Abstract
Background
Decreased serum concentrations of vitamin B12 are associated with Alzheimer’s type dementia. The transcobalamin II gene (TCN2) 776C→G polymorphism affects transcobalamin II function as a carrier of vitamin B12 and might modify its availability. The association of the TCN2 776C→G polymorphism with Alzheimer’s type dementia is unclear and was investigated in the present study.
Methods
Case–control study including 27 individuals diagnosed with Alzheimer’s type dementia and 28 healthy controls. Serum concentrations of vitamin B12, homocysteine and other analytes were determined and the presence of TCN2 776C→G and 5, 10-methylenetetrahydrofolate reductase 1298A→C polymorphisms genotypes was ascertained by polymerase chain reaction–restriction fragment length polymorphism.
Results
Serum concentrations of vitamin B12 were lower while those of homocysteine were higher in patients than in controls (P < 0.05). The frequency of individuals carrying at least one 5, 10-methylenetetrahydrofolate reductase 1298C allele was higher (59% versus 32%) while frequency of individuals harbouring at least one TCN2 776G allele was lower (58% versus 86%) in patients than in controls (P < 0.05). Univariate logistic regression showed negative association of TCN2 776CG genotype with Alzheimer’s type dementia (OR = 0.17 versus CC genotype, P < 0.02). Multivariate logistic regression identified TCN2 776C→G polymorphism as independent predictor of Alzheimer’s type dementia together with higher concentrations of homocysteine, cholesterol and uric acid and lower concentrations of oestradiol. Association of TCN2 776C→G polymorphism with Alzheimer’s type dementia was observed for individuals carrying the 5,10-methylenetetrahydrofolate reductase 1298AA genotype but not the AC or CC genotypes, indicating interaction between the two polymorphisms.
Conclusions
The TCN2 776C→G polymorphism is negatively associated with Alzheimer’s type dementia, suggesting a protective role against the disease in subjects with the 5, 10-methylenetetrahydrofolate reductase 1298AA genotype.
Keywords
Introduction
Alzheimer’s disease (AD) is a multifactorial neurodegenerative pathology involving a gradual deterioration of cognitive functions and its increasing prevalence constitutes a burden to contemporary society.1,2 Histologically it is characterized by amyloid-β peptide deposits, neurofibrillary tangles of hyperphosphorylated tau protein and increased neuronal loss. 1 Alterations in several metabolic and hormonal parameters and impairment of some physiological functions are associated with AD and other dementias and have been proposed as risk factors for these diseases. Specifically, high serum concentrations of cholesterol, low serum concentrations of oestradiol as well as increased concentrations of uric acid and urea (probably reflecting moderate impairment of renal function) have been observed in AD patients.3–8
Impaired homocysteine and one-carbon metabolism are also commonly observed in AD patients. Homocysteine is derived from hydrolysis of S-adenosylhomocysteine, a product of S-adenosylmethionine-dependent methyltransferases catalysed reactions. S-adenosylmethionine is in turn synthesized from methionine. 9 Hyperhomocysteinemia was reported as an independent risk factor for AD10,11 and may result from dietary deficiencies (especially vitamin B12 and folate) and/or genetic alterations in enzymes involved in one-carbon metabolism (for review see Stipanuk 9 ). 5, 10-methylenetetrahydrofolate reductase (MTHFR) catalyses the reduction of 5, 10-methylenetetrahydrofolate to 5-methyltetrahydrofolate, the main circulating form of folate and the methyl donor for the vitamin B12-dependent remethylation of homocysteine to methionine. 9 Low concentrations of folate and vitamin B12, as well as genetic variants of MTHFR, are frequently found in individuals with AD and have been proposed as risk factors for the disease.11–13
Circulating vitamin B12 is bound to haptocorrin and transcobalamin II (TC). About one-third of circulating B12 is bound to TC (forming holo-TC) constituting the biological active fraction of vitamin B12 which is delivered to all tissues in the body.14–16 TC is encoded by the TCN2 gene for which have been described several single nucleotide polymorphisms (SNP). Of the TCN2 SNPs, the 776C→G (dbSNP ID: rs1801198) base substitution, originating the P259R missense mutation in TC, has been reported as common and potentially modifying vitamin B12 availability and homocysteine concentration,15,17–19 though controversy exists about the effect of this SNP. 20
The potential of the TCN2 776C→G polymorphism to modify vitamin B12 availability, and therefore interfering with homocysteine and one-carbon metabolism, might link this SNP to AD. However, the association between this SNP and AD is unclear. With the present work we intended to investigate the association of the TCN2 776C→G polymorphism with Alzheimer’s type dementia (ATD) in a Portuguese elderly population. We also studied if the association of this SNP with ATD is independent of serum concentrations of homocysteine, cholesterol, oestradiol and other related biochemical and genetic parameters associated with ATD. Since both the TCN2 776C→G and MTHFR 1298A→C polymorphisms influence one-carbon metabolism, the possibility of gene–gene interaction between the two polymorphisms should be considered and was also investigated in the present study.
