA system is suitable for the detection of the PDA039/044 in serum.
Comparison between sandwich ELISA and quantitative MALDI-TOF-MS analyses by Bland–Altman difference plots
The peptide (PDA039/044) concentrations in serum (randomly selected [n = 45] from our serum stock) were assayed using the sandwich ELISA and MALDI-TOF-MS systems. Each serum peptide concentration was plotted two-dimensionally; the correlation assay values were as follows: PDA039; slope = 0.765, intercept = 1.665, and r2 = 0.898; PDA044; slope = 0.570, intercept = 10.977, and r2 = 0.656 (Figure 3(a)). Although PDA044 showed a relatively low correlation among these two assay systems, peptide assay values converted by Bland–Altman difference plot
20
with a correction method reported by Dewitte et al.
21
demonstrated that the most peptide concentrations provided by ELISA and MS analysis were distributed within limits of agreement (LOA) (Figure 3(b)).
Comparison of serum peptide concentrations assayed by the sandwich ELISA and MALDI-TOF-MS systems. Two-dimensional correlation of assay values between the peptide concentration by ELISA and that by MALDI-TOF-MS (a) and Bland–Altman plots comparing data obtained from both assay systems (b). PDA039: mean difference (solid line) = 0.583, + LOA/−LOA (dotted lines) = 2.214/−1.047, and PDA044: mean difference = 0.065, + LOA/−LOA = 1.711/−1.581, respectively.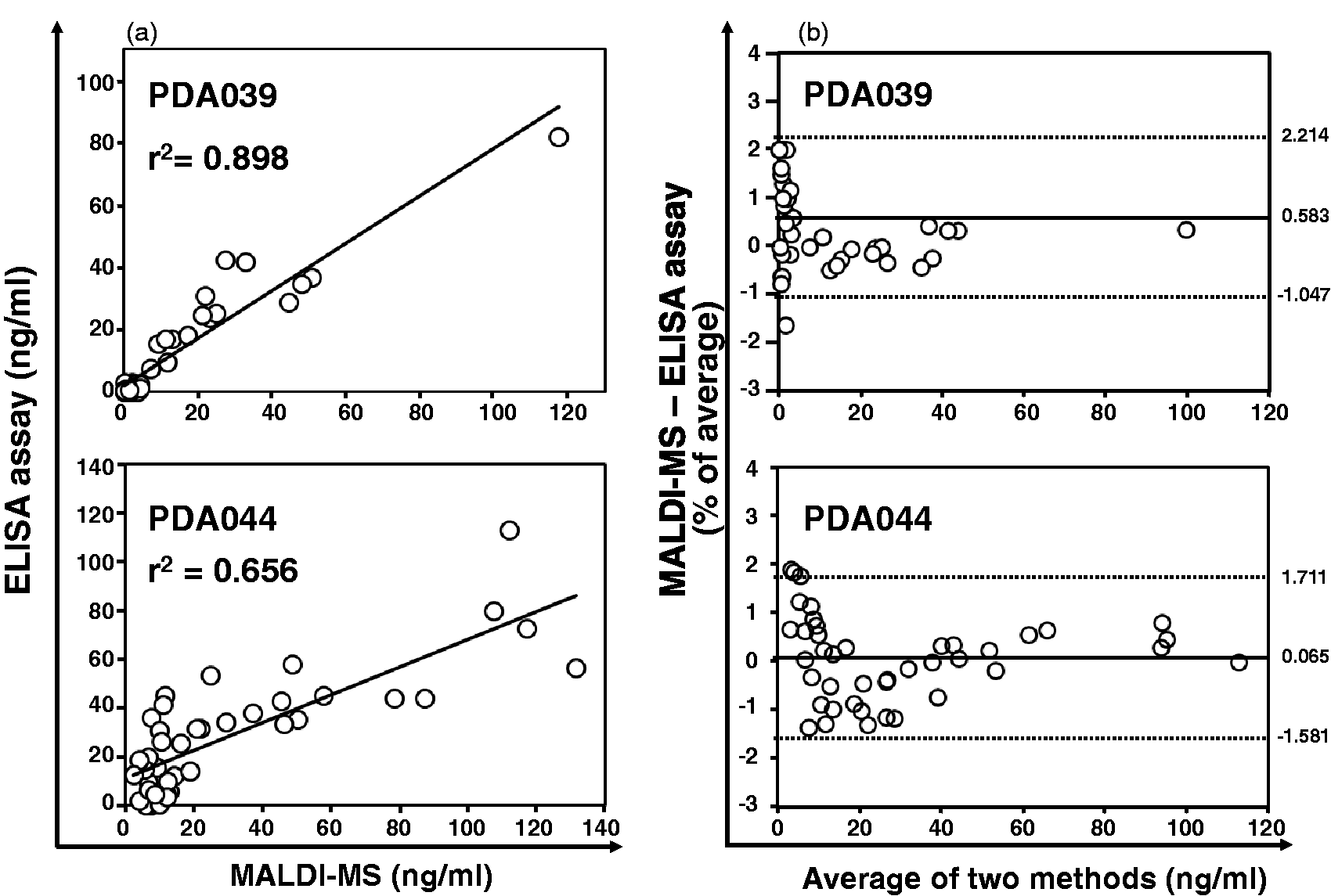
