Abstract
Background
We developed a novel, ultrasensitive enzyme immunoassay (immune complex transfer enzyme immunoassay) for determination of glutamic acid decarboxylase autoantibody concentrations in serum samples from patients with type 2 diabetes.
Methods
We developed an immune complex transfer enzyme immunoassay for glutamic acid decarboxylase autoantibody and measured glutamic acid decarboxylase autoantibody from 22 patients with type 1 diabetes, 29 patients with type 2 diabetes, and 32 healthy controls.
Results
A conventional ELISA kit identified 10 patients with type 1 diabetes and one patient with type 2 diabetes as glutamic acid decarboxylase autoantibody positive, whereas 15 patients with type 1 diabetes and six patients with type 2 diabetes were identified as glutamic acid decarboxylase autoantibody positive using immune complex transfer enzyme immunoassay.
Conclusions
Immune complex transfer enzyme immunoassay is a highly sensitive and specific assay for glutamic acid decarboxylase autoantibody and might be clinically useful for diabetic onset prediction and early diagnosis.
Keywords
Introduction
In some patients with type 2 diabetes, specifically those who are non-obese and older than age 30, the disease can be controlled initially with diet and oral agents, but these cases gradually progress to insulin dependency within an average of three years.1,2 In these patients, glutamic acid decarboxylase autoantibody (GADA), insulin autoantibody (IAA), and tyrosine phosphatase-like insulinoma-associated antigen-2 antibody (IA-2A) are detected. GADA is common in autoimmune diabetes in adults, whereas IA-2A and IAA have been found to be far less prevalent in these patients.3,4 Kobayashi et al. introduced the term ‘slowly progressive insulin-dependent diabetes mellitus’ (SPIDDM) to describe this subgroup of adult phenotypic type 2 diabetes patients who are positive for an autoantibody to GAD. 5 SPIDDM also has been referred to as latent autoimmune diabetes of adults 6 or type 1.5 diabetes. 7 Epidemiological studies suggest that SPIDDM may account for 2–12% of all cases of diabetes.8,9 Treatment of type 2 diabetes with insulin therapy from an early stage is effective in preventing progression to SPIDDM. 10 For this reason, early detection of SPIDDM in patients who are diagnosed with type 2 diabetes is highly desirable.
Among pancreatic autoantibodies (including islet cell autoantibodies (ICA), GADA, IA-2A, and IAA), ICA and GADA are the most reliable predictive markers of SPIDDM. Furthermore, SPIDDM appears to progress only in patients who are GADA positive.11,12 Thus, the detection of GADA is important for the diagnosis of SPIDDM.
Several methods have been developed for the detection of GADA, and some assays are available commercially.
13
ELISA, which has proven reliable and is widely used, is performed as follows. Serum samples are applied to an ELISA plate that has been precoated with glutamic acid decarboxylase 65 (GAD65) antigen. After washing, GAD65-biotin conjugates are added to the ELISA plate. Next, avidin conjugated to horseradish peroxidase is added. Subsequently, a substrate solution is added to the plate, and the antibody concentration is quantified by measurement of enzyme activity (Figure 1(a)). However, this conventional ELISA kit has two problems. (1) Peroxidase–avidin can adsorb non-specifically and generate high background, reducing the sensitivity of the assay. (2) Because the assay measures all materials (immunoglobulin and proteins) that bind to GAD65, specificity is reduced, sometimes making it unclear whether anti-GAD65 IgG antibody is present. We previously developed a novel, highly sensitive enzyme immunoassay (immune complex transfer enzyme immunoassay, ICT-EIA) for IgG autoantibodies to solve these problems. We found that ICT-EIA for IAA is 1000-fold more sensitive than the conventional ELISA kit. In addition, this ICT-EIA is able to detect IAA in subjects with type 2 diabetes who had been classified as negative by the conventional ELISA kit.