Methods
Subjects
The participants in the study were selected from individuals living in the Cova da Beira region in Portugal and consisted of 27 patients diagnosed with ATD (14 female and 13 male, mean age of 75 years) and 28 healthy older adults (16 female and 12 male, mean age of 71 years). All individuals were over 50 years of age. All females were postmenopausal and had not taken hormone replacement therapy. Females were classified as postmenopausal once they had experienced at least 12 consecutive months of amenorrhoea.
The patients were recruited from the Department of Neurology of the Cova da Beira Hospital. Cognitive impairment was evaluated by several tests, including Mini-Mental State Examination. 21 Diagnosis was based on neurological and neuropsychological examination and complemented with neuroimaging information (brain CT and/or MRI scans). The diagnostic of ATD was made according to the criteria of the ‘National Institute of Neurological and Communicative Disorders and Stroke/Alzheimer’s Disease and Related Disorders Association’, 22 for probable AD. Patients with concomitant cerebrovascular disease were excluded from the study.
The control group was composed of elderly volunteers, without symptoms of memory impairment or suffering from neurological diseases, which were recruited from healthy non-blood relatives accompanying patients to clinical appointment at the hospital and also (the majority) from general practices in the Cova da Beira Health Authority area. Control individuals were included in the study after providing information on their health status and undergoing routine biochemical measurements and physical examination. In order to eliminate known conditions that interfere with serum concentrations of homocysteine, oestrogens and vitamins, both patients and healthy individuals were excluded from the study if they suffered from hypertension, cardiovascular disease, renal or hepatic diseases, diabetes, hypo or hyperthyroidism or hypercholesterolemia; if they were smokers, had alcohol dependence or had undergone recent surgery and if they have taken corticosteroids or vitamin supplements within the previous two months. Individuals suffering from malnutrition or serious feeding problems were also excluded from the study.
The study was approved by the local ethical committee. The subjects volunteered for the study and gave their informed consent.
Biochemical assays
Blood samples were collected between 8 am and 9 am after overnight fast and a minimum period of rest of 8 h. Blood was collected either into plain (for biochemical analysis) or EDTA (for DNA extraction) tubes. Serum was promptly separated from blood samples without EDTA by centrifugation (15 min, 4000 rotations per minute [r/min]) at 4 ℃, divided into aliquots and stored at −20 ℃ until analysis. Serum homocysteine concentration was measured by fluorescence polarization immunoassay, using an Abbott AxSYM® system auto-analyser (Abbott Laboratories, North Chicago, IL, USA). Serum folate and serum vitamin B12 concentrations were determined by competitive binding assays with electrochemiluminescence detection, using an Elecsys 2010 auto-analyser (Roche Diagnostics, Mannheim, Germany). Serum creatinine and urea concentrations were measured using an IL-600® auto-analyser (Instrument Laboratory, Warrington, UK), serum glucose concentration was measured using a COBAS INTEGRA® 400 plus auto-analyser (Roche Diagnostics, Mannheim, Germany), serum concentrations of uric acid and total cholesterol were measured using a COBAS® C 501 auto-analyser (Roche Diagnostics, Mannheim, Germany) and serum oestradiol concentration was measured using an Abbott AxSYM® system auto-analyser (Abbott Laboratories, North Chicago, IL, USA). All measurements were performed as per the manufacturers’ instructions and with reagents supplied by the manufacturers.