Measurement of serum PDA039/044 peptide concentrations by sandwich ELISA in patients with GH/PE and normal pregnant females at 23/33 weeks of gestation
Concentrations of the PDA039/044 peptides in serum from GH/PE patients and normal pregnant females were examined using sandwich ELISA. The PDA039 concentration (median, 6.42; interquartile range [IQR], 3.35–16.62; n = 34) was significantly higher in the serum of GH/PE patients compared to that of normal pregnant females at 23 (median, 2.18; IQR, 0.63–3.40; n = 50, P < 0.0001) and 33 (median, 1.08; IQR, 0.11–2.46; n = 50, P < 0.0001) weeks of gestation (Figure 4(a)). It should be noted that a case of quite high concentration of serum PDA39 was identified in the GH/PE patient group (Figure 4(a)). This value was from PIH syndrome 16 patient in Table 1. We confirmed the serum concentrations of PDA039 peptides with GH/PE patients without the PIH syndrome 16 case, but statistical significance of PDA concentration was observed between GH/PE patients without PIH syndrome 16 and other groups (P < 0.0001; data not shown), like the data shown in Figure 4. Although the median PDA044 concentration in the serum from GH/PE patients (26.18 ng/mL) was higher than that in the serum of normal pregnant females (medians, 15.25 and 20.64 ng/mL at 23 and 33 weeks of gestation, respectively), the PDA044 concentration in serum samples