Abstract
Background
Analysis of lipoprotein profile gives important clinical information for lipid-lowering therapy which prevents atherosclerotic diseases. The lipoprotein classes can be isolated from serum with ultracentrifugation, which inevitably consumes a long time and needs large serum volume. We have established a method with anion-exchange chromatography with 1.0 µL of the injected volume in 5.2 min for assay of one sample.
Methods
One-hundred-forty-one male volunteers without overt diseases were divided three groups (Group 1, non-dyslipidemia with LDL-cholesterol [LDL-C] <120 mg/dL and HDL-cholesterol (HDL-C) ≥40 mg/dL; Group 2, borderline dyslipidemia with 120 ≤ LDL-C < 140 mg/dL and HDL-C ≥40 mg/dL; Group 3, dyslipidemia with LDL-C ≥ 140 mg/dL or HDL-C < 40 mg/dL). Their lipoprotein profiles were evaluated by rapid anion-exchange chromatography, which measured concentrations of HDL-C, LDL-C, IDL-cholesterol, VLDL-cholesterol, and other fraction (chylomicron + lipoprotein [a])-cholesterol (other-C).
Results
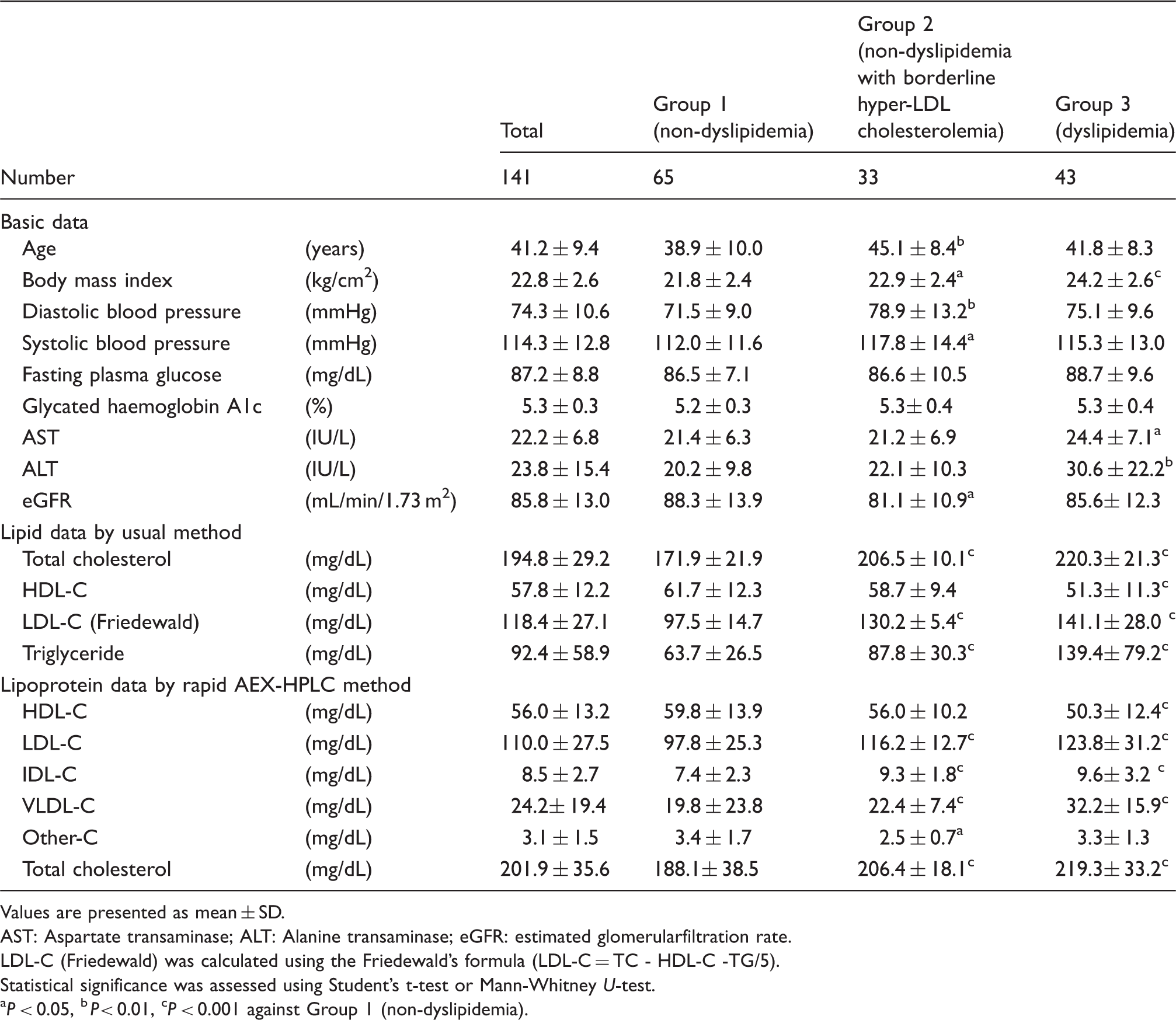
The within-day and between-day assay coefficients of variation of lipoprotein cholesterol values were 0.33–4.31% and 2.37–9.19%, respectively. The correlation coefficients between values of HDL-C, LDL-C, IDL-C and VLDL-C by the anion-exchange chromatography and those by ultracentrifugal method were 0.97, 0.92, 0.58 and 0.94, respectively. Group 3 had significantly lower HDL-C and higher concentrations of IDL-C, VLDL-C and other-C than did Group 1. Group 2, borderline dyslipidemia, had significantly higher concentrations of IDL-C and VLDL-C than did Group 1.
