Abstract
Background
Cardiac troponins (cTns) are the ‘gold standard’ biomarker for the diagnosis and prognosis of acute coronary syndrome. Analytical performance is critical at low concentrations of cTn, and many of the current assays do not meet the guideline requirement of a 10% coefficient of variation (CV) at the 99th percentile concentrations. The aim of the study was to establish if the newly released Access® AccuTnI®+3 (AccuTnI+3) cardiac troponin I assay (Beckman Coulter Inc., Brea, CA, USA) reached this objective.
Methods
All AccuTnI+3 assays were performed on UniCel® DxI800 analyzer (Beckman Coulter Inc). Limit of Blank (LoB), Limit of Detection (LoD) and Limit of Quantitation (LoQ) were determined according to Clinical Laboratory Standard Institute EP17-A and EP5-A2 protocols. The 99th percentile upper reference limit (URL) was determined by analysing serum samples from 330 apparently healthy blood donors (260 men, 70 women, age range 18–70 years, median age 36 years).
Results
LoB and LoD values were 2.6 and 12 ng/L, respectively. The 10% CV was at 18 ng/L (95% confidence interval [CI] 8–25). The 99th percentile URL was 22 ng/L (95% CI 11–34).
Conclusions
The newly released assay has improved low-end analytical performance and reaches the goal of having a total imprecision ≤10% at 99th percentile of a healthy reference population (guideline acceptable). With this assay, it is now possible to utilize the 99th percentile as decision level for myocardial injury detection.
Introduction
Cardiac troponins (cTns) are the standard biomarkers for the detection of myocardial injury, risk stratification in patients suspected of acute coronary syndrome (ACS) and myocardial infarction (MI) diagnosis. 1
The third Universal Definition of MI establishes that cTns (I or T) are the preferred biomarker overall and for specific category of MI based on high myocardial tissue specificity as well as high clinical sensitivity. 2
An increased cTn concentration is defined as a value exceeding the 99th percentile of normal reference population consisting of apparently healthy subjects free from heart disease.2,3 Guidelines recommend that such a decision level must be measured with an imprecision less than or equal to 10% coefficient of variation (%CV).2–6 Apple 7 , in his scorecard, defined a test with total imprecision ≤10% at the 99th percentile guideline as acceptable. In our study, we evaluated the low-end analytical performance of the newly released Access® AccuTnI®+3 cardiac Troponin I assay from Beckman Coulter Inc. (Brea, CA, USA) according to Clinical Laboratory Standard Institute (CLSI) EP5-A and CLSI EP17-A protocols.8,9 We also calculated the 99th percentile of an apparently healthy reference population using this novel version of the AccuTnI assay. The aim of the study was to establish if the Access® AccuTnI®+3 (AccuTnI+3) assay reached the objective of having optimal analytical performance (%CV ≤ 10) at the 99th percentile, as recommended by Apple 7 in his scorecard approach.
Methods
Access®AccuTnI® (AccuTnI) is a chemiluminescence immunoassay 10 used in our laboratory for cTnI detection for the last 10 years. On September 2013, Beckman Coulter Inc. released a new AccuTnI+3 assay for UniCel® DxI Immunoassay System. AccuTnI+3 is not a reformulated assay: all reagents, antibody pairs and calibrators remained the same as those used in the AccuTnI assay. The differences between the two assays are as follows: modifications to the UniCel® DxI calibrator value assignment to harmonize patient results with Access immunoassay platform, addition of an algorithm to adjust results for changes in room temperature and modifications to the assay protocol file which alter mixing and probe wash processes to improve analytical performance and lot-to-lot consistency. 11
All tests were performed on UniCel® DxI800 analyzer (Beckman Coulter Inc.).
All serum pools were prepared using anonymous routine residual material from patients’ samples collected in Vacuette® 5 mL Serum Sep Clot Activator, Becton–Dickinson Vacutainers. After an appropriate clotting time, tubes were centrifuged for 6 min at 3500 rotations per minute and 18℃, pooled in 500 μL aliquots and stored at −80℃.
Limit of Blank (LoB), Limit of Detection (LoD) and Limit of Quantitation (LoQ) were determined according to CLSI EP17-A and EP5-A2 protocols.8,9 LoB was assayed utilizing four samples (S1–S4, twice over 10 days): S1 and S2 were zero calibrators without cTnI from two different cTnI calibrator lots; S3 and S4 were cTnI Sample Diluent certified free from cTnI. LoD was assayed with six sample pools with TnI values ranging from the LoB to four times the LoB (twice over 10 days). LoQ was determined by testing 11 serum pools (twice over 20 days) with TnI values ranging from 9 to 236 ng/L.