14
Schematic diagrams summarising the use of conventional ELISA (a) and ICT-EIA (b) for the detection of GADA. DNP: 2,4-dinitrophenyl; ELISA: enzyme-linked immunosorbent assay; GADA: glutamic acid decarboxylase autoantibody; ICT-EIA: immune-complex transfer enzyme immunoassay.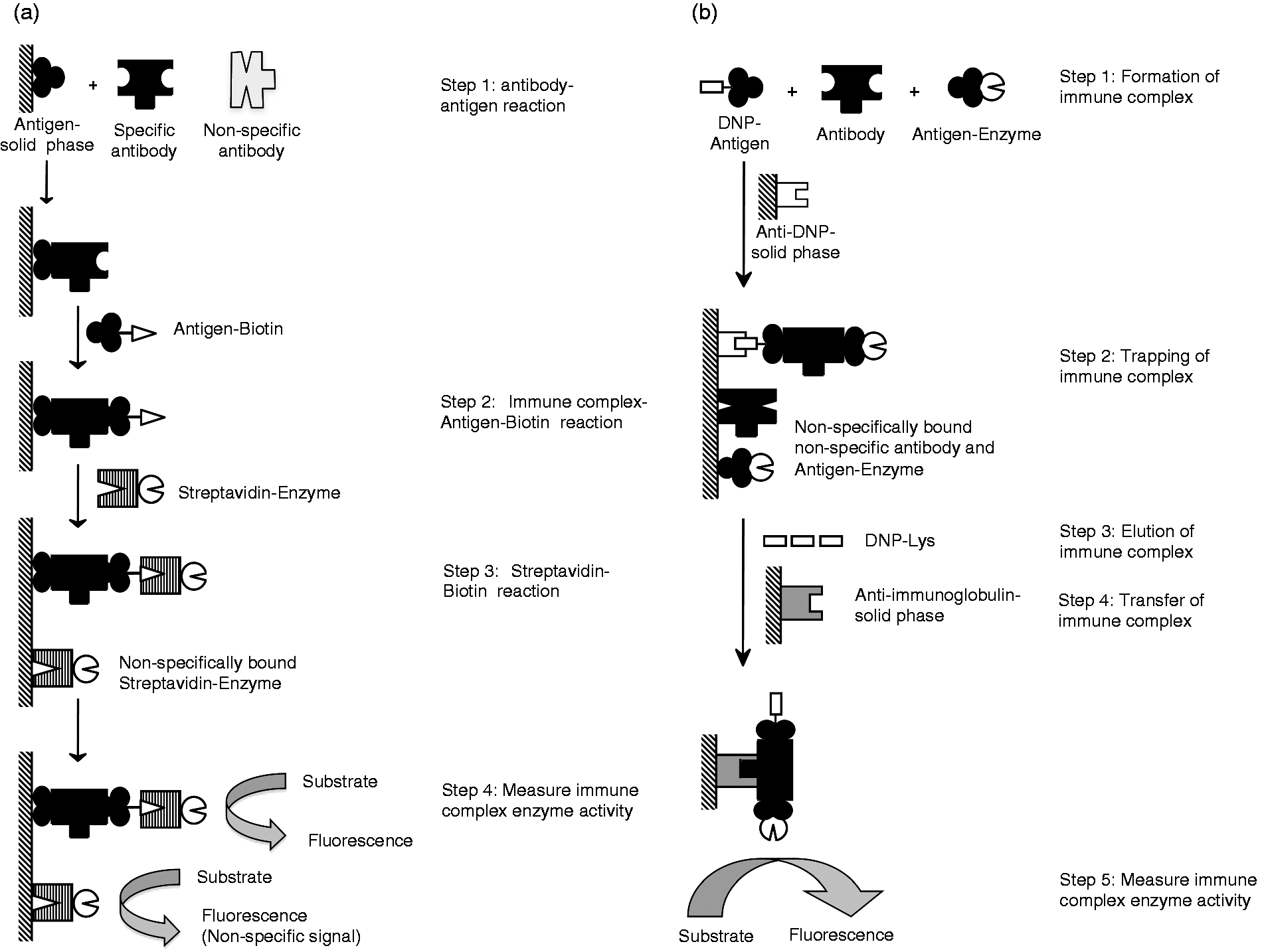
In the present study, we report on the development of a novel, highly sensitive enzyme immunoassay for GADA, which we used to determine anti-GAD65 IgG antibody concentrations in serum samples from patients with type 2 diabetes.
Materials and methods
Buffers
Buffer A consisted of 10 mM sodium phosphate (pH 7.0) containing 0.1 M NaCl, 1.0 mM MgCl2, 1.0 g/L NaN3, and 1.0 g/L bovine serum albumin (BSA; fraction V). Buffer B consisted of 10 mM sodium phosphate (pH 7.0) containing 0.1 M NaCl, 1.0 mM MgCl2, 1.0 g/L NaN3, and 0.1 g/L BSA (fraction V). Both buffers were obtained from Intergen Co. (Purchase, NY).
Antibodies
Rabbit anti-2,4-dinitrophenyl (DNP)-BSA serum and rabbit anti-human-IgG γ-chain IgG were obtained from Shibayagi Co., Ltd (Gunma, Japan) and Medical and Biological Laboratories Co., Ltd (Nagoya, Japan), respectively.
Antigen and ELISA kit
Recombinant human GAD65 was obtained from RSR Limited (Cardiff, UK). The ELISA kit for anti-GAD65 antibody also was obtained from RSR Limited.
Subjects
Serum samples were obtained from patients with type 1 diabetes (n = 22) or type 2 diabetes (n = 29), as well as from non-obese, healthy control subjects (n = 32). Patients with type 1 or type 2 diabetes had been diagnosed based on clinical and laboratory findings, using the criteria defined by the Japan Diabetes Society (http://www.jds.or.jp/).
Blood sampling
Blood samples were drawn from the antecubital veins of subjects. Serum was prepared by centrifugation at 1500 g for 15 min at room temperature and then kept frozen at −20 ℃ until analysis.
Capture antigen and antigen-labelled enzyme preparations
Synthesis of capture antigen (DNP-GAD65)
Thiol groups were introduced into recombinant human GAD65 by reaction with N-succinimidyl S-acetylthioacetate (Pierce Biotechnology Inc., Rockford, IL). Thiols then were reacted with maleimide groups introduced into DNP-lysine (Sigma-Aldrich, St. Louis, MO) using N-(6-maleimidocaproyloxy) succinimide (Dojindo Laboratories, Kumamoto, Japan).