of all subjects (GH/PE patients and normal pregnant females at 23 and 33 gestational weeks) did not differ markedly among the three groups (Figure 4(b)). Seemingly, the serum PDA044 concentration varied randomly during normal pregnancy from 23 to 33 gestational weeks (Figure 4(b)).
Serum concentration of disease biomarker candidate peptides for GH/PE by sandwich ELISA. Concentration of PDA039 (a) and 044 (b) in the pregnant women as shown by box plots. Open/closed circles indicate peptide concentration in the healthy pregnant women and patients with GH/PE, respectively. Change of the peptide concentrations within a pair serum is represented by dotted lines. Asterisks indicate significant differences statistically between two groups.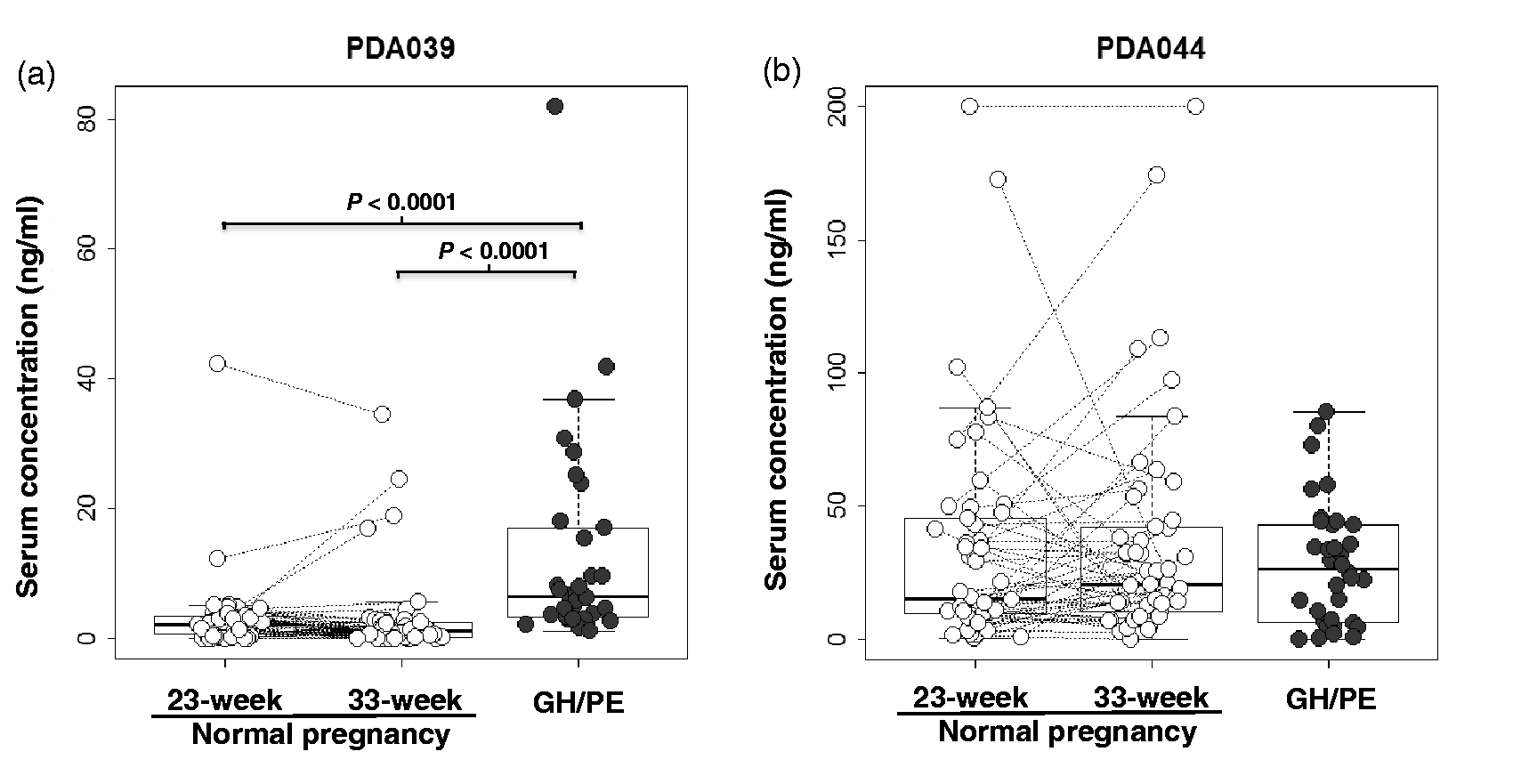
Diagnostic accuracy of PDA039 in patients with GH/PE
Since PDA039 has a potential utility as DBM for GH/PE (Figure 4), we evaluated its diagnostic accuracy in the patients using ROC curve analysis. The ROC values of serum PDA039 concentrations in GH/PE patients vs. those in normal pregnant females were: SN = 58.8%, SP = 96.0%, PPV = 90.9%, NPV = 77.4%, AUC (95% CI) = 0.844 (0.760–0.927), and cut-off value = 5.02 ng/mL (vs. 23 weeks of gestation; Figure 5(a)); SN = 85.3%, SP = 86.0%, PPV = 80.6%, NPV = 89.6%, AUC (95% CI) = 0.890 (0.817–0.961), and cut-off value = 2.81 ng/mL (vs. 33 weeks of gestation; Figure 5(b)).
