Abstract
This review focuses on the effect that cross-gender sex steroid therapy has on metabolic and hormonal parameters. There is an emphasis on those changes that result in significant clinical effects such as the positive effects of the development of secondary sexual characteristics and negative effects such as haemostatic effects and thromboembolism in transwomen or dyslipidaemia in transmen. There is also a description of the current hormonal regimens used at the largest UK gender identity clinic. The overall safety of these treatments in the context of long-term outcome data is reviewed.
Keywords
Introduction
Gender dysphoria is not an uncommon condition. Present estimates of its incidence vary between 1:7440
1
and 1:30,000 for natal males and 1:31,153 to 1:10,000 for natal females. The condition is defined as: a marked difference between the individual’s expressed/experienced gender and the gender others would assign him or her, and it must continue for at least six months. In children, the desire to be of the other gender must be present and verbalized. This condition causes clinically significant distress or impairment in social, occupational, or other important areas of functioning.
2
The International Classification of Diseases 10 (ICD-10) published by the World Health Organization extends the definition to include the desire of individuals to alter their somatic appearance to be more congruent with their internal sense of gender. To make this diagnosis, the condition needs to have been present for at least two years and not a symptom of a major psychiatric disorder. The diagnostic phase of assessment is performed by a mental health practitioner but the delivery of care in the UK is in the context of a multidisciplinary team including psychiatrists, psychologists, endocrinologists and surgeons.
The nomenclature of gender identity in this area is constantly evolving but the present preferred terms are transwoman (natal male transitioning to female), transman (natal female transitioning to male) and gender queer or neuter for those who see themselves as not part of the traditional gender binary.
This review will look at the effects of cross-sex hormone biochemical parameters and review the available longer term outcome data in transpeople.
Hormone regimens used
There has been a multitude of hormone regimens used and therefore assessing the effects of cross-sex hormone therapy on biochemical parameters in transpeople has been difficult.3,4 This is especially so for transwomen where a much broader range of oestrogen preparations is available, compared with testosterone therapy for transmen. The situation is compounded by the fact that transwomen usually need testosterone suppression either in the form of a gonadotropin releasing hormone (GnRH) analogue to suppress testicular testosterone production or an anti-androgen to reverse the effects of circulating testosterone. There have been recent guidelines to good practice published by the Joint Royal colleges which provide a guide to the commonly used hormonal regimens. 5
This review examines what is known about the metabolic effects of cross- sex hormone therapy in humans and examines the outcomes and side-effects of these treatments.
Physical effects of hormone regimens
The study by Meyer et al. was the first to demonstrate the course of the physical changes that occur during cross- sex hormone therapy. 6 The physical changes mimic natal puberty in that the pubertal development of breast was complete by 18 to 24 months after initiation of hormone therapy in transwomen but virilization in transmen was slower taking four to five years to complete in transmen, as it does in natal males.
Transwomen
Physical effects
The physical effects of oestrogen treatment in those born with a male phenotype have been well documented. The aim of the treatment is to give an escalating dose of oestrogen until upper follicular phase oestradiol concentrations are achieved over a period of six to nine months. 7 Breast development begins in about two to three months after the initiation of treatment and the maximum effect of oestrogen on breast development is not seen until two years of oestrogen therapy. 8 The average breast development is 14.5 ± 1.2 cm; this equates to a B cup brassier size.
Despite adequate oestrogen exposure, 60% of transwomen progress to breast augmentation surgery. 9 In the past, use of large doses of oestrogen early in breast development induction led to rapid growth of the breast but premature termination of ductal branching, resulting in breast development that is arrested at Tanner stage III/IV giving small conical breasts. 7 This problem is reduced with modern hormone regimens but we must bear in mind that the programming for breast development each individual has is genetic and this determines the breast response to oestrogen stimulation. The regimen used does not appear to influence the final breast outcome. 10 There are however differences in anatomy between the male and female thorax; therefore, despite maximal response to oestrogen therapy, more than half the patients require breast augmentation so that the breast contour appears proportionate for their larger body frame. 9
With oestrogen treatment the skin texture becomes finer, and there is a reduction in the growth of facial hair. Both facial and truncal hair shaft diameters decrease and this effect is maximal after four months of treatment. 11 Hormone therapy itself is not adequate to control facial hair growth and local measures such as electrolysis, waxing, shaving, sugaring or laser therapy are needed to reduce the appearance of facial hair and facilitate female presentation.