The 99th percentile upper reference limit (URL) was determined by analysing serum samples from 330 healthy blood donors (260 men, 70 women, age range 18–70 years, median age 36 years). Samples were from a previous study 10 stored frozen at −80℃. The inclusion criteria for the apparently healthy subjects were in accordance with the CLSI standard protocol for recruitment of healthy individuals for reference interval determination. 12 No participant reported a current or past history of coronary artery disease or cardiac related medical conditions on the basis of a health questionnaire. Surrogate testing was performed [urea, electrolytes, liver function tests, glucose, B type natriuretic peptide, electrocardiogram and thoracic X-ray]. All subjects included in healthy population had normal results and values of the surrogate tests.
Informed consent was obtained from all subjects and, in accordance with the Declaration of Helsinki, the study protocol was approved by the study site’s Ethical Committee.
Statistical analysis was performed using both Statistical Package for Social Science (SPSS, Chicago, IL, USA, version 13.0) and dedicated software developed in Visual Basic language.
Results
LoB and LoD calculations are derived parametrically or non-parametrically as appropriate, using both concentration and raw Relative Light Units data fixing both alpha and beta at 0.05 level. 9 The calculated LoB and LoD values were 2.6 and 12 ng/L, respectively. Imprecision profile and LoQ 10% was determined by fitting data with hyperbolic regression; the 95% confidence interval (95% CI) was evaluated by inverse regression method, after linearization of the hyperbolic function. The 10% CV was at 18 ng/L (95% CI 8–25).
Figure 1 reported hyperbolic and linearized imprecision profiles. The 99th percentile URL in our apparently healthy reference population, determined by bootstrap techniques, was 22 ng/L (95% CI 11–34). Measurable values (between LoD and 99th percentile) were found on 62% of samples. There were no significant differences between gender and no correlation between cTnI and age.
Hyperbolic and linearized imprecision profiles of cTnI. (a) Imprecision profile of cTnI by hyperbolic regression analysis. x-axis: cTnI average concentration (ngL−1) and y-axis: %CV. (b) Imprecision profile of cTnI hyperbolic regression analysis after linearization. x-axis: 1/cTnI average concentration (ngL−1)−1, y-axis: %CV.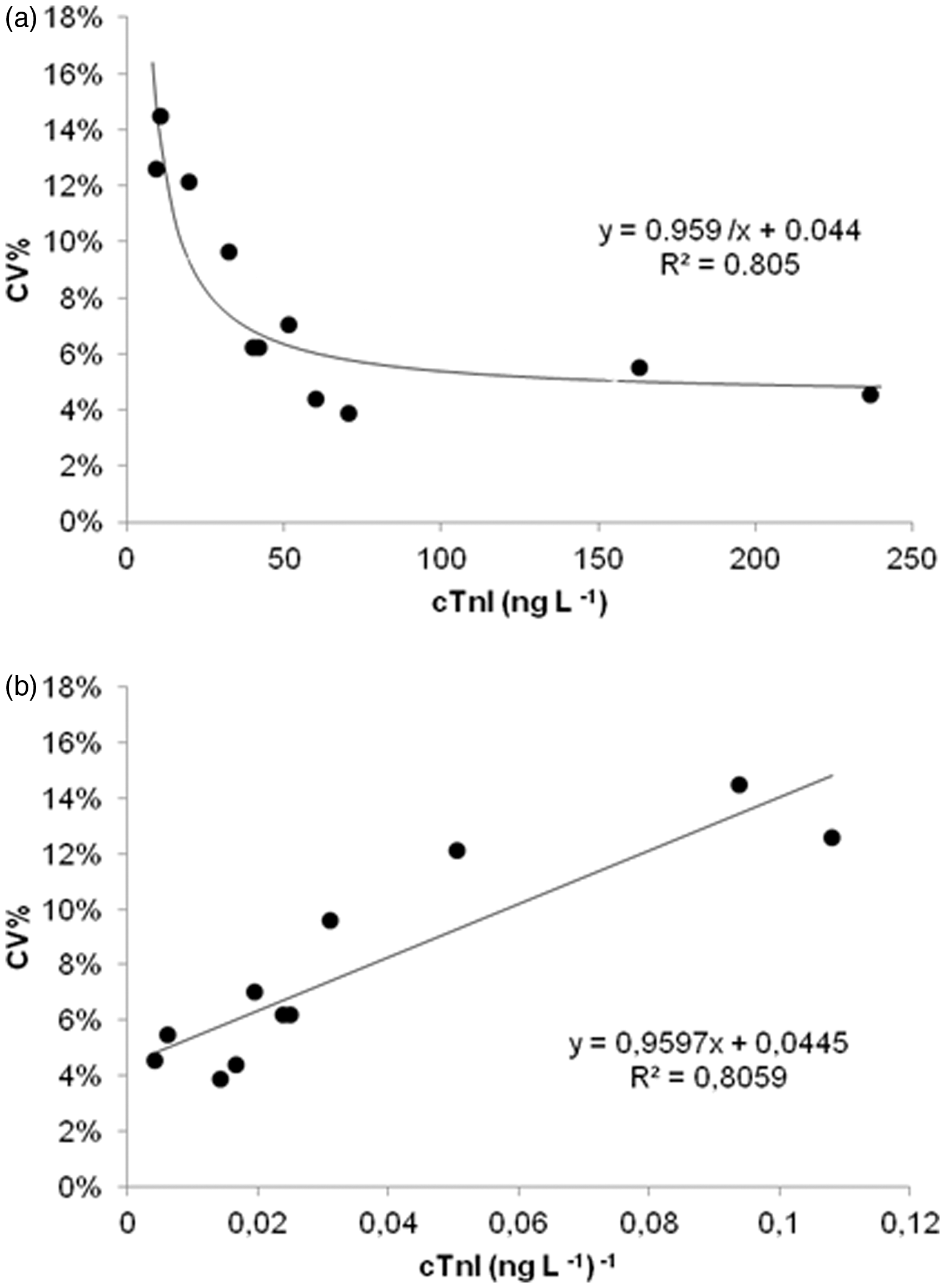
Discussion
Cardiac troponins are well established as the standard biomarkers for myocardial injury detection and prognostic evaluation of patients with and without ACS. 7 International guidelines1–4 have defined increased cTns above the 99th percentile of a normal reference population as abnormal result. This discriminatory 99th percentile must be determined for each specific assay with appropriate quality control in each laboratory.3,13 Optimal precision, as described by %CV at 99th percentile URL for each assay, should be defined as ≤10%. 2
Based on Apple’s scorecard 7 , only cTn assays with total imprecision %CV ≤ 10 at the 99th percentile are guideline acceptable, tests with %CV between 10 and 20% are clinically usable and tests with %CV greater that 20% are not suitable for routine use.
On September 2013, Beckman Coulter Inc. released the new AccuTnI+3, replacing AccuTnI which was designated clinically usable by Apple’s scorecard. 7 The manufacturer declared an improved analytical performance in the instructions for use (package insert), claiming an LoB, LoD and LoQ 10% <10 ng/L, <10 ng/L and 40 ng/L, respectively.