Genetic analysis
Leucocytes were isolated from peripheral blood by adding five volumes of Red Blood Cells (RBCs) Lysis Buffer (NH4Cl 154 mmol/L, NaHCO3 14 mmol/L, EDTA 0.1 mmol/L, pH 7.3) to a blood sample collected with EDTA, and incubated at room temperature for 3–5 min. During the incubation the sample was vortexed several times. The sample was then centrifuged at 2000 r/min for 5 min and the supernatant decanted. An additional volume of the RBCs Lysis Buffer was then added to the pelleted white blood cells and the process repeated twice. The pelleted leukocytes were then used for DNA extraction. Genomic DNA was isolated from peripheral blood leucocytes and the MTHFR 1298A→C and TCN2 776C→G polymorphisms genotypes were analysed by PCR-RFLP, as previously described.17,23 Briefly, 500 ng of genomic DNA was incubated in a total reaction volume of 50 µL containing final concentrations of 500 nmol/L of the forward and the reverse primers, 200 µM each dNTP, 10 mM Tris–HCl pH 8.3, 50 mmol/L KCl, 2 mmol/L MgCl2, 0.05% detergent and 1 unit Taq DNA polymerase (Life Technologies, Grand Island, NY, USA). For the MTHFR 1298A→C polymorphism, the forward primer was 5′-CTT TGG GGA GCT GAA GGA CTA CTA C and the reverse primer was 5′-CAC TTT GTG ACC ATT CCG GTT TG, while for the TCN2 776C→G polymorphism the forward primer was 5′-TCA GGG GTG GAG TGG TCA G and the reverse primer was ATT TTG GCT GCT GTG TCC C. PCR conditions were optimized for the Omnigene (Hybaid, Middlesex, UK) apparatus and included denaturation at 94 ℃, annealing at 55 ℃ and extension at 72℃. The MTHFR 1298A→C variant abolishes an MboII site and the TCN2 776C→G variant abolishes a ScrF1 site. After restriction enzyme digestion, PCR products were evaluated by gel electrophoresis analysis.
Statistical analysis
Statistical analysis was performed using SPSS for Windows program version 16.0 (SPSS Inc., Chicago, IL, USA). Data are expressed as mean (normal variables) or median (non-normal variables) with 95% confidence intervals. Normality of the data was assessed by the Shapiro–Wilk test. Differences between two means were analysed by the two-tailed unpaired Student’s t-test or by multi-way ANOVA when adjusting for one or more factors. Differences between more than two means were analysed by one-way or multi-way ANOVA. Differences between medians were analysed by the Mann–Whitney U test. Differences between relative frequencies were analysed by the χ2 and the Fisher’s exact tests.
Association of each studied variable with ATD was analysed by logistic regression analysis. Multiple logistic regression analysis was also performed to determine which variables are the best predictors of ATD. Interaction between pair of variables, in the association with ATD, was evaluated by introducing interaction terms, consisting of cross-products of the variables, in a multiple regression model. Statistical significance of the calculated odds ratios and interaction coefficients were analysed by the Wald and likelihood ratio tests. Levels of statistical significance were set at P ≤ 0.05.
Correction for multiple testing was performed by the method described by Storey and Tibshirani, 24 setting the false discovery rate (δ) equal to 0.05, using the Qvalue version 1.38.0 package for the R program version 3.1.0 (The R Foundation for Statistical Computing, Vienna, Austria).
Results
Characteristics of the studied subjects.