Conclusion
The rapid anion-exchange chromatography assay may be sufficiently applied to the assessment of borderline dyslipidemia.
Introduction
Elevated low-density lipoprotein cholesterol (LDL-C) has been shown to be a risk factor for the development of atherosclerosis and associated with an increased risk of cardiovascular disease (CVD).1,2 High-density lipoprotein cholesterol (HDL-C) is an inverse predictor of atherosclerosis risk.3,4 Moreover, increased triglyceride (TG)-rich lipoproteins, including intermediate-density lipoprotein (IDL), very low-density lipoprotein (VLDL) and chylomicron remnant, are related to the risk of atherosclerotic diseases.5–7 Thus, dyslipidemia is a risk factor for atherosclerosis, and is classified as either familial or acquired disorder of lipoprotein metabolism. World Health Organization (WHO) classification of hyperlipidemia is a biochemical categorization based on raised lipoprotein patterns. 8 Hence, analysing lipoproteins in detail provides important clinical information for the prevention of atherosclerosis and the therapy of dyslipidemia.
Major classes of lipoproteins can be isolated from serum with ultracentrifugation, but the method is time-consuming and needs large volumes of serum. 9 Our previous study showed a new method for the measurement of cholesterol in the five major classes of serum lipoproteins by anion-exchange chromatography (AEX-HPLC) with perchlorate ion-containing elute, and confirmed cholesterol concentrations of lipoproteins fractionated by the AEX-HPLC were significantly correlated with the lipoprotein cholesterol values measured by ultracentrifugation method. 10 The AEX-HPLC method required 20 min per one measurement, and therefore shortening the measurement time was needed to use as a routine test in clinical laboratory.
We have developed a rapid AEX-HPLC for the measurement of cholesterol in the five major lipoprotein fractions (HDL, LDL, IDL, VLDL and other fraction [Other], including chylomicron + lipoprotein(a)). The measurement time of the new method could be shortened by the improvement of column size, eluent composition, and flow rates of eluent and reaction reagents, and reached 5.2 min to assay one sample. We evaluated the lipoprotein profiles measured by the new AEX-HPLC method in male volunteers without overt diseases.
Materials and methods
Samples
This study was approved by the ethics committee at Jikei University and TOSOH Corporation. At entry, written informed consent was obtained from all participants.
Thirty-five dyslipidemic serum samples obtained from the volunteers (male/female = 31/4, aged 56.5 ± 16.4 years, total cholesterol (TC) = 236.8 ± 48.8 mg/dL, TG = 250.5 ± 147.8 mg/dL) of Tokyo Research Center of TOSOH Corporation were used to examine correlations between the data measured by the rapid HPLC method and those obtained by an ultracentrifugation method. VLDL + chylomicron fraction (d < 1.006 g/mL) was prepared by the ultracentrifugation using 40 chylous serum samples derived from patients (male/female = 24/16, aged 55.8 ± 15.5 years, TC = 221.3 ± 49.7 mg/dL, TG = 941.1 ± 546.8 mg/dL) of Jikei University Kashiwa Hospital. These VLDL + chylomicron fraction samples were used to compare VLDL cholesterol (VLDL-C) and chylomicron cholesterol values of the rapid HPLC measurement with agarose gel electrophoresis determination.
Fasting serum samples (n = 141) were obtained from the male volunteers without overt disease in Tokyo Research Center of Tosoh Corporation. Subjects taking lipid-lowering medications and those with heart, liver or kidney disease were excluded. They were divided into three groups: Group 1, non-dyslipidemia (LDL-C < 120 mg/dL, HDL-C ≥ 40 mg/dL and TG < 150 mg/dL); Group 2, borderline dyslipidemia (120 ≤ LDL-C < 140 mg/dL, HDL-C ≥40 mg/dL and TG <150 mg/dL); Group 3, dyslipidemia (LDL-C ≥ 140 mg/dL, HDL-C < 40 mg/dL or TG ≥150 mg/dL). In the Japan Atherosclerosis Society guideline, diagnostic criteria for dyslipidemia are defined as LDL-C ≥140 mg/dL, TG ≥150 mg/dL and/or HDL-C < 40 mg/dL, and the borderline hyper-LDL cholesterolemia is defined as LDL-C 120–139 mg/dL.
