Abstract
Background
Direct oral anticoagulant dabigatran was first introduced as a fixed-dose drug without routine coagulation monitoring, but current recommendations suggest that diluted thrombin time can be used to estimate plasma drug level. The aim of this study was to assess a diluted thrombin time assay based on the same thrombin reagent already used for traditional thrombin time measurements that reliably measure low to intermediate plasma dabigatran levels.
Methods
We included 44 patients with atrial fibrillation who started treatment with dabigatran 150 mg (23 patients) or 110 mg (21 patients) twice a day. Blood samples were collected at baseline (no dabigatran) and 2–4 weeks after the beginning of dabigatran therapy at trough and at peak. Plasma dabigatran levels were measured with diluted thrombin time and compared to liquid chromatography with tandem mass spectrometry as the reference method. The performance of the diluted thrombin time was compared to Hemoclot® Thrombin Inhibitor and Ecarin Chromogenic Assay.
Results
In
Conclusion
We conclude that the diluted thrombin time assay presented in this study reliably detects dabigatran and that it is superior to the Hemoclot® Thrombin Inhibitor assay in the low to intermediate range.
Introduction
Dabigatran is a direct thrombin inhibitor developed for the prevention and treatment of patients with atrial fibrillation and venous thromboembolic disease. Oral administration of the prodrug dabigatran etexilate produces a predictable pharmacodynamic effect; therefore, fixed-dosing without routine coagulation monitoring or dose adjustment is recommended. However, plasma concentrations of the drug vary widely across patient population, depending on absorption, renal function and other patient characteristics.
1
Therefore, in certain clinical situations such as bleeding or thromboembolic event, renal failure, potential overdose and major surgery, measurements of the dabigatran effect may be desirable and even important.
2
The reference method for dabigatran concentration measurement is liquid chromatography with tandem mass spectrometry (LC-MS/MS).
3
However, this method is only available in specialized centres and usually only during weekdays. Therefore, coagulation assays that estimate plasma dabigatran concentrations from its anticoagulant effect are currently used.3,4 Routine coagulation assays such as activated partial thromboplastin time (aPTT) and prothrombin time (PT) were shown to be inadequate for measuring anticoagulant effect of dabigatran, while thrombin time and ecarin clotting time showed linear and steep dose–response relationship in an
Patients and methods
Patients and blood sampling
We included 44 patients with atrial fibrillation who started treatment with dabigatran 150 mg (23 patients) or 110 mg (21 patients) twice a day at the Anticoagulation clinic (University Medical Centre, Ljubljana, Slovenia). A lower dose of dabigatran was prescribed to patients with moderately impaired renal function (estimated glomerular filtration rate 30–50 mL/min), elderly patients (>75 years), those with a history of major bleeding, those on amiodarone, verapamil or antiplatelet drugs and to frail patients. All patients signed a consent form agreeing to participate in the study. The study was approved by the Medical Ethical Committee of the Slovenian Ministry of Health.
Blood was collected from each patient at baseline (no dabigatran) and then again 2–4 weeks after dabigatran therapy initiation at trough (12 ± 1 h after the previous dabigatran ingestion) and at peak (120 ± 5 min after the last dabigatran ingestion) into 4.5 mL vacuum tubes containing 0.11 mol/L sodium citrate (9:1 v/v) (Becton Dickinson, Vacutainer System Europe, Heidelberg, Germany). Plasma was prepared with 30 min centrifugation at 2000
Methods
The in-house dTT was performed on the CS2100i coagulation analyzer (Sysmex Europe GmbH, Norderstedt, Germany) with the following protocol: 10
dTT results of
dTT method validation and statistical analysis
Linearity of the calibration curve was tested with linear regression and reported as R2. R2 was also calculated between the tested methods (dTT, HTI and ECA-T) and the LC-MS/MS. Limit of Blank (LoB) was reported as the highest result of a blank sample (NPP), lower limit of detection (LoD) was calculated as an average of + 3 standard deviations and Limit of Quantification (LoQ) as an average of + 10 standard deviations of NPP.
