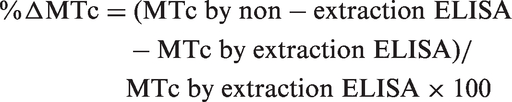Abstract
Background
The aim of the present study is to evaluate the utility of extraction versus non-extraction-based commercial melatonin ELISA kits for determining the melatonin concentration in amniotic fluid obtained in early and late pregnancy.
Methods
Pregnancy duration less than 28 weeks was defined as early and from 28 weeks until delivery as late gestation. Nine samples were obtained in early and 18 in late pregnancy. Two commercially available melatonin ELISA kits (melatonin ELISA RE54021, including methanol-based extraction and direct saliva melatonin ELISA RE 54041, not including an extraction step, both from IBL-International, Germany) were used to determine melatonin concentrations in amniotic fluid.
Results
The mean melatonin concentration in ELISAs assayed by the non-extraction was significantly lower than those assayed after extraction. Subgroup analysis showed that there was no significant difference between melatonin concentration measured by non-extraction versus extraction ELISA in early pregnancy (11.2 ± 7.4 vs. 12.2 ± 7.7, respectively, P = 0.463) but that the mean melatonin concentration in late pregnancy was significantly lower when assayed by non-extraction ELISA than when assayed by extraction ELISA (14.8 ± 9.3 vs. 145.1 ± 179.3, respectively; P < 0.001). Agreement between both measurements in late pregnancy was rather poor (r2 = 0.271, P = 0.022), as opposed to the good correlation found in early pregnancy (r2 = 0.929, P < 0.001).
Conclusions
The present study revealed that a melatonin assay without an extraction step, such as direct saliva ELISA, does not seem to be a valid method to determine the melatonin concentration of amniotic fluid, especially in late gestation.
Introduction
Melatonin (N-acetyl-5-methoxytryptamine) is a hormone secreted mainly by the pineal gland. In addition to its major role as a chronobiotic, melatonin is thought to exhibit modulatory and protective effects on other body systems, including the antioxidant, gastrointestinal and immune systems. Once synthesized, it is not stored in pineal cells but is rapidly released into the bloodstream by diffusion. 1 Because of its high lipophilic and partial hydrophilic property, melatonin has access to various fluids, tissues and cellular compartments, allowing its detection in saliva, 2 urine, 3 cerebrospinal fluid, 4 semen 5 and breast milk. 6
Measurements of melatonin concentrations (MTcs) in plasma or serum, as well as in several other body fluids, have helped to understand its multiple molecular functions in these compartments. For this goal, several analytical techniques have been developed to determine MTcs in different biological fluids.2,4–8 These include radioimmunoassay (RIA); 2 enzyme linked immunoassay (ELISA), which uses an iodinated tracer and solid-phase second antibody; 9 high-performance liquid chromatography tandem mass spectrometry; 10 column-switching semi-microcolumn liquid chromatography-mass spectrometry; 11 and recently, automated solid-phase extraction, high-performance liquid chromatography and fluorescence detection. 12 Some methods, including those based on sample extraction plus chromatography and mass spectrometry, are generally thought to be highly accurate, but they require very costly apparatus, complex sample preparation and a large sample volume and are not suitable for measuring large numbers of samples. RIA is widely used for the quantitative measurement of melatonin in plasma and serum samples because of its high sensitivity, but it is associated with potential health hazards, expensive radiolabelled ligands and the need for a costly radioisotope license to purchase and perform the assays.2,13 Therefore, ELISA is an attractive alternative to the RIA method for melatonin measurement because of its simplicity and rapidity and because of its potential for widespread distribution on account of the standard technology used.13,14 Recently, two ELISA methods, including melatonin direct saliva ELISA (non-extraction), used for the measurement of melatonin in saliva and melatonin ELISA (with extraction), used for the measurement of melatonin in serum and plasma, have been commercialized. The direct ELISA, which can be applied without preliminary treatment of amniotic fluid samples, is technically less complex, very fast and requires a smaller sample volume than the melatonin ELISA with extraction steps (100–200 μL vs. 500–800 μL).