Diagnostic accuracy of PDA039 for GH/PE. ROC curves of PDA039 in patient with GH/PE versus normal pregnant women at 23- (a) or 33-week of gestation (b). SN, SP, PPV and NPV were calculated as described in the “Materials and Methods”.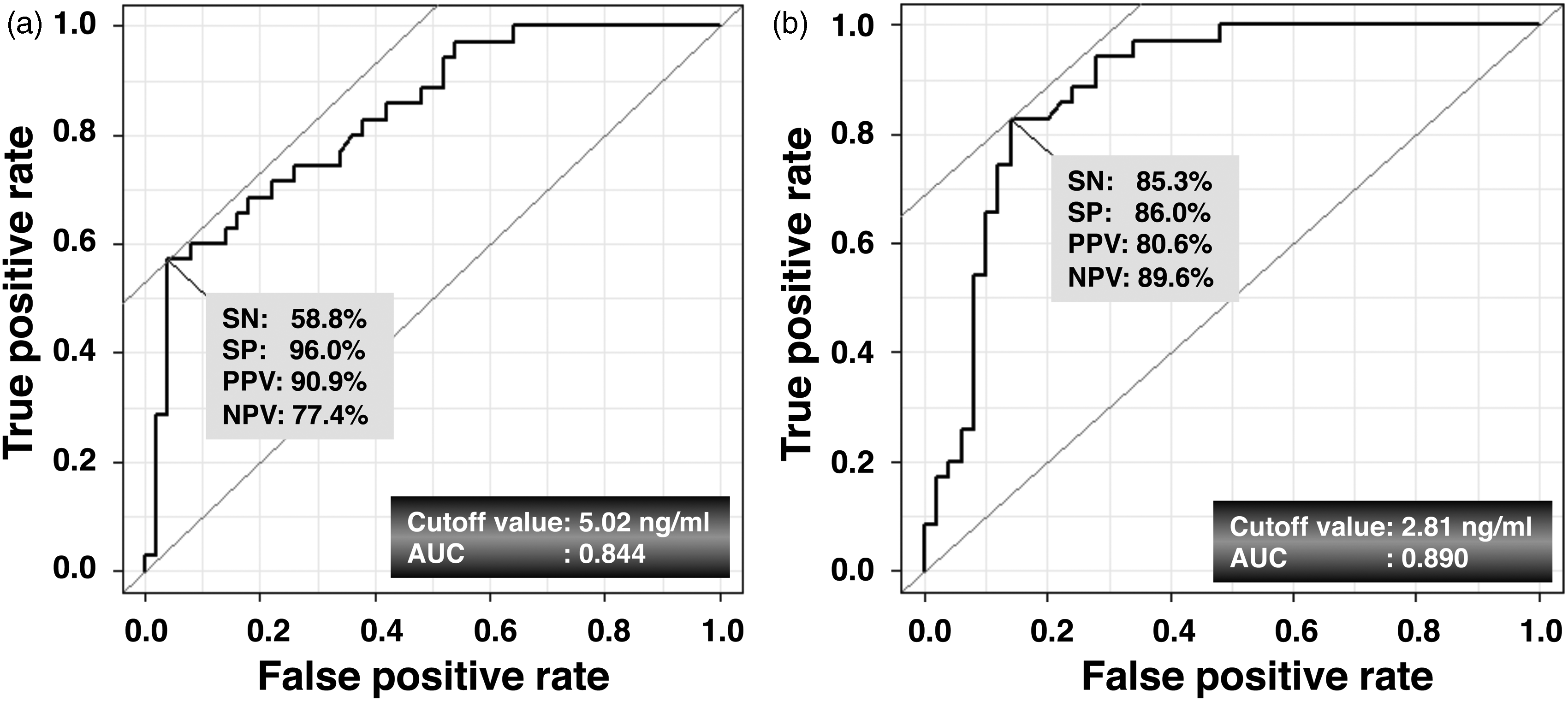
Discussion
Data obtained from the present study suggested that PDA039, a peptide fragment derived from kininogen-1, shows clinical usefulness as a DBM for at least diagnosis of GH/PE (Figures 4(a) and 5), whereas PDA044 (also derived from kininogen-1) did not (Figure 4(b)). In the inspection process of quantitation PDA044, we found that sodium adduct (+22) of the peptide (PDA039/PDA044) was overlapped with the stable-isotope labelled peptide (PDA039H21/PDA044H16) in the linear mode of MALDI-TOF-MS. For the purpose of removing the sodium-adduct interference, we selected potassium-containing buffer as a washing buffer instead of PBS with sodium chloride and measured peptides in the reflector mode for peptide quantitation. In addition, stable-isotope labelling efficiency of the synthesized peptide (PDA039H21/PDA044H16) affected the quantitative value of the peptides. Therefore, we calculated accurate height values resulting in more accurate measurement of the peptides (see Supplementary methods). However, the absolute values of quantitation for PDA044 were somewhat different between ELISA and MS methods. According to our preliminary experimental data, PDA044 as well as PDA039 possess binding affinity to serum major proteins such as albumin and IgG. PDA044 has much high affinity, approximately 1.5-fold affinity to serum protein compared to that of PDA039. 16 Although PDA044 possesses only one additional amino acid residue at the N-terminus compared to PDA039 (Supplementary Table S1), the detection ELISA system for PDA044 did not show cross-reactivity for that of PDA039 (Supplementary Figure S3). This implies that the pAb against N-terminus of PDA044 has quite high specificity to the epitope. Based on the experimental data, we speculate that the reason for the absolute values between quantitative MS analysis and that by ELISA in the case of PDA044 might be due to the effect of partial inhibition by serum unknown factor(s) including abundant proteins on the immunoreaction of the pAb to N-terminus of PDA044 in the ELISA system.