Synthesis of enzyme-labelled antigen (β-D-galactosidase-GAD65)
Thiol groups were introduced into recombinant human GAD65 by reaction with N-succinimidyl S-acetyl (PEG)4 (Pierce Biotechnology Inc.). Thiols then were reacted with maleimide groups introduced into Escherichia coli β-
Preparation of protein-coated polystyrene beads
Polystyrene beads (6.4 mm in diameter; Immunochemical Inc., Okayama, Japan) were coated with 10 mg/L rabbit anti-DNP-BSA IgG or 100 mg/L affinity-purified rabbit anti-human-IgG γ-chain IgG by physical adsorption. 15
ICT-EIA for anti-GAD65 antibody
An aliquot (5 μL) of serum sample diluted to 95 µL with buffer A was incubated overnight at 4 ℃ with 100 μL of buffer A containing 30 fmol of a capture antigen conjugate (DNP-GAD65), 30 fmol of an enzyme-labelled antigen conjugate (β-
The specific FI of ICT-EIA for GADA was calculated by subtracting the fluorescence signals obtained from serum with added GAD antigen from the fluorescence signals obtained from serum not supplemented with GAD.
Expression of the detection limit of GADA
The detection limit for GADA was expressed as the minimal amount that gave a bound β-
Statistical analysis
The cut-off value was set at the mean + 3 SDs of the FI or the absorbance in healthy control subjects; this cut-off provided coverage of almost all of the GADA-negative healthy subjects. Statistical analysis was performed using SPSS version 20.0.0. The ICT-EIA method was compared with the ELISA method by correlation analysis using Spearman’s correlation coefficient.
Results
Sensitivity and specificity of ICT-EIA for GADA
In order to compare the sensitivity of GADA detection via ICT-EIA with that of the conventional ELISA kit, we assayed GADA concentrations in diluted serum samples from patients with type 1 diabetes; for this purpose, the serum samples were serially diluted with serum from healthy control subjects. ICT-EIA was found to be 100-fold more sensitive than conventional ELISA for the detection of GADA serum concentrations (Figure 2).
Comparison of the sensitivity of detection of GADA via conventional ELISA and ICT-EIA. Serum from a type 1 diabetic patient was diluted with serum from a healthy control subject, and GADA in the serum was assayed using conventional ELISA (⋄) or ICT-EIA (○). In this assay, pooled serum samples from six diabetic patients were diluted with pooled serum samples from five healthy control subjects. Vertical bars indicate standard deviations for five replicate determinations.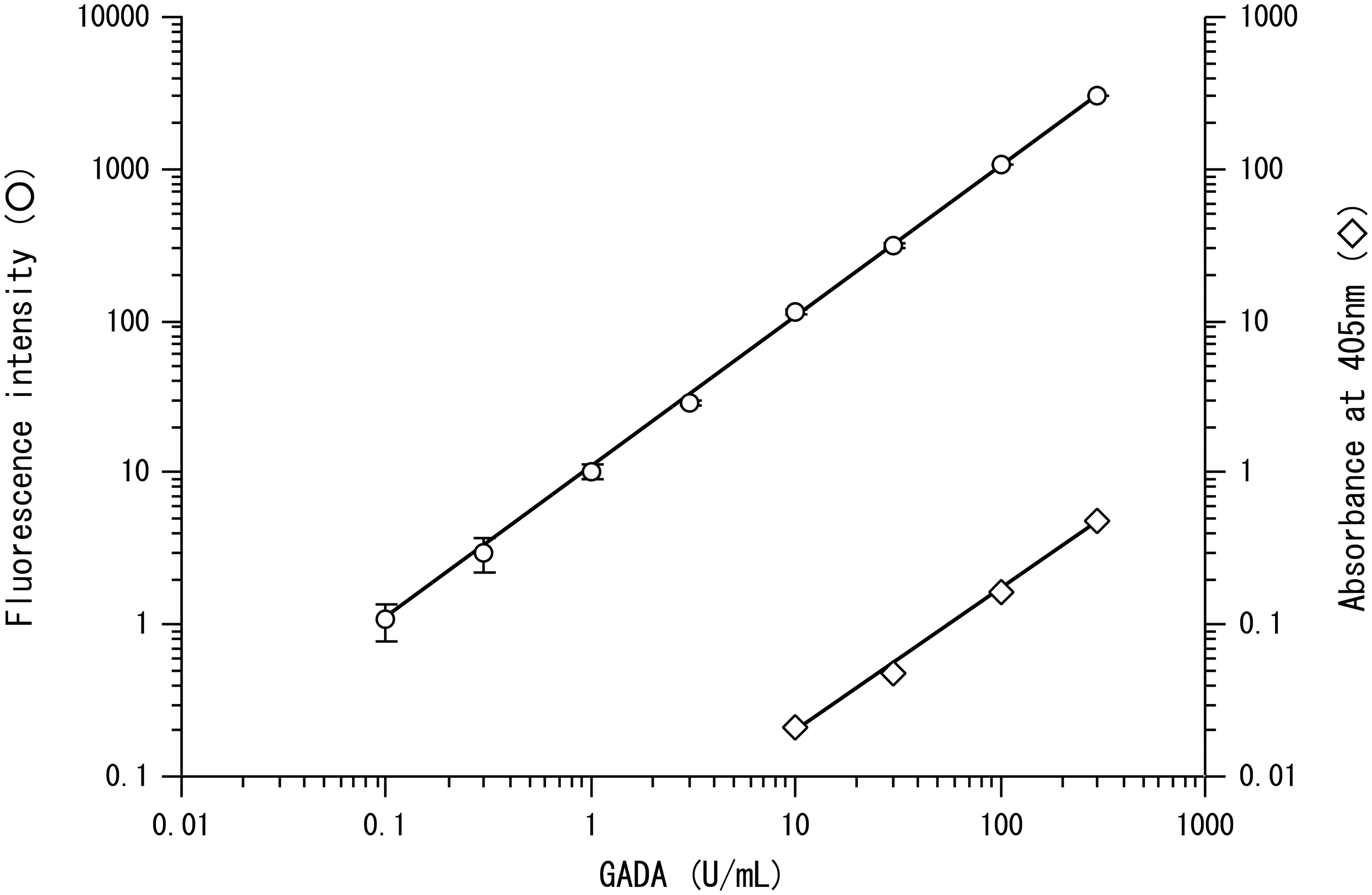
In order to determine the specificity of GADA detection by the ICT-EIA method, GAD (3 pmol/specimen) was added to serum samples from patients with GADA, and the samples then were analysed using ICT-EIA. FIs were decreased greatly (86.5–100.0%) by preincubation with GAD. Thus, most of the fluorescence signal measured in ICT-EIA was shown to be specific for GADA.