Since the development of validated assays for measuring melatonin in serum or plasma, there has been continuous interest in the evaluation of MTcs in other body compartments to understand its local physiological role. Amniotic fluid is crucial for fetal health not only because it provides a physical barrier against fetal trauma and a defense against fetal infection and is essential for lung development but also because it assists to maintain acid/base balance and contains nutritive components and growth factors. While the existence of melatonin in human amniotic fluid has been previously demonstrated using RIA 8 and high-pressure liquid chromatography, 15 there is little information about its concentration and physiological functions in amniotic fluid throughout pregnancy. One important reason for this may be that there is no validated commercial kit on the market to determine melatonin concentrations in amniotic fluid. Because the concentrations of proteins and other constituents can vary greatly in body fluids depending on the presence of disease or the sampling time or methods, the matrix effects of these constituents can be expected to influence the measurement of melatonin. It is known that composition of amniotic fluid change as pregnancy progresses. 16 Therefore, it remains unclear whether the commercial ELISA kits developed for plasma, serum and saliva are suitable to measure melatonin content in amniotic fluid. The aim of the present study is to evaluate the utility of extraction versus non-extraction melatonin ELISA for determining the melatonin content in amniotic fluid obtained in early and late pregnancy.
Material and methods
Twenty-seven pregnant females were included in the study. The mean age (SD) of the females was 33 ± 5 y (range 22–43 y) and mean gestational age (SD) 31.1 ± 8.5 weeks (range 12–39.1 weeks). In accordance with the Declaration of Helsinki, the study was approved by the Ethics Committee of the University of Bonn. We obtained written informed consent from all females.
Gestational age was evaluated using the last menstrual period and ultrasonographic fetal biometry. Early pregnancy was considered as <than 28 weeks and late pregnancy from 28 weeks until delivery. Nine samples (33.3%) were obtained in early pregnancy and 18 (66.7%) in late pregnancy. Amniotic fluid was obtained from seven pregnant females undergoing amniocentesis and from 20 pregnant females undergoing caesarean delivery under general anaesthesia. Amniotic fluid samples were obtained by needle aspiration that was visually free of blood contamination. The amniotic fluid samples were centrifuged at 3000 g, aliquoted and stored at –80℃ until melatonin analysis.
Melatonin assays
MTc in amniotic fluid was measured using commercial ELISA KITs (melatonin ELISA, RE54021 and direct saliva melatonin ELISA, RE54041, IBL-International, Germany) analysed at the IBL-International laboratory, in Hamburg, Germany. Standards and all samples were incubated in duplicate wells of microtitre plates.
Briefly, melatonin ELISA (Extraction ELISA) is based on the competition principle and microtitre plate separation. First, each sample (500 μL) was passed through a C18 reversed phase column, extracted with methanol, evaporated to dryness and reconstituted with water. Each sample was then added to the corresponding well, which was coated with the goat-antirabbit antibody. Each microtitre plate was filled with either 50 μL of blank reagent, calibrators, extracted samples or standard solutions containing 0, 3.5, 9.5, 40, 140 and 380 pg/mL of melatonin. In total, 50 μL of melatonin biotin and 50 μL of rabbit-antiserum were added into each well, shaken carefully, sealed with adhesive foil and left to incubate overnight (14–20 h) at 2–8℃. After washing, 150 μL of antibiotin conjugate to alkaline phosphatase was added into each well and incubated for 2 h at room temperature. The reaction was developed using p-nitrophenyl phosphate, and optical densities were determined at 450 nm in an automatic microplate reader. According to the manufacturer’s protocol, the intra- and inter-assay coefficients of variation for serum were 3.0–11.4% in the range of 8.8–151.7 pg/mL and 6.4–19.3% in the range of 5.6–134.3 pg/mL. The mean recovery of melatonin was 102.4%, and the sensitivity of the assay was 1.6 pg/ml.
Direct saliva melatonin ELISA (non-extraction ELISA) is also based on the basic principle of a competitive ELISA, such as melatonin ELISA. But there are no extraction steps, and tetramethylbenzidine was used as substrate instead of p-nitrophenyl phosphate in this assay. According to the manufacturer’s protocol, intra-assay variation was 10.8% at 1.7 pg/mL, 6.1% at 5.1 pg/mL and 8.7% at 33.2 pg/mL. Inter-assay variation for saliva samples was 12.7% at 2.1 pg/mL, 7.6% at 4.9 pg/mL and 13.0% at 14.7 pg/mL. The mean recovery of melatonin for saliva samples was 97%, and the sensitivity of the assay was 0.3 pg/ml.
