Abstract
Background
For the early identification of patients at risk of developing diabetic nephropathy, we have developed an ultrasensitive immune complex transfer enzyme immunoassay to measure adiponectin in urine.
Methods
We developed immune complex transfer enzyme immunoassay for adiponectin and measured urinary adiponectin from 70 healthy subjects, 35 obese non-diabetic subjects and 20 patients with diabetes.
Results
The urinary adiponectin concentrations in patients with diabetes (3.3 ± 10.7 ng/mg creatinine) were significantly higher than those in obese subjects (0.54 ± 0.44; P < 0.01) and healthy subjects (0.46 ± 0.42; P < 0.001). The gel filtration elution profile of urine from healthy subjects showed traces of four immunoreactive peaks (high-, medium-, low-molecular weight and monomer molecules), despite the majority of blood adiponectin being high-molecular weight. However, urinary adiponectin molecules were more frequent in low-molecular weight as the estimate glomerular filtration rate decreased. Furthermore, as blood glucose concentrations rose, middle-molecular weight and high-molecular weight increased in urine. Further, urinary adiponectin concentrations correlated with estimate glomerular filtration rate (r = −0.61, P < 0.001), but not urinary albumin. In addition, our analysis showed a significantly (P < 0.001) higher value for urinary adiponectin in the G2 stage of chronic kidney disease classification where urinary albumin is not elevated.
Conclusion
Adiponectin increases in urine as renal function decreases, and urinary adiponectin may be useful as a surrogate marker for diabetic nephropathy risk.
Introduction
Recent reports have described diabetic patients with progressive early decline in renal function, the so-called glomerular filtration rate (GFR) decliners.1–4 Despite the absence of persistent albuminuria and having renal function within a reference range, these diabetic patients have progressively declining renal function that can lead to renal failure. In GFR decliners without increased albuminuria, treatment initiation is delayed, possibly precluding adequate prevention of progression to renal failure. Therefore, if GFR decliners can be identified early by periodic evaluation of renal function, renoprotective treatment can be started while renal function is still within the reference range. This may reduce the decline in, or even improve, subsequent renal function. This requires development of a more sensitive clinical marker for earlier detection of diabetic nephropathy as a predictive factor for determining which patients will be GFR decliners.
Adiponectin is a protein consisting of 244 amino acids, which is an adipocytokine secreted from adipocytes and exists as a trimer and three multimer forms, low-molecular weight (LMW), middle-molecular weight (MMW), high-molecular weight (HMW) as a basic structure in the blood.5–7 It is present in blood at relatively high concentrations (5 to 10 µg/mL) and it has been reported that it has physiological activities such as antiarteriosclerotic effect and an insulin resistance improving action. In recent years, the physiological effects and role of multimer structures have attracted attention, with the HMW concentration or the HMW/total ratio being elevated in visceral fat-type obesity, diabetes mellitus, glucose tolerance and insulin resistance, coronary artery disease, metabolic syndrome8–12 and other diverse diseases such as dietary therapy and surgery to reduce weight effects.13–15 Furthermore, the importance of quantitative molecular determinations of adiponectin has been demonstrated.
Waki et al. 5 have reported that humans with mutations that specifically prevent the formation of HMW adiponectin become diabetic. Furthermore, it has been found that HMW adiponectin is particularly involved in obesity-induced insulin resistance. 16 Urine albumin excretion, glomerular hypertrophy and tubulointerstitial fibrosis were significantly worse in adiponectin knockout mice. 17 In addition, according to a study examined on adiponectin and renal injury, fusion of the podocyte podoplasm of the kidney was observed in adiponectin-deficient mice, and albuminuria was observed. Supplementation with adiponectin resulted in normalization of the pathological kidney tissue and improvement of albuminuria. 18 From these facts, it is suggested that adiponectin may have a protective effect against kidney damage.
Urinary adiponectin from healthy humans has been reported to be below the limit of detection with ELISA kits, 19 and monomers, dimers and trimers of the adiponectin molecule have been detected in urine from patients with albuminuria by Western blotting. 20 A small amount of HMW adiponectin has been reported to be detected in the urine of proteinuria patients. 21
The authors of the present report have used direct measurement of urinary adiponectin by an ultrasensitive immune complex transfer enzyme immunoassay (ICT-EIA) to investigate urinary adiponectin as a possible surrogate marker for renal impairment. We found differences in urinary adiponectin concentrations due to differences in estimate glomerular filtration rate (eGFR) among diabetic patients. Moreover, urinary adiponectin, multimer forms, especially MMW and LMW adiponectin, was detected prior to microalbuminuria. Furthermore, urinary HMW adiponectin increased with the deterioration of renal function.
This paper discusses the possibility of urinary adiponectin as a novel surrogate marker, different from albuminuria, for early detection of renal dysfunction.
Materials and methods
Buffers
The following buffers were used. Buffer A: 10 mM sodium phosphate buffer (pH 7.0), containing 0.1 M NaCl, 1.0 g/L bovine serum albumin (BSA), 1.0 mM MgCl2 and 1.0 g/L NaN3; buffer B: 10 mM sodium phosphate buffer (pH 7.0), containing 0.4 M NaCl, 0.1 g/L BSA, 1 mM MgCl2 and 0.1% NaN3 and buffer C: 10 mM sodium phosphate buffer (pH 7.0), containing 0.1 M NaCl, 0.01 g/L BSA, 1 mM MgCl2 and 0.1% NaN3. BSA (fraction V) was obtained from Sigma-Aldrich Co. LLC. (St. Louis, MO, USA).
