Abstract
Magnesium is one of the most abundant cations in the body and is essential for a wide variety of metabolically important reactions. Serum magnesium concentration is regulated by the balance between intestinal absorption and renal excretion. Hypomagnesaemia is relatively common, with an estimated prevalence in the general population ranging from 2.5 to 15%. It may result from inadequate magnesium intake, increased gastrointestinal or renal loss or redistribution from extracellular to intracellular space. Drug-induced hypomagnesaemia, particularly related to proton-pump inhibitor (PPI) therapy, is being increasingly recognized. Although most patients with hypomagnesaemia are asymptomatic, manifestations may include neuromuscular, cardiovascular and metabolic features. Due to the kidney’s ability to increase fractional excretion to nearly 100% when the renal magnesium threshold is exceeded, clinically significant hypermagnesaemia is uncommon, generally occurring only in the setting of renal insufficiency and excessive magnesium intake. Symptoms include hypotension, nausea, facial flushing, ileus and flaccid muscle paralysis. In most cases, simply withdrawing exogenous magnesium is sufficient to restore normal magnesium concentrations, although occasionally administration of intravenous calcium or even dialysis may be required.
Introduction
Magnesium is the second most abundant intracellular cation after potassium and the fourth most abundant cation in the body. It serves several important functions in the human body; it is a co-factor for DNA and protein synthesis, oxidative phosphorylation, neuromuscular excitability, enzyme activity and regulation of parathyroid hormone (PTH) secretion. 1 The normal adult human body contains approximately 1000 mmols of magnesium (22–26 g). 2 Approximately 99% of total body magnesium is intracellular, stored predominantly in bone (85%), muscle and soft tissues, with only 1% in the extracellular space. 3 Up to 70% of serum magnesium exists in the ionized (free), physiologically active form, which is important for physiological processes, including neuromuscular transmission and cardiovascular tone. 4 The normal total serum magnesium in adults ranges between 0.70 and 1.10 mmol/L, with a typical reference range for serum ionized magnesium concentration of 0.54–0.67 mmol/L. 2 However, the Pathology Harmony Group has recently suggested that the reference range for total magnesium should be standardized to 0.7–1.0 mmol/L. 5 Magnesium deficiency can result from a variety of causes including gastrointestinal and renal losses; it may be asymptomatic or present with a wide variety of features. Hypermagnesaemia occurs less frequently, usually due to failure of excretion and/or increased intake.
This article reviews physiological magnesium homeostasis and also discusses the causes, diagnosis and management of hypo- and hypermagnesaemia.
Physiological functions of magnesium
Magnesium is essential for a wide variety of metabolically important reactions in the body. It is a co-factor in over 300 enzymatic reactions, in particular those involving enzymes that use nucleotides as co-factors or substrates, such as ATPases, kinases and cyclases. 6 Magnesium influences the activity of enzymes via a number of mechanisms including binding to ligands and active sites of enzymes, causing conformational change during catalytic processes, and promoting aggregation of multienzyme complexes.6,7 Magnesium also plays an important role in protein and nucleic acid synthesis, the cell cycle, cytoskeletal and mitochondrial integrity and the binding of substances to the plasma membrane, as well as modulating ion transport by pumps, carriers and channels.2,8
Since magnesium acts as a calcium channel antagonist, it affects processes regulated by intracellular calcium concentration fluxes and is thereby essential for normal neurological and muscular function.2,9
Sources of magnesium
The recommended dietary allowance (RDA) for magnesium is 350 mg/day for male adults and 280 mg/day for females, rising during pregnancy and lactation to 355 mg/day. 2 The richest dietary sources of magnesium are whole grain cereals, green vegetables, beans, nuts and seafood. 10 Chocolates, vegetables, fruits, meats and fish have intermediate magnesium content, while dairy products are poor in magnesium. Magnesium in drinking water is thought to account for only about 10% of the daily magnesium intake. 2 Refining, processing or cooking food can significantly deplete magnesium content. 6
Regulation of magnesium balance
Serum magnesium concentration is regulated by the balance between intestinal absorption and renal excretion (Figure 1). Magnesium absorption occurs predominantly in the small intestine, via a saturable transcellular active pathway and a non-saturable paracellular passive pathway.