Albumin and total protein assay
Total protein concentrations were measured using Dimension Vista 1500® (Siemens Healthcare Diagnostics, Inc., Newark, Del., USA) and albumin concentrations by BN ProSpec® (Siemens Healthcare Diagnostics, Inc., Newark, Del., USA).
Statistical analyses
The statistical analyses were performed using SPSS 22.0 (SPSS Inc., Chicago, IL, USA). All data are presented as means ± standard deviation (range). The variables were tested for normality using the Kolmogorov-Smirnov test. Because MTc albumin and total protein concentrations were not normally distributed, we used the two-tailed non-parametric Mann–Whitney U-test for group comparison. Percentage difference (%ΔMTc) between the MTc measured using the non-extraction ELISA and extraction ELISA was calculated using the equation:
The association and agreement between both ELISA methods were analysed using correlation coefficients. For all analyses, P values of less than 0.05 were considered significant.
Results
Recovery of melatonin from amniotic fluid assayed by the non-extraction and extraction ELISA
Pooled amniotic fluid samples (two samples from early pregnancy and three samples from late pregnancy) were enriched with 7.5, 11.25, 25, 37.5, 90 or 135 pg/mL melatonin. The analytical recoveries were estimated at six different concentrations using the expected and measured values. The mean recovery of MTc in amniotic fluid assayed by extraction ELISA was 96.8% (coefficient of variation, CV = 15.6%), with a range of 78.2 to 116.8%. The mean recovery of MTc in amniotic fluid assayed by non-extraction ELISA was 74.5% (CV = 34.4%), with a range of 39.0 to 133.0%.
Assay of MTc using the non-extraction and extraction ELISA