Assay variation
The assay variation of ICT-EIA was examined using 20-, 50-, and 100-fold dilutions (with buffer A) of serum samples from a patient with type 1 diabetes. The coefficients of within-assay variations of ICT-EIA (as determined using the 50- and 100-fold diluted samples) were 4.8–5.4% (n = 10). The coefficient of between-assay variations of ICT-EIA (as determined using the 20-fold diluted sample) was 9.0% (n = 10).
Detection of human GADA using ICT-EIA
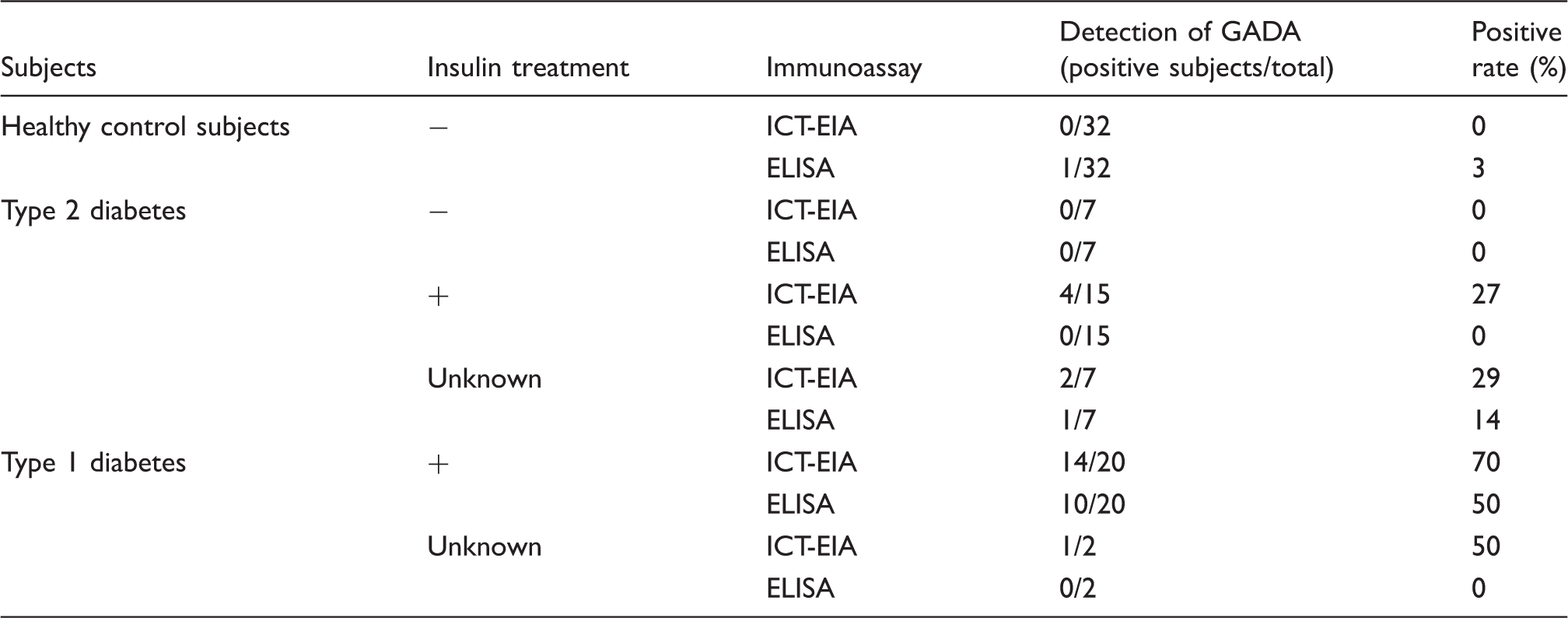
GADA concentrations in serum samples obtained from healthy control subjects and diabetic patients also were analysed using ICT-EIA. We used a cut-off value of 2.4 FI (mean + 3 SDs), which was determined based on data obtained from healthy control subjects. Positive fluorescence signals (i.e. those exceeding a value of 2.4) were confirmed as GADA-specific signals following the induction of a significant decrease in the signal (40–100%) via preincubation with excess GAD. Using ICT-EIA, GADA was detected in 12 out of 22 (55%) patients with type 1 diabetes, and three out of 29 (10%) patients with type 2 diabetes (Figure 3(a)). We also determined the specific GADA concentrations (specific signal) by subtracting the fluorescence signals of serum samples with added GAD from the fluorescence signals of samples without exogenous GAD. The cut-off value for this specific signal was 1.0 FI (mean + 3 SDs). Using this measure, GADA was detected in 15 out of 22 (68%) patients with type 1 diabetes, and in six out of 29 (21%) patients with type 2 diabetes (Figure 3(b)) (Table 1).