Human adiponectin antigen and antiadiponectin antibodies
The human adiponectin antigen used was recombinant human adiponectin derived from an HMW adiponectin determination kit (Fujirebio Inc., Tokyo, Japan). Mouse antihuman adiponectin monoclonal antibody (capture: MAB10651, detecting: MAB1065) was purchased from R&D Systems (Minneapolis, MN, USA). Rabbit (anti-2,4-dinitrophenyl (DNP)-BSA) serum was purchased from Shibayagi Co., Ltd (Gunma, Japan). Human adiponectin ELISA was purchased from Fujirebio Inc. and Otsuka Pharmaceutical Co., Ltd (Tokyo, Japan). Recombinant human globular adiponectin and recombinant mouse adiponectin were purchased from R&D Systems and Bio Vendor (Brno, Czech Republic), respectively.
Subjects
Seventy Japanese non-diabetic, non-obese (NDNO; age 20.1 ± 0.2 years, body mass index [BMI] 21.0 ± 2.0 kg/m2, HbA1c 4.6 ± 0.4%), 35 non-diabetic obese (NDO; age 48.4 ± 7.0 years, BMI 27.0 ± 4.1 kg/m2, HbA1c 5.1 ± 0.3%) volunteers and 20 Japanese diabetic patients (DM; age 62.8 ± 12.9 years, BMI 25.5 ± 3.6 kg/m2, HbA1c 7.6 ± 1.6%) from our clinic participated in the study. Criteria for obese subjects in this study included: BMI of ≥25 kg/m2 or % body fat ratio of ≥25% for men and ≥30% for women.
Blood and urine sampling
Blood samples were drawn early in the morning from an antecubital vein of subjects who had fasted overnight. Samples were then transferred into chilled glass tubes and kept on ice for <30 min. Plasma was separated from the samples by centrifugation at 3000 r/min for 15 min at 4℃, and serum was kept frozen at −30℃ until analysis.
Early morning urine samples were collected from non-diabetic and diabetic subjects who had fasted for 16 h. The urine samples (10 mL) were mixed with 0.1 mL of both 10% BSA and NaN3 and dialysed against 10 mM sodium phosphate buffer (pH 7.0) containing 0.1 M NaCl at 4℃ overnight. Dialysed urine samples were kept frozen at −30℃ until analysis. Urinary albumin and creatinine were also measured. Urine samples with micro- and macro-albuminuria were excluded. Urinary adiponectin concentrations were expressed as a ratio of urinary concentration to milligrams of urinary creatinine.
Capture antibody and detection antibody preparation
Thiol groups were added to monoclonal mouse IgG2b (MAB10651) using N-succinimidyl S-acetylthioastate (Pierce Biotechnology Inc., Rockford, IL, USA) and conjugated with 6-maleimidohexanoyl-DNP-biotinyl-BSA and used as a capture antibody.22,23
The detecting mouse antiadiponectin IgG1 monoclonal antibody (detection: MAB1065) was digested with pepsin to F(ab′)2, which was further reduced to produce Fab′. Fab′ was conjugated with β-D-galactosidase from Escherichia coli (EC 3.2.1.23) using o-phenylenedimaleimide and used as an antibody-labelled enzyme.22,23
Preparation of protein-coated polystyrene beads
Polystyrene beads (6.4-mm diameter; Immuno Chemical, Inc., Okayama, Japan) were coated with rabbit anti-DNP-BSA IgG (0.05 g/L) and biotinyl-BSA (0.01 g/L) by physical adsorption 22 . Biotinyl-BSA-coated polystyrene beads were then reacted with streptavidin (0.01 g/L). 22
Two-site ICT-EIA for adiponectin
ICT-EIA for adiponectin
An aliquot (100 µL) of standard human adiponectin, serum or urine sample, was incubated overnight at 4℃ with 100 µL buffer B containing 10 fmoL of capture antibody conjugated with DNP-biotinyl-BSA and 30 fmoL of detecting antibody conjugated with β-D-galactosidase conjugate (Formation). Thereafter, one polystyrene bead coated with IgG (anti-DNP-BSA) was added to the mixture and incubated for 30 min (Entrapment). After removing the incubation mixture, the polystyrene bead was washed twice and then incubated in 150 µL buffer A containing 2 mM ɛN-2,4-DNP-L-lysine for 30 min (Elution). After removal of the polystyrene bead, one streptavidin-coated polystyrene bead was added to the eluate and incubated for 30 min (Transfer). The bead was then washed and the bound β-D-galactosidase activity was assayed fluorometrically with 4-methylumbelliferyl-β-D-galactoside (0.2 mM) as a substrate for 20 h at 30℃. The incubations with polystyrene beads were performed with shaking at 210 strokes/min at room temperature throughout.22,23
ICT-EIA for albumin
Antialbumin-1 Fab′ and antialbumin-2 Fab′-β-D-galactosidase, both conjugated with DNP-biotinyl-BSA, were used for ICT-EIA for albumin.
Characterization of adiponectin in urine
A urine sample (1.0 mL) was subjected to gel filtration using an AKTA explorer 10S (GE Healthcare, Tokyo Japan) on a column of HiLoad 16/60 Superdex 200 prep grade (1.6 × 60 cm) (GE Healthcare) using buffer A. An aliquot (100 µL) of each fraction (fraction volume, 1.0 mL) was used to measure the adiponectin concentration by ICT-EIA. For comparison, standard adiponectin, human serum and Gel Filtration Calibration Kit (GE Healthcare) were applied to the column, and their concentrations and absorbance were measured.
Measurements of urinary albumin and creatinine concentrations
Albumin and creatinine concentrations of initial urine samples in the morning were measured. Albumin was assessed using ICT-EIA with a specific antialbumin monoclonal antibody as described above, and creatinine was assessed by reaction with picric acid using a commercial kit (Creatinine-test Wako, Wako Pure Chemical Industries, Ltd, Osaka, Japan).