The mean MTc measured by the non-extraction ELISA was significantly lower than in extraction ELISA (13.6 ± 8.8 vs. 100.8 ± 158.5, respectively; p < 0.001). The mean %ΔMTc was −51.5 ± 39.1% (range 19.5 to −96.1%). There was a significant negative correlation between %ΔMTc and gestational age (Pearson correlation’s coefficient, r = −0.814, P < 0.001) (Figure 1).
Correlation between gestational age and percentage difference (%ΔMTc) between melatonin concentrations measured using non-extraction and extraction ELISA.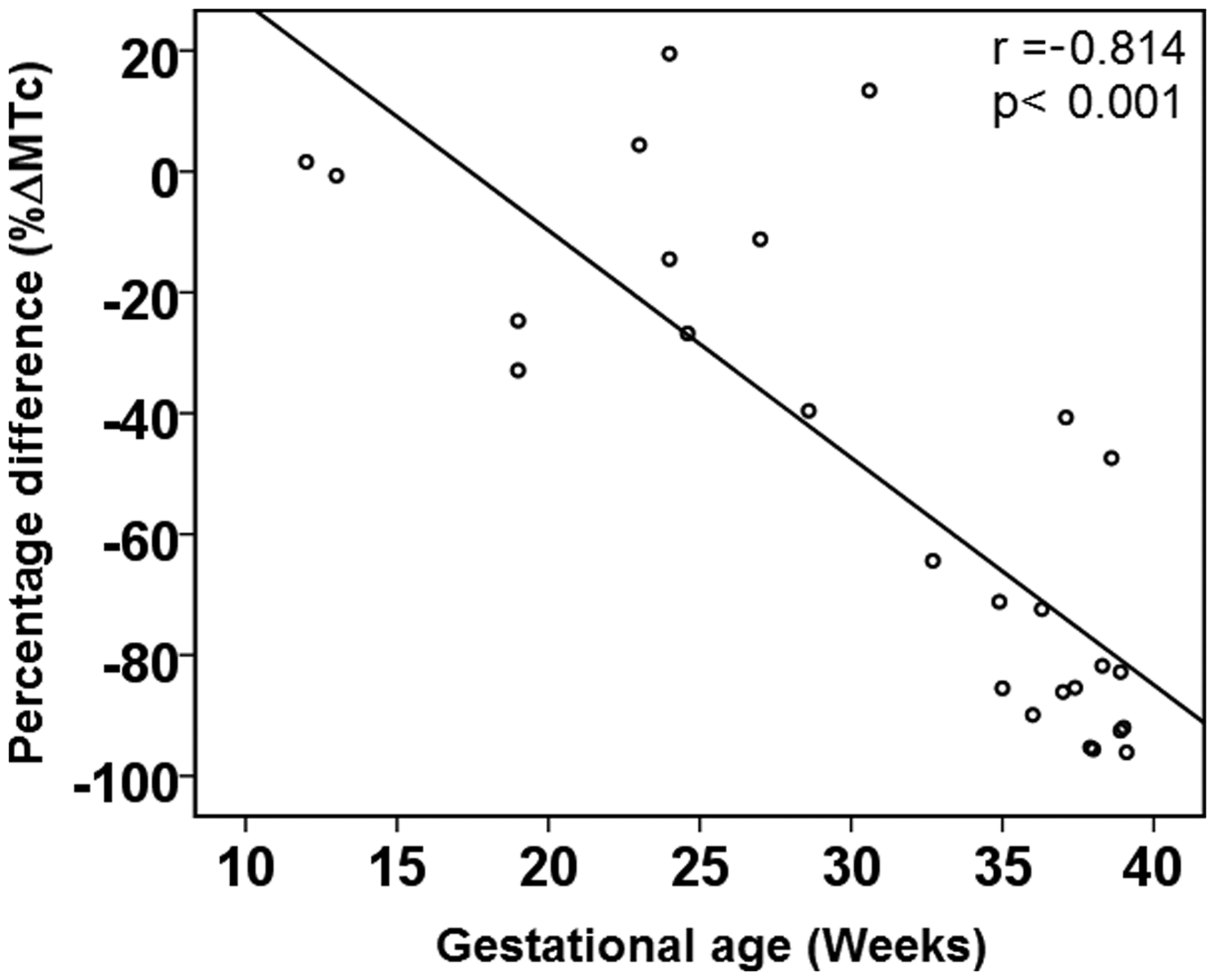
Assay of MTc using the non-extraction and extraction ELISA in early and late pregnancy
A subgroup analysis showed that there was no significant difference between early pregnancy MTc using non-extraction versus extraction ELISA (11.2 ± 7.4 vs. 12.2 ± 7.7, respectively; P = 0.463). However, mean MTc in late pregnancy amniotic fluid samples was significantly lower using non-extraction ELISA than by extraction ELISA (14.8 ± 9.3 vs. 145.1 ± 179.3, respectively; P < 0.001).
In late pregnancy, applying non-extraction and extraction ELISA resulted in greatly different results. The mean %ΔMTc between both methods in late pregnancy was −72.5 ± 28.3% (range 13.4 to −96.1%), compared with −9.5 ± 17.1% (range 19.5 to −32.9%) in early pregnancy. Agreement between both measurements (non-extraction (x) and extraction (y) ELISA) in late pregnancy was rather poor (y = −1.26 + 9.8 × x; r2 = 0.271, P = 0.022), as opposed to a good correlation between both measurements in the second trimester (y = 0.99 + 1.01 × x; r2 = 0.929, P < 0.001) (Figure 2).
Correlation between melatonin concentration (MTc) assayed by non-extraction ELISA (direct saliva ELISA) and extraction ELISA in early (a) and late (b) pregnancy.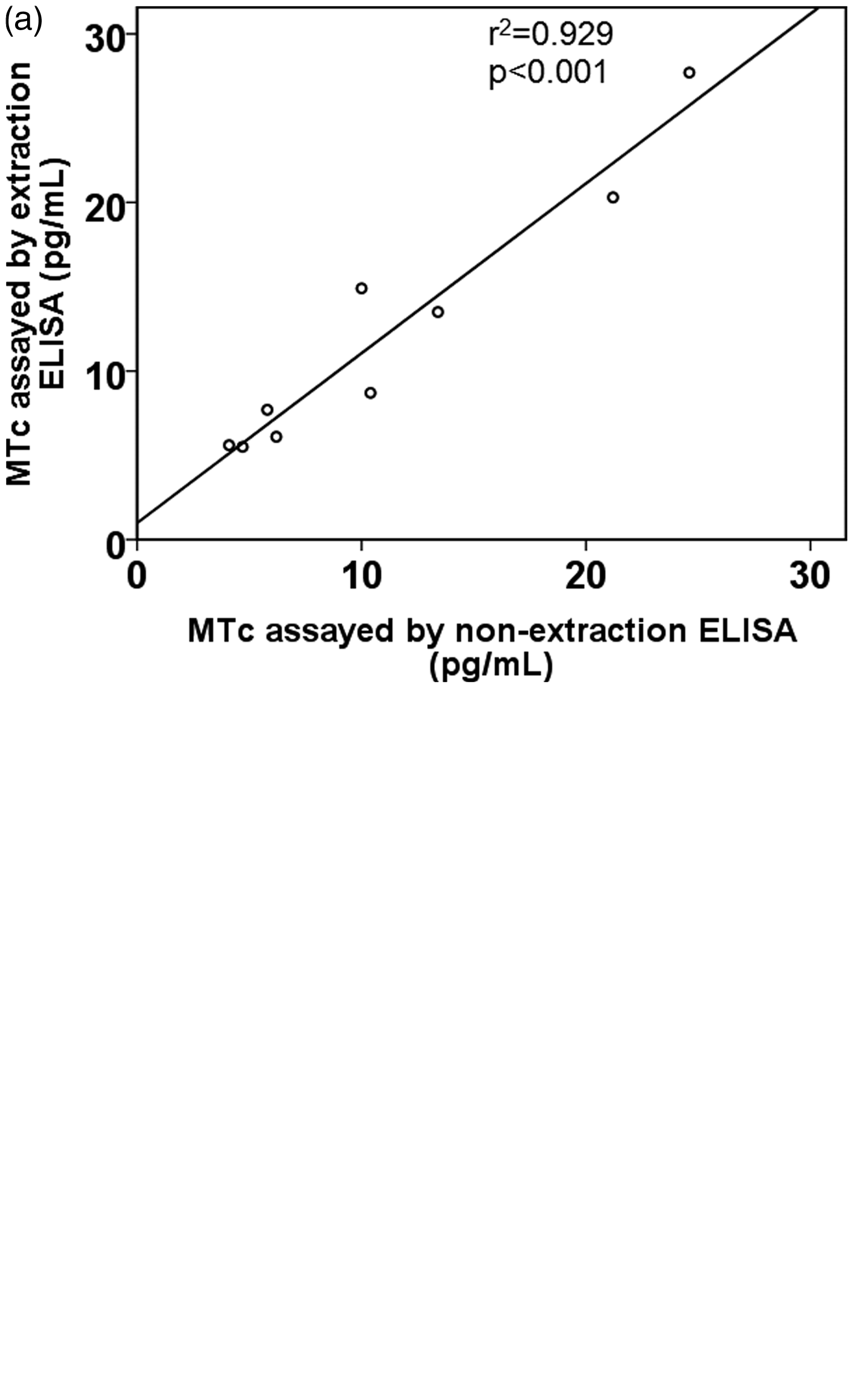