11
Smaller amounts are absorbed in the colon. At low intraluminal concentrations, magnesium is absorbed primarily via the transcellular route, which involves the active transport of magnesium to the blood through the interior of the epithelial cell; the passive paracellular pathway comes into play as magnesium concentrations rise.
3
Paracellular magnesium absorption is a passive process which involves the absorption of magnesium through the small spaces between the epithelial cells; it relies on tight junction permeability, which is still poorly understood. Claudins are small transmembrane proteins which are key components of the paracellular channel. Paracellular magnesium transport occurs predominantly in the ileum and distal parts of the jejunum, because of the relatively low expression of ‘tightening’ claudins 1, 3, 4, 5 and 8.
12
Claudins 16 and 19, known to be involved in magnesium permeability, are not expressed in the intestine.12,13
Schematic representation of magnesium regulation. Relative width of arrows represents relative flux of magnesium across compartments. The skeleton provides a large exchange reservoir for magnesium while regulation of gut absorption and renal exchange provides dynamic responsiveness to changes in serum magnesium concentration. Adapted from Ayuk and Gittoes.
53
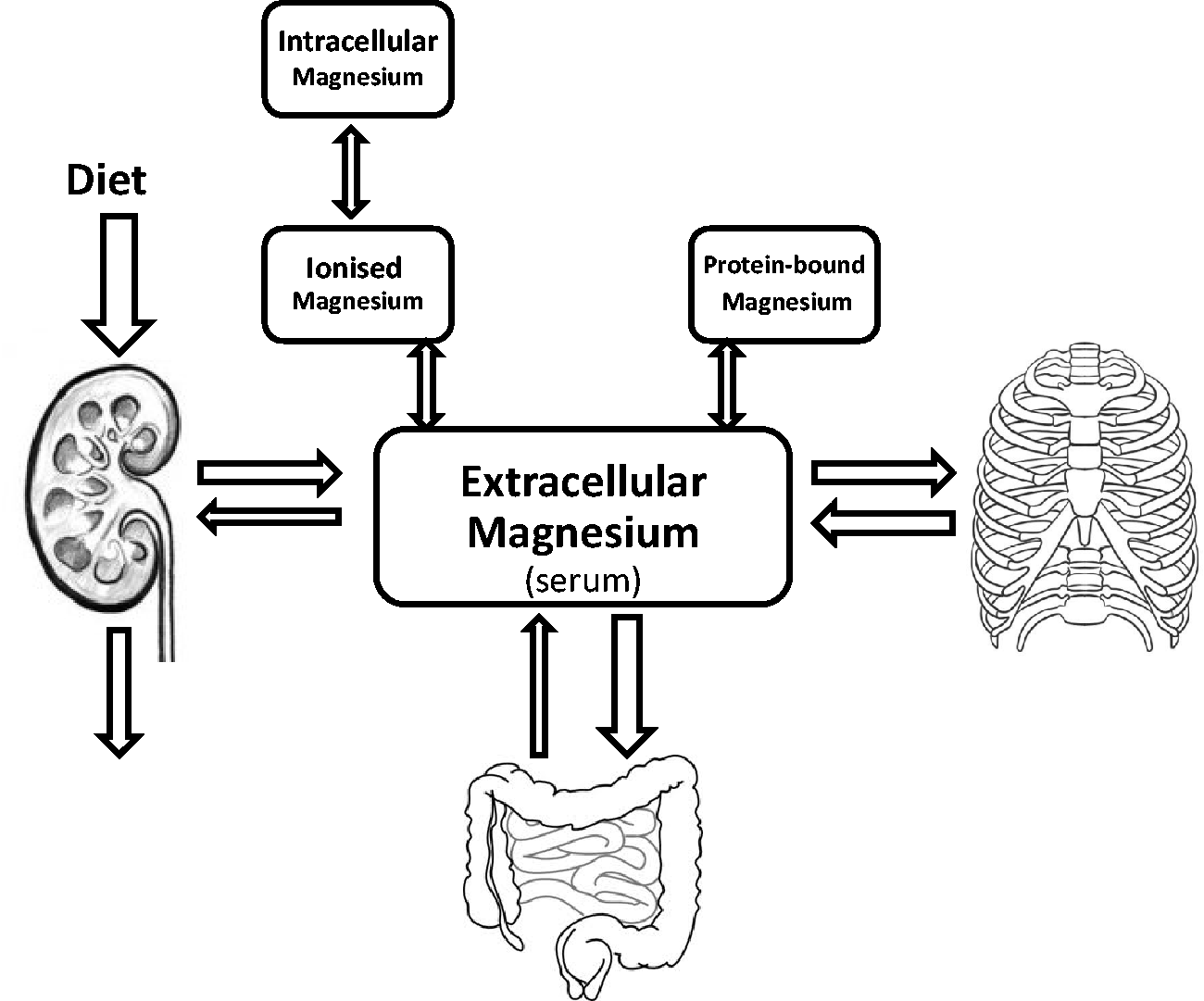