Statistical analysis
All data are presented as means ± SD. The detection limit for adiponectin was expressed as the minimal amount of bound β-D-galactosidase activity that was significantly higher than the non-specific activity in the absence of adiponectin (background). Statistics were performed using SPSS for Windows version 24 J (SPSS, Chicago, IL, USA). Correlations were performed using Spearman’s correlation coefficient. Non-parametric statistics were used to identify significant differences. Differences between two and three independent groups were compared using the Mann-Whitney’s U and Kruskal-Wallis’ H tests, respectively, with the statistical significance level set at 5%.
Results
An ultrasensitive ICT-EIA for adiponectin in urine
The detection limit and sensitivity for adiponectin were 0.1 pg and 1 pg/mL of dialysed urine, using 100 -µL samples, respectively, for complete recovery of the added adiponectin. The detection limit was a few thousand-fold less than that of conventional ELISA kit. The assay range (coefficient of variation [CV] <10%) for urine was 1–1000 pg/mL, using 100 -µL dialysed urine samples.
The reproducibility and recovery of ICT-EIA were estimated for urine using three samples containing different concentrations of adiponectin (range: 5–250 pg/mL). The within-assay and between-assay CVs for adiponectin were 2.5–7.0% (n = 10) and 4.5–7.5% (n = 10), respectively. Analytical recovery of exogenously added adiponectin to 100 -µL urine samples was 90–105%. Therefore, 100 µL of urine was used in all experiments described below. In ICT-EIA for adiponectin, cross-reactions on a molar basis with human adiponectin, human globular adiponectin and mouse adiponectin were 5% and <0.1%, respectively.