Purified lipoprotein(a) (Lp(a)) sample from human plasma is purchased from Biomedical Technologies Inc., USA.
Rapid anion-exchange chromatographic method
For shortening the time of assay, we modified the method reported previously,
10
and developed the new rapid AEX-HPLC method (Figure 1). The new method can measure levels of HDL-C, LDL-C, IDL cholesterol (IDL-C), VLDL-C and other lipoprotein faction cholesterol (Other-C) including cholesterol of chylomicron, chylomicron remnant and Lp(a) within 5.2 min after injecting sample.10–12 Table 1 shows the improved points compared with the previous method.
Schematic diagram of the rapid AEX-HPLC system. Modification of rapid AEX-HPLC method from a previously reported method.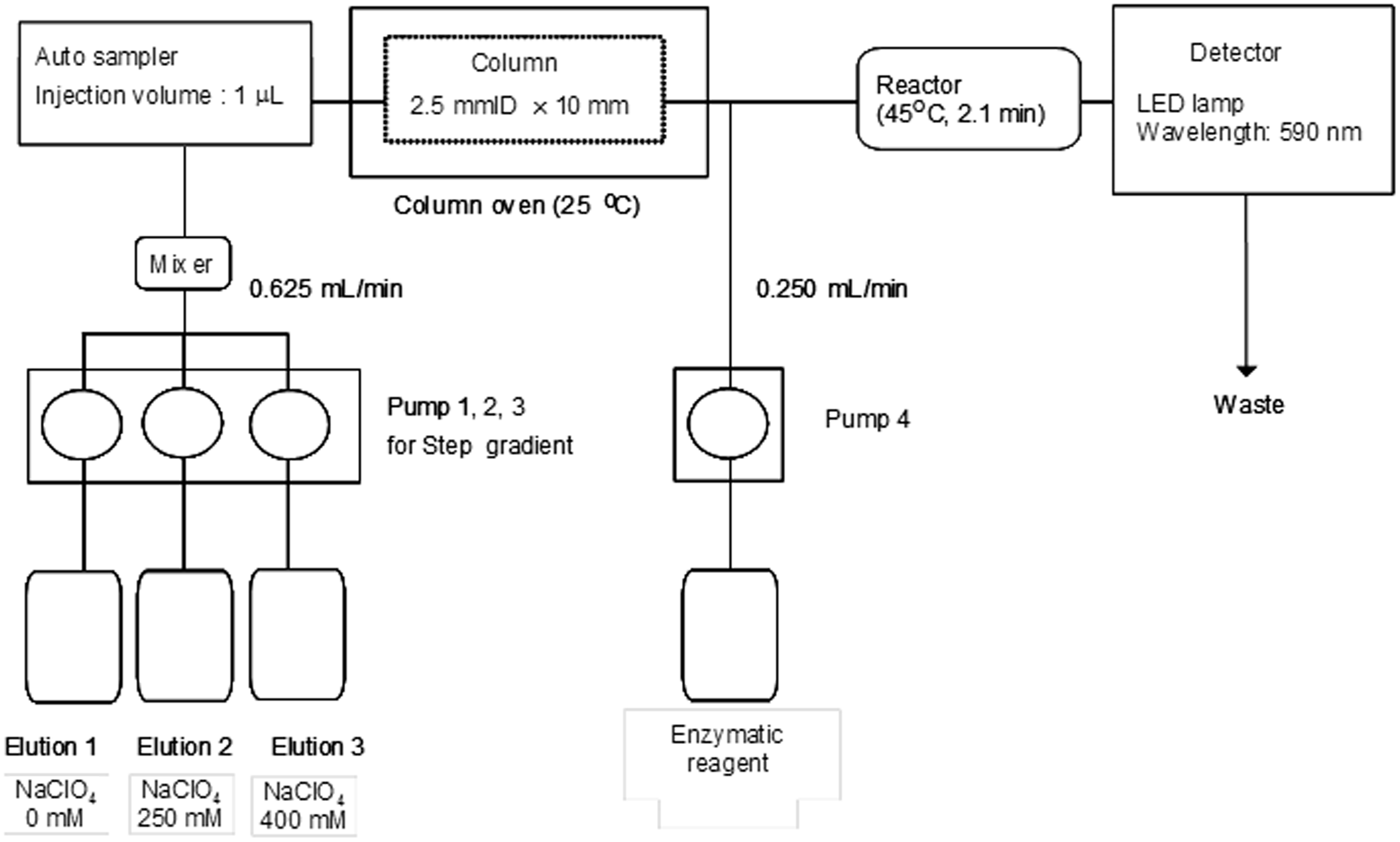
To separate lipoproteins rapidly, an optimized column containing 2.5 µm of non-porous gel with diethylaminoethyl ligand is used, and its column size is 2.5 mm I.D. × 10 mm. Then, three eluents are used, and respectively sent with three pumps for the gradient elution method. These eluents are composed as follows: Eluent-1 (50 mM Tris-HCl, pH 7.5), Eluent-2 (50 mM Tris-HCl + 250 mM sodium perchlorate, pH 7.5) and Eluent-3 (50 mM Tris-HCl + 400 mM sodium perchlorate, pH 7.5). The flow rate is 0.625 mL/min, and the step gradient patterns for the separation of the lipoproteins are designed in a stepwise fashion (59.5% Eluent-1, 40.5% Eluent-2 and 0.0% Eluent-3 for 0–1.10 min; 50.5% Eluent-1, 49.5% Eluent-2 and 0.0% Eluent-3 for 1.10–2.25 min; 44.5% Eluent-1, 55.5% Eluent-2 and 0.0% Eluent-3 for 2.25–3.06 min; 30.0% Eluent-1, 70.0% Eluent-2 and 0.0% Eluent-3 for 3.06–3.83 min; 0.0% Eluent-1, 0.0% Eluent-2 and 100.0% Eluent-3 for 3.83–4.27 min; 80.0% Eluent-1, 20.0% Eluent-2 and 0.0% Eluent-3 for 4.2–5.2 