Abstract
Background
‘Add-on’ tests are often requested for samples already in the laboratory. This study was carried out to assess the stability of common analytes in uncapped samples stored in different conditions, and therefore their suitability for ‘add-on’ requests.
Methods
Storage conditions evaluated included initial storage at room temperature for 16 hours, followed by storage at 4℃ (Group 1: current conditions employed in the laboratory), compared with storage at 4℃ immediately following analysis (Group 2: optimum storage conditions).
Results
Some analytes were not suitable for ‘add-ons’ when samples were stored in either storage condition, whereas some were suitable for ‘add-ons’ irrespective of storage condition. Storage conditions influenced the suitability of ‘add-on’ tests for a proportion of analytes including urea, amylase, total protein, alkaline phosphatase, adjusted calcium, lactate dehydrogenase, triglycerides, HDL cholesterol and total cholesterol; these analytes were stable in optimum conditions (Group 2) but not in current conditions (Group 1).
Conclusions
‘Add-on’ tests can only be safely performed on a proportion of routine analytes. For some analytes, storage conditions affect their suitability for delayed analysis.
Introduction
‘Add-on’ tests are often requested for samples already in the laboratory. This saves a further venepuncture, but how stable are the samples in modern laboratory environments? Laboratories now involve sample managers attached to robotic tracking systems. At Leeds General Infirmary, samples are stored uncapped in the sample manager module postanalysis, sometimes for up to 16 hours until they are removed for cold storage, leaving the samples susceptible to deterioration. This study was carried out to assess the stability of common analytes in samples stored in such conditions, and therefore their suitability for ‘add-on’ requests.
Methods
Samples collected in serum separator tubes (Greiner Bio-One, Stonehouse, Gloucestershire, UK) from primary care which contained excess serum (>2 mL) after all requested analyses were performed were identified and anonymized. Two groups of 10 samples were used: samples in Group 1 were stored in conditions used currently, whereas samples in Group 2 were stored in optimum conditions. Group 1 samples were analysed immediately following uncapping, left on the sample track at room temperature, and after 16 hours all analyses were repeated. A 16-hours measurement point was chosen, as this represented the longest time period any individual sample would remain on the sample manager in routine practice. Thereafter, Group 1 samples were transferred to the cold room. Group 2 samples were analysed immediately following uncapping and placed in the cold room at 4℃ without delay. Samples in Groups 1 and 2 were reanalysed after 4 days. All samples were subjected to the same transport and handling conditions and were uncapped at the same time (t = 0).
All analytes routinely analysed in our laboratory were included in the study. All analyses were performed on Siemens ADVIA or Centaur XP analysers with manufacturer’s reagents and using manufacturer’s protocols (Siemens Healthcare Diagnostics, Frimley, Surrey, UK). Although the laboratory at Leeds General Infirmary has multiple analysers, the same analyser was used for all measurements of each individual analyte. Internal quality control (IQC) samples (Biorad Laboratories Ltd., Hemel Hempstead, Hertfordshire, UK) were analysed every 2–4 hours for all analytes apart from for total triiodothyronine (T3), follicle stimulating hormone (FSH) and luteinizing hormone (LH) (analysed every 24 hours), and for cortisol (analysed every 12 hours). All assays were monitored by external quality assurance schemes and remained in control.
Comparison of the mean baseline concentrations of analytes in patients’ samples with IQC concentrations used to define acceptability limits.