In the kidney, around 80% of total serum magnesium is filtered in the glomeruli, with >95% being re-absorbed along the nephron. Under normal conditions only 3–5% of the filtered magnesium is excreted in the urine. Approximately 10–15% of filtered magnesium is re-absorbed in the proximal convoluted tubules, while 60–70% is passively re-absorbed in the thick ascending loop of Henle.9,14 Renal re-absorption of magnesium varies widely to maintain magnesium homeostasis, declining to nearly zero in the presence of hypermagnesaemia or reduced glomerular filtration rate (GFR), and rising to >99% to minimize urinary losses in response to magnesium depletion or decreased intake. 9 Stimuli that increase urinary magnesium excretion include high natriuresis, osmotic load, metabolic acidosis and phosphate depletion, while metabolic alkalosis, PTH, glucagon, antidiuretic hormone and aldosterone reduce urinary magnesium excretion.9,14
The exact mechanisms underlying transepithelial magnesium transport have not been fully elucidated, but recent clinical and molecular genetic studies have considerably increased understanding of this process. Transient receptor potential channel melastatin member 7 (TRPM7) and TRPM6 were the first magnesium channels to be identified in mammalian cells.15,16 While TRPM7 (previously LTRPC7) is widely expressed and is thought to control magnesium homeostasis in individual cells, TRPM6 is specifically localized in the intestine (duodenum, jejunum, ileum and colon) and the distal convoluted tubule of the nephron, where it functions as a divalent cation channel with a higher affinity for magnesium than for calcium.3,8,17 This distribution strongly suggests TRPM6 regulates magnesium homeostasis via intestinal and renal re-absorption.