min). It takes 5.2 min to complete the assay of one sample. The eluate from the column is mixed with an enzymatic cholesterol reagent which contains cholesterol esterase, peroxidase and cholesterol oxidase as major ingredients. The flow rate of the enzymatic cholesterol regent is set as 0.25 mL/min by using a pump. The mix solution reacts at 45℃ for 2.1 min. The reactivity of the new method is similar to the previous method. The reactivity of cholesterol at 45℃ for 2.1 min is 97.7% compared to the condition at 37℃ for 5.0 min in the previous method. The absorbance is detected at 590 nm with a light emitting diode (LED) lamp. There was little difference in absorbance of cholesterol between at 590 nm and at 600 nm, and the ratio of 590 nm to 600 nm was 102.9%. The injected sample volume of the rapid method is 1.0 µL. We have confirmed that the sensitivity of the rapid method is similar to that of the previous method, and the limit of detection for lipoprotein peaks on a signal-to-noise ratio of 3 by the rapid method and the previous method were 0.92 mg/dL and 0.93 mg/dL, respectively.
Figure 2(A)a and (A)b shows representative chromatograms of healthy serum assayed with the new rapid method and the previous method during the changes in concentration of sodium perchlorate of eluate solution under the step gradient patterns indicated by a hatched line.
Chromatograms of the rapid AEX-HPLC.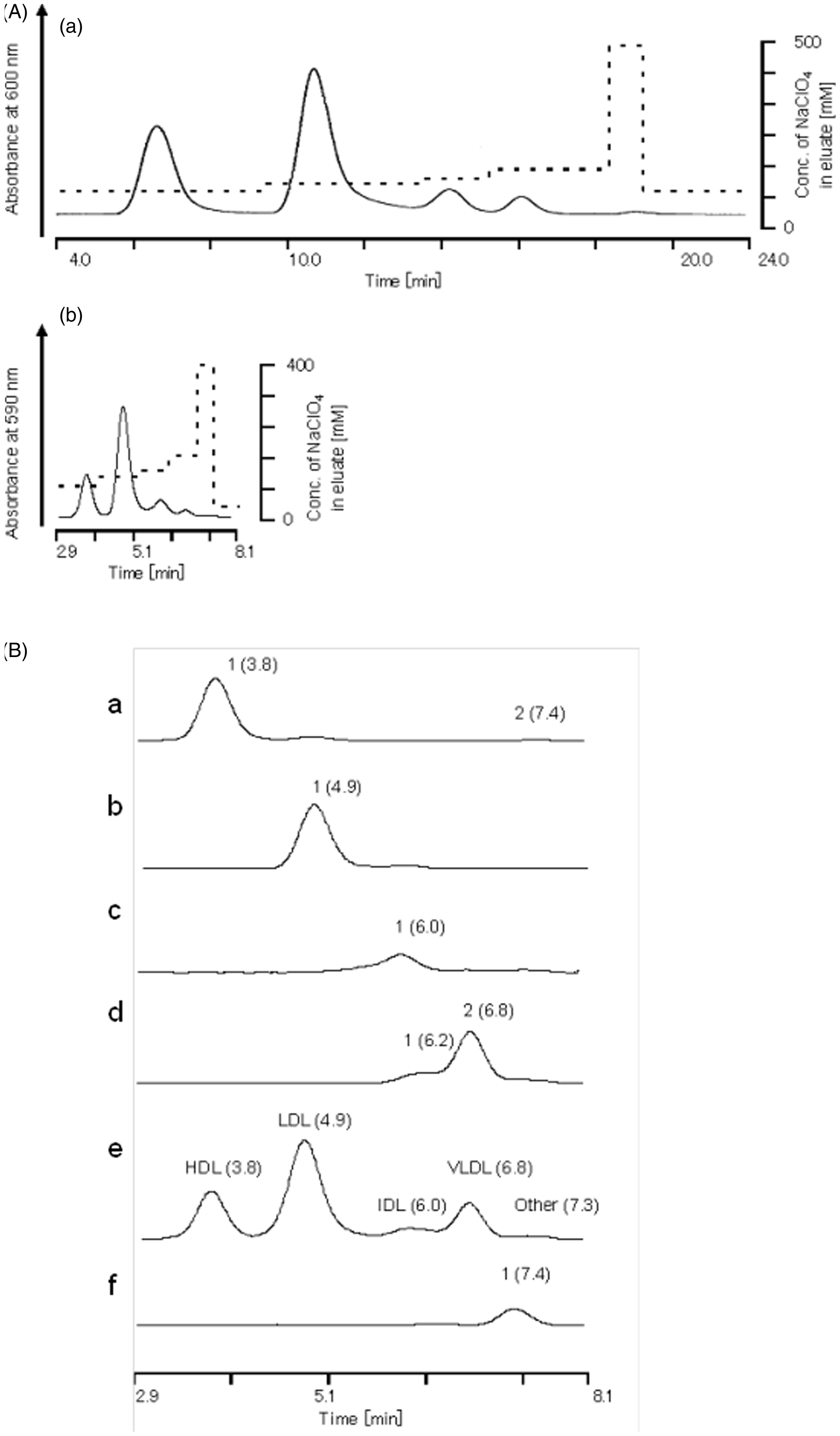
Ultracentrifugation method
Sequential ultracentrifugation of serum lipoproteins was performed by the method reported previously.9,10 Densities of VLDL and chylomicrons, IDL, LDL, and HDL were set as follows: d < 1.006 g/mL, 1.006 < d < 1.019 g/mL, 1.019 < d < 1.063 g/mL and 1.063 g/mL < d, respectively. We used an ultracentrifugation instrument (Himac CS100GXII, Hitachi Koki Co.) and an ultracentrifuge fixed-angle rotor (S58A-0112, Hitachi Koki Co.).