ICT-EIA analysis of GADA in patient serum samples. GADA concentrations in the serum samples of patients with the indicated diseases were measured using ICT-EIA. (a) Conventional ICT-EIA: GADA concentrations in the serum of each patient were detected as FI obtained from the serum without GAD antigen. Then nominally positive serum samples, as determined by the cut-off value, were confirmed for positivity by preincubation with excess GAD. (b) Advanced ICT-EIA: GADA concentrations in each serum sample were measured by ICT-EIA with or without GAD antigen. Specific GADA concentrations (specific signal) were defined as the value of the FI obtained from serum samples incubated with GAD less than the FI obtained from serum samples incubated without GAD. True positive serum samples then were determined by comparison to the cut-off value of specific FI. The positivity was defined by the induction of a significant (40–100%) decrease in the signal via preincubation with excess GAD. The dashed lines represent the cut-off values. The circle, diamond, and square symbols represent the healthy control subjects, the patients with type 2 diabetes, and the patients with type 1 diabetes, respectively. Black symbols, GADA-positive samples; white symbols, GADA-negative samples. Comparison of GADA detection by conventional enzyme linked immunosorbent assay (ELISA) and immune-complex transfer enzyme immunoassay (ICT-EIA).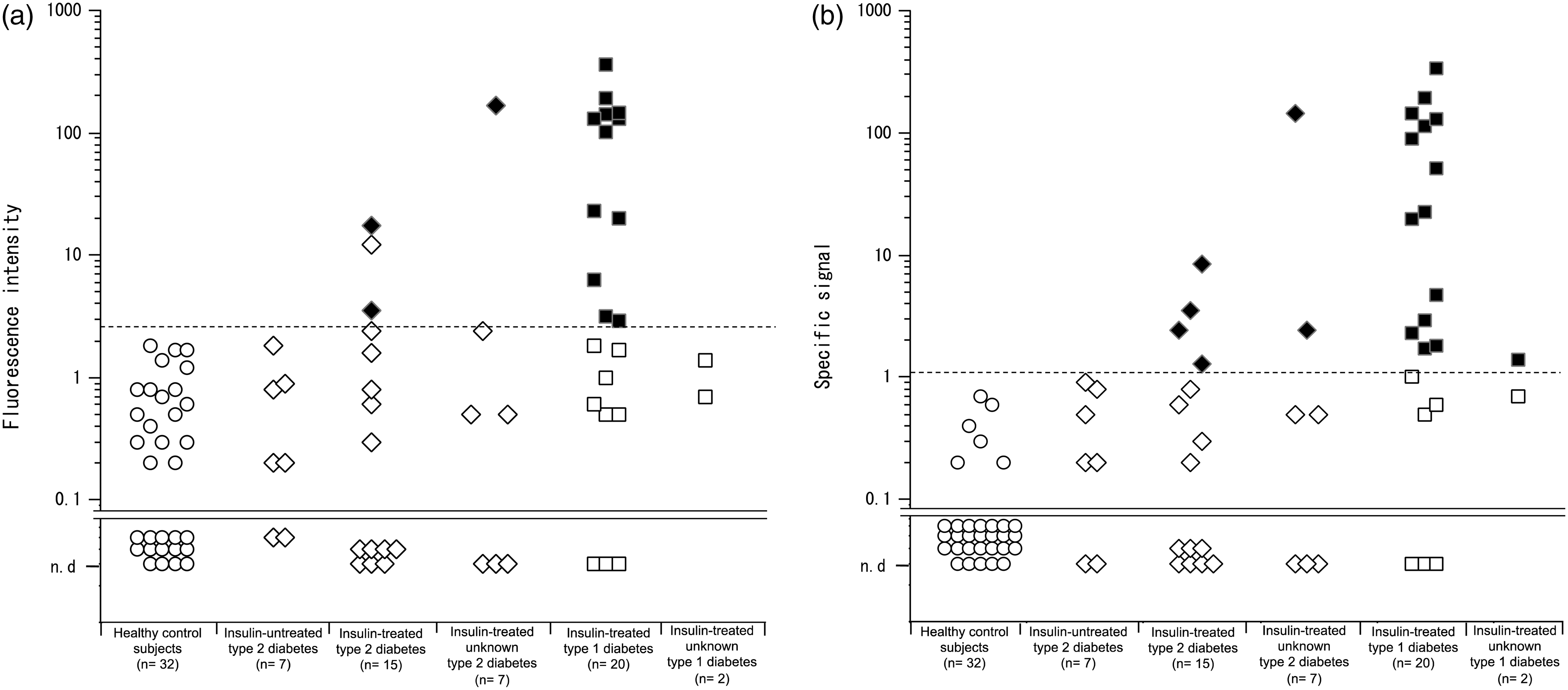
Detection of human GADA using a conventional ELISA kit
Serum samples from healthy control subjects and from diabetic patients were analysed for GADA using a conventional ELISA kit. We used a cut-off value of 0.047 (mean + 3 SDs), which was determined based on data obtained from healthy control subjects; absorbance values higher than 0.047 therefore were considered positives. Using this conventional ELISA kit, we detected GADA in 10 out of 22 (45%) patients with type 1 diabetes, in one out of 29 (3%) patients with type 2 diabetes, and in one out of 32 (3%) healthy control subjects (Figure 4) (Table 1).