From October to December 2013, we evaluated the newly released assay, according to CLSI EP5-A2 and EP17-A, prior to its introduction into routine use. We also decided to confirm the URLs to ensure proper representation of specific populations and sample types. Our evaluation was conducted incorporating several analytical variables during the three-month evaluation period: a UniCel® DxI in routine use, environmental changes (e.g. fluctuating room temperature), involved variety of operators, and two calibrators and reagents lots changes. Serum pools were at critical concentration levels and were used as proposed by CLSI and International Federation of Clinical Chemistry (IFCC).8,9,14
During the study, both cTnI assays satisfied our laboratory internal quality control rules.
In 2009, we evaluated the performance of the AccuTnI assay using the same methodology. At that time, LoB, LoD and LoQ 10% CV were shown to be 6, 20 and 57 ng/L, respectively. 10
In this current study, the LoB, LoD and LoQ 10% of AccuTnI+3 were found to be lower with values of: 2.6, 12 and 18 ng/L, respectively.
Comparison of data obtained in this study compared with our previous work 10 suggests improved analytical performance for the newly released AccuTnI+3 assay.
We also determined the 99th percentile for AccuTnI+3 in a healthy reference population and compared it with those suggested by manufacturer.
The 99th percentile ideally should be determined for each specific assay and in each laboratory3,13,15; however, it is difficult to carefully recruit and select a large and appropriate target population. In our study, inclusion criteria and demographic characteristics of the apparently healthy reference population only partially reflect those of patients who present to the emergency department (ED) with chest pain and suspected ACS.
In spite of this, the reference population calculated 99th percentile 22 ng/L (95% CI 11–34) is consistent with reported by manufacturer in the Clinical Information Bulletin. 16 The Bulletin shows two independent studies carried out to establish 99th percentile for AccuTnI+3 assay. One study based on an adult European population (998 subjects, age 40–99 years) reported 99th percentile at 40 ng/L which reflects the population likely to attend the ED, while the other based on US population (527 subjects, age 18–94 years) reported 99th percentile at 20 ng/L is a healthy population with a lower mean age.
Analytical performance (%CV) and clinical data (99th percentile) of AccuTnI+3 shows that this assay satisfied the imprecision quality specification recommended in the guidelines. With this assay, it is now possible to utilize the 99th percentile as decision level for myocardial injury detection. The assay hits the target of having a total imprecision %CV at 99th percentile <10% (Apple’s scorecard7 guideline acceptable). AccuTnI+3 is able to recognize low but meaningful signal of cTnI from the inherent system noise. 17 Finally, this assay is suitable for better clinical evaluation of patients with or suspected of having cardiac disease.
Footnotes
Acknowledgements
The authors thank Beckman Coulter Inc., Italy for supplying cTnI reagents.
Declaration of conflicting interests
None declared.
Funding
This research received no specific grant from any funding agency in the public, commercial, or not-for-profit sectors.
Ethical approval
Not applicable.
Guarantor
MM.
Contributorship
MM and BP wrote the paper, DS and MBR performed all data analysis. All authors reviewed and edited the final version of the manuscript.