9
Within-run and between-run imprecision and accuracy of the dTT were determined by performing duplicate low and high control measurements in 20 consecutive days and reported as coefficient of variation (CV). Accuracy was calculated as the difference between the target and the average measured value divided by the target value (×100%). For statistical analysis all negative dabigatran values obtained from
Results
dTT calibration curve was linear, with R2 of 0.9996 (Figure 1). There was no deviation from linearity up to 920 Average ± SD of three dTT calibration curves obtained four months apart (28.0 ± 0.3 s at 0 The within- and between-run imprecision and accuracy of dTT.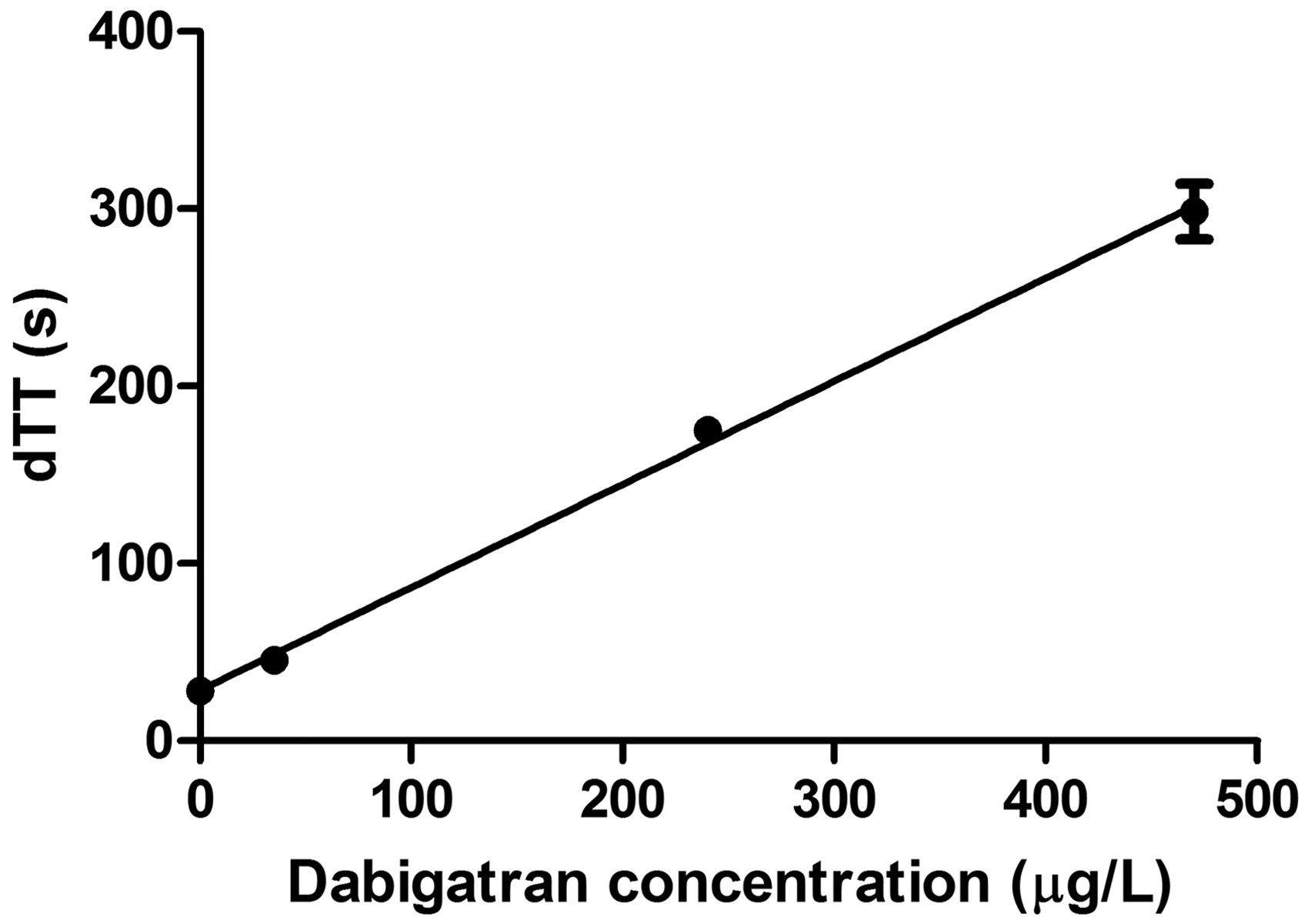

When Comparison of dTT, HTI and ECA-T assays with the LC-MS/MS (reference method) for the whole measuring range (first column), low to intermediate range (≤100 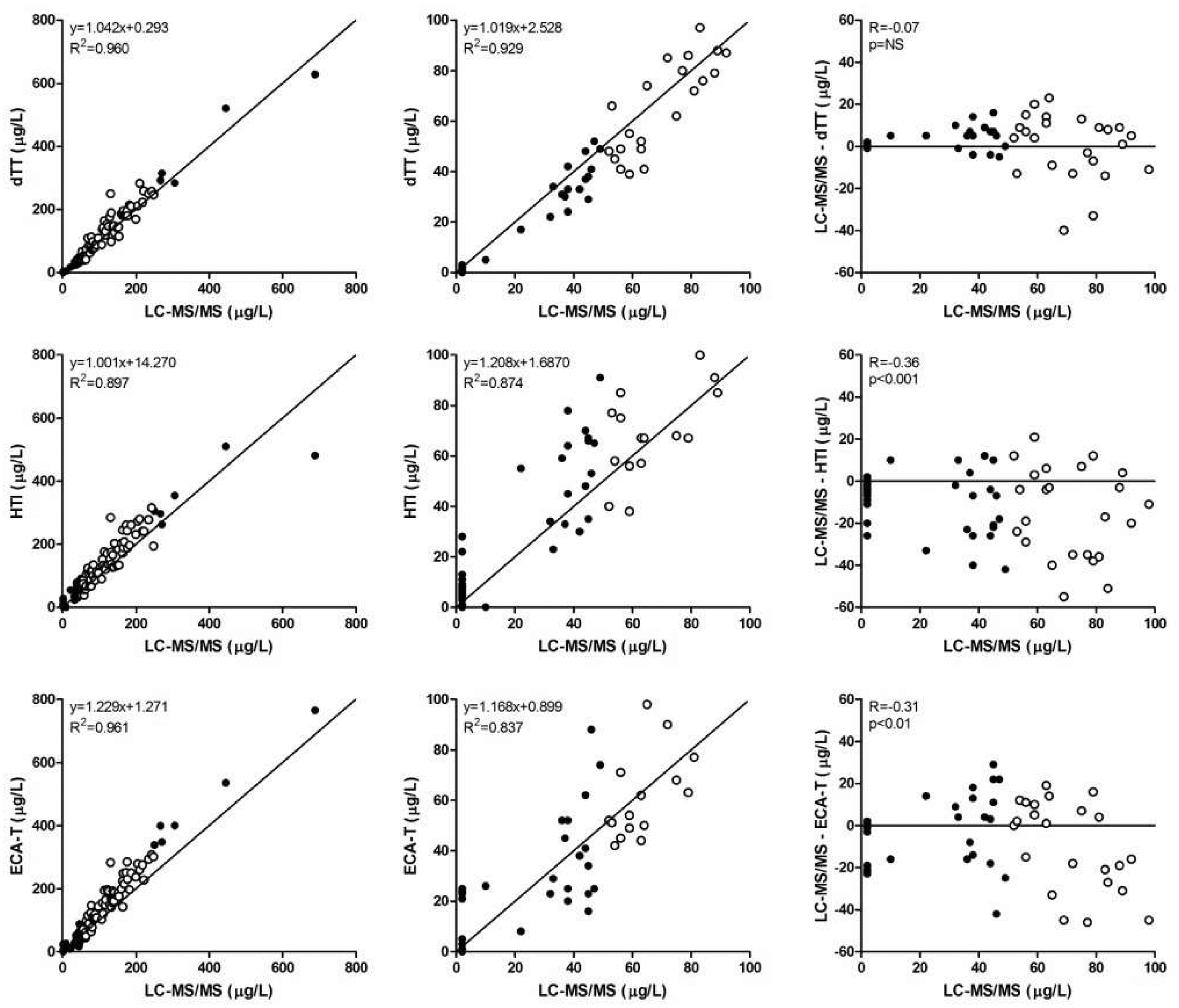
Dabigatran concentrations at baseline (no dabigatran), trough and peak measured with LC-MS/MS (reference method), dTT, HTI and ECA-T.
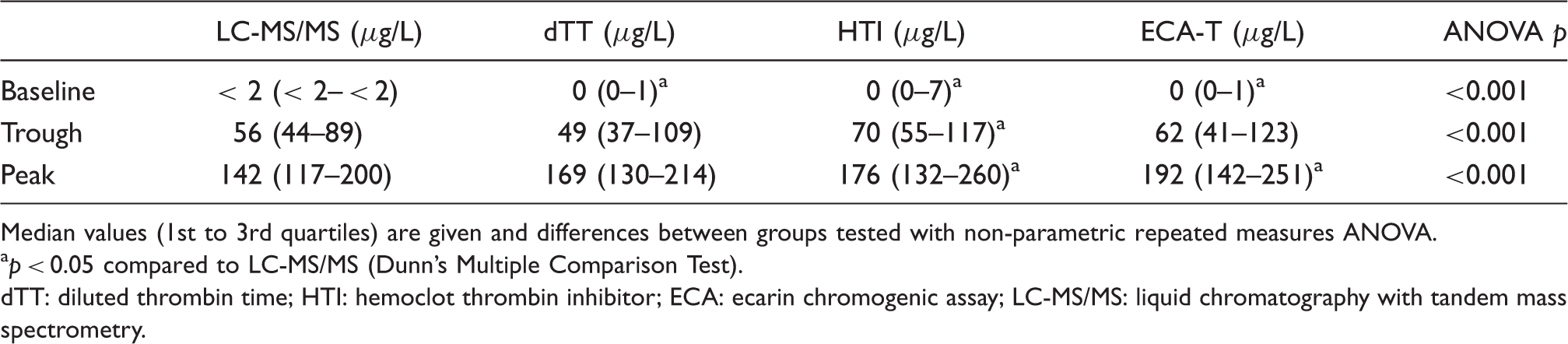
Median values (1st to 3rd quartiles) are given and differences between groups tested with non-parametric repeated measures ANOVA.