In the thick ascending loop of Henle, passive paracellular magnesium re-absorption is driven by the transepithelial voltage gradient, and facilitated by the presence of claudins 16 and 19, which form a cation-selective tight junction. 13 It is currently hypothesized that the lumen-positive potential is created by ion transport across the cells; NaCl enters the thick ascending loop cells via the apical Na+–K+–2Cl− cotransporter (NKCC2) and K+ is recycled into the luminal space via the renal outer medullary K+ (ROMK) channel, while Na+ and Cl− are extruded from the cell basolaterally via the Na+/K+-ATPase and the kidney-specific Cl− channel (CLC-Kb), respectively.18,19
In the distal convoluted tubule, the TRPM6 magnesium channel allows the influx of magnesium into the cell but the basolateral magnesium extrusion mechanism remains to be identified. 18 TRPM6 consists of six membrane-spanning domains with a pore-forming region between the fifth and sixth segment and intracellular C- and N-termini; its channel activity is tightly regulated by intracellular magnesium concentrations. 17 The TRPM6 protein contains a C-terminal α-kinase domain, which appears to have a regulatory role in the modulation of channel functioning. Interactions between the α-kinase domain and a number of regulatory factors have been shown to modulate TRPM6 activity.17,18 Receptor for activated C-kinase 1 (RACK1) interacts with the α-kinase domain to inhibit TRPM6 channel activity, thereby preventing magnesium overload during active magnesium re-absorption. 17 More recently, repressor of oestrogen receptor activity (REA) has also been demonstrated to inhibit TRPM6 channel activity by binding to the α-kinase domain. 20
Hypomagnesaemia
Biochemical hypomagnesaemia is common, with an estimated prevalence in the general population ranging from 2.5 to 15%, rising to as high as 65% in patients in intensive care settings.21,22 However, hypomagnesaemia with clinical consequences is far less common. Low serum magnesium concentration generally indicates total body magnesium depletion, but it is noteworthy that serum magnesium concentration may be normal in the presence of total body magnesium depletion; this has implications for clinical practice.
Clinical manifestations
Most patients with hypomagnesaemia are asymptomatic, with symptoms usually not manifesting until the serum magnesium concentration is <0.50 mmol/L. 22 Clinical manifestation may depend more on the rate of development of magnesium deficiency or the total body deficit than the actual serum magnesium concentration. 6 Symptomatic magnesium depletion is often associated with multiple other biochemical abnormalities, including hypokalaemia, hypocalcaemia and metabolic acidosis. As a result, it is often difficult to ascribe specific clinical features solely to hypomagnesaemia. Anorexia, nausea, vomiting, lethargy and weakness are typical early symptoms of magnesium deficiency; manifestations of severe hypomagnesaemia include neuromuscular, cardiovascular and metabolic features. 2
Neuromuscular manifestations
Neuromuscular irritability occurs commonly in magnesium depletion. It manifests as muscular weakness, apathy, tremors, paraesthesia, tetany, vertical nystagmus and positive Chvostek and Trousseau signs.9,23 Although many of these features could be attributed to co-existing metabolic abnormalities, similar findings have been demonstrated when isolated magnesium depletion was induced in volunteers. 24
Several mechanisms may contribute to the neuromuscular manifestations seen in magnesium deficiency. Hypomagnesaemia lowers the threshold for axon stimulation and increases nerve conduction velocity, causing hyper-responsive neuromuscular activity. 6 Magnesium deficiency also leads to increased release of calcium from the sarcoplasmic reticulum of the muscle cell and reduced re-uptake of calcium, resulting in increased contractility to a given stimulus and reduced ability to recover from the contraction, thereby making it prone to tetany. 6
Severe effects such as seizures, drowsiness, confusion and coma occur at magnesium concentrations <0.4 mmol/L. 25
Cardiovascular manifestations
Intracellular magnesium deficiency results in a decrease in intracellular potassium. This leads to disturbance of the resting membrane potential and re-polarization phase of the myocardial cells. 26 Electrocardiographic changes seen with hypomagnesaemia include flattened T-waves, U-waves, prolonged QT interval and widened QRS complexes. 9 Hypomagnesaemia has been associated with both atrial and ventricular arrhythmias, although the underlying mechanism is unknown and it is unclear whether this association exists in subjects without underlying cardiac disease. 22 There is some evidence to suggest that magnesium deficiency may play an important role in the pathogenesis of ischaemic heart disease and congestive heart failure.2,22 The most life-threatening cardiovascular manifestation of hypomagnesaemia is ventricular arrhythmia.
