Abstract
Familial hypercholesterolaemia, one of the most common inherited diseases in the general population, is associated with mutations in at least three different genes including the low density lipoprotein receptor (
Background
Hypercholesterolaemia is a major risk factor for atherosclerosis and its cardiovascular complications. Familial hypercholesterolaemia (FH; OMIM#143890), usually an autosomal dominant condition, is characterized by very high plasma concentrations of cholesterol and LDL cholesterol, tendon xanthomas and increased risk of premature coronary heart disease. Mutations in at least three genes have been identified to be associated with FH, including the low density lipoprotein receptor (
The majority of FH cases are associated with mutations in the
Case report and family history
The proband (II.3; Figure 1), a Caucasian male, was initially referred to the lipid clinic at the age of 38 years after his brother’s fatal heart attack in his early 40s. At the time of presentation, the proband was asymptomatic with an unremarkable past medical history. On examination, he was found to have bilateral corneal arcus and thickening was detectable in his Achilles tendons. Blood pressure was 118/76 mmHg; 12-lead electrocardiography was unremarkable before and after a vigorous exercise tolerance test. His initial fasting serum total cholesterol concentration was 12.8 mmol/L with HDL-C, triglycerides (TG) and LDL-C concentrations of 1.1, 1.2 and 11.2 mmol/L, respectively. A clinical diagnosis of FH was made on the basis of the Simon Broome criteria, which are commonly used in the UK for the management of patients suspected to have FH.
7
His best lipid profile results to date (total cholesterol 5.3, HDL-C 1.1, TG 1.3 and LDL-C 3.6 mmol/L) were achieved using a combination of rosuvastatin 40 mg, ezetimibe 10 mg and colestyramine 12 g daily. Very little is known about the proband’s parents except that his mother (I.2) died of myocardial infarction at the age of 75 years, whereas the age and cause of his father’s death are unknown. The proband’s only sibling (II.1; Figure 1) had his first ischemic heart attack at the age of 43 years and died approximately a year later as a result of a second episode