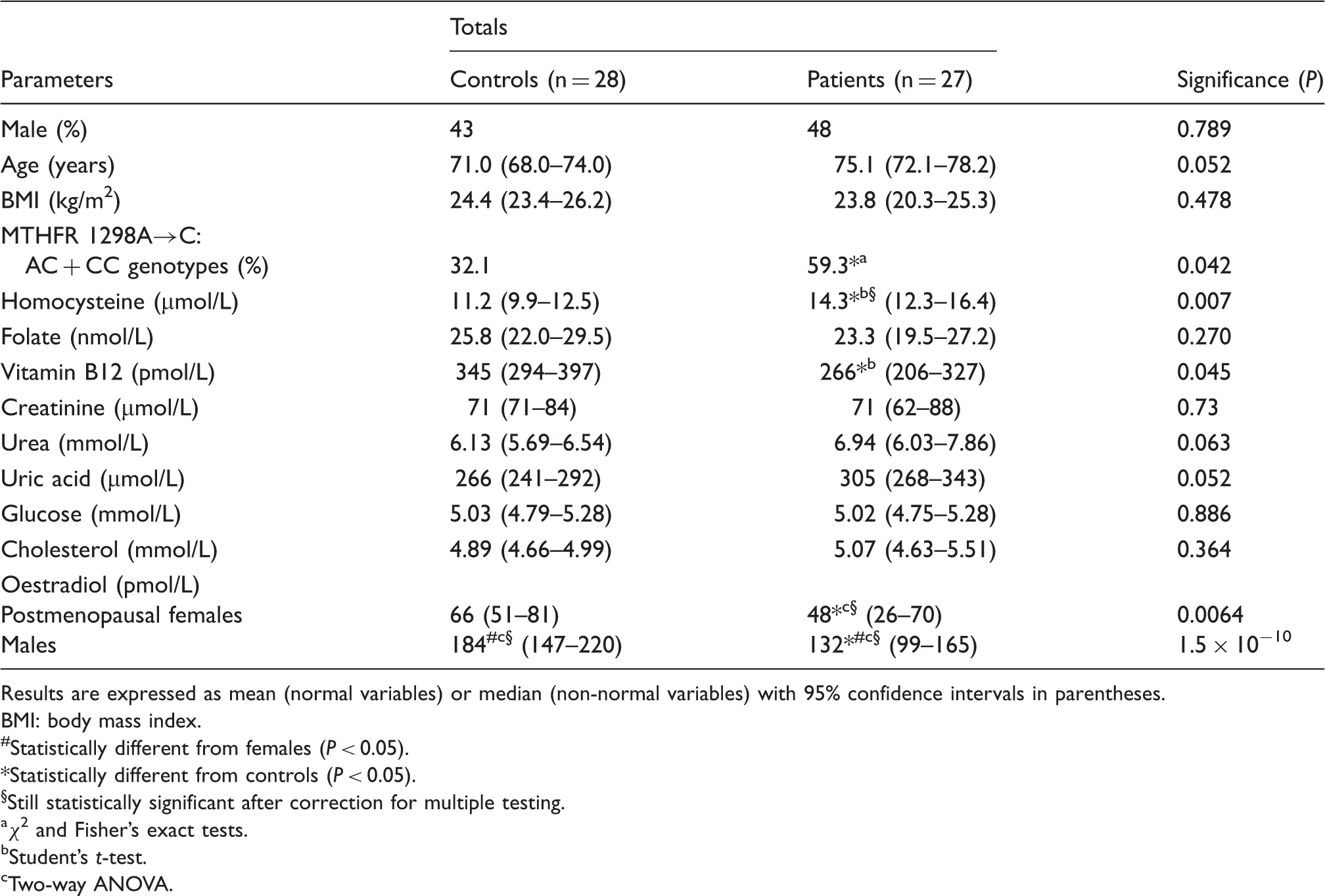
Results are expressed as mean (normal variables) or median (non-normal variables) with 95% confidence intervals in parentheses.
BMI: body mass index.
Statistically different from females (P < 0.05).
*Statistically different from controls (P < 0.05).
§Still statistically significant after correction for multiple testing.
χ2 and Fisher’s exact tests.
Student’s t-test.
Two-way ANOVA.
The relative frequencies of genotypes for the MTHFR 1298A→C polymorphism were 54.5% (AA homozygotes), 43.6% (AC heterozygotes) and 1.8% (CC homozygotes). The overall relative frequency of the C allele was 23.6%. The observed frequency distribution of the genotypes did not significantly deviate from the Hardy–Weinberg equilibrium (P > 0.3). Since the observed frequency of the MTHFR 1298CC genotype was very low, the individuals with at least one C allele (AC or CC genotypes) were combined for further analysis. The percentage of individuals carrying at least one C allele of the MTHFR 1298A→C polymorphism tended to be higher in patients than in controls (Table 1).