Using ICT-EIA and a commercially available ELISA Kit (Otsuka Pharmaceutical) to measure total adiponectin, adiponectin concentrations in 17 serum samples were measured and compared, resulting in the observation of a good correlation (y = 0.022 x2 + 0.27 x + 0.77, r = 0.94, P < 0.001). However, ICT-EIA showed values lower than the total adiponectin concentration in samples below 10 μg/mL. On the other hand, we examined the correlation between HMW adiponectin and the measurement ELISA kit (Fujirebio), showing a strong correlation (y = 0.81 x + 0.95, r = 0.96, P < 0.001). Even in the low concentration range, there was no difference between the methods. From these results, it was suggested that measurements using ICT-EIA mainly represents HMW adiponectin.
Correlation between urinary adiponectin and albumin concentrations
Urinary adiponectin and albumin concentrations (n = 77) are expressed as a ratio of urinary concentration to milligrams of urinary creatinine. A strong correlation was found between urinary adiponectin and albumin concentrations (r = 0.80, P < 0.001). However, in diabetic patients who do not exhibit microalbuminuria, a subject showing a high concentration of urinary adiponectin was observed.
Characterization of adiponectin in serum and urine
The molecular weight and immunoreactivity of adiponectin in serum and urine were examined by gel filtration and ICT-EIA. The elution profiles for serum and urine by gel filtration using AKTA explorer 10S on a column of HiLoad 16/60 Superdex 200 prep grade (1.6 × 60 cm) are shown in Figures 1 and 2.
Gel filtration of immunoreactive adiponectin in serum. Gel filtration of immunoreactive adiponectin in urine.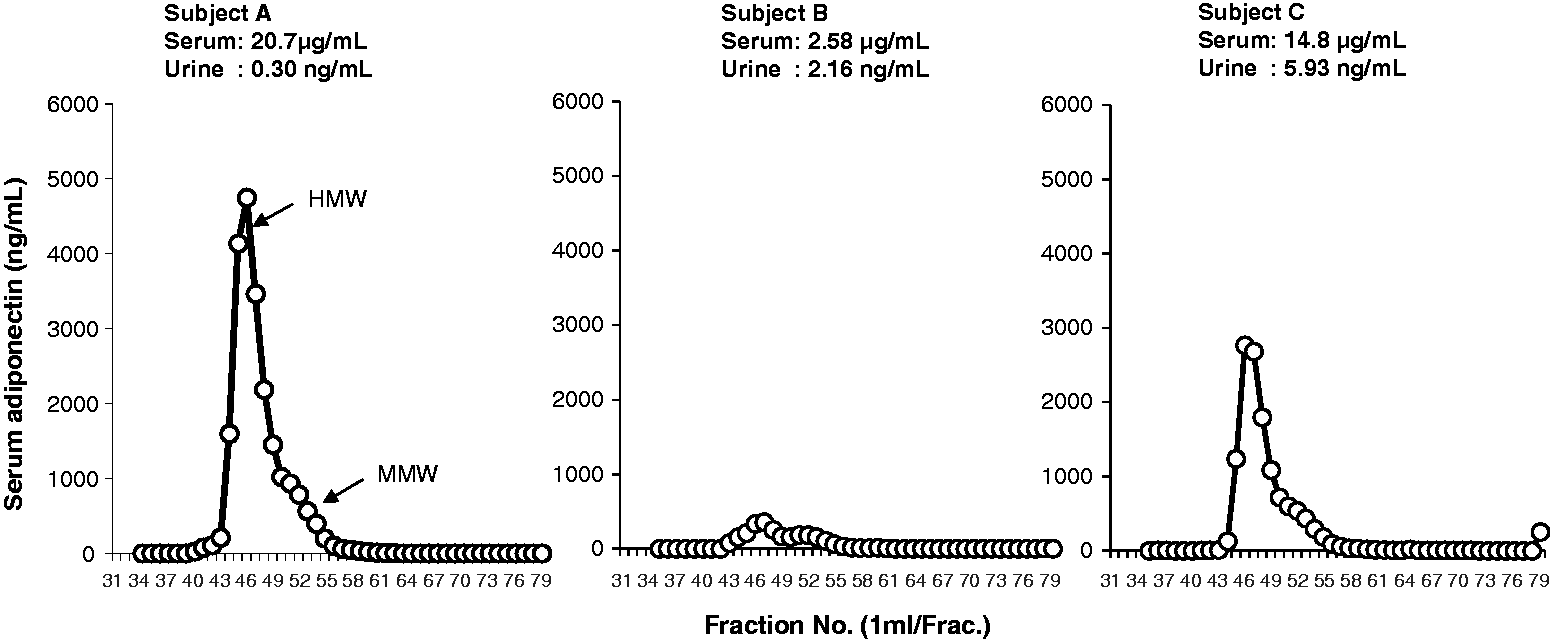
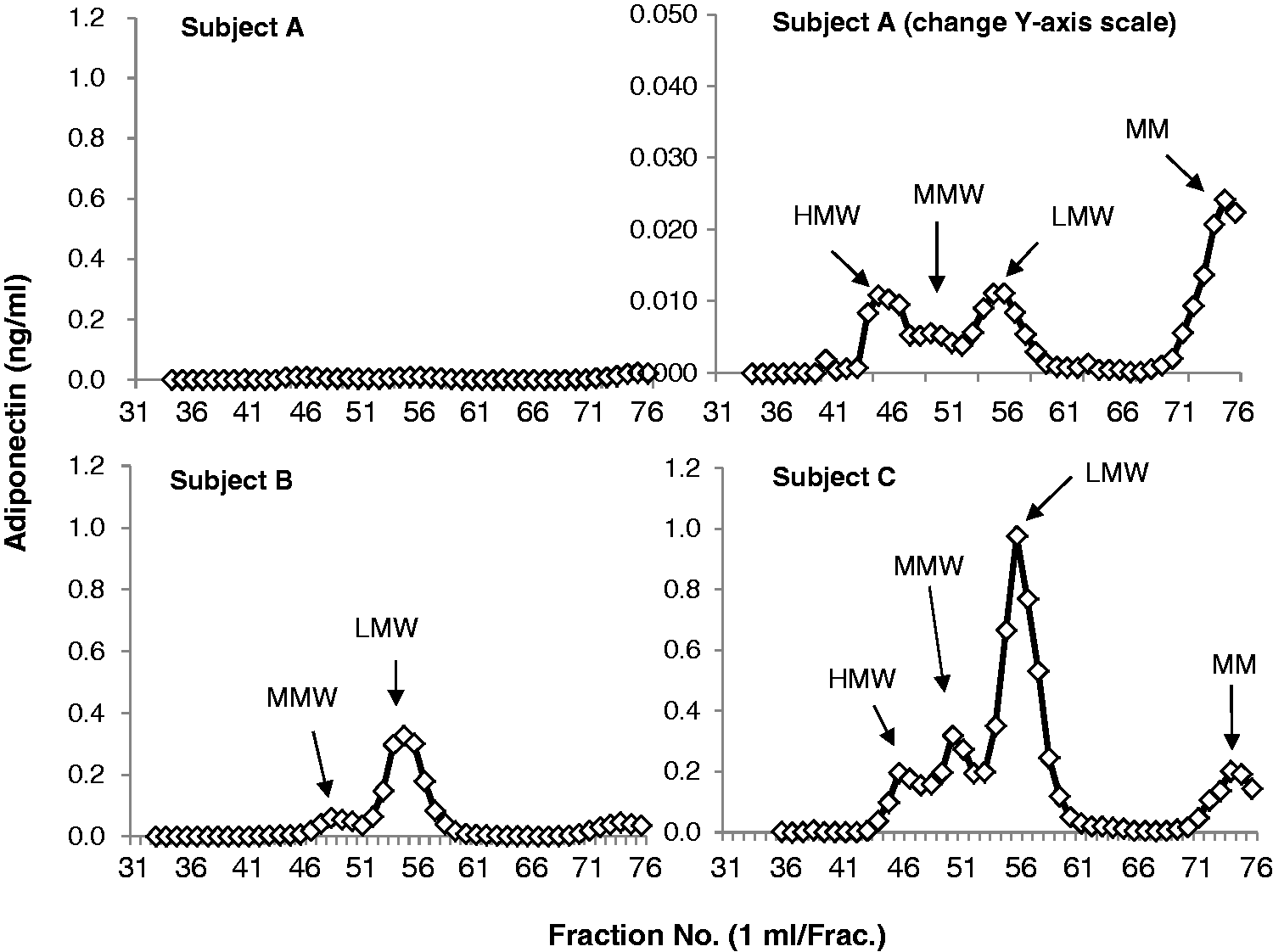
In serum, two immunoreactive peaks were observed. Molecular weight markers showed that serum contains HMW (670 kDa) adiponectin and MMW (440 kDa) adiponectin. On the other hand, in addition to these two adiponectin molecules, further LMW immunoreactive substances were detected in the urine. These LMW molecules are probably LMW (140 kDa) adiponectin and monomer adiponectin molecule (28 kDa).
Characteristics of three subjects and serum or urinary adiponectin concentrations.