Conventional ELISA analysis of GADA in patient serum samples. GADA concentrations in the serum samples of patients with the indicated diseases were measured using conventional ELISA. The dashed line represents the cut-off value. The circle, diamond, and square symbols represent the healthy control subjects, the patients with type 2 diabetes, and the patients with type 1 diabetes, respectively. Black symbols, GADA-positive samples; white symbols, GADA-negative samples.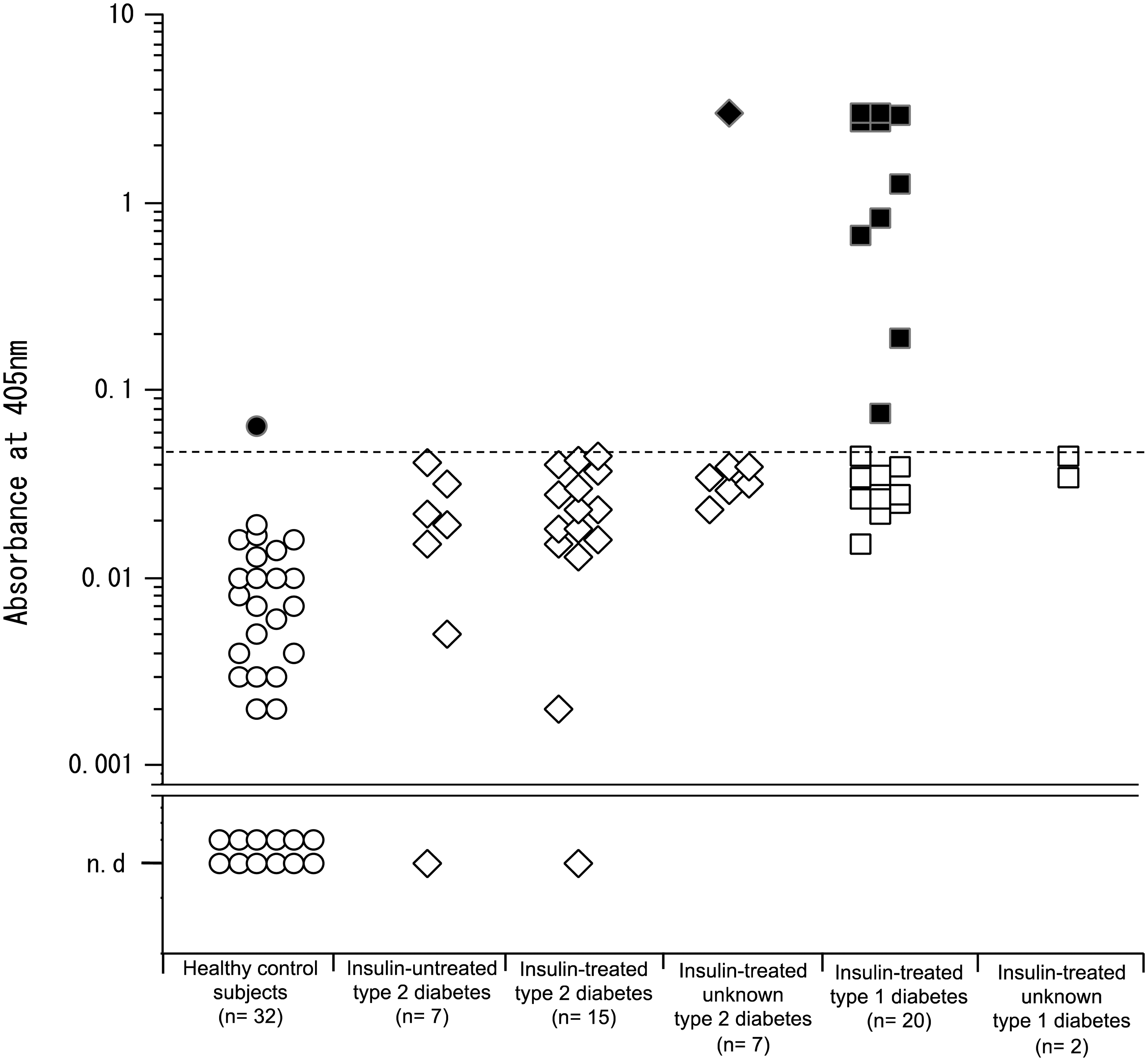
Correlation between ICT-EIA and conventional ELISA kit
We performed correlation analysis in the subset of 37 patients with type 1 or type 2 diabetes in whom GADA was detected by both ICT-EIA and conventional ELISA kit. Also, correlation of ICT-EIA was determined by using the specific signal method. There was a significant correlation between ICT-EIA and conventional ELISA kit data (r = 0.621, P < 0.001) (Figure 5).