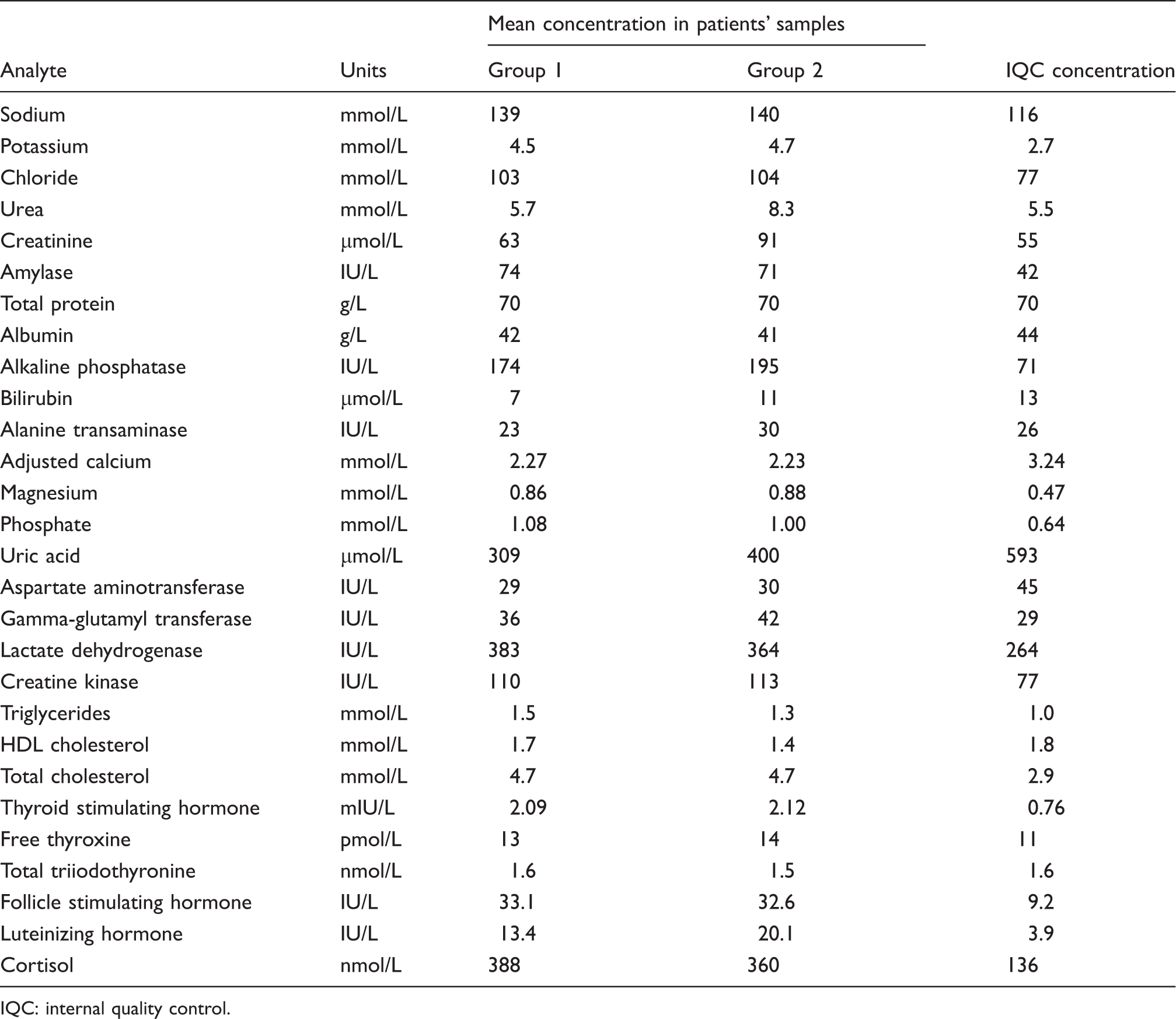
IQC: internal quality control.
Results
Mean difference in concentration between baseline and either 16 hours or 4 days for Group 1 and between baseline and 4 days for Group 2.
