Abstract
Background
Intravenous iron is commonly administered to anaemic patients to treat iron deficiency, but due to its ferric colouration, it may interfere with the spectrophotometric assessment of haemoglobin concentrations. This paper investigates the potential interference of three clinically used intravenous iron preparations on the measurement of haemoglobin.
Methods
Haemoglobin concentration was measured for neat and Hartmann’s solution-diluted iron polymaltose, carboxymaltose and sucrose solutions using bedside (Radiometer HemoCue®), point-of-care (Radiometer ABL800 Flex) and laboratory (Abbott CellDyne Sapphire™) devices. Haemoglobin concentration was then assessed with the same devices utilizing anaemic whole blood with the iron solutions added.
Results
Neat iron preparations registered clinically significant haemoglobin concentrations on bedside and laboratory measurements. When intravenous iron preparations were diluted to clinical concentrations, their effect on haemoglobin measurements, either in isolation or mixed with anaemic blood, was negligible.
Conclusion
Although neat preparations of intravenous iron do interfere with spectrophotometric analysis of haemoglobin, concentrations likely to be seen post iron infusion do not significantly interfere with haemoglobin measurement.
Introduction
Total haemoglobin (Hb) concentration is a commonly used laboratory measurement with results informing clinical decision making on blood transfusion and need for further testing to investigate anaemia. Intravenous iron is increasingly used in the treatment of anaemia and is a significant component of the Australian National Blood Authority’s treatment algorithm for perioperative anaemia. 1
There are three commonly used options available for measuring Hb, namely portable bedside devices such as HemoCue®, point-of-care devices such as benchtop blood gas analysers and laboratory-based Hb measurement. All three measurement devices utilize spectrophotometric analysis in the assessment of Hb concentrations. Theoretically, the presence of other light absorbing substances, such as intravenous iron, in blood samples may interfere with the measurement of Hb as it does with other processes utilizing this technique.2,3
In this study, the impact of neat and clinically used concentrations of three preparations of intravenous iron was assessed on the three methods for measuring Hb. Bedside (HemoCue®), point-of-care arterial blood gas (ABL800 Flex), and laboratory-based (CellDyne Sapphire™) devices were investigated.
Materials and methods
All samples were analysed using HemoCue®, ABL800 Flex and CellDyne Sapphire™ devices maintained and standardized as per PathWest Laboratory Medicine, Western Australia protocols. Iron polymaltose and ferric carboxymaltose are presented as 50 mg/mL solutions, and iron sucrose is presented as 20 mg/mL.
Hb measurement was determined for each device and for each iron preparation with sequential testing of (1) Hartmann’s solution, (2) neat iron solution, (3) diluted iron solution (with Hartmann’s solution), (4) anaemic blood and (5) anaemic blood with dilute iron present.
Anaemic blood was prepared by diluting whole blood with Hartmann’s solution to Hb of approximately 70 g/L. The dilute iron concentration of 0.2 mg/mL used for this study was determined by a dose of 1000 mg of iron (maximum iron carboxymaltose single dose) administered to a 70 kg, 180-cm male with an expected blood volume of 5 L as calculated by the Nadler formula. 4
Each sample was analysed in triplicate on each of the devices with mean apparent Hb concentration recorded. Due to the variability of Hb content in the anaemic blood samples prior to adding dilute iron, the measured Hb has been standardized to a haematocrit of 0.2 as measured by the CellDyne Sapphire™.
Unpaired t-tests were used to compare the mean Hb measurements in anaemic blood with and without iron added. Due to multiple statistical comparisons, a Bonferroni correction has been applied, and a P value of 0.01 considered significant.
Results
Neat and dilute iron preparations
Apparent haemoglobin concentrations.
