Abstract
A middle-aged woman with neutropenia and ataxia was found to have raised plasma zinc and profoundly low plasma copper concentrations. When found that she had been prescribed 135 mg zinc/day for seven years, a diagnosis of zinc-induced copper deficiency was made. After the zinc prescription was stopped, her copper and zinc concentrations and neutropenia normalized but she only had partial improvement in neurological status. The diagnosis of zinc-induced copper deficiency can be facilitated by the laboratory through measurement of plasma zinc concentration in patients with a low plasma copper concentration.
Introduction
The body requires small amounts of copper (1.0 mg/day) 1 and since copper is plentiful in the diet, copper deficiency is rare. However, it occurs occasionally either from inadequate provision in patients receiving enteral and parenteral nutrition but more commonly from malabsorption of copper. Copper absorption mainly takes place in the jejunum and duodenum and so copper deficiency can occur in untreated coeliac disease and following gastric/duodenal surgery. An additional important cause of copper deficiency is excessive oral intake of zinc which interferes with copper absorption. 2 High oral zinc intake stimulates the intestinal enterocyte production of metallothionein which binds zinc making it less available for export from the cell so controlling zinc homeostasis. 3 However, copper has a greater affinity for metallothionein 4 and if high zinc ingestion is maintained for long enough (usually several months or years), copper deficiency may result as enterocytes are sloughed off into the gut lumen and lost in the faeces. With long-term ingestion of excess zinc, the body’s stores of copper are depleted with the ultimate result of deficiency.
Early pathological findings of copper deficiency are usually haematological with anaemia and neutropenia. These changes are reversible and respond well to stopping the source of excess zinc and commencing copper supplementation.5,6 In unrecognized and untreated cases, myelodysplasia may ensue as well as neurological sequelae such as ataxia and peripheral neuropathy.5,6 These may be stabilized with treatment but are not usually reversible. 6
Case history
A woman in her mid-50s presented with stocking loss of sensation, neuropathic pain and poor balance and was found to be neutropenic on routine blood testing. She had a history of Roux-en-Y anastomosis 7 years previously and following a postoperative plasma zinc result of 9.9 μmol/L (reference interval 10–18 μmol/L; Varian Vista inductively coupled plasma optical emission spectrometer, Agilent Technologies, Santa Clara, US) had been prescribed three Solvazinc tablets (Galen Ltd, Craigavon, UK) per day each containing 45 mg of elemental zinc. Over a period of several months, her neurological status deteriorated and she became unable to walk. Plasma zinc measured at this time was 21 μmol/L, and the plasma copper concentration (which had not been specifically requested but was measured simultaneously by inductively coupled plasma technology) was found to be 1.0 μmol/L (reference interval 10.7–25 μmol/L). The biochemist contacted the requesting clinician, a presumptive diagnosis of zinc-induced copper deficiency (ZICD) was made and her zinc prescription was stopped. Further, blood samples collected 7 and 10 weeks after discontinuation of zinc showed plasma copper concentrations of 7 μmol/L and 15 μmol/L, respectively, with plasma zinc falling to 17 and 18 μmol/L, respectively. This resulted in resolution of the neutropenia and although her neurological symptoms partially improved, she remained wheelchair-bound.
Discussion
The daily zinc requirement in humans is 5.5 to 9.5 mg/day for male and 4 to 7 mg/day for female. 1 Different agencies and organizations have attempted to calculate the upper tolerable daily intake level but it is clear from the large variation between estimates, from 25 mg/day (by the UK Food Standards Agency 1 and European Commission 7 ) to 45 mg/day (World Health Organization 8 ) and 60 mg/day (US Environment Protection Agency 9 ), that it is difficult to assign a safe level with any degree of accuracy. The lowest reported daily dose of zinc associated with ZICD is 121 mg/day 10 ; however, negative copper balance is induced in Wilson’s disease patients with zinc doses of 75 mg/day 11 and in a further study, a raised intake of oral zinc of only 18.5 mg/day over a 2-week period resulted in negative copper balance. 12
The first described case of copper deficiency resulting from over-treatment with zinc was published in 1978. 13 Since then it has been reported on several occasions. 14 It has even been described in patients with Wilson’s disease treated with high doses of zinc. 15 More unusually, the source of zinc may be from ingested coins.