Body composition alters such that there is an average 3.8 kg weight gain, with a 38% increase in subcutaneous fat deposits. 12 This fat is mainly centred on the hips and buttocks giving a more female fat distribution. There is a concomitant decrease in muscle mass. 12
Oestrogen therapy in transwomen leads to a reduction in libido and erectile function. In transwomen, this is often seen as a positive outcome of treatment. Due to gonadotropin suppression there is a decrease in testicular volume. 8 Sex steroid therapy also has powerful effects on cognition. In peri- and postmenopausal women, oestrogen therapy has mood-modulating effects, as it increases feelings of well-being and decreases depression scores.13,14 Similarly, in transwomen, oestrogen therapy produces a positive effect on mood.15,16 Anecdotally, many patients report an increased feeling of femininity on oestrogen therapy and a calmer mood in reaction to a reduction in their feelings of being male.
Endocrine and metabolic effects
The administration of exogenous sex-steroids results in inhibition of gonadotropin production. 17 This results in inhibition of erectile function and reduced spermatogenesis17,18; there is also shrinkage of the testes. It is therefore recommended that the impact of therapy on fertility be discussed with the client before hormonal intervention is commenced and also that the client be offered gamete storage before treatment.4,5,19
The metabolic effects of oestrogen therapy are centred around alteration in liver function and the effects of oestrogen on lipid parameters. There appears to be a decrease in hepatic lipase activity by 64% and of lipoprotein lipase by 23%. 12
The effects of oestrogen therapy on cardiovascular risk in transpeople have been reviewed elsewhere. There appears to be no effect on either total cholesterol or high-density lipoprotein (HDL) cholesterol in a meta-analysis of the studies published. 20 Triglyceride concentrations do, however, increase by 0.26 mmol/L. These results are surprising as we might expect oestrogen therapy to increase concentrations of HDL cholesterol and lower those of total cholesterol.
We know from long-term outcome studies that the use of ethinylestradiol in transwomen is associated with increased cardiovascular risk and this risk is not seen in past users of either ethinylestradiol or current users of oestradiol preparations and in past users of conjugated equine oestrogen (CEE). 21 The problem with this meta-analysis is that only 9% of the clients were treated with oestradiol, whereas the majority were treated with either ethinylestradiol or CEE and only 7% were receiving oestrogens and GnRH analogues. This is an area where further research is needed to characterize the effects of oestradiol preparations rather than ethinylestradiol or CEE on lipid parameters.
In natal female HRT, also there is a move away from CEE in light of the Women’s Health Initiative (WHI) 22 and the Heart and Estrogen/Progestin Replacement Study (HERS) 23 clinical trials with a move towards transdermal oestradiol preparations as a safer alternative for HRT. 24
Oestrogen treatment can result in an obstructive pattern of liver dysfunction, especially with orally delivered oestrogen.25,26 If the liver function disturbance is mild (<3 times the upper limit of normal), using topical oestrogen or dose alteration is usually all that is needed. 7 In the largest trial so far published, 4.3% of subjects had a transient rise in liver enzymes but in all cases this was less than 2.5 times the upper limit of normal and none required cessation of the treatment. However, other groups have reported an incidence of abnormal liver function of 2%, which was the same as that observed in controls. 27 Oestrogen treatment in transwomen increases the incidence of gallstones with a standardized incidence ratio of 5.26 (95% confidence interval 1.09 to 15.38). 28
The physiological response of the lactotroph to high oestrogen concentrations is hyperplasia and hypersecretion of prolactin. In normal physiology, this is only seen in pregnancy suggesting that very high oestrogen concentrations are required for this response. The incidence of significant hyperprolactinaemia has been reported to be up to 15% in transwomen treated with oestrogen. 29 In a larger series of patients, the incidence of prolactin concentrations greater than 1000 mU/L was reported to be 3.7 to 7.2% per treatment year. 30 In these studies, however, oestrogen was used in combination with cyproterone acetate, which may increase the incidence of hyperprolactinaemia.28,29