Relationship between albumin and total protein concentrations and mean percentage difference
Mean albumin concentration was 1.9 ± 1.1 g/L (range 0.8–6.5 g/L) and total protein concentration was 3.0 ± 1.9 g/L (range 1.1–10.3 g/L). The albumin to total protein ratio was 63 ± 11%. There was a significant positive correlation between albumin and total protein concentrations (r = 0.920, P < 0.001). We found no significant difference in albumin concentration (1.8 ± 0.8 g/L vs. 1.9 ± 1.3 g/L, respectively; P = 0.857), total protein concentration (2.7 ± 1.3 g/L vs. 3.1 ± 2.2 g/L, respectively; P = 0.625) or albumin to total protein ratio (67 ± 13 g/L vs. 61 ± 11 g/L, respectively; P = 0.217) between early and late pregnancy. There was no significant correlation between mean %ΔMTc and albumin (r = −0.160, P = 0.425) or total protein concentration (r = −0.156, P = 0.436).
Stepwise multiple linear regression analysis was used to analyse associations between mean %ΔMTc and maternal age, gestational age, total protein concentration and albumin concentration. Only gestational age was found to significantly predict %ΔMTc (P < 0.001), indicating that %ΔMTc between both methods tends to decrease throughout pregnancy.
Discussion
Our analysis showed that the measurement of MTc in amniotic fluid using non-extraction saliva ELISA method is not valid in late pregnancy. A high %ΔMTc between non-extraction and extraction ELISA is independent of the albumin and total protein concentrations in late pregnancy but might depend on other factors that change during pregnancy, e.g. affinity of melatonin to so far unknown binding proteins. Hence, it remains unknown whether the validity of non-extraction ELISA is compromised by a ‘matrix’ effect.