TCN2 776C→G polymorphism genotype frequencies.
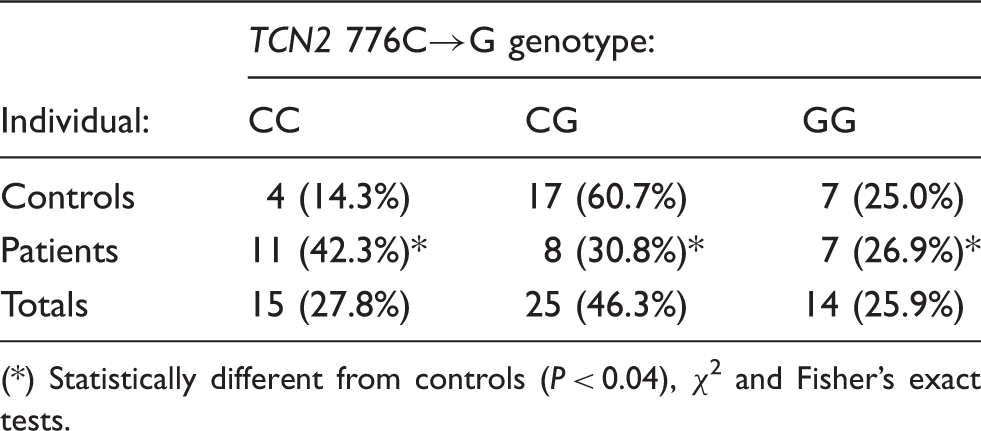
(*) Statistically different from controls (P < 0.04), χ2 and Fisher’s exact tests.
Serum concentrations of vitamin B12 did not vary significantly across TCN2 776C→G genotypes (P > 0.4, one-way ANOVA), even after adjusting for gender, age and subject (patient or control) (P > 0.4, multi-way ANOVA). Vitamin B12 serum concentrations (pM) were 267 (187–347) (mean and 95% confidence interval), 326 (272–380) and 317 (218–415), respectively, for the CC, CG and GG genotypes of the TCN2 776C→G polymorphism.
Odds ratios (OR) of ATD for different factors, obtained by univariable logistic regression (27 patients, 28 controls).
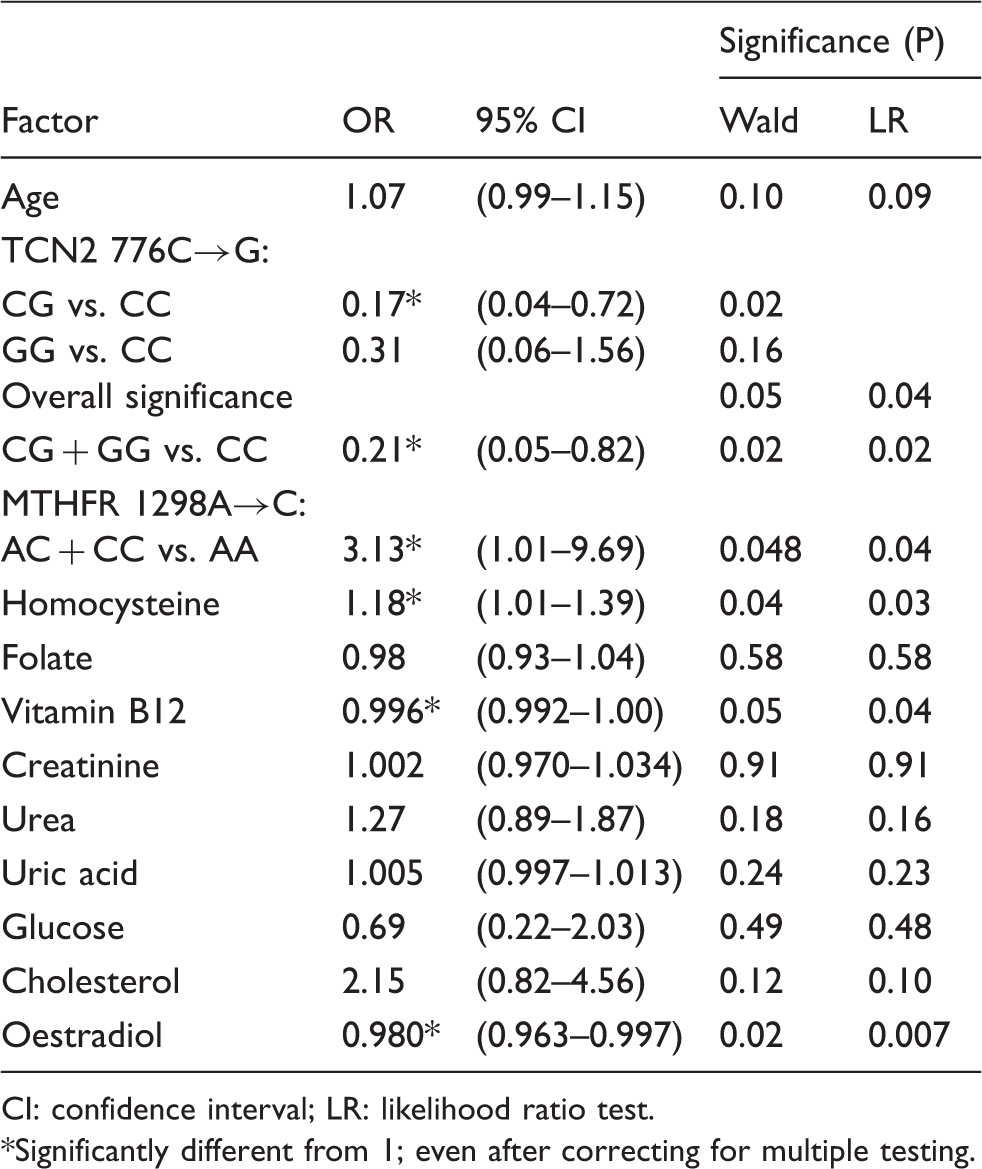
CI: confidence interval; LR: likelihood ratio test. *Significantly different from 1; even after correcting for multiple testing.