Correlation between serum GADA concentrations measured by ICT-EIA and by ELISA. The GADA concentrations by ICT-EIA and ELISA were measured in 19 patients with type 1 diabetes and 18 patients with type 2 diabetes. The diamond and square symbols represent the patients with type 2 diabetes and the patients with type 1 diabetes, respectively. The dashed lines represent the cut-off values of ICT-EIA and ELISA. The correlation coefficient for analysis by ICT-EIA and ELISA (n = 37, r = 0.621, P < 0.001) is shown. Black symbols indicate GADA-positive samples; white symbols indicate GADA-negative samples.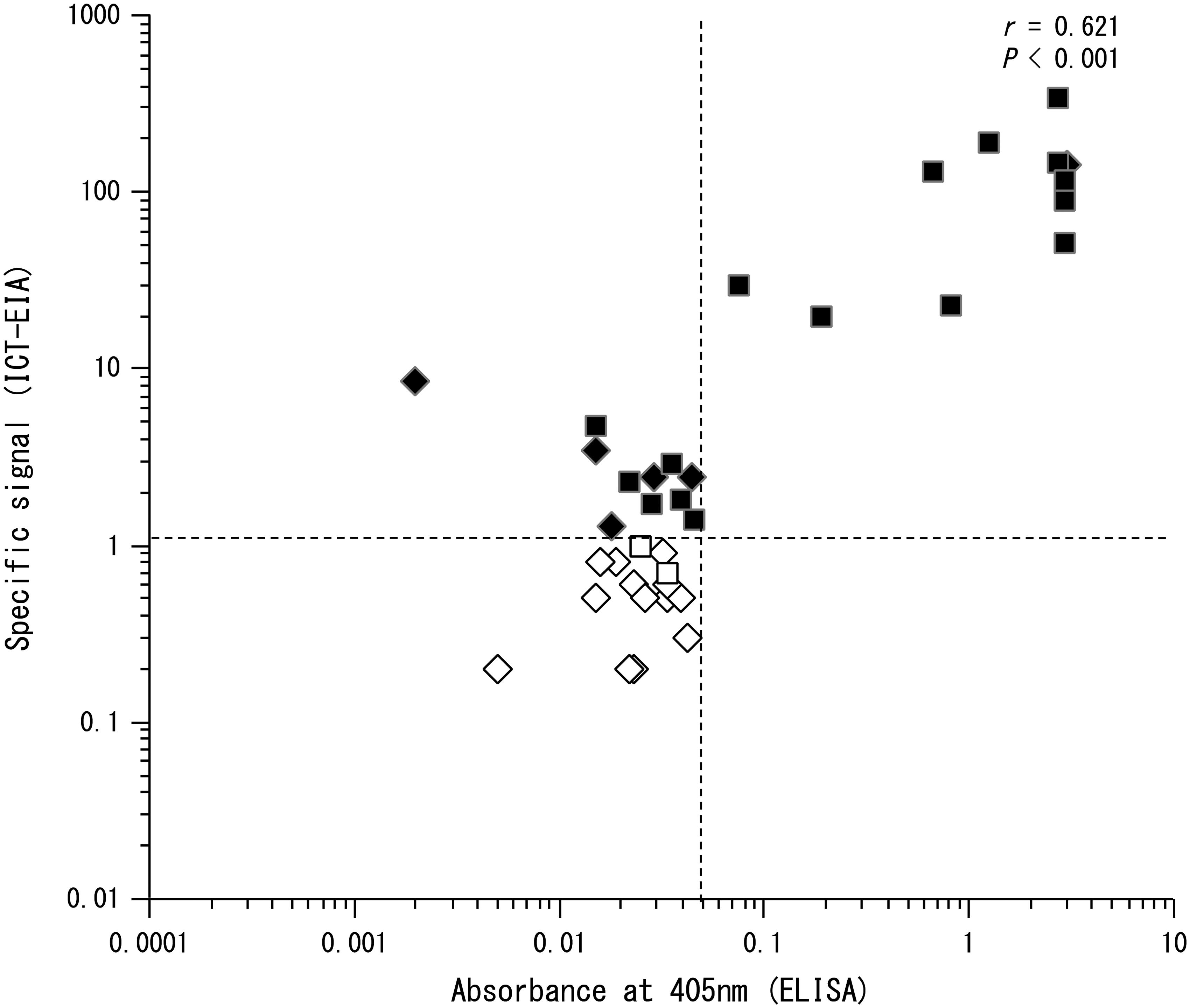
Discussion
It has been reported that the progression to SPIDDM can be controlled by insulin treatment at an early stage. 10 Therefore, it is important to diagnose SPIDDM early. Autoantibodies to pancreatic beta cell antigens, including insulin, GAD, and IA-2, are important serological markers of type 1 diabetes and SPIDDM. In SPIDDM, GADA concentrations remain elevated over the long-term compared to IA-2 A and IAA concentrations. In the past, radioimmunoassay (RIA) methods typically were used to detect GADA, but such techniques required the use of a radioisotope, which was inconvenient. Therefore, ELISA for GADA was developed for clinical use. However, conventional ELISA does not have sufficiently high sensitivity due to signals from enzyme-labelled antigens that are non-specifically adsorbed to the solid phase. For this reason, we developed an ultrasensitive ELISA (ICT-EIA) for GADA. In this method, the immune complexes of antibodies bound to labelled GAD are transferred from solid phase to solid phase to eliminate substances, including non-specifically bound enzyme-labelled antigens, that limit the assay sensitivity.