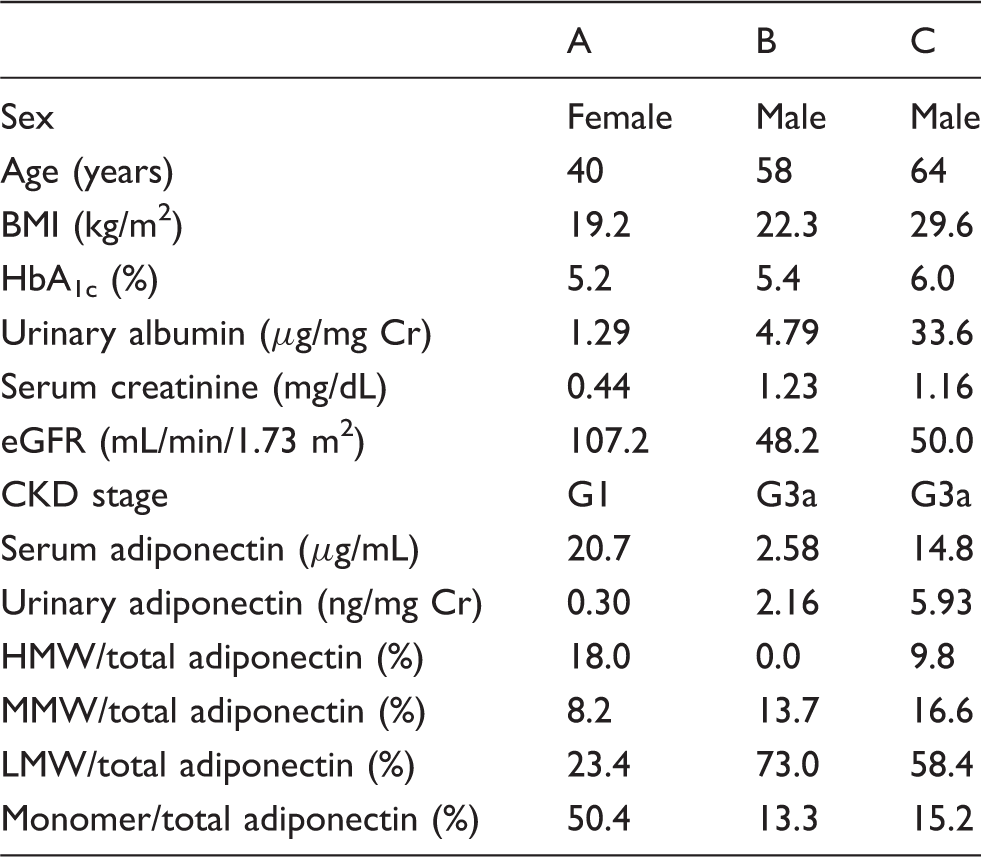
CKD: chronic kidney disease; eGFR: estimate glomerular filtration rate; MMW: middle-molecular weight; HMW: high-molecular weight; LMW: low-molecular weight; BMI: body mass index; HbA1c: haemoglobin A1c; Cr: creatinine.
Association of urinary adiponectin concentrations with diabetic nephropathy risk
The adiponectin concentrations in urine from NDNO and NDO subjects and DM patients, excluding patients with micro- and macro-albuminuria, were determined to be 0.46 ± 0.42, 0.54 ± 0.44 and 3.3 ± 10.7 ng/mg creatinine, respectively (Figure 3). The adiponectin concentration in DM patients was significantly higher than in NDNO and NDO subjects (P < 0.001 and P < 0.05), respectively.
Adiponectin concentrations in serum and urine from NDNO subjects, NDO subjects and DM patients.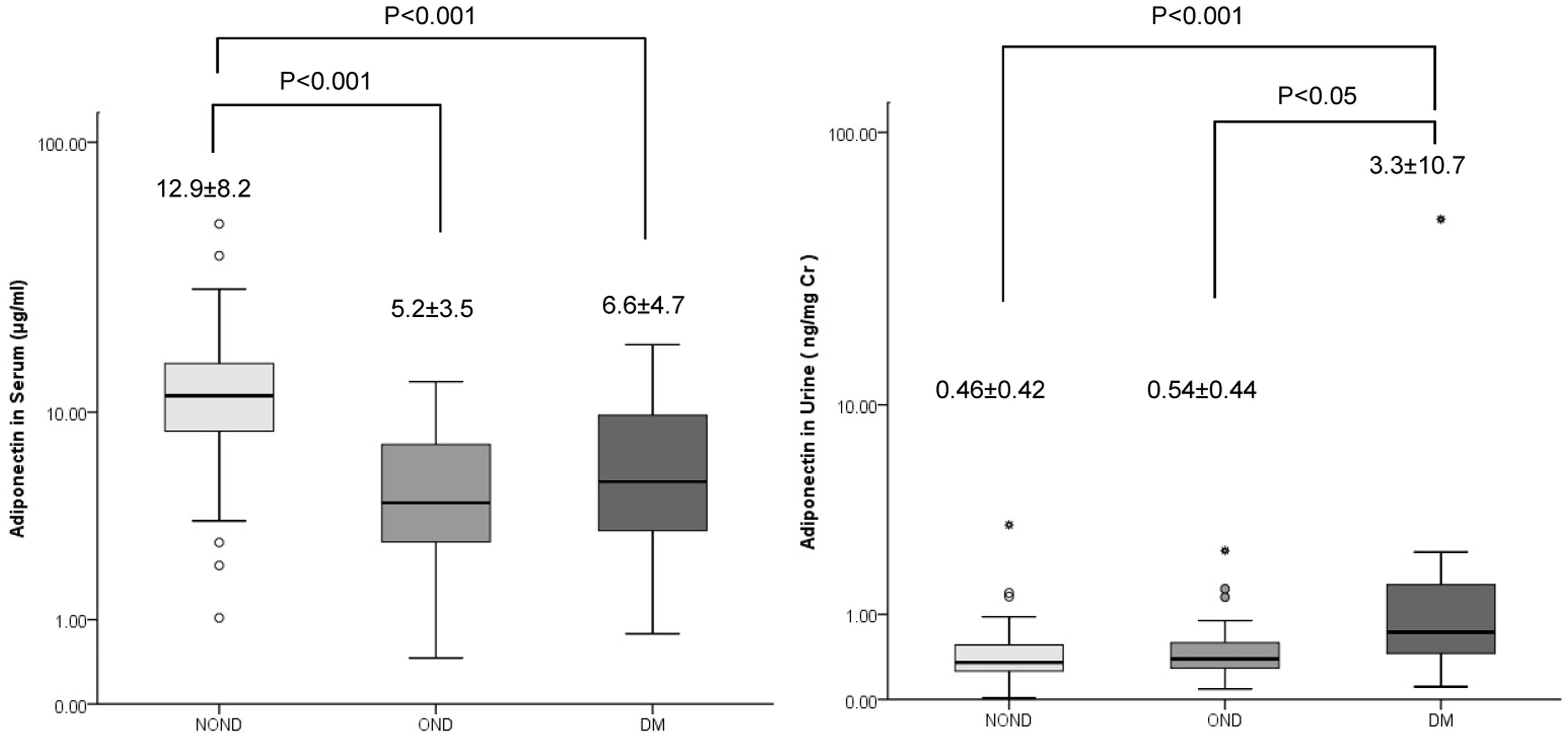
We analysed the correlations of urinary adiponectin concentrations to eGFR in 70 subjects. The urinary adiponectin concentrations were significantly and positively correlated to eGFR (r = 0.61, P < 0.001). On the other hand, urinary albumin concentrations were not correlated to eGFR (r = 0.16, P = 0.198) (Figure 4).