of myocardial infarction. Mutation analysis was not carried out for the proband’s brother. Unfortunately, the latter’s medical records are not accessible; his lipid profiles and treatment are also unknown to the existing family members. He was survived by two daughters (Figure 1). The proband’s elder niece (III.2) was found to have a serum total cholesterol concentration of 7.9 mmol/L at the age of 26 years but no detailed lipid profile was performed before commencement of her lipid-lowering treatment. At the time of writing, she was 43 years old and well. The proband’s grandniece (IV.1) was also found to have relatively high fasting cholesterol concentrations at the age of 12 years (total cholesterol 7.3 mmol/L, HDL-C 1.5 mmol/L, TG 1.3 mmol/L and LDL-C 5.2 mmol/L). The proband’s younger niece (III.3) has declined testing of her cholesterol concentrations and genetic mutation. Lipid profiles of other family members are not available.
Kindred with c.1813C>T variant in the low density lipoprotein receptor (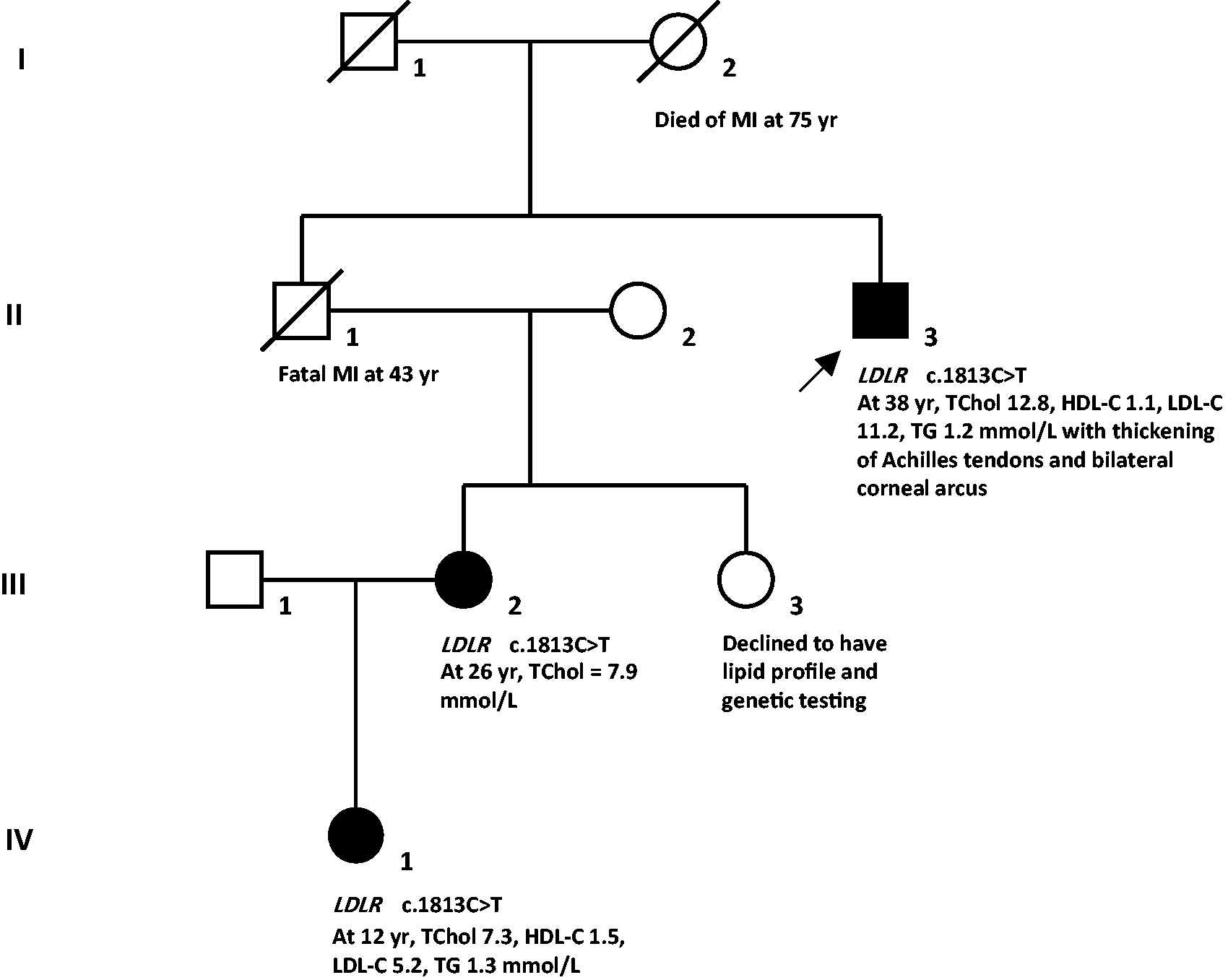
Peripheral blood samples were collected from patients with informed consent. Materials and methods are available as supplementary information on the journal’s website. Sequence analysis of all exons of the
Given the above synonymous mutation and potential cryptic donor splice site, total RNA was extracted from the proband’s peripheral blood and reverse transcribed. Subsequent analysis of cDNA confirmed that the sequence variant c.1813C>T resulted in the creation of a donor splice site, leading to the loss of 34 base pairs from exon 12 in the coding sequence (Figure 2). The latter truncated mRNA is predicted to cause a frameshift leading to a premature stop at codon 652 and thus early termination of LDLR translation.
Sanger sequence analysis of cDNA from control (upper panel) and proband (lower panel) showing the heterozygous cDNA variant as a result of the loss of 34 bases from exon 12 in the 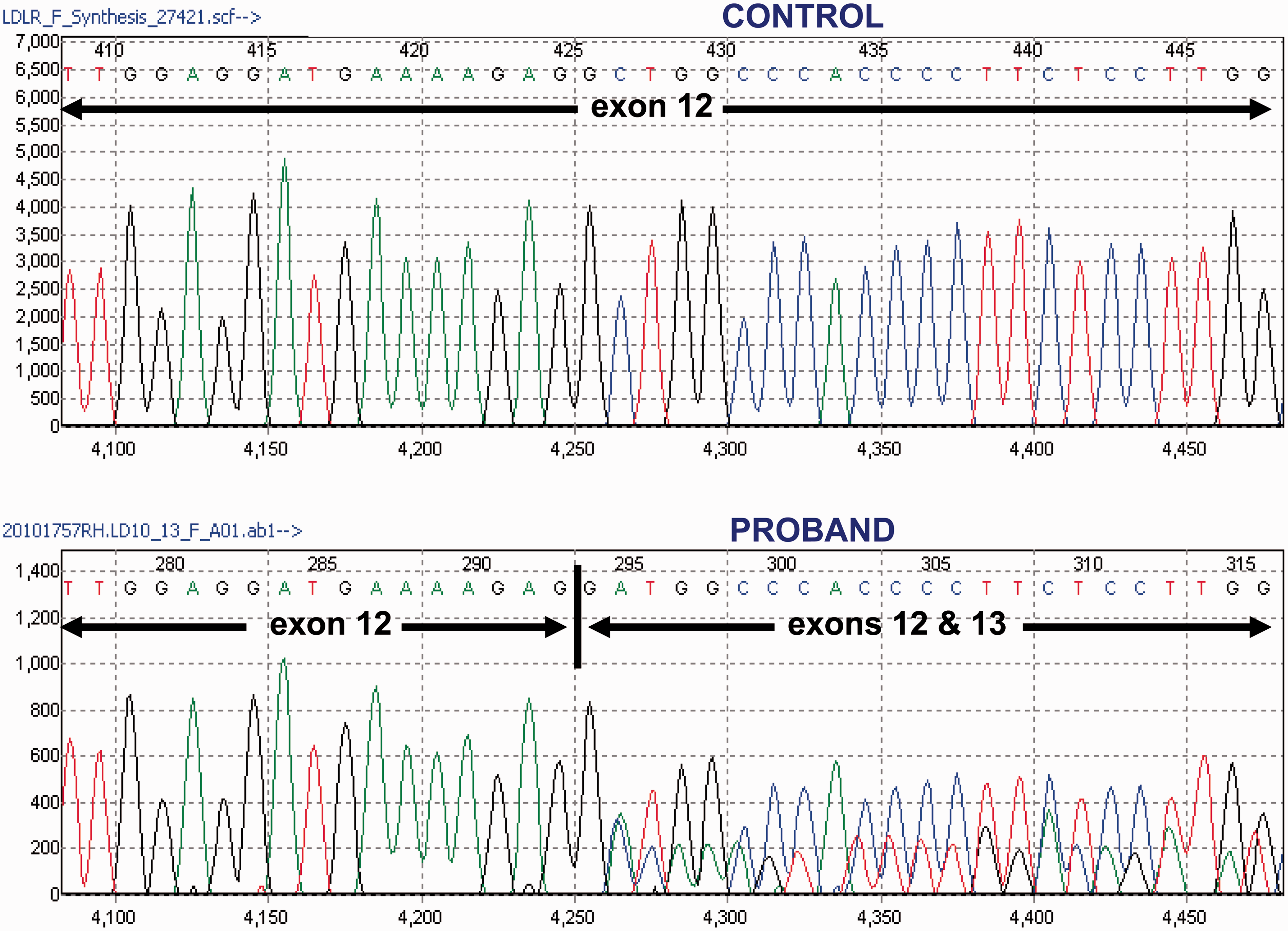
Discussion
To our knowledge, the