A more recent study suggested, however, that with newer regimens using oestradiol and GnRH analogues, the incidence of hyperprolactinaemia is 2.3%. 27 The incidence of hyperprolactinaemia appears to be related to the dose of oestrogen used, and if the plasma prolactin concentration becomes elevated, then reduction of the oestrogen dose usually resolves the hyperprolactinaemia. 28
Despite the occurrence of hyperprolactinaemia, there have only been five case reports of prolactinomas in transwomen and none have needed withdrawal of oestrogen treatment.31–34 The development of prolactinomas seems to be associated with self-medication using high-dose oestrogen preparations. Indeed, one of the patients who developed a prolactinoma had self-administered intramuscular oestrogen benzoate in addition to her prescribed oestrogen therapy. 33
Haemostatic effects of oestrogen therapy
There has been a great alteration in the reported incidence of venous thromboembolism (VTE) in transwomen with changes in hormonal practice over time. The original study of Asscheman et al. reported a 45-fold increased risk of VTE when ethinylestradiol and cyproterone acetate were used for the feminization of transwomen, with a clear age-related bias where those over 40 years old had a DVT rate of 12% and those under 40, 2.1%. 35 This led to a change in practice at the clinic to the use of transdermal preparations in those over 40 years old and consequently a drop in the VTE incidence. In their subsequent study, they reported a VTE incidence of 2.6% in oestrogen treated transwomen, which is a risk of DVT that is 20 times that of the general Dutch population. 28 The majority of these incidents occurs during the first two years of treatment. There is, however, an ongoing risk of 0.4% per year which continues. 28
The risk of DVT is affected by several factors such as the oestrogen type, the route of delivery and the co-administered androgen-lowering medications. It has been demonstrated that ethinylestradiol alters the plasma concentrations of protein S, C and prothrombin, which results in a procoagulant haemostatic profile in transwomen. Oral ethinylestradiol 100 μg daily in combination with cyproterone acetate 100 mg/day caused a significant increase in activated protein C (APC) resistance (nAPCsr 1.2 + 0.8 to 4.1 + 1.1); however, transdermal oestradiol 100 μg× 2 per week and cyproterone acetate 100 mg or cyproterone acetate monotherapy had a minor effect (1.3 ± 0.6 to 2.0 ± 2.0 and 1.4 ± 0.6 to 1.8 ± 0.9, respectively). Furthermore, ethinylestradiol and cyproterone acetate also decreased Protein S concentrations, an inhibitor of coagulation. 36 These changes were not observed when either transdermal oestrogen or oral oestrogen valerate was used. 36 The assumption from this study was that the route of administration of oestrogen was important in reducing VTE risk and so this clinic began a policy of using transdermal oestrogen after the age of 45 years, and they have noticed a decrease in the incidence of DVT in their clinic population from a 40-fold to a 20-fold increased risk. 28
Recently, however, studies have challenged this view and suggest it is the type of oestrogen used and not the route which may be more important. A study from my own group demonstrated that the incidence of VTE in a population of 330 transwomen was 0.6% for those treated with oral oestradiol in doses of up to 10 mg daily but that the incidence of VTE was eight-fold higher in those treated with CEE strongly suggesting that it is the type of oestrogen used and not the route of delivery that is important in determining the VTE risk of oestrogen therapy in transwomen. 27 It is also being recognized as an important issue in contraceptive practice where the prothrombotic effect of ethinylestradiol has been ascribed to its molecular structure rather than to the first-pass liver effect again suggesting that it is the chemical structure rather than route that is important in this effect. 37
Another important issue is the interaction between oestrogen therapy, body weight and smoking status. We know from the combined contraceptive pill studies that the incidence of VTE is increased in smokers by approximately two-fold, 38 and in obesity this risk increases to nine-fold. 38 An important feature of the protocol at the largest UK clinics is that the client must stop smoking in order to receive high-dose oestrogen, 39 which may act to reduce the VTE risk of this patient group.