Linearity and precision tests
To test for linearity, the serum of a dyslipidemic patient (male; aged 49 years; TC = 143 mg/dL; TG = 272 mg/dL; HDL-C = 40.9 mg/dL; LDL-C = 77.2 mg/dL; IDL-C = 11.1 mg/dL, VLDL-C = 28.6 mg/dL, Other-C = 159.9 mg/dL) was recruited from Jikei University Kashiwa Hospital. The samples were diluted serially with 5% bovine serum albumin (BSA) solution. For within-day and between-day assay precision tests, a freeze-dried pooled human serum (TC = 281 mg/dL and TG = 179 mg/dL) was used.
Statistical analysis
Static analyses of the present data were performed by the Stat Flex Ver6.0 software (Artec Inc, Osaka, Japan). The data were presented as mean ± standard deviation (SD). Student’s t-test or Mann-Whitney U-test was used to compare variables between Group 1 (non-dyslipidemia) and Group 2 (borderline hyper-LDL dyslipidemia) or Group 3 (dyslipidemia). P values <0.05 were considered significant.
Results
Chromatograms of lipoproteins separated from dyslipidemic serum by ultracentrifugation, and Lp(a) purified from human plasma
Five peaks were identified in the chromatogram of serum from a healthy subject (Figure 2(B)e) and detected at the retention times of 3.8, 4.9, 6.0, 6.8 and 7.3 min, respectively. The expressed peaks at these retention times corresponded to the HDL fraction peak (1.063 g/mL < d), LDL fraction (1.019 d < 1.063 g/mL), IDL fraction (1.006 < d < 1.019 g/mL) and VLDL + chylomicrons fraction (d < 1.006 g/mL) (Figure 2(B)a–d). A single peak was detected at 7.4 min in the chromatogram of Lp(a) sample which was purified from human plasma using affinity chromatography and commercially available (Figure 2(B)f).