Correlation of urinary adiponectin and albumin concentrations to eGFR.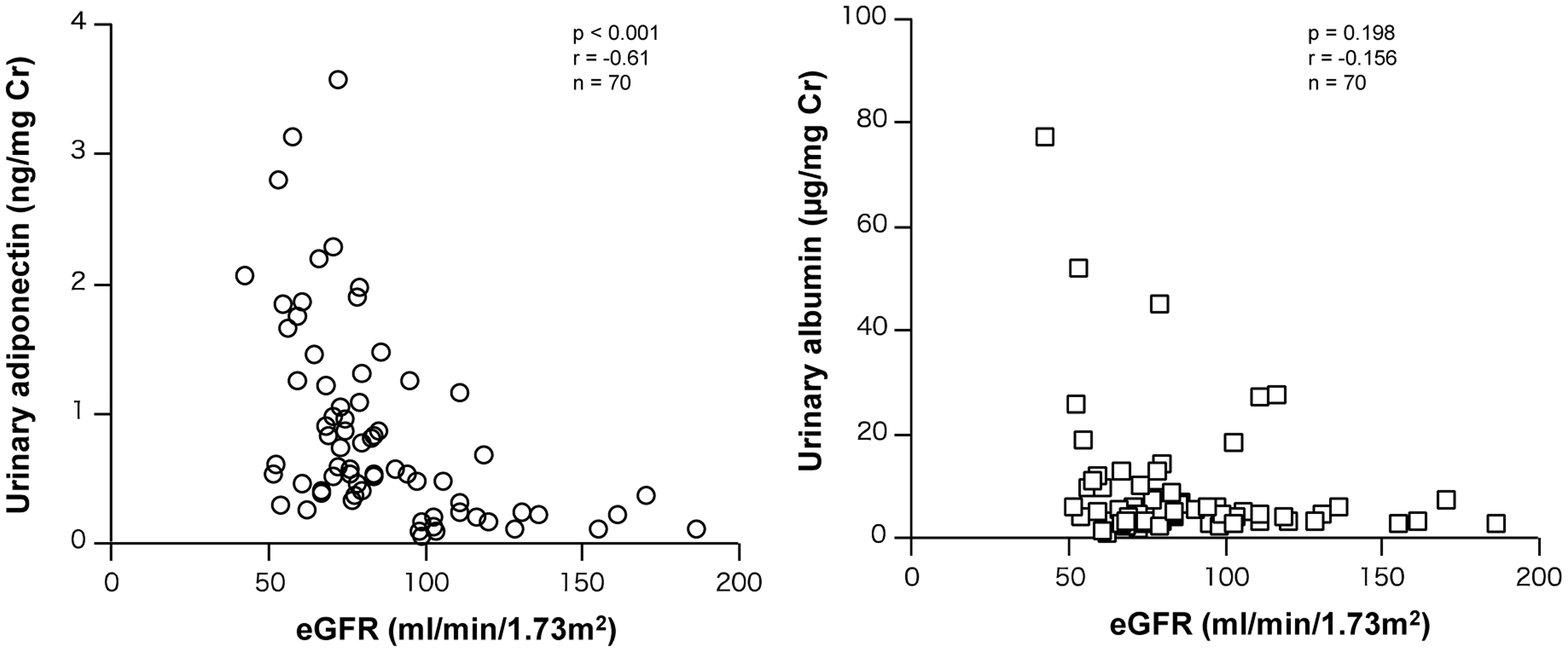
Changes in urinary adiponectin in chronic kidney disease (CKD) classification were examined (Figure 5). As a result, urinary adiponectin increased with decreasing renal filtration function and showed a significantly increased value in stage G2 (P < 0.001). On the other hand, urinary albumin was delayed relative to adiponectin and was significantly increased in stage G3a (P < 0.01).