In 2003, Hedera et al. 16 described 11 patients with clinical symptoms characteristic of copper deficiency and who had high concentrations of plasma zinc. These patients were not taking zinc supplements, and it was hypothesized at the time that they may have a genetic disorder characterized by zinc overload of unknown origin. However, 2 years later, Willis et al. 15 described a patient with copper deficiency associated with excessive use of dental fixatives, and Nations et al. 17 subsequently described more such patients. These publications prompted Hedera et al. 18 to review their original 11 patients and they found that in all cases, dental fixatives were applied in excess. The zinc content of the three main brands of dental fixative, Poligrip Original, Poligrip Polyseal (GlaxoSmithKline, Brentford, UK) and Fixodent (Procter and Gamble, Weybridge, UK) is high; 34 mg/g, 27.5 mg/g and 17 mg/g, respectively. 17 (In 2010, a zinc-free formulation of Poligrip was introduced.) Using these data and the rate of usage of tubes of dental fixative, Hedera et al. 18 calculated the average daily intakes of zinc in their 11 patients and found that they ranged from 350 mg to 1700 mg. Since then, several publications have reported similar cases of copper deficiency caused by zinc-containing dental fixatives.14,19 Application of zinc-containing dental fixatives is usually safe but the characteristic feature of affected patients is that they apply unusually large amounts of fixative, usually because of poorly fitting dentures or, in our experience, consumption of large amounts of hot beverages.
Clinical awareness of copper deficiency is often lacking and so the diagnosis is frequently missed or significantly delayed. For example, in two retrospective studies, 34 of 40 (85%) and 12 of 13 (92%) of patients developed neurological symptoms by the time of diagnosis.5,14 In the same two studies and a third report, 20 the median duration of symptoms from initial presentation to diagnosis was 13 months, 12 months and 21 months, respectively. It is also likely that copper deficiency is undiagnosed in patients prescribed high doses of zinc. 21
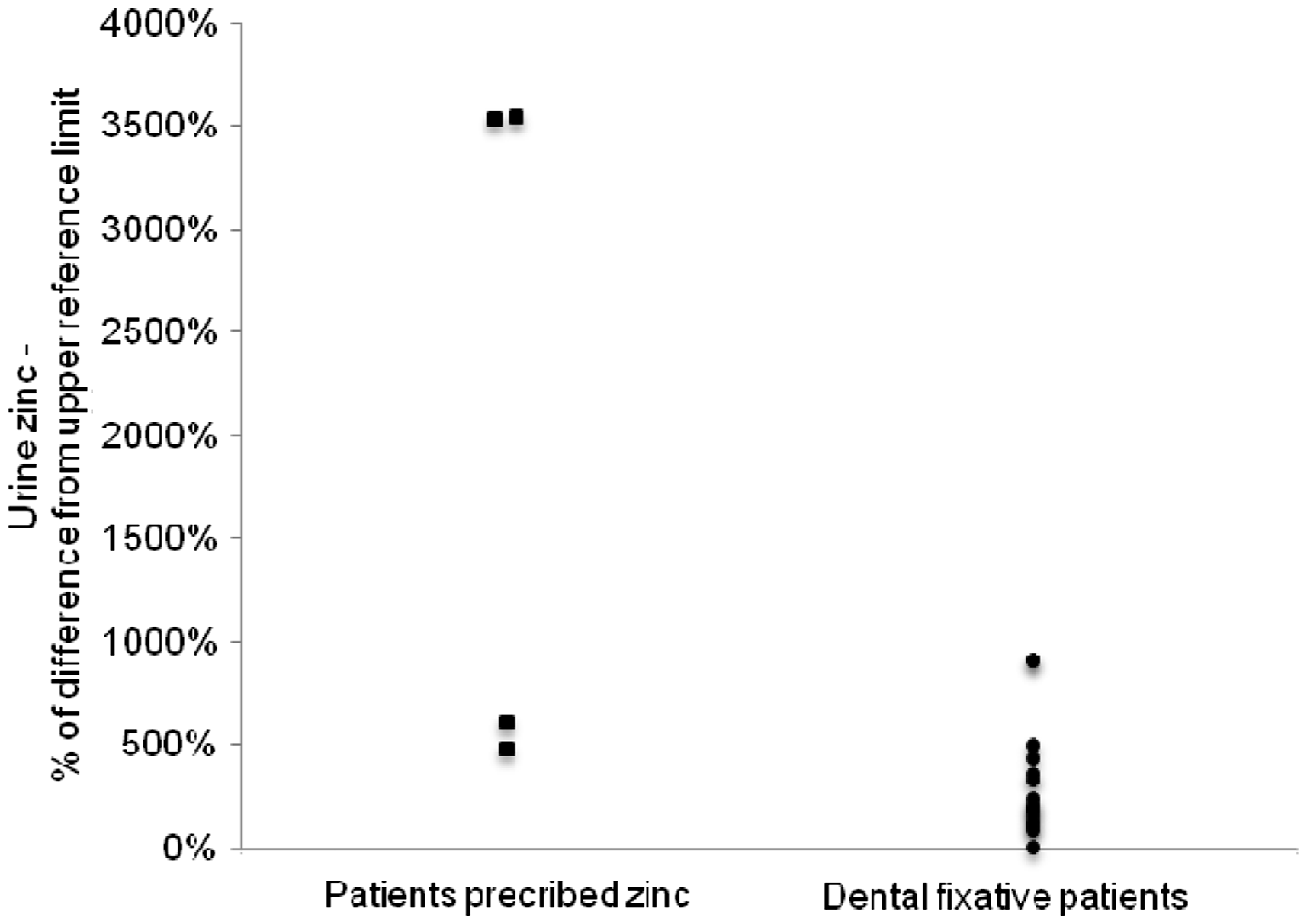