Osteoporosis
In transwomen, oestrogen therapy appears to maintain bone mineralization despite testosterone withdrawal.40–43 There is an initial increase in bone mineral density (BMD) on commencing treatment which settles to baseline values by one year in one report. 42 One group, however, has shown a decrease in trabecular bone area and mineral content in transwomen 44 ; this was associated with a lower amount of exercise performance in this group. It is of note that the bone mass is inversely proportional to gonadotropin concentration suggesting that under-treatment with sex steroids (as suggested by high gonadotropin concentrations) may explain the differences in the findings of this study compared with other published reports. 42
Cancer risks
The incidence of breast cancer with standard HRT in natal females is estimated at an excess of 3.2/1000 aged 50 to 59 years and 4/1000 aged 60 to 69. 45 This is based on large population-based studies. We know from both the WHI 22 and HERS 23 that the inclusion of progesterone in the HRT regimen increases this risk. There have only been five case reports of breast tumours occurring in treated transsexual patients, suggesting that the risk of breast cancer secondary to feminizing hormone therapy is very low.7,46
In the literature, there is only one recent study examining this issue in the transsexual population. This large cohort study (n = 2307) reported the incidence of breast cancer as 4.1 per 100,000 person-years (95% CI = 0.8 to 13.0). This rate is the same as the background rate of breast cancer in males. These data suggest that the oestrogen therapy does not increase breast cancer risk in transwomen. 47 Some units do use progestins routinely as part of their hormonal regimen and have not reported excess breast cancer incidence in comparison with other transsexual populations; however, there are no studies published examining the effects of progestins on cancer risks in transwomen.
In view of the fact that progestins are not involved in normal pubertal breast development, are of no proven benefit in breast development induction in transwomen, 47 and that there is evidence that they may increase breast cancer risk in natal females, their use for the feminization of transsexual women has to be seriously questioned.
The incidence of prostate cancer in transwomen is extremely low. At the present time there are only four cases reported in the world literature.48–51 In view of the high incidence of this malignancy within the male population, e.g. up to 50% by eight decades, it is difficult to attribute these cases to hormonal therapy. 52 A recent study looking at long-term follow-up of a cohort of transwomen, half of whom had 30 years of follow-up, demonstrated only one case of prostate cancer in a cohort of 303 people giving an incidence of 0.04% in this population. 50
Adjunctive therapies
Anti-androgen therapy as either cyproterone acetate or spironolactone is used by some centres but generally not in the UK. Cyproterone acetate is a progesterone derivative and is metabolized in the liver. Its use is associated with hepatic dysfunction, and although it rarely leads to discontinuation of the drug it does require regular monitoring of the liver function.53–55 More significantly for this patient group where depression is a common psychiatric co-morbidity, 27 the use of cyproterone is significantly associated with depression. 27 53 The same spectrum of side-effects occurs with the use of finasteride; both depression and liver function disturbance have been described as adverse effects of this drug,56, 57 although depression is not as prominent in the transfemale population.27 There have also been reports of the development of meningioma associated with the use of high-dose cyproterone acetate, 58 with an adjusted relative risk of 11.4 (95% CI = 4.3 to 30.8) as compared with non-users. 58
Spironolactone is a mineralocorticoid receptor antagonist that acts as an androgen receptor partial antagonist as well as an oestrogen receptor agonist. As such, in addition to blocking the androgen receptor (which is its primary purpose in this situation), it also has a significant oestrogenic action at the doses used in transwomen. It is a potassium sparing diuretic and can cause renal dysfunction and hyperkalaemia as well as liver dysfunction. Worryingly, there have been reports of spironolactone use being associated with upper gastrointestinal bleeding. 59 Importantly, its use may result in poorer breast outcome in transwomen. 27
Anti-androgens have been necessary in the past, as many patients fail to suppress their production of testosterone with oestrogen therapy. Now, we have the availability of GnRH analogues that completely suppress gonadal testosterone production. These drugs work by over-stimulating the luteinizing hormone (LH) receptors on the gonadotropin cells of the pituitary resulting in tachyphylaxis and reduced testosterone production. There has been extensive experience in using these drugs both in the treatment of prostate cancer and infertility, and they have an excellent side-effect profile. The use in the hormonal treatment of transwomen appears safe with minimal side-effects. The usual side-effects of hot flushes and tiredness are obviated by the oestrogen treatment 27 40 and bone mineralization is maintained when sex steroids are used with GnRH analogues. 40
Progesterone is used by some centres and is widely purported by self-help trans websites to improve breast development. In the large European centres, progestins are not used. A recent meta-analysis looking at breast development in transwomen found no evidence that there is any additional benefit of progesterone on breast growth over oestrogen alone. 10 Moreover, progestin in combination with oestrogen may increase the incidence of cardiovascular events and has a trend towards increasing levels of breast cancer when we look at the large-scale trials of hormone replacement in natal females.22,23 These risks were not seen in the oestrogen-only arm of these trials, suggesting that progesterone is detrimental for both cardiovascular and breast health.22,23
Fertility