dTT: diluted thrombin time; HTI: hemoclot thrombin inhibitor; ECA: ecarin chromogenic assay; LC-MS/MS: liquid chromatography with tandem mass spectrometry.
Discussion
In this study, a dTT assay that utilizes the same thrombin reagent used in traditional thrombin time measurements is presented. The analysed dTT assay was accurate and reproducible, particularly for detecting plasma dabigatran concentrations below 100
The dTT assay presented in this study showed a wide range of linearity, similar to the traditional thrombin time.
1
The within- and between-run imprecision (all CVs < 10%) and the accuracy (<3%) of the dTT were satisfactory and comparable to previously reported imprecisions of HTI
10
and ECA-T.
11
In
At baseline, trough and peak plasma samples, we have shown that HTI significantly overestimated trough and peak levels, ECA-T significantly overestimated peak, but not trough levels, while no significant deviation was noted for dTT at either trough or peak. It has previously been proven that HTI reflects free dabigatran levels, while ECA-T reflects total dabigatran, the latter being on average 20% higher due to pharmacologically active dabigatran acylglucuronides. 15 Since the concentration of free dabigatran was highly predictable from the total dabigatran and was easier to measure (no need for alkaline hydrolysis), it was concluded that free dabigatran concentrations provide the relevant information needed for the monitoring of dabigatran treatment. 15 In both the previous 15 and the present study, the regression slopes between free dabigatran and HTI were close to 1 and for ECA-T close to 1.2. We therefore expected similar trough and peak HTI values reflecting free dabigatran levels measured with the LC-MS/MS and 20% higher ECA-T values reflecting total dabigatran. However, it was not so in the low to intermediate range. The regression slope of HTI in this range rose from 1.0 to 1.2, and average trough HTI was overestimated compared to LC-MS/MS. For ECA-T, an overestimation of around 20% was expected and confirmed for peak levels, but in the low to intermediate range the overestimation was lower than expected (10%, Table 2). The discrepancies found require further elucidation, but they may lie in the imprecision of the HTI and ECA-T. For dTT, however, both the regression slope of 1 (whole and low to intermediate range) and comparable trough and peak values confirmed that dTT reliably detects free dabigatran levels. At baseline all functional assays significantly differed from LC-MS/MS measurements, but these differences can be attributed to mathematical conversions of very low or negative values needed for statistical analysis.
This study has some limitations. Firstly, only one combination of reagent and analyser at a single site has been tested. The same protocol may not be applicable to other analysers. Secondly, we used NPP prepared in-house. This may not be possible for many laboratories. However, commercially available NPP can be used in the dTT assay, yielding slightly longer coagulation times (data not shown).
This study also has strengths. We performed our study on real-world patients, not on healthy individuals or
In conclusion, the results of our study showed high accuracy and good reproducibility with wide range of linearity of the dTT assay. It was shown to be superior to HTI for measuring trough dabigatran levels.
Footnotes
Declaration of conflicting interests
JPA has received speaker’s honoraria from Siemens and Stago. Other authors declare no conflict of interest related to this work.
Funding
The study was supported by the Slovenian Research Agency (Grant No. P3-0308).
Ethical approval
Slovene National Medical Ethics Committee approved this study (REC number 123/04/13).
Guarantor
MBM.
Contributorship
MBM, NV and AM researched literature and conceived the study. MBM, PŠ and AM developed protocol and gained ethical approval. PŠ, NV and AM recruited patients and analysed patient data. MBM, REM, PM and JPA analysed samples and the data. MBM wrote the first draft of the manuscript. All authors reviewed and edited the manuscript and approved the final version of the manuscript.