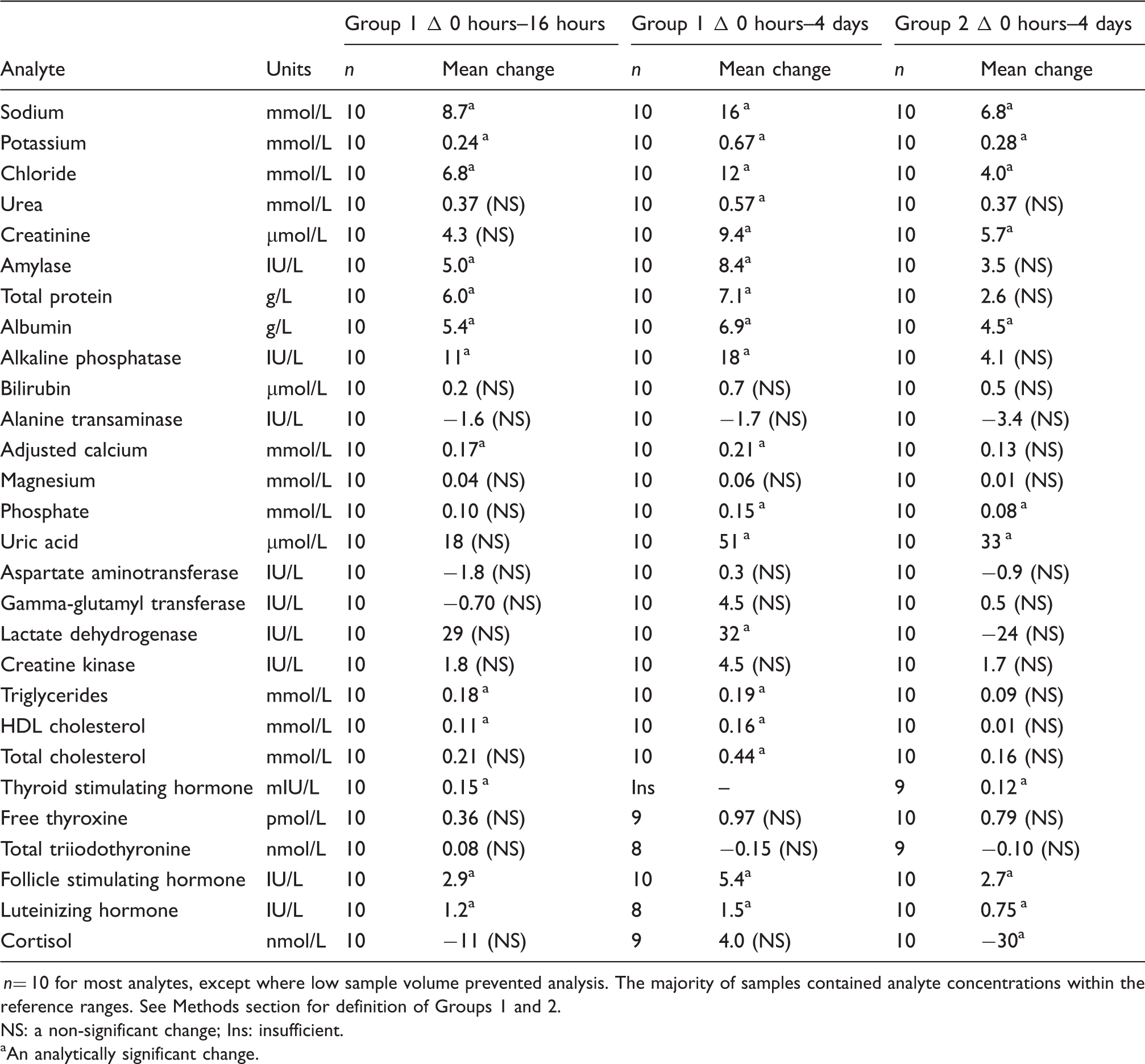
n= 10 for most analytes, except where low sample volume prevented analysis. The majority of samples contained analyte concentrations within the reference ranges. See Methods section for definition of Groups 1 and 2. NS: a non-significant change; Ins: insufficient.
An analytically significant change.
Discussion
Sodium, potassium, chloride, albumin, FSH and LH are not suitable for ‘add-on’ requests if samples are stored in either storage condition, whereas bilirubin, ALT, magnesium, AST, GGT, CK, free T4 and total T3 are stable, and therefore suitable for ‘add-ons’, in both current and optimum storage conditions. Creatinine, phosphate and uric acid are stable for up to 16 hours only; a change to storage of samples in optimum conditions does not significantly increase stability of these analytes and allow analysis after 4 days. However, a change in storage conditions may prevent significant changes in concentration of urea, amylase, total protein, ALP, adjusted calcium, LDH, triglycerides, HDL cholesterol and total cholesterol. These tests could therefore be added on to samples up to 4 days after uncapping if a change in storage conditions was implemented.