Multivariable logistic regression analysis was performed (stepwise backward method) to determine which variables are best independent predictors of ATD. The variables selected as candidates were all variables that had P values less than 0.25 in the univariable logistic regression analysis (Table 3) plus gender. The best model obtained identified the TCN2 776C→G polymorphism (CG + GG versus CC genotypes) as an independent predictor of ATD (OR = 0.043; 95% CI: 0.004–0.51; P < 0.003, Likelihood Ratio test) together with homocysteine (OR = 1.43; CI: 1.01–2.04; P < 0.013), uric acid (OR = 1.020; CI: 1.003–1.038; P < 0.007), cholesterol (OR = 9.5; CI: 1.5–56.5; P < 0.001) and oestradiol (OR = 0.95; CI: 0.92–0.99; P < 0.0002). The results were not modified after adjusting for age, gender and vitamin B12.
Dependence of the TCN2 776C→G polymorphism association with ATD on the MTHFR 1298A→C genotype.
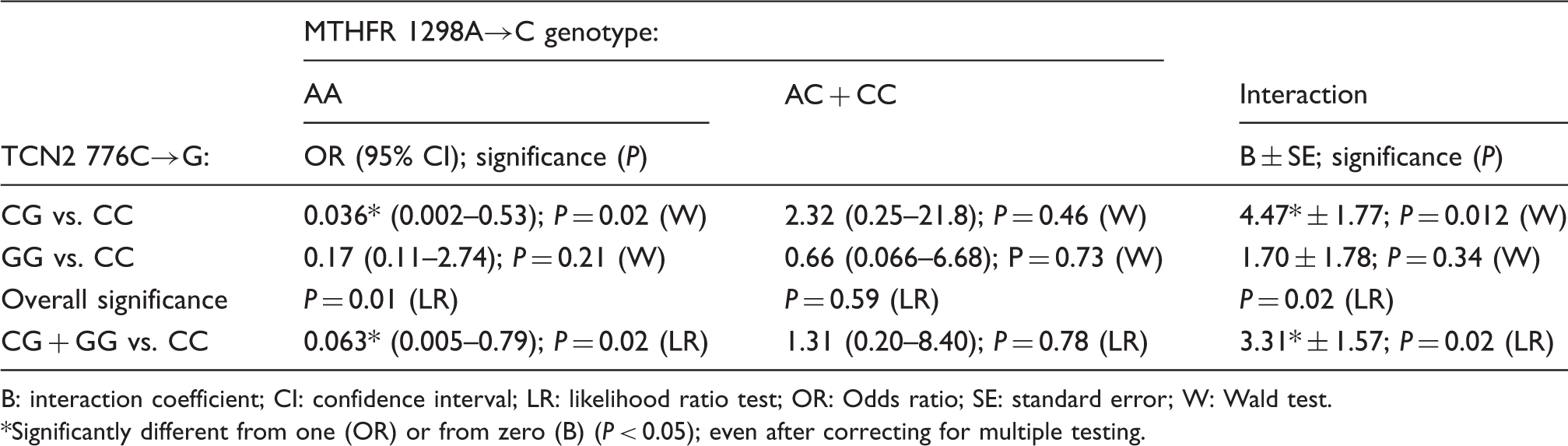
B: interaction coefficient; CI: confidence interval; LR: likelihood ratio test; OR: Odds ratio; SE: standard error; W: Wald test. *Significantly different from one (OR) or from zero (B) (P < 0.05); even after correcting for multiple testing.