Sensitivity of ICT-EIA for GADA was compared with that of an ELISA kit. In our hands, ICT-EIA was 100-fold more sensitive than conventional ELISA. The technical documents included with the commercial GAD antibody kits indicate detection limits for ELISA and RIA of 5 and 25 U/mL, respectively. In the work described here, the detection limits for ICT-EIA and the ELISA kit were 0.1 and 10 U/mL, respectively. Based on these results, we speculate that our ICT-EIA for GADA is ≥100-fold more sensitive than RIA for this autoantibody.
Serum samples from healthy control subjects, patients with type 1 diabetes, and patients with type 2 diabetes were assayed using both the conventional ELISA kit and ICT-EIA. We defined the signal that specifically responded to GAD as the specific FI (advanced ICT-EIA), calculated as the net FI (the FI obtained from serum samples without added GAD subtracted from the FI obtained from serum samples with added GAD). A cut-off value for the specific FI was used to distinguish GADA-positive and -negative status. Specific FIs of the advanced ICT-EIA for GADA were measured as described in the ‘Materials and Methods’ section. The advanced ICT-EIA detected lower concentrations of GADA than the conventional ICT-EIA, since the latter measured any signal that bound to GAD (Figure 3(a) and (b)). In conventional ICT-EIA (Figure 3(a)), GADA was initially measured without using the GAD antigen. Nominally positive serum samples, determined by the cut-off value, were confirmed for positivity by preincubation with excess GAD antigen, with positivity defined by the induction of a marked decrease (40–100%) in the signal via preincubation with excess GAD antigen. As a result, pseudo-positive reactions may occur in temporary positive samples by the first measurement. Conversely, in advanced ICT-EIA (Figure 3(b)), these samples are not missed because advanced ICT-EIA always specifically measured GADA.
We analysed the correlation of signals between the ELISA kit and ICT-EIA in 19 patients with type 1 diabetes and 18 patients with type 2 diabetes (Figure 5). There was a significant correlation between the results of the ICT-EIA and those of the ELISA kit (r = 0.621, P < 0.001). Among patients with type 1 and type 2 diabetes, positive detection by ICT-EIA occurred in 11 out of 27 (41%) individuals who were negative by the ELISA kit. In serum samples from patients with type 1 diabetes, GADA was detected in 50% (10 samples) and 80% (16 samples) of the samples using the ELISA kit and ICT-EIA, respectively. In serum samples from patients with type 2 diabetes, GADA was detected in 6% (1 sample) and 33% (six samples) of samples using the ELISA kit and ICT-EIA, respectively. The ELISA kit yielded a positive signal in one individual among the healthy control subjects, whereas ICT-EIA did not detect any positive signal in the same group. These differences presumably reflected the distinct substances detected by each method. ICT-EIA detects only anti-GAD IgG, whereas the ELISA kit detects all substances that react with GAD, which could include IgG, IgA, or the GAD receptor. Since it is possible that the ELISA kit detected substances other than IgG that bind to GAD, a false-positive was suspected for the one sample that was positive by the ELISA kit, but negative by ICT-EIA. In fact, ICT-EIA showed positive detection in 11 of 40 samples that were negative by the ELISA kit. We speculate that ICT-EIA detected low-concentration GADA that was not detected by the ELISA kit (Figure 5). An ultrasensitive immunoassay is necessary for patients with type 2 diabetes that have low-concentration GADA; this higher sensitivity is expected to permit detection of changes in GADA over time.
Specific treatment strategies have been proposed for low-concentration GADA-positive patients.17,18 We have initiated a prospective cohort study for patients with type 2 diabetes and those with prediabetes. We expect that our study may clarify the significance of low-concentration GADA status in the clinical context.
In summary, we developed a novel, ultrasensitive enzyme immunoassay for GADA (advanced ICT-EIA). This method permits the detection of low-concentration GADA that cannot be detected via ELISA in patients with type 2 diabetes. However, this advanced ICT-EIA for GADA is insufficient for independently diagnosing type 1 diabetes or SPIDDM, precisely because the GADA-positive status can represent extremely low antibody concentrations. The presence of GADA (as well as those of IAA, IA-2A, and ZnT8 antibodies) has been proposed as a criterion for the diagnosis of SPIDDM and type 1 diabetes.19,20 It also has been reported that anti-GAD and -IA-2 antibodies reflect distinct immune responses. 21 Multiplex assays (combining the detection of several distinct auto-antibodies) likely will be required. We note that an ICT-EIA method for IAA already has been developed. 14 We are currently developing a novel ultrasensitive enzyme immunoassay for anti-IA-2 antibody. We believe that these ICT-EIA methods together will lead to improved diagnosis of SPIDDM and type 1 diabetes.
Footnotes
Acknowledgements
The authors thank T. Tominaga (University Tokushima, Tokushima, Japan) for assistance in blood sampling.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Ethical approval
The study was approved by the ethics committee of Tokushima Bunri University (Approval number: 4), and all participants provided written informed consent.
Guarantor
SH.
Contributorship
SN was in charge of research design, research planning, and all experiments. SH guided research design and research planning. HK provided the serum of healthy control subjects and patients with diabetes. SI and HS provided the serum of patients with diabetes.