Thyroid peroxidase antibodies could not be included in the statistical analysis, as the majority of concentrations were reported as <28 IU/mL. However, the qualitative nature of the result (positive or negative) did not change for any sample at any time point, and changes were therefore considered to be insignificant.
The thyroid stimulating hormone (TSH) assay requires a large sample volume, and as a result many samples in Group 1 did not contain sufficient volume for measurement of TSH after 4 days. However, significant changes in concentration were observed after 16 hours in Group 1 and after 4 days in Group 2. It is therefore likely that significant changes in concentration of TSH would be observed at all time points, and that samples would not be suitable for ‘add-ons’ of TSH if stored in either storage condition.
For the majority of analytes, mean concentrations increased upon delayed analysis. Delayed analysis might be expected to result in a degree of evaporation and therefore an increase in concentration of the analyte, but a significant decrease in concentration was demonstrated in cortisol (in samples from Group 2), while some analytes did not display significant changes in concentration. Net decreases in concentration may be caused by the predominance of analyte degradation over sample evaporation.
There have been other reports in the literature evaluating sample stability.2–4 These studies have assessed analyte stability in capped sample tubes, and it is therefore not surprising that some of the results of these studies do not agree with the findings described here. For example, Dirar et al. 2 state that urea, creatinine, albumin, total protein, total cholesterol, calcium and triglycerides are stable for up to 72 hours following venepuncture, and that storage temperature (4℃ vs. room temperature) did not have any effect on stability. Cuhadar et al. 3 report similar findings by concluding that albumin, total protein, creatinine, cholesterol, triglycerides, GGT, ALP, ALT, CK and LDH are stable for up to 72 hours at both room temperature and at 4℃. However, in contrast, they report that urea is only stable for up to 30 hours, and this is only at 4℃. Oddoze et al. 4 state that storage temperature does not significantly affect stability of the majority of analytes, including potassium, phosphate, magnesium and FSH, but this study was limited to the evaluation of stability over a period of only 24 hours. However, the authors did note that LDH was only stable for a period of 6 hours at 25℃, compared to 24 hours at 4℃.
Differences in reported stabilities may arise for several reasons, such as differences in storage conditions and tube types, but also due to differences in the statistical techniques used to define significant change. Although we have used a method recommended by the Clinical and Laboratory Standards Institute in this study, other methods are available. For example, statistically significant change can be determined by using the Wilcoxon matched-pairs test on non-parametric paired observations. However, this technique does not consider the imprecision of the analytical method and may therefore give misleading results. The total change limit, incorporating both analytical and biological variation, is perhaps one of the most useful determinants of change, as it can be used to determine the clinical significance of any change in result. This approach was not used in this study, as samples were collected only once from each individual.
Although our study evaluates the suitability of ‘add-on’ tests within the time frames of 16 hours and 4 days after uncapping, it does not suggest that the unstable analytes would not be stable for shorter periods of time. This would need to be investigated further. Moreover, it does not answer the question of whether resealing the sample tubes would preserve analyte stability.
Conclusions
Significant differences in concentration were observed for many analytes at all time points in Group 1 and in Group 2, indicating their lack of suitability for ‘add-on’ requests irrespective of storage condition. In current storage conditions, bilirubin, ALT, magnesium, AST, GGT, CK, free T4, total T3 and thyroid peroxidase antibodies are suitable for analysis up to 4 days after uncapping. Creatinine, phosphate and uric acid are stable for up to 16 hours only. A change in laboratory procedure to storage of samples at 4℃ immediately following analysis may allow for ‘add-ons’ of urea, amylase, total protein, ALP, adjusted calcium, LDH, triglycerides, HDL cholesterol and total cholesterol up to 4 days after uncapping. These results may be applicable to other clinical laboratories where samples are stored uncapped at room temperature for significant periods of time.
Footnotes
Declaration of conflicting interests
None declared.
Funding
This research received no specific grant from any funding agency in the public, commercial, or not-for-profit sectors.
Ethical approval
Not required.
Guarantor
JHB.
Contributorship
RLK and JHB researched the literature, conceived the study and compiled the manuscript. CB was involved in study design. CB, RL, FB, CK and JB provided technical advice. All authors reviewed and edited the manuscript and approved the final version.