In both transmen and transwomen loss of reproductive capability will accompany the use of cross-sex hormones which, although usually reversible if the person elects to stop taking the hormones, may be irreversible.4,5 Should their treatment lead to gonadectomy then there would be a permanent loss of reproductive capacity. Consequently, a discussion about the implications of this is vital in the decision-making processes concerning trans people’s transition. 19 It is recommended as an important part of the assessment and counselling process for gender transition by several of the main bodies working in this field.5,60 It is also recommended that the individual be offered gamete storage ideally before they commence hormone therapy.5,19,60
Transmen
The virilization of transmen is achieved by the administration of testosterone at standard doses used in hypogonadal males. 58 The physical changes that occur mimic male puberty and therefore take two to five years to complete.4,7,8 Traditionally, testosterone has been administered in the form of mixed testosterone esters given as Sustanon injections 250 mg two to four weekly.7,61 Indeed, this product is the only preparation that is licensed for use in the field of transgender medicine in the UK. 62 At this dose, testosterone suppresses ovarian function in preoperative patients, obviating the need for other endocrine manipulation in the vast majority of cases.
Physical effects
The genital changes induced by testosterone therapy are the first to be noticed by transmen. There is an increase in clitoral size; this onsets early by three to four months and is complete by one year with a usual final clitoral length of 4 to 5 cm; 6 however, this growth is never of a degree that will allow penetrative intercourse. There is an increase in ovarian stromal tissue and cyst formation identical to the changes seen in polycystic ovarian syndrome.63–65 These changes occur after six months of therapy; their significance is unclear.
For the majority of transmen, menstruation is distressing as it is a powerful manifestation of their birth gender. The suppression of menses is often found to be the most psychological beneficial effect of hormonal therapy. Menses usually cease within two to three cycles from the commencement of testosterone therapy because gonadotropin production and therefore ovarian function are suppressed by the high concentrations of testosterone. 7
Very occasionally, testosterone therapy is not sufficient to suppress menstruation; in this situation, progestins such as medroxyprogesterone acetate 10 mg three times daily or norethisterone 5 mg three times a day can be used to suppress menstruation. More recently, GnRH analogues have also been used to suppress ovarian function in this situation.
Testosterone treatment in transmen results in the development of male pattern facial and body hair. There is an overall increase in body hair and a change of the genital hair to a masculine pattern, with hair growth on the face, chest, abdomen, sacrum and inner thighs.6,8 In susceptible individuals, there is a loss of capital hair in a male pattern baldness distribution, with temporal thinning and crown hair loss. The onset of facial hair development is typically six to nine months and is complete by 48 to 56 months. 4 This increase in facial hair is accompanied by a coarsening of the skin texture of the face.
As with hormone replacement for hypogonadal men,61,66,67 testosterone therapy in transmen results in an increase in lean body mass and upper body strength; there is a concurrent decrease in body fat, resulting in a more masculine body shape with increased muscle definition and a decrease in hip-to-waist ratio.6,8
Testosterone therapy has important effects on cognition. Aggression and general drive are increased when testosterone is given to men, 58 testosterone having a general activating role in cognition. 68 Transmen report that they have more energy, aggression and there is an increase in libido. 58 Psychologically, patients feel more masculine and generally more settled in their new gender role once testosterone therapy has commenced. There is also an improvement in visio-spatial ability in these individuals. 69
Testosterone promotes growth in the laryngeal cartilage and vocal cords. The lengthening of the vocal cords decreases the fundamental frequency of the voice, which deepens and takes on a more masculine timbre. The vocal cords also thicken, further decreasing the pitch. Typically, this occurs after about 9 to 12 months of testosterone treatment but can take up to two years to complete. 4
Metabolic and hormonal effects
There is significant gender difference in lipid parameters, with males having higher total cholesterol, low-density lipoprotein (LDL) cholesterol and triglyceride, with lower plasma HDL cholesterol. As expected, testosterone therapy in transmen adversely alters their lipid parameters. A recent meta-analysis of the available data demonstrated that there was no change in total cholesterol or LDL cholesterol but there was a minor increase in triglyceride (0.35 [95% CI = 0.09 to 0.62] mmol/L) and a decrease in plasma HDL concentration (−0.73 [%CI = −0.3 to 0.02] mmol/L), both of which are pro-atherogenic. 20 It is interesting that these changes in lipid profile do not appear to translate into an alteration in cardiovascular risk, as there is no increase in cardiovascular mortality in treated transmen; indeed the rate of myocardial infarction is expected to be approximately one-third in the male population. 28 These findings have been confirmed in long-term follow-up studies where the standard mortality ratio for transmen is not different from the general population. 21
Testosterone induces the production of erythropoietin and so increases the production of red blood cells. 70 Testosterone replacement therapy can be associated with polycythaemia and this increase in blood viscosity can lead to an increased incidence of stroke in those who have a haematocrit above 48%. 71 This can occur even in young subjects; both stroke and myocardial infarction have been reported in anabolic steroid abusers. 72
The development of polycythaemia is more common with injectable forms of testosterone suggesting that the length of time that the testosterone concentration is supraphysiological is important in this effect. 73 There are no reports on the incidence of polycythaemia in transmen receiving testosterone treatment in the current literature.