Correlation of lipoprotein cholesterol values between the rapid AEX-HPLC data and the ultracentrifugation method data
Figure 3(A)a–d indicates the correlations between the cholesterol concentrations of HDL, LDL, IDL and VLDL + chylomicron measured by the rapid AEX-HPLC and the ultracentrifugation method in 35 dyslipidemic serum samples. The linear regression equations and the coefficients of correlation between cholesterol values of HDL, LDL, IDL and VLDL + chylomicron found by these two rapid methods were y = 0.924 x + 0.133 (r = 0.971), y = 0.990 x + 7.992 (r = 0.918), y = 0.584 x + 7.675 (r = 0.584) and y = 0.868 x + 2.505 (r = 0.935), respectively.
Correlation between cholesterol concentrations in each lipoprotein classes measured by rapid HPLC method and those measured by ultracentrifugation method or measured by agarose gel electrophoresis.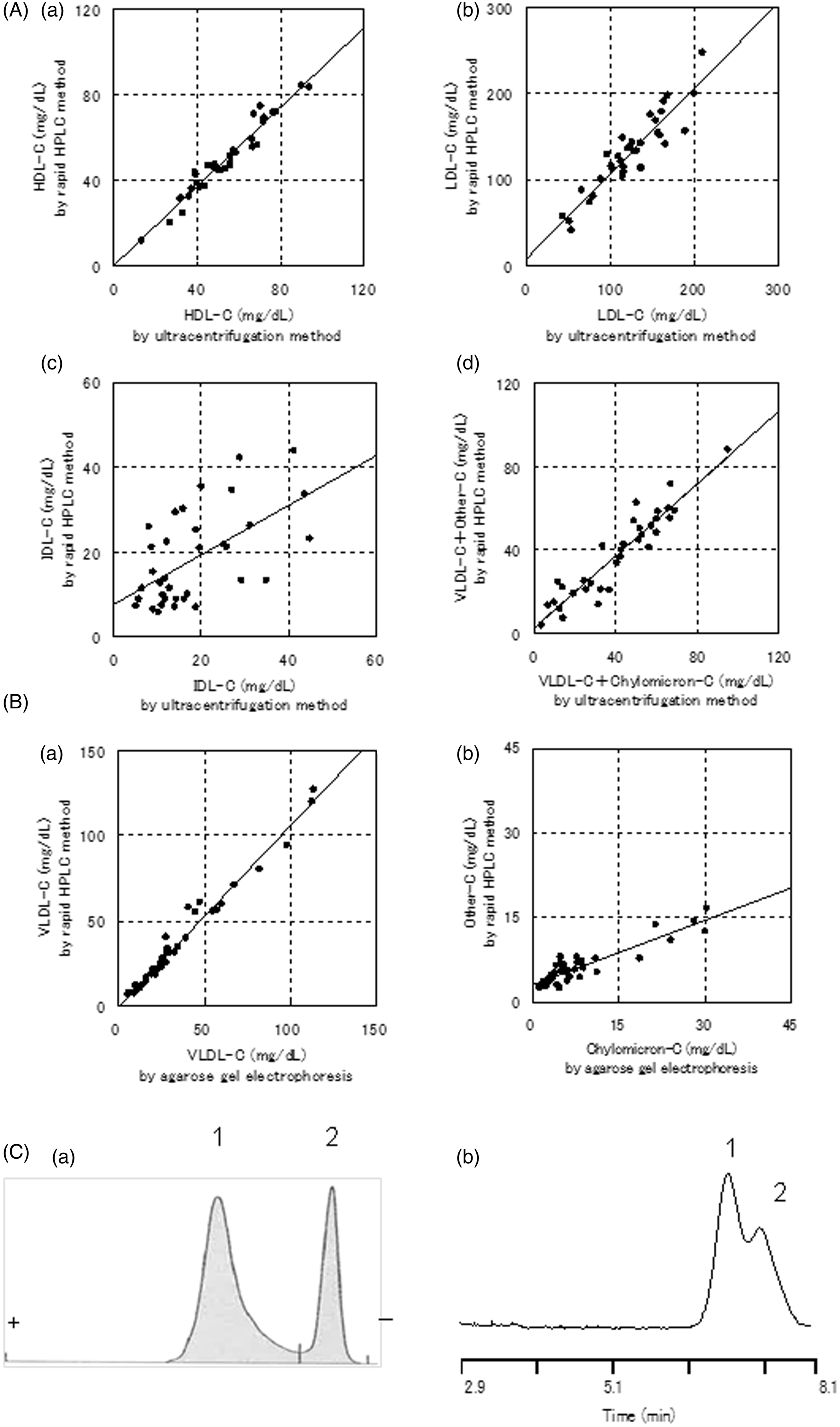
Correlation of VLDL and chylomicron cholesterol values between the rapid AEX-HPLC data and the agarose gel electrophoresis data
VLDL and chylomicron cannot be separated using ultracentrifugation method, because their densities are same range (<1.006 g/mL). The flotation rates (Sf) of VLDL and chylomicron in a solution of density 1.006 g/mL were Sf 20–400 and Sf > 400, respectively. Due to the difference of Sf, they were separated using ultracentrifuge but it is difficult to separate with precision.