Urinary adiponectin and albumin concentrations in non-diabetic volunteers.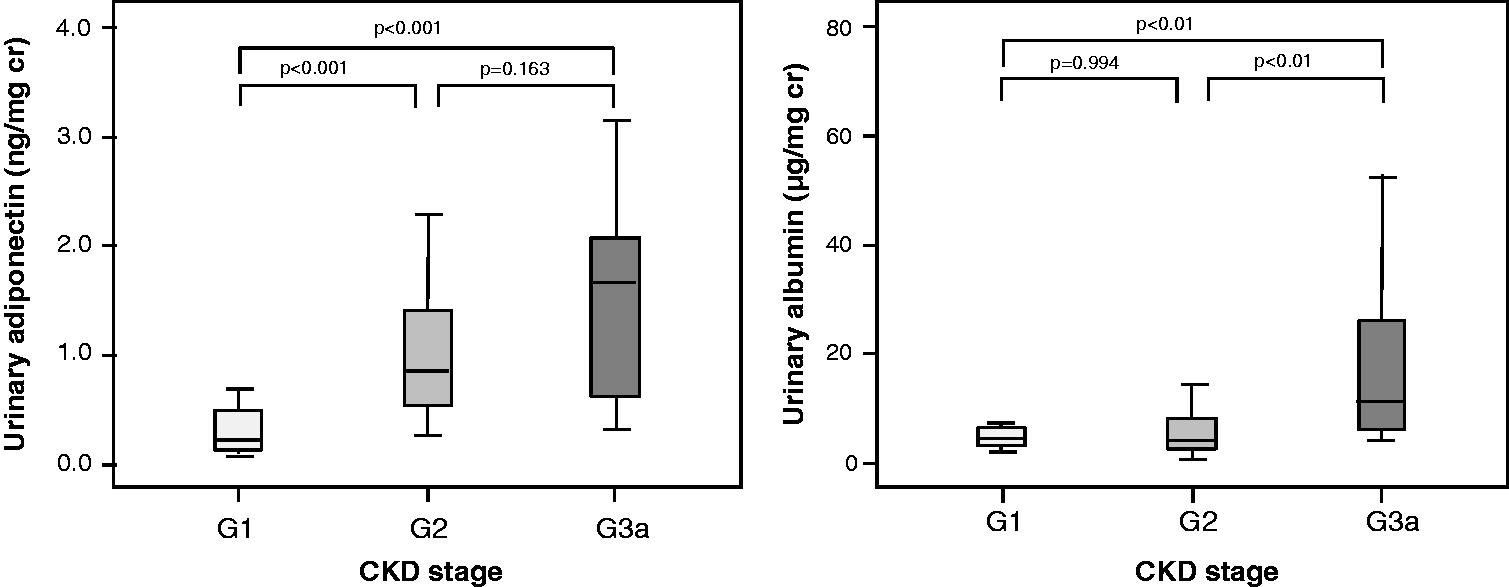
Discussion
We previously developed a highly sensitive ELISA for measuring urinary growth hormone (GH) in 1987 24 that showed the efficacy of nocturnal urinary GH as a supporting diagnostic index of the pituitary dwarfism. Recently, we have developed an ultrasensitive ICT-EIA for urinary soluble insulin receptor-α(sIRα), resistin, insulin and leptin found that these concentrations were significantly correlated with blood concentrations. 25 Urine samples with micro- and macro-albuminuria were excluded. Urinary sIRα, resistin, insulin and leptin concentrations are increased in patients with diabetes and increased with obesity. Therefore, urinary biomarkers can be useful for both physiological and clinical studies.
We developed a new technique for measuring adiponectin with ultrasensitivity by employing an ICT-EIA. This new method can measure adiponectin at concentrations of 0.1 pg, with a sensitivity of 1 pg/mL, using 100 -µL urine samples. Comparing adiponectin concentrations in serum from NDNO subjects, NDO subjects and DM patients revealed significantly lower levels in NDO subjects and DM patients, excluded micro- and macro-albuminuria, compared with those in NDO subjects (P < 0.001 for both comparisons) (Figure 3). In contrast to blood concentrations, urinary adiponectin from DM patients were significantly higher than in NDNO subjects and NDO subjects (P < 0.001 and P < 0.05, respectively) (Figure 3). These results indicate that an increase in urinary adiponectin concentrations may be a clinically important biomarker for diabetic nephropathy risk.
Saraheimo et al. 26 reported that in type 1 diabetics from the Finnish Diabetic Nephropathy Study, patients with macro-albuminuria on average had a higher level of adiponectin when compared with patients with normo-albuminuria and microalbuminuria, and high adiponectin concentrations in urine predicted the progression of macro-albuminuria to end-stage renal disease (ESRD). On the other hand, Panduru et al. 27 reported that in 2090 patients with type 1 diabetes followed for a median of 5.8 (4.4–6.9) years, urinary adiponectin was a strong independent predictor of diabetic nephropathy progression from macro-albuminuria to ESRD. Thus, the increase in urinary adiponectin is identified to be associated with ESRD risk in type 1 diabetes. Kacso et al. 28 found that low adiponectin concentrations predicted increased urine albumin to creatinine ratios over the one-year follow-up of patients with type 2 diabetes. These results indicate that in diabetic patients, an increase in urinary adiponectin is associated with a decrease in renal function.
Unfortunately, the molecular morphology of native adiponectin in the urine is not clearly understood. In urinary adiponectin of healthy subject, the native form could not be analysed using a conventional kit because the concentration of the adiponectin was very low. Therefore, we developed an ultrasensitive measurement method for adiponectin and analysed the molecular morphology using the size exclusion chromatography method. The molecular weight of adiponectin in serum was shown to be mainly HMW and MMW (Figure 1), although there is a difference in their amounts. However, trace amounts of LMW and monomer were detected. On the other hand, the molecular pattern of adiponectin in urine varied greatly depending on the level of kidney function (Figure 2 and Table 1). In subject A, with normal renal function, the urinary adiponectin concentration was very low, but there were four molecular forms detected (HMW, MMW, LMW and monomer). Furthermore, it was shown that about half of the monomers were occupied. Subject B was in stage G3a, where urinary albumin was not observed, and MMW, LMW and monomer were detected in the urine, with the majority being LMW (Figure 2 and Table 1). Subject C was in stage G3a, exhibiting a somewhat higher blood glucose concentration with microalbuminuria, with high urinary adiponectin concentration, expressing all four types of adiponectin (HMW, MMW, LMW and monomer). Koshimura et al. 29 reported that the 30 kDa form of adiponectin, identified by Western blot analysis, was present in the urine from patients with diabetic nephropathy with macro-albuminuria. Kopf et al. 30 reported that HMW adiponectin (∼380 kDa; by ELISA) was excreted to urine from patients with diabetic kidney disease.