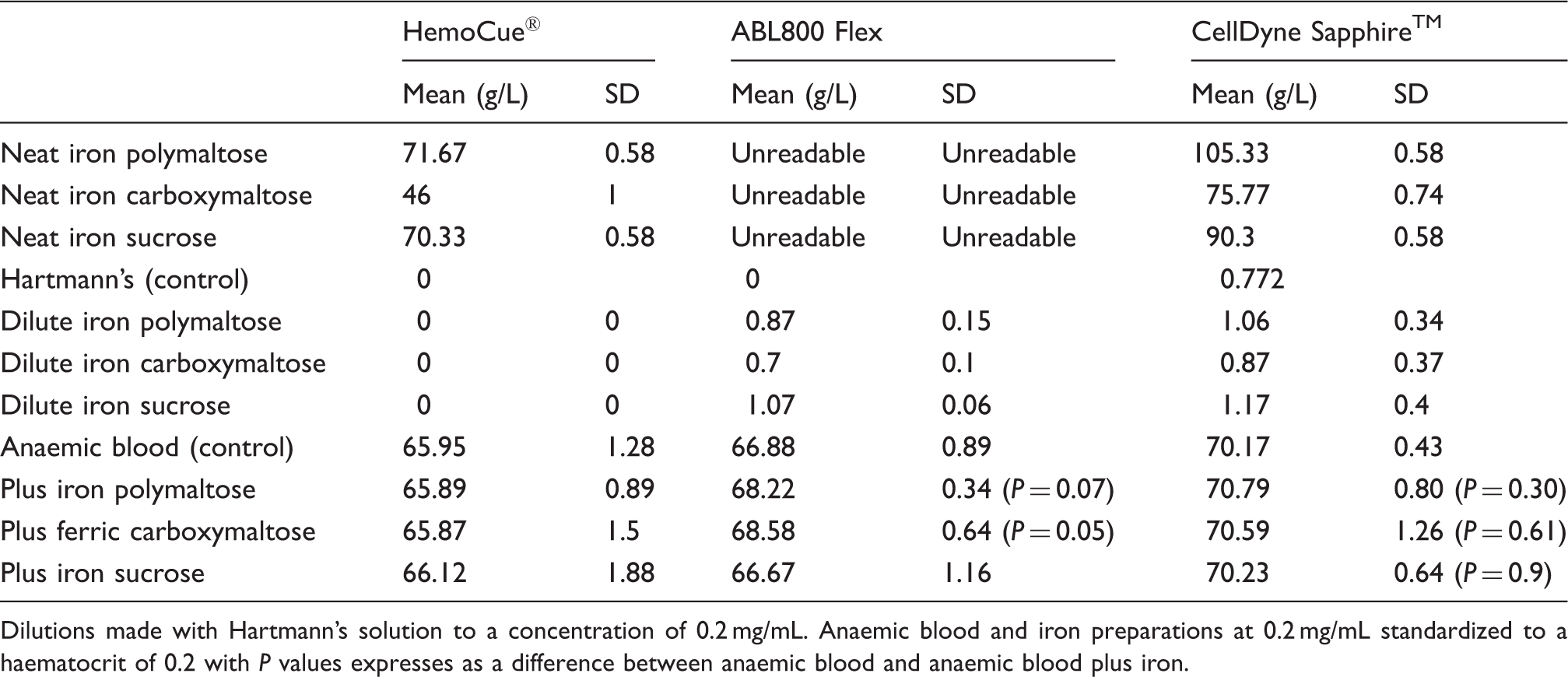
Dilutions made with Hartmann’s solution to a concentration of 0.2 mg/mL. Anaemic blood and iron preparations at 0.2 mg/mL standardized to a haematocrit of 0.2 with P values expresses as a difference between anaemic blood and anaemic blood plus iron.
Iron in anaemic blood
All three measurement devices recorded Hb readings for the iron preparations diluted with anaemic blood. The results are summarized in Table 1 with comparison against the control of anaemic blood free of added iron.
Discussion
Apparent Hb measurements were recorded in neat iron by HemoCue® and CellDyne Sapphire™, despite the absence of blood in these samples. This highlights the potential of intravenous iron to interfere with the spectrophotometric analysis of Hb.
Significant Hb recordings were not made when iron was diluted by Hartmann’s solution to a concentration of 0.2 mg/mL. Nor was a significant difference observed between measurements of Hb in samples containing iron in anaemic blood compared to control. This suggests that iron at estimated clinically relevant concentrations does not interfere with the measurement of Hb by HemoCue®, CellDyne Sapphire™ and ABL800 Flex analysis. Variations in Hb measurement between the measuring devices are noted, which are consistent with published studies. 5
There are a number of limitations of this study. Small sample group sizes limit the ability of this study to detect small differences in measured Hb concentrations. Likewise, a single concentration of iron has been tested with a presumed volume of distribution (Vd) of 5 L. This assumes that intravenous iron distributes throughout the 5 L of blood volume, rather than 3 L of plasma volume, which may actually reflect a more accurate pattern of distribution in vivo. It is also recognized that relatively higher doses of iron can on occasion be given to smaller patients, potentially increasing interference with Hb measurement. Nevertheless, the absence of an increase in measured Hb suggests any artefactually high Hb measurement is unlikely to be clinically significant.
Conclusion
Available intravenous iron preparations are unlikely to significantly interfere with the spectrophotometric analysis of Hb at clinical concentrations and common levels of anaemia. The impact of the presence of intravenous iron may become significant at profound levels of anaemia and at larger doses than tested in this study, although these situations are likely to be rare.
The accurate measurement of Hb is essential in diagnosis, monitoring of disease progression and treatment in everyday clinical practice. It is important to remain vigilant about potential sources of interference or error when interpreting any medical test, including Hb results.
Footnotes
Acknowledgements
We thank Drew Craig from PathWest Laboratories, Fremantle Hospital.
Declaration of conflicting interests
EO has received unrestricted trial funding support from Vifor for an unrelated study, the ‘IronNOF’ trial.
Funding
This trial was funded by the Anaesthetic Department of Fremantle Hospital and PathWest Laboratories, Fremantle Hospital.
Ethical approval
Not applicable.
Guarantor
EO.
Contributorship
EO devised the study. PG and NMF prepared the manuscript with supervision and additional input from EO. RW provided review and laboratory assistance.