As a functional peptide derived from kininogen-1, bradykinin regulates blood pressure via the nitric oxide signaling pathway in vascular smooth muscle. 22 Conversely, angiotensin-converting enzyme, a key molecule in the pathway of renin-angiotensin-aldosterone pathway, has an alternative activity as kininase II (a peptidase) that inactivates bradykinin. 23 These results imply that hyper- and hypo-tensive mechanisms display both complex and multidirectional molecular interaction. Indeed, an imbalance between the levels of vasoconstrictor and vasodilator substances in the placenta, including kininogen, in patients with PIH syndrome was reported. 24 However, the pathophysiology of patients with hypertensive disorders of pregnancy remains unclear, and the physiological functions of circulating peptides (including fragments derived from kininogen-1), other than bradykinin for blood pressure, are far from being completely understood.
Our previous study revealed that molecular mechanism as to production of PDA039/044 might be different from that of bradykinin from kininogen-1, since no significant difference in serum bradykinin concentration was observed in between the patients and control groups. 16 Therefore, monitoring of these peptide fragments derived from kininogen-1 using the simple quantitation method developed in the present study may provide a new insight into pathophysiology of GH/PE. Recent studies of the serum peptidome have suggested that clinical serum peptidomic analyses reflect various pathological disorders involving systematic changes, including hypertensive disorders of pregnancy.25,26 Numerous peptides in serum are considered to be not simply products of protein degradation. Several peptides have been identified to have biological activity as hormones, growth factors, neurotransmitters and antibiotic agents. 27 As a biological event, it is generally considered that the contentious stressful environment induces variety of stressors, and they sometimes cause pathophysiological changes of organs resulting in various disorders. Because production of peptide fragments in the blood is thought to regulate cellular metabolism, the homeostatic balance in between proteases and their inhibitors may be essential to maintain a healthy conditions; this is also likely to be the case in pregnancy.
Conclusion
The present study clearly shows the clinical usefulness of PDA039 as a DBM for GH/PE using a simple quantitation system. The DBM candidates we identified in the previous study are quite unique molecules by specific plate, BLOTCHIP® for the MALDI-TOF/MS system. 16 Using this novel technology, the DBM candidates identified in our previous study have positive potential as the DBM biomarkers compared to that identified by conventional omics methods. 26 Therefore, the special conclusion of the present study is not only the establishment of the simple quantitation technology by the ELISA, but also clinical utility of PDA039, at least, is validated using clinical samples. Although development of a simple quantitation system for other potential DBM peptides 16 is necessary, monitoring of the serum PDA039 concentration during pregnancy would provide novel information regarding the pathophysiology of GH/PE. Further multidimensional evaluation in combination with PDA039 and other peptides, or other proposed DBMs for PE, such as placental growth factor, soluble fms-like tyrosine kinase 1 receptor and soluble endoglin28–30 would enhance our understanding of the molecular mechanisms underlying, and provide information regarding the clinical manifestations for hypertensive disorders of pregnancy.
Footnotes
Acknowledgements
The authors are indebted to clinical staff of Yamagata Saisei Hospital and Juntendo Urayasu Hospital for their assistance. They gratefully acknowledge Drs. Kyoichi Asada (Protosera Inc.) and Hiroshi Yoshitake (Juntendo University) for their helpful discussion, technical supports and encouragement throughout the course of this study.
Competing interests
None.
Funding
This study was funded in part by Grants-in-Aid for General Scientific Research/‘High-Tech Research Center’ Project for Private Universities: matching fund subsidy from the Ministry of Education, Culture, Sports, Science & Technology, Japan; a grant (A-STEP) from Japan Science and Technology Agency; and Juntendo University Young Investigator Joint Project Award.
Ethical approval
The Ethic Committee board members of Juntendo University and Yamagata Saisei Hospital approved this study (registration #19-017 and #144, respectively).
Guarantor
YA.
Contributorship
KH, DN, MY, KTan, KTak, ST and YA researched the literature, conceived the study, managed grant application and data analysis; KH, DN and LJL collected experimental data; HI, MB, MN and KY collected samples, analysed clinical data and reviewed the statistical protocol; MY and YA contributed to design the study protocol; KH and YA wrote the manuscript. All authors reviewed and edited the manuscript and approved the final version of the manuscript.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