Agarose gel electrophoresis is a method for separating lipoproteins, and α peak, β peak, pre β peak, and origin correspond to HDL, LDL, VLDL and chylomicron, respectively.13,14 Other peak of AEX-HPLC contains Lp(a) and chylomicron.10–12 We prepared VLDL + chylomicron fraction samples separated from 40 chylous serum samples by using ultracentrifugation and compared the cholesterol values of VLDL and other peaks by the rapid AEX-HPLC with those of VLDL and chylomicron by agarose-gel electrophoresis, respectively. Figure 3(B)a and b indicates the correlations (VLDL-C, y = 1.064 x–0.177 [r = 0.987]; chylomicron cholesterol, y = 0.383 x + 3.163 [r = 0.904]). An electrophoresis pattern stained for cholesterol by agarose gel electrophoresis using VLDL + chylomicron fraction sample and a chromatogram by the rapid AEX-HPLC was shown in Figure 3(C)a and b, respectively.
Linearity and precision of the rapid HPLC method
Precision data for rapid AEX-HPLC method.
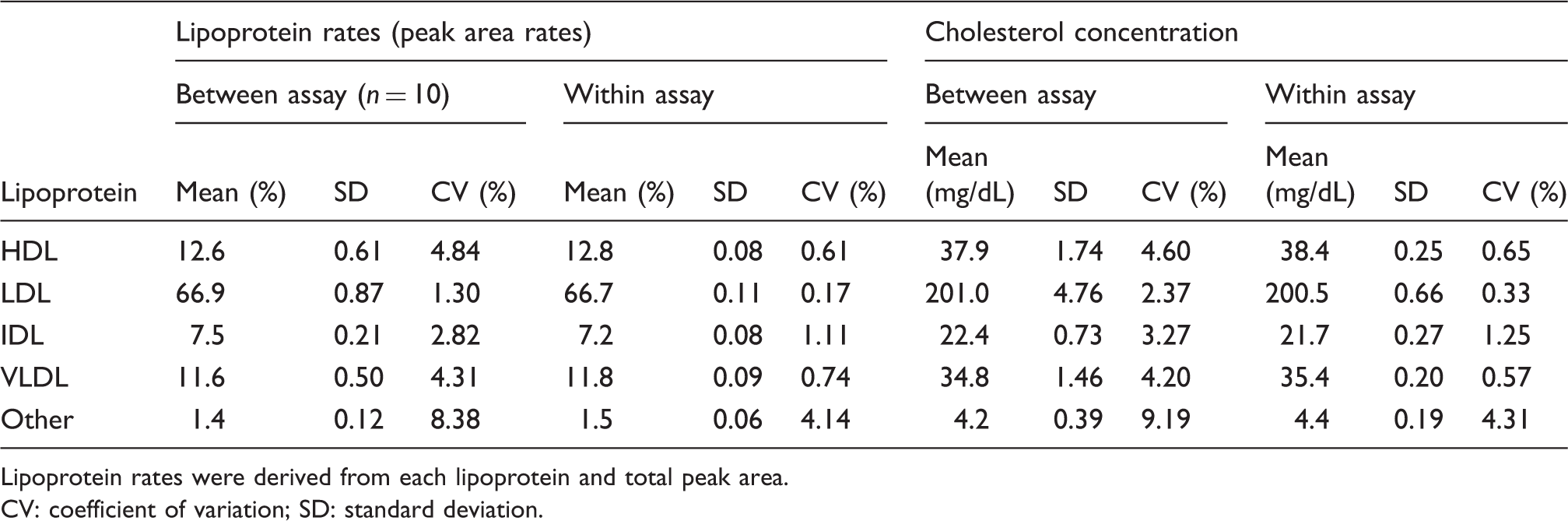
Lipoprotein rates were derived from each lipoprotein and total peak area. CV: coefficient of variation; SD: standard deviation.
Lipid profiles of three groups (non-dyslipidemia, borderline hyper-LDL cholesterolemia and dyslipidemia)
Basic data and lipid profiles of three groups (non-dyslipidemia, non-dyslipidemia with borderline hyper-LDL cholesterolemia and dyslipidemia).
Values are presented as mean ± SD.
AST: Aspartate transaminase; ALT: Alanine transaminase; eGFR: estimated glomerularfiltration rate.
LDL-C (Friedewald) was calculated using the Friedewald’s formula (LDL-C = TC ‐ HDL-C ‐TG/5).
Statistical significance was assessed using Student’s t-test or Mann-Whitney U-test.
P < 0.05, b P< 0.01, cP < 0.001 against Group 1 (non-dyslipidemia).