In the present study, the proband’s only sibling (II.1) died at 43 years of age due to an ischaemic heart event with an unknown lipid phenotype. Since the latter’s daughter (III.2) and brother (proband; II.3) carry the same c.1813C>T variant and were hypercholesterolaemic before treatment, it is highly probable that he also had the same variant with consequent hypercholesterolaemia, contributing to his premature ischaemic heart disease. However, severe hypercholesterolaemia caused by the heterozygous c.1813C>T mutation might not have been the only factor leading to the premature ischaemic heart disease and death of the proband’s brother. It is possible that, in addition to hypercholesterolaemia, other cardiovascular risk factors such as cigarette smoking, hypertension, diet and obesity might have contributed to the latter’s premature ischaemic heart disease. He might also have mutations in other gene(s) that can increase the risk of myocardial infarction. 9
Compared to the other family members who are also known to be
The present report also highlights the importance of RNA studies for patients who are suspected of carrying genetic mutations that may affect mRNA splicing and thus subsequent protein translation. In recent years, isolation of stable and high-quality RNA from patients’ peripheral blood has become clinically feasible because of the availability of improved technology for blood collection and RNA extraction. Blood collection tubes suitable for RNA transport at room temperature with minimal RNA degradation are now widely available. It is anticipated that RNA studies will become increasingly used in routine clinical lipidology service and clarify the significance of some unclassified variants for the molecular diagnosis ofFH.
In conclusion, in this report we have identified a previously unclassified variant of the
Footnotes
Declaration of conflicting interests
None declared.
Funding
This research received no specific grant from any funding agency in the public, commercial, or not-for-profit sectors.
Ethical approval
Informed consent was obtained from the patients. No ethical approval was required.
Guarantor
SWW.
Contributorship
CKMH wrote and edited the manuscript. FRM wrote the first draft of the manuscript. CB performed the molecular assays. SWW conceived the study. All authors reviewed the manuscript and approved the final version.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