Metabolic effects
Hypokalaemia is associated with hypomagnesaemia in up to 60% of cases. 27 This is due partly to underlying common aetiologies that cause magnesium and potassium losses and partly to a specific disorder of renal potassium wasting as a result of hypomagnesaemia. 9 Hypomagnesaemia is thought to lead to an increase in the number of open potassium channels in the thick ascending limb and cortical collecting tubule, and to directly increase the activity of potassium channels of ascending limb cells, thereby promoting potassium secretion from the cell into the lumen and enhanced urinary losses. 22 The exact underlying mechanism was previously unexplained, but Huang and Kuo 28 recently hypothesized that intracellular magnesium is a critical determinant of ROMK-mediated potassium secretion in the distal nephron, binding and blocking the pore of the channel from the inside and thereby inhibiting outward potassium flux. A decrease in intracellular magnesium caused by magnesium deficiency releases the magnesium-mediated inhibition of ROMK channels and increases potassium secretion into luminal fluid; this, along with increase in distal sodium delivery or elevated aldosterone concentrations, is likely to explain the hypokalaemia associated with magnesium deficiency. 28 The hypokalaemia in this setting is relatively refractory to potassium supplementation and requires correction of the magnesium deficit.
Hypocalcaemia is also commonly associated with hypomagnesaemia; hypomagnesaemia suppresses the release of PTH and also induces end-organ resistance to PTH, possibly through interference with G protein activation in response to PTH, thereby minimizing the stimulation of adenylate cyclase.22,29 Symptomatic hypocalcaemia is almost always associated with plasma magnesium concentrations below 0.5 mmol/L. 22 Hypocalcaemia associated with hypomagnesaemia is refractory to correction until the magnesium deficit has been corrected.
Diagnosis and interpretation
Serum magnesium concentration
Measurement of total serum magnesium is the method of choice for determining clinical magnesium status. Measurement of ionized magnesium is feasible using ion-selective electrodes but unlike ionized calcium its clinical utility above and beyond measuring total serum magnesium is unproven. Most laboratories use automated spectrophotometric techniques, the reference method being atomic absorption spectrophotometry.
A number of factors must be taken into account when interpreting serum magnesium results to diagnose magnesium deficiency. Potassium EDTA is a sample tube anticoagulant used for many laboratory analyses. Gross potassium EDTA contamination of blood samples can cause spurious hypomagnesaemia. 30 In addition, as extracellular fluid space contains only 1% of total body magnesium, serum magnesium may not always accurately reflect the intracellular magnesium status and a subject with normal serum magnesium concentrations may have total body magnesium depletion. 3 Finally, 30% of serum magnesium is bound to albumin and is therefore inactive; as most methods of assessing serum magnesium content measure total magnesium concentration, hypoalbuminaemic states may lead to spuriously low magnesium values. Conventionally, however, serum magnesium concentrations are not ‘adjusted’ for albumin concentrations, as there is generally a high correlation between serum total and ionized magnesium concentrations. 31
In adults, serum magnesium concentration is generally not influenced by sex or age. It may be slightly higher in the very elderly, after intense short-lived exercise and in subjects on a vegetarian diet, and lower during the third trimester of pregnancy and after endurance exercise. 6
Once hypomagnesaemia is confirmed, in many cases the cause can be obtained from the history. If no cause is apparent, the distinction between gastrointestinal and renal losses can be made using some of the diagnostic tests below.