Discussion
TG-rich lipoproteins are related to the development of atherosclerosis and coronary artery disease risk as well as LDL. Analysing lipoproteins in detail is clinically important for preventing the development of atherosclerotic disease.5–7 Ultracentrifugation can be used to separate or collect major lipoprotein classes including TG-rich lipoproteins, but consumes a long time, and needs large serum volume. 9 We developed the rapid AEX-HPLC based on the method reported previously, which can measure cholesterol concentrations of five lipoprotein classes: HDL, LDL, IDL, VLDL and other fraction (chylomicron and Lp[a]). For shortening analysis time to 5.2 min from 20 min, column size, sample injection volume, reaction condition, flow rates of eluents and reaction reagents were improved and optimized. The consumed volumes of eluent and reaction reagents were reduced by 20% and 16%, respectively, as compared to the previous AEX-HPLC. Therefore, the rapid AEX-HPLC may be adopted for not only laboratory research use but also for routine clinical laboratory use.
The eluted time of major peaks of HDL, LDL, IDL and VLDL of VLDL + chylomicron fraction separated by ultracentrifugation was similar to those of the four peaks in the chromatogram of a serum (Figure 2(B)a–e). We also indicated that Lp(a) purified from human plasma was identified in other peaks of healthy serum (Figure 2(B)f). The HDL fraction (1.063 g/mL < d) contains the major part of Lp(a) whose density is 1.040–1.125 g/mL. 16 Therefore, we thought that the small peak (retention time 7.4 min) in the chromatogram of HDL fraction, ultracentrifugally separated, was likely to be Lp(a) peak (Figure 2(B)e). These results indicated that the rapid AEX-HPLC has a similar ability of lipoprotein separation as compared to the previous AEX-HPLC.10,11 The cholesterol concentrations of serum lipoproteins (HDL, LDL, IDL and VLDL + chylomicron) by the rapid AEX-HPLC were correlated with those by the ultracentrifugation method (Figure 3(A)a–d). Moreover, the VLDL-C and chylomicron cholesterol in the VLDL + chylomicron samples measured by the rapid AEX-HPLC were correlated with those by the agarose gel electrophoresis, respectively (Figure 3(B)a and b). Measuring cholesterol concentrations of serum lipoproteins by the rapid AEX-HPLC was highly repeatable and had the good dilution linearity. These results suggest that the rapid AEX-HPLC may be substituted for ultracentrifugal method.
In this study, we examined the cholesterol concentrations of five lipoproteins in non-dyslipidemia (Group 1), borderline hyper-LDL cholesterolemia (Group 2) and dyslipidemia (Group 3). The HDL-C of non-dyslipidemia (Group 1) was similar to that of healthy subjects as previously reported in the general Japanese males (55 ± 14 mg/dL). 15 A previous report showed IDL-C concentrations of control subjects and type III hyperlipoproteinemia were 7.7 ± 3.9 mg/dL and 42.5 ± 19.3 mg/dL, respectively. 17 The IDL-C concentration of non-dyslipidemic serum samples (7.4 ± 2.3 mg/dL) (Table 3) in our report was consistent with those in the previous paper.
Low HDL-C and high LDL-C are associated with CAD risk, and some studies suggested the relevance of increased IDL and VLDL to atherosclerosis.6,12 The present study shows that IDL-C and VLDL-C were significantly different between non-dyslipidemia (Group 1) and borderline hyper-LDL cholesterolemia (Group 2). These results suggest that IDL-C and VLDL-C determined by the rapid AEX-HPLC may be useful for clinical assessment of Japanese males with borderline hyper-LDL cholesterolemia.
In conclusion, this study showed that the five major classes of lipoproteins in human serum samples can be separated in 5.2 min, and the cholesterol concentrations of lipoprotein classes can be determined by the rapid HPLC method. Furthermore, IDL-C and VLDL-C measured by the rapid AEX-HPLC were significantly different between non-dyslipidemia and borderline hyper-LDL cholesterolemia. These results suggest that the rapid AEX-HPLC may be sufficiently applied to the assay for the clinical assessment of borderline hyper-LDL cholesterolemia.
Footnotes
Acknowledgements
We would like to thank Ms Syuko Onodera for excellent technical assistance.
Declaration of conflicting interests
DM is an employee of TOSOH Corp (Tokyo, Japan).YH was employed with TOSOH Corp (Tokyo, Japan) until March 2015.
Funding
Research funds were provided in part by the Jikei University Research Fund (to HY) and in part by the Tosoh Research Fund (to DM and YH).
Ethical approval
Jikei University Ethics Council (no. 15-166), TOSOH Corporation Ethics Council (no. 14-04).
Guarantor
YH.
Contributorship
DM corrected data, contributed to the discussion and wrote the manuscript. YH and HY designed the study protocol, contributed to the discussion and reviewed and edited the manuscript.