Discussion
The present study shows, for the first time, that the TCN2 776C→G polymorphism in the TC gene is negatively (inversely) associated with ATD, suggesting that the allele G of this polymorphism might be protective against the disease. This association was independent of other studied risk factors also associated with ATD but dependent on the MTHFR 1298A→C polymorphism genotype.
Altered one-carbon metabolism in ATD
Transmethylation reactions are required for maintenance of normal function of the nervous system and increasing evidence of altered one-carbon metabolism in ATD has been reported.10–13 Maintaining S-adenosylmethionine availability – the main methyl group donor for methyltransferases catalysed reactions – requires remethylation of homocysteine back to methionine through the action of methionine synthase, a vitamin B12-dependent enzyme which uses 5-methyltetrahydrofolate as methyl donor. 9 Thus, reduced concentrations of vitamin B12 and folate, as well as genetic variants of the MTHFR (which converts 5, 10-methylenetetrahydrofolate into 5-methyltetrahydrofolate, the main circulating form of folate) with reduced activity/stability, impair remethylation of homocysteine back to methionine, therefore increasing homocysteine concentrations, and have been proposed as risk factors for ATD.11–13 Accordingly, in the present study, a higher serum concentration of homocysteine, a lower concentration of vitamin B12 and the MTHFR 1298A→C genetic polymorphism were associated with ATD, supporting previous findings.3,11,13
Association of the TCN2 776C→G genetic polymorphism with ATD
Variants of TC might potentially influence cellular availability of vitamin B1215,17,18 and thus contribute to the association between disrupted one-carbon metabolism and ATD. Therefore, the association of the TCN2 776C→G polymorphism with ATD, together with other factors involved in one-carbon metabolism, was addressed in the present study.
The frequency of the G allele of the TCN2 776C→G polymorphism found in the elderly population sample studied in the present work (49%) as well as the genotype distribution (28% for CC, 46% for CG and 26% for GG genotypes) was very similar to those reported for another independent Portuguese population sample (48% for G allele frequency; 31% for CC, 41% for CG and 28% for GG genotypes 17 ) of younger subjects. The 776G allele frequency found for the Portuguese population was also similar to those reported for the French (45%) or Dutch (47%) Caucasian populations,25,26 but higher than that reported for Sicilian population of Italy (35%; see Guéant et al., 2007 25 ).