Liver dysfunction and even fulminant hepatic failure have been reported as side-effects of testosterone therapy. 74 This was, however, seen when alkylated anabolic steroid was used especially in the context of drugs of abuse by body builders and athletes. A very high incidence of liver dysfunction at 32% was reported when these compounds were used in transmen. 75 Current treatment protocols do not use these anabolic steroids for testosterone replacement therapy.
There has been one series reporting the incidence of hepatic dysfunction in transmen treated with modern testosterone replacement therapy that reported the incidence of transient liver dysfunction (<6 months) at 4.4% of transmen and this prolonged dysfunction (>6 months) in 6.8%. 28 It is commented in that report that no subject had liver enzyme dysfunction of more than 2.5 times the upper limit of normal; we can infer from that finding, that the disturbance was not great enough to merit cessation of therapy. Routine monitoring of the liver function in patients on testosterone replacement is recommended.4,5,7
Testosterone if used in excess, such as steroid abuse in athletes, increases insulin resistance; 76 it had therefore been assumed that the high doses used in the masculinization of transmen would lead to an increase in insulin resistance. This does not appear to happen; in the only published study on this topic, insulin resistance did not change following one year of testosterone treatment. 12
Cancer risk
Testosterone can be aromatized to oestradiol and so in transmen there maybe excess oestrogen action unopposed by the action of cyclical progesterone. As oestradiol is mitogenic for the endometrium, in theory this could lead to a risk of developing endometrial cancer in the long term. Reassuringly, there has only been one case of endometrial cancer reported in a transman on testosterone therapy, suggesting that this risk is low. 77
A small study reported a high risk of endometrial hyperplasia in transmen at 15% 29 ; however, this risk is probably overstated as the findings of this study have not been repeated and the histological studies on the endometria of hysterectomy specimens taken from transmen consistently demonstrate endometrial atrophy. 65 Monitoring of the endometrial thickness by ultrasound scanning biannually is currently recommended. It is usual practice to recommend hysterectomy after two years of testosterone therapy.5,7
Ovarian cancer risk appears to be very low; there have been only three cases reported following testosterone therapy for a prolonged period. 58 Exogenous testosterone treatment is also associated with the development of a polycystic morphology,63–65 the implications of this in the longer term are unknown and so oophorectomy is usually performed at the time of hysterectomy.
Breast cancer risk appears to be very low in transmen. The background risk is that of the natal male population. 47 It is to be remembered that male chest reconstruction is not a total mastectomy and that the residual breast tissue should be checked regularly by transmen. Overall, there are reports of five cases of breast cancer in transmen on testosterone therapy. 78
Osteoporosis
Testosterone therapy in transmen appears to maintain bone mineralization effectively. 78 Androgen therapy increases cortical bone size, 79 thickness 41 and BMD 80 in transmen. There has been one study suggesting that a decrease in BMD in transmen occurs postovariectomy, 42 but as in transwomen there was a clear inverse relationship between gonadotropin concentration and bone mass suggesting that low sex steroid concentrations may explain the differences in these findings.