Is adiponectin excreted in urine, by (1) leakage from glomeruli or (2) secretion? We have reported that insulin (6 kDa), leptin (16 kDa), resistin (20 kDa), GH (22 kDa) and sIRα (370 kDa) leak into the urine with intact molecular weight, as confirmed by the gel filtration method. Furthermore, the blood and urine concentrations have been shown to be well correlated. The urine/blood concentration ratio for insulin was almost 1, while the ratios for GH and leptin were 1/100 and sIRα was 1/1,000 (data not shown). The urinary/blood concentration ratio for adiponectin is approximately 1/10,000. In addition, ferritin (440 kDa) was hardly detected in the urine. Therefore, we believe that glomerular filtration membranes are permeable at a certain rate for proteins up to 20–30 kDa. At this time, trace amounts of LMW (140 kDa) and monomer (28 kDa) adiponectin in the serum were also filtered out at a constant rate, and as a result, it is thought that monomers adiponectin was high in healthy subjects. Additionally, MMW (440 kDa) and HMW (670 kDa) do not easily leak into the urine due to the filtration function of the glomerulus. It is inferred that MMW starts to leak due to abnormalities in the filtration function of the glomeruli, and HMW starts to leak owing to further functional deterioration.
Kopf et al. 30 reported that HMW-adiponectin with a size >250 kDa would not pass the filtration barrier. On the other hand, Shimotani et al. 31 showed that adiponectin monomers (28 kDa) and dimers have a molecular weight small enough to cross the normal functioning glomerular filtration barrier and thereby adiponectin can be traced in the urine of healthy subjects. von Eynatten 20 showed a lower accumulation of adiponectin in glomerular capillaries of patients with type 2 diabetes and nephropathy compared with control kidneys, with increased vascular stress, glomerular damage and podocyte depletion, resulting in a significant loss of adiponectin binding sites and increased adiponectin in the urine. Benigni et al. 32 reported that nephrin, a podocyte associated protein, is important in maintaining the size selective barrier of the kidney, and the renal expression of nephrin is impaired in diabetic nephropathy. In this way, we speculate that elevated urinary HMW, MMW and LMW-adiponectin may be the result of a collapse of the glomerular molecular barrier and is involved in the pathomechanism of diabetic kidney disease.
On the other hand, what is the possibility of adiponectin secretion into the urine? Rovin et al. 19 described that adiponectin is normally present on the endothelium and in the smooth muscle cells of intrarenal arteries/arterioles and on the endothelium of glomerular and peritubular capillaries, while a weaker signal for adiponectin can be found in proximal and distal tubular epithelial cells. Inoue et al. 33 suggested that human mesangial cells express adiponectin and that the expression of adiponectin and the adiponectin receptor were downregulated under inflammatory conditions, such as IgA nephropathy. Christou et al. 34 reviewed that adiponectin crossing the glomerular filtration barrier possibly inhibits inflammation, fibrosis and oxidative stress in kidneys through the activation of AMP-activated protein kinase and provides protection against albuminuria and increased podocyte permeability. Thus, the production of adiponectin in the kidney has been demonstrated; however, there is no clear answer on the physiological significance of adiponectin secreted into the urine, and further studies will be needed to determine whether adiponectin is secreted in urine in response to kidney damage.
Since albumin also has a molecular weight of about 66 kDa, it begins to leak upon deterioration of renal filtration function; however, it seems that resistance attributable to the charge of the basement membrane delays the appearance of albumin in the urine. As a result, it is inferred that albumin behaves differently from urinary adiponectin. While secretion is a possibility, secretion cannot explain the difference in molecular weight.
Perkins et al. 3 reported that 50% of patients with type 1 diabetes showed progression of renal disease even in the absence of obvious albuminuria. Ha et al. 35 reported that urinary adiponectin might play a role in the identification of diabetic patients at increased risk of nephropathy progression.
Based on these findings, we are currently developing an ultrasensitive measurement method for HMW adiponectin specifically for the purpose of more accurately monitoring the progression of diabetic kidney disease.
In summary, we present a new ultrasensitive enzyme immunoassay for adiponectin, ICT-EIA. ICT-EIA can specifically measure adiponectin in urine, without any requirement for sample extraction or concentration. The urinary adiponectin concentrations are increased in patients with diabetes and nephropathy. In the initial stage, LMW adiponectin is increased in urine, with urinary concentrations of MMW and HMW adiponectin increasing as the disorder progresses. Thus, urinary adiponectin may be useful as a surrogate marker for diabetic nephropathy risk.
Footnotes
Acknowledgments
The authors thank the following laboratory researchers: Dr. Asako Umehara (Naruto Hospital), Ms. Naoki Kanematsu (Hayashi Hospital) and Yumiko Hisabae (Tokushima Bunri University). The authors also thank Prof. Hiroshi Shiota (Tokushima University) for participating in the recruitment of patients and discussion of the research.
Declaration of conflicting interests
The author(s) declare no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was supported in part by Grant-in-Aids for Scientific Research (C) from the Ministry of Education, Culture, Sports, Science and Technology, Japan.
Ethical approval
The study protocol was approved by the ethical committees of Tokushima Bunri University (No. 24) and Tokushima University (No. 259); all participants gave written informed consent.
Guarantor
S. Hashida.
Contributorship
MY contributed to the study concept and design, data collection and follow-up, data research, statistical analyses, and writing of the manuscript. YH contributed to the data collection and discussion. S. Hayashi contributed to the sample collection and discussion. S. Hashida contributed to the study concept and design, data research, discussion, and review and editing of the manuscript. All authors reviewed and edited the manuscript and approved the final version of the manuscript.