The negative association of the TCN2 776C→G polymorphism with ATD was independent of the association with ATD of other studied factors (related or not to one-carbon metabolism). This conclusion was supported by the observation that the TCN2 776CC genotype, together with higher concentrations of homocysteine, cholesterol and uric acid, and lower concentrations of oestradiol, emerged as independent predictors of ATD in a multiple logistic regression model. The fact that the negative association of the TCN2 776C→G polymorphism with ATD was independent of the association of homocysteine with ATD suggests that the influence of this SNP on ATD might not be mediated by modification of homocysteine concentration. 20
The observation that TCN2 776G allele is negatively associated with ATD, suggesting that it might have a protective role against ATD onset, is somewhat unexpected, since this SNP has been previously reported to be associated with lower holo-TC and higher homocysteine serum concentrations,17,18,27 though controversy exists.20,28 However, the findings obtained in the present study agree with a recent worldwide study reporting that the TCN2 776G allele might confer an adaptive advantage to some populations. 25 In fact, the frequency of this allele shows worldwide variability, ranging from a low prevalence in African population (18%) to an high prevalence in Asian population, namely in central China (61%) with an intermediate prevalence in Afro-American, Caucasian and Hispanic populations. 25
To our knowledge only two previous studies have investigated the association of the TCN2 776C→G polymorphism with ATD. One of these reported a positive association of the TCN2 776C→G polymorphism with ATD 29 in a combined UK (clinical study) and Swedish (histological study) populations. However, in this study most subjects included as controls had died from cardiac or cancer-related diseases. Another study did not observe any association of this SNP with ATD in a Sicilian population. 30
The total vitamin B12 concentration did not differ across the TCN2 776C→G genotypes, in accordance with previously reported results,17,18 suggesting that a potential protective effect of this SNP on ATD might not be mediated by modification of total vitamin B12 concentrations (but see Stanisławska-Sachadyn et al., 2010 31 ). Interestingly, a recent study involving a North American Hispanic elderly population 28 reported lower concentrations of homocysteine associated with the TCN2 776G allele in individuals with lower concentrations of total vitamin B12 or holo-TC, suggesting a potential protective role of this allele as an effect modifier of the relationship between vitamin B12 status and homocysteine.
In the present study, statistically significant association between the TCN2 776C→G polymorphism and ATD was evident even after correction for multiple testing, which underlines the relevance of this association.
Interaction between TCN2 776C→G and MTHFR 1298A→C polymorphisms
Since both TCN2 776C→G and MTHFR 1298A→C polymorphisms were associated with ATD and both influence one-carbon metabolism, the possibility of gene–gene interaction should be considered and was investigated in the present study. Negative association of the TCN2 776C→G polymorphism with ATD was only observed in individuals carrying the MTHFR 1298AA genotype, while it was absent in subjects harbouring the 1298C allele (AC + CC genotypes), suggesting an interaction between the two polymorphisms. The results therefore suggest that a potential protective effect of the TCN2 776G allele might be offset in populations with high frequency of the MTHFR 1298C allele. However, the occurrence of an interaction between the two polymorphisms will require confirmation in further studies with larger sample sizes.
The MTHFR gene 1298 A→C mutation, in the exon 7, causes a glutamate to alanine substitution in the MTHFR protein, probably in a regulatory domain, resulting in a decreased MTHFR activity but not modifying the thermostability of the protein. 32 In the case of the TCN2 gene, the 776 C→G mutation, located in exon 6, produces a proline to arginine substitution in position 259 of the TC protein (P259R), which probably will not affect either the binding of cobalamin to TC, the stability of the protein or the recognition of holo-TC by its cell surface receptor.33,34 However, the molecular mechanism explaining the interaction between the two polymorphisms cannot be explained with the present knowledge and deserves further investigation.
Conclusions
The present study shows that the frequency of genotypes with at least one TCN2 776G allele is lower in individuals with ATD. The association of this SNP with ATD was independent of other metabolic and hormonal risk factors but dependent on the MTHFR 1298A→C polymorphism genotype, suggesting a protective role of the TCN2 776C→G polymorphism on the onset of ATD in individuals with the MTHFR 1298AA genotype.
Footnotes
Declarations of interests
None declared.
Funding
This research was funded by the University of Beira Interior.
Ethical approval
The ethics committee of ‘Centro Hospitalar Cova da Beira’ approved this study (ethical approval number: 50/2006).
Guarantor
JFC.
Contributorship
JFC, RC, FCD and SA researched literature and conceived the study. MG and MB obtained the data. RC and SA were involved in protocol development. MP was responsible for patient recruitment. JFC and FCD were involved in gaining ethical approval. JFC, AS and ACD were involved in data analysis. JFC wrote the first draft of the manuscript. All authors reviewed and edited the manuscript and approved the final version of the manuscript.