In published reports of ZICD, plasma zinc concentrations were high in 60 of 65 cases (92%; Figure 1). (References for the data from which the graphs are derived are included in the supplementary material.) It is relevant to note the large variation of the upper reference limits quoted in these reports, from 15.4 to 24.5 μmol/L (median = 18.0 μmol/L). For this reason, Figure 1 shows the percentage difference from the upper reference limit used in the reporting laboratory. Fewer publications document urine zinc concentrations in ZICD but in all 17 cases in which it was measured, the 24-h output exceeded the upper reference limit (Figure 2). Again the upper reference limits varied considerably between reports from 7.6 to 18.4 μmol/L (median = 9.2 μmol/L).
Plasma zinc (expressed as percentage difference from the upper reference limit) in published cases of zinc-induced copper deficiency. Urine zinc (expressed as percentage difference from the upper reference limit) in published cases of zinc-induced copper deficiency.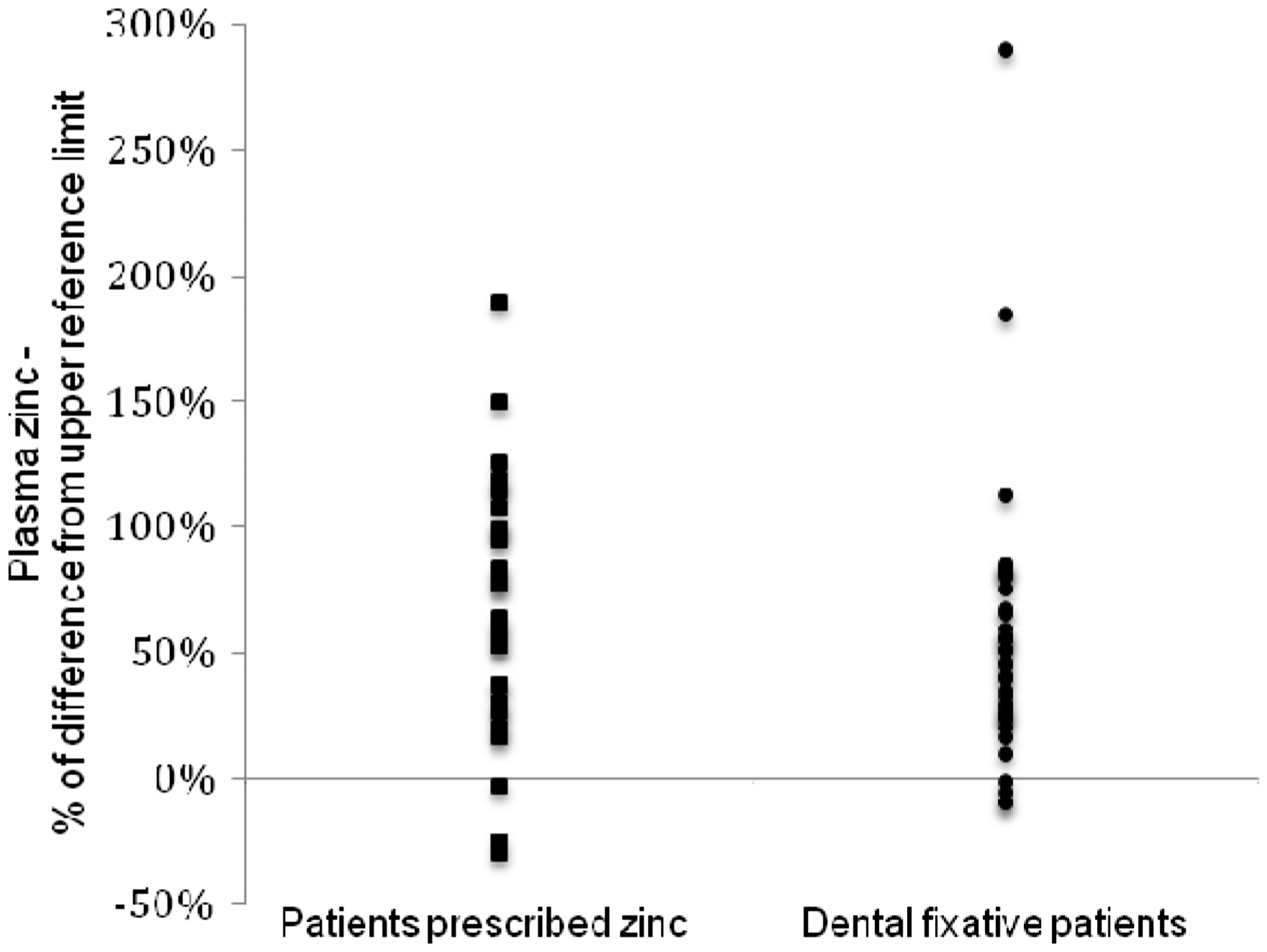
Recommendation for early diagnosis
The finding of a low plasma copper in conjunction with a raised plasma zinc concentration offers a potential opportunity for the clinical biochemistry laboratory to facilitate earlier diagnosis of ZICD. Since 1992, our laboratory has measured requests for plasma copper and zinc simultaneously by inductively coupled plasma technology, initially using optical emission spectrometry and since 2006, mass spectrometry. If a plasma copper or plasma zinc analysis is requested, we measure both and only report the analyte requested. In 2004, we introduced a policy that on finding a plasma copper ≤6.0 μmol/L we routinely check the plasma zinc result; similarly, plasma copper is checked if the zinc concentration is elevated (>18 μmol/L). By this means, the reporting biochemist was able to alert the clinician to the diagnosis of ZICD in 7 of 16 (44%) cases. 14
This case history highlights a typical example of ZICD. The initial diagnosis of zinc deficiency based on a plasma zinc of 9.9 μmol/L may have been inappropriate because the sample was taken postoperatively. Plasma zinc falls by up to 40% following a systemic inflammatory response in patients admitted for elective knee surgery 22 and relatively modest increases in C-reactive protein of over 20 mg/L are associated with statistically significant decreases in plasma zinc. 23 Such misdiagnoses of zinc deficiency have been described before and are more common than might be expected; in a retrospective survey, around 65% of patients were probably inappropriately prescribed zinc as a result of a low plasma zinc concentration which was secondary to the systemic inflammatory response rather than zinc deficiency. 21 Consequently, in an attempt to avoid an inappropriate diagnosis of zinc deficiency, the interpretive protocol in our laboratory includes a warning that plasma zinc concentration may fall independently of zinc status in patients with a systemic inflammatory response (CRP > 20 mg/L). This case report also demonstrates that even after excess zinc ingestion is stopped, copper absorption may remain low for several weeks until metallothionein production is destimulated and enterocytes are lost into the gut lumen. For this reason, it may be necessary to replace copper intravenously, especially if the patient is severely copper deficient, in order to prevent the development or worsening of neurological pathology.
More recently, our laboratory policy has been slightly modified so now the combination of a plasma copper concentration of ≤7.0 μmol/L, rather than ≤6 μmol/L, and a plasma zinc >18 μmol/L is used as the trigger to contact the clinician. This change was introduced following the finding of ZICD in a patient whose diagnosis might have been expedited had an initial low plasma copper of 7 μmol/L been acted on. 19 It is also useful to request a 24-h urine collection for copper and zinc; a urine copper of ≤0.6 μmol/24 h helps to rule out Wilson’s disease 24 and a urine zinc less than 19 μmol/24 h helps to exclude ZICD when the plasma zinc is normal, a finding in around 8% of published cases. In summary, by following this approach, the clinical biochemistry laboratory has an opportunity to facilitate the clinician in expediting the diagnosis of ZICD. This simple intervention may potentially allow earlier instigation of treatment and so prevent the development of irreversible neurological sequelae.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Ethical approval
The North Glasgow Hospitals Trust Ethics Group confirmed that ethical approval was not required. Written consent from the case history patient has been obtained.
Guarantor
AD.
Contributorship
AD and GG reviewed the relevant literature and AD wrote the manuscript. LW provided clinical overview. All authors checked the final manuscript.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