A common problem in the determination of melatonin concentrations in body fluids is the difference in composition between the standard probes and the sample matrix. The advantage of using extraction steps is that methanol extraction liberates melatonin bound to proteins in the sample and removes potentially interfering molecules, including proteins that can lead to errors in other assays without extraction steps.17,18 It is generally believed that amniotic fluid in the first and early second trimesters of pregnancy is isotonic with maternal or fetal plasma 19 and represents a transudate of plasma from either the fetus or the mother. 20 However, its composition is known to change as pregnancy progresses. In late pregnancy, amniotic fluid is a mixture of mainly fetal urine, transudate of maternal plasma through the uterine decidua and fluids from the lower fetal respiratory and alimentary tract. 16 Therefore, amniotic fluid does not only contain water and electrolytes but also more than 200 fetal and maternal proteins, fetal and maternal peptides, carbohydrates, lipids and growth factors.21–23 Studies have shown that approximately 60–85% of serum melatonin is reversibly bound to plasma albumin with a low binding affinity.24,25 In contrast to serum, the albumin concentration in amniotic fluid ranges from 0.6 to 6.0 g/L and total protein concentration from 0. 2 to 10.0 g/L, both of which are 10–20 fold lower than their serum concentrations.19,26,27 Burghard et al. 26 demonstrated that the albumin constituted the main fraction of amniotic fluid proteins and accounted for approximately 64% (range 48–85%) of total protein at any stage of gestation. Albumin and total protein concentrations and the albumin to total protein ratio in our samples were consistent with these previous results. Measurements of the protein content showed that the total protein concentration of amniotic fluid increases between 7 and 25 weeks of gestation and decreases thereafter until 35 weeks with a late increase again after 38 weeks.19,27–30 On the other hand, Burghard et al. 26 showed that the concentrations of the entire low-molecular-weight protein fraction were fairly constant between the 17th and 23th week of gestation and decreased progressively during the last trimester, while high-molecular weight protein concentrations were unchanged throughout the last two trimesters. In our analysis, amniotic fluid in late pregnancy tended to have a lower albumin concentration, but there was no significant difference comparedwith the early pregnancy.
Although melatonin binds mainly to albumin and not to alfa-, beta-, or gama-globulin or fibrinogen, a recent study by Morin et al. 31 provided new evidence that an acute phase protein, alpha-1-acid glycoprotein, binds melatonin with a high affinity. 32 The presence of alpha-1-acid glycoprotein in amniotic fluid has also been previously demonstrated. 33 Because albumin was found unchanged in our collective, it is difficult to explain the percentage difference between both methods in late pregnancy on the basis of melatonin binding to albumin. However, it should be noted that our present knowledge of the complete profile of amniotic fluid proteins is rudimentary at best. The proteomic analysis of amniotic fluid by Cho et al. 23 demonstrated that there are significant differences between the plasma proteome and amniotic fluid proteome in early second trimester. Only 36% of the amniotic fluid proteins were found in the plasma proteome. Their results suggest that the amniotic fluid is composed differently than plasma and that it serves different functions. Therefore, it remains unexplained whether melatonin may be bound to other amniotic fluid proteins in late pregnancy. Another possibility is that a number of physiological conditions leading to alterations in the affinity of melatonin to these proteins may change free melatonin concentrations in amniotic fluid in late pregnancy. It is known that recovery during the non-extraction procedure is variable and uncontrollable because of non-specific binding proteins. 17 Moreover, the methanol-extraction procedure, which was used for melatonin extraction in our study, extracts free and bound melatonin from all sources. 17
In summary, the present study revealed that a non-extraction melatonin assay, such as direct saliva ELISA, does not seem to be valid when determining the melatonin content of amniotic fluid in late gestation. One explanation for our findings might be that the affinity of melatonin to binding proteins increases throughout pregnancy. Therefore, a methanol-based extraction for amniotic fluid samples obtained in late gestation is essential to obtaining a valid MTc of amniotic fluid. Future research is needed to determine the factors affecting the affinity of melatonin to binding proteins in amniotic fluid, and an investigation evaluating these factors would be desirable in future studies.
Footnotes
Acknowledgments
We thank Gabriele Strackbein and Dr Jochen Reinsberg for their contributions.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
This study was funded by the Department of Neonatology, Children's Hospital, University of Bonn.
Ethical approval
This study approved by the ethics committee of the university of Bonn (REC number 077/13).
Guarantor
SB.
Contributorship
SB and PB conceptualized and designed the study; SB and DK drafted the initial manuscript, analysed data and performed statistics; ÖA, CB and AW collected samples; BZ performed laboratory detection; PB, AM and HR reviewed and revised the manuscript and approved the final manuscript as submitted.