Long-term studies
The current data suggest that long-term treatment with oestrogen in transwomen is associated with a slight increase in the standard mortality ratio.21,81 The increase in mortality appears to be associated with an increase in the risk of suicide81 in vulnerable individuals and also an increase in cardiovascular deaths. 21 The increase in suicide deaths appears to be historical, with mortality rates in a more-recently treated cohort not different from those found in normal populations.81 However, psychological health should nevertheless be assessed and support given when needed.
The increase in vascular disease, however, appears to be associated with the use of ethinylestradiol but not other oestrogen types and so this oestrogen type should be avoided. 21 Breast cancer is extremely rare in transwomen and therefore life-long hormone treatment can be administered.
The current data suggest that long-term treatment with testosterone in transmen is not associated with any increased risk of cardiovascular disease, and the standard mortality ratio of this patient population is normal at 1.21,28
Aims of hormonal therapy
There are many different hormonal regimens used for cross-gender sex steroid therapy. Below is an outline of the regimen used at the largest UK clinic. 7
Transwomen
A list of the oestrogen preparations commonly used in UK practice and the monitoring requirements of these.
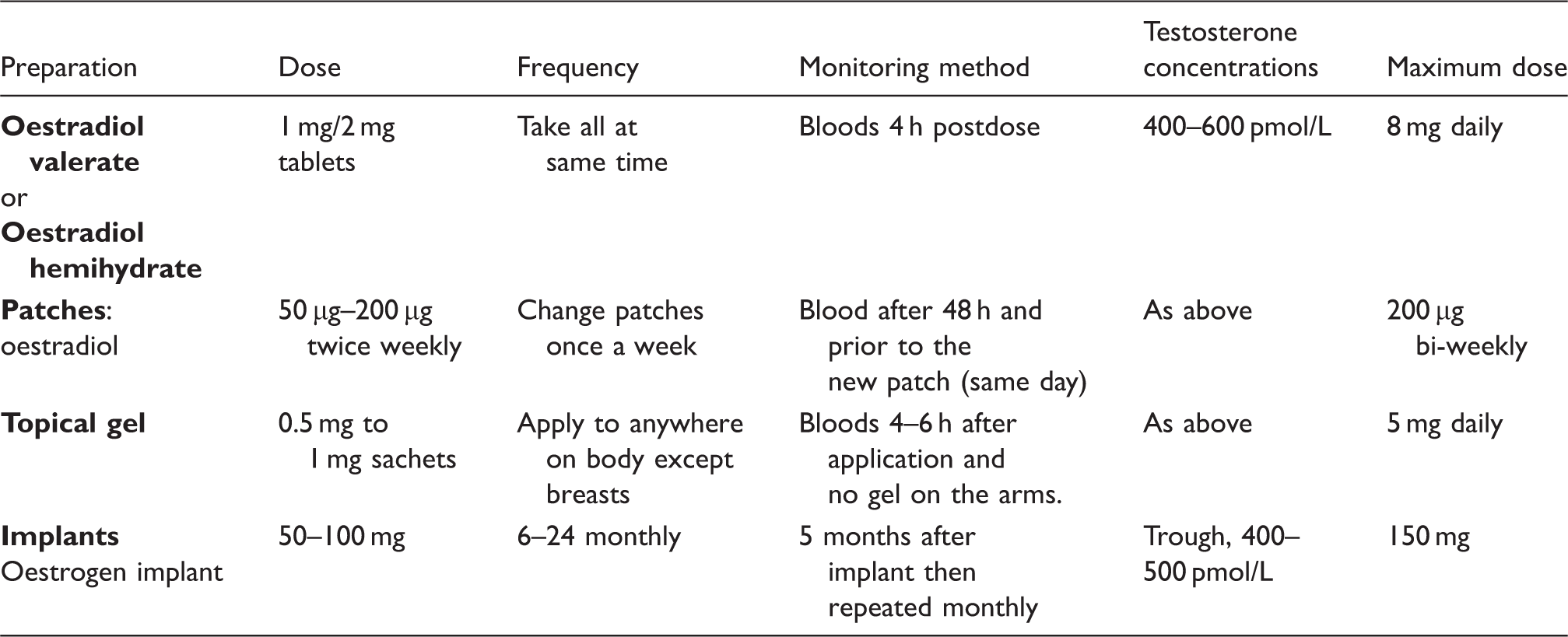
If oestrogen therapy does not suppress testosterone concentrations to the female range (<3 nmol/L), then GnRH analogues are used to suppress testicular function until orchidectomy is performed.
Transmen
A list of the testosterone preparations commonly used in UK practice and the monitoring requirements of these.
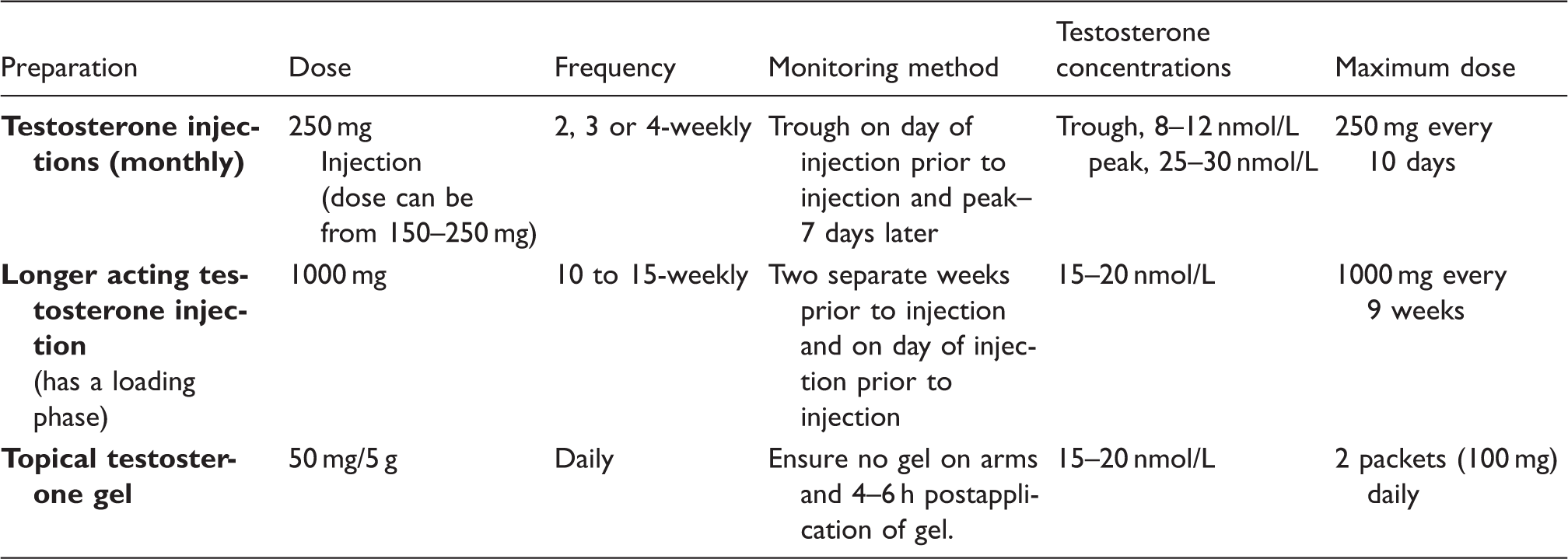
Testosterone at these doses is sufficient in the vast majority of cases to suppress ovarian activity but if menstruation does not cease, then progestins such as norethisterone or medroxyprogesterone can be used. More recently, GnRH analogues have been used to suppress ovarian function until oophorectomy.
Conclusions
Cross-sex hormone therapy is integral to the treatment of the majority of people with gender dysphoria and is effective in inducing the secondary sexual characteristics of the person’s desired gender. Cross-gender sex steroid replacement can be associated with side-effects, for transwomen thromboembolism and for transmen an adverse lipid profile but provide psychological benefit to the individual being treated. With careful monitoring and the use of sex steroid preparations with more physiological properties, these therapies appear to be safe in the longer term.
Footnotes
Acknowledgements
This article was prepared at the invitation of the Clinical Sciences Reviews Committee of the Association for Clinical Biochemistry and Laboratory Medicine.
Declaration of conflicting interests
None.
Funding
This research received no specific grant from any funding agency in the public, commercial, or not-for-profit sectors.
Ethical approval
None.
Guarantor
LJS.
Contributorship
LJS wrote the manuscript.