24-h urinary magnesium excretion
The 24-h urine collection for total magnesium is a useful indication of renal magnesium avidity. In the presence of hypomagnesaemia, urinary magnesium excretion <12 mg (0.5 mmol) is consistent with an intact renal response, while concentrations >24 mg (1 mmol) indicate abnormal renal wasting. 9
Fractional excretion of magnesium (FEMg)
This is occasionally used to distinguish between gastrointestinal and renal magnesium loss using the formula FEMg = [(uMg × sCr) / (sMg × uCr × 0.7)] × 100, where u and s refer to the urine and serum concentrations of magnesium (Mg) and creatinine (Cr). In the presence of hypomagnesaemia, FEMg >2% indicates inappropriate renal wasting, while FEMg <2% indicates appropriate renal handling of magnesium, implicating extra-renal magnesium losses, decreased intake or absorption. 9
Magnesium infusion test (magnesium tolerance test)
Measurement of magnesium retention after acute magnesium loading can be used as a surrogate measure for intracellular magnesium, but this method is useful only when the clinical suggestion of magnesium deficiency is strong in the setting of normomagnesaemia. Reduced excretion (<80% over 24 h) of an infused magnesium load (2.4 mg/kg of lean body weight given over the initial 4 h) suggests magnesium deficiency. 22
Red cell, mononuclear blood cell and skeletal muscle magnesium concentrations can also be measured, but are not used in routine clinical practice. 6
Causes of hypomagnesaemia
Causes of hypomagnesaemia.

Mechanism involves impaired intestinal absorption of magnesium and renal magnesium wasting.
Exact mechanism unclear.
Inadequate magnesium intake
Hypomagnesaemia due exclusively to reduced dietary intake is very uncommon. Magnesium deficiency may be seen in patients with alcoholism, on magnesium-free intravenous fluids or on total parenteral nutrition, but in most reported cases it is associated with gastrointestinal or urinary magnesium losses.6,9 In patients with alcoholism, as well as decreased dietary intake, pancreatitis, intermittent diarrhoea and vomiting and excessive urinary magnesium excretion due to alcohol-induced tubular dysfunction also contribute to hypomagnesaemia.9,22
Redistribution of magnesium
Hypomagnesaemia due to the shift of magnesium from extracellular to intracellular space can occur in a number of clinical situations, including re-feeding syndrome, hungry bone syndrome following parathyroidectomy, correction of metabolic acidosis and acute pancreatitis.32–34
Increased gastrointestinal loss
Conditions like inflammatory bowel disease and short bowel syndrome which are associated with diarrhoea, malabsorption or steatorrhoea can result in magnesium loss through reduced re-absorption of intestinal secretions. In general, the degree of magnesium depletion correlates with the severity of the diarrhoea and the stool fat content, as fat may form insoluble magnesium soaps.6,9 Hypomagnesaemia with secondary hypocalcaemia (HSH) is an inherited disorder of selective magnesium malabsorption caused by a mutation in the TRPM6 gene.3,35 It usually presents in the neonatal period and is associated with hypocalcaemia, tetany and seizures.
Renal losses
Hypomagnesaemia due to inappropriate urinary magnesium loss can occur via a variety of mechanisms. Renal tubular damage due to chronic renal impairment, recovery from acute tubular necrosis and postobstructive diuresis can all cause symptomatic hypomagnesaemia. Several congenital disorders of magnesium renal tubular re-absorption have been described. Bartter syndrome, caused by a mutation in the CLCNKB gene, presents in infancy and is associated with hypokalaemia, metabolic alkalosis and in up to 50% of patients, hypomagnesaemia. Gitelman syndrome, caused by a defect in the gene coding for the NaCl co-transporter, presents in childhood or adolescence and is associated with hypomagnesaemia and hypocalciuria.6,9 Other molecular defects underlying hereditary tubular disorders of renal magnesium handling have been identified in recent years. 3
Drug-induced hypomagnesaemia
Drugs can cause magnesium wasting by interfering with renal re-absorption of magnesium (loop and thiazide diuretics) or by causing tubular damage (Amphotericin B, aminoglycosides and cisplatin). With nephrotoxic drugs, the impairment in loop and distal magnesium re-absorption may occur before the onset of overt tubular necrosis and acute renal failure, and persist after kidney function has normalized. 22 The mechanism via which proton-pump inhibitor (PPI) drugs cause hypomagnesaemia has not yet been fully elucidated, but it has been hypothesized that they may interfere with the active transport pathway across the intestinal wall. PPI-induced hypomagnesaemia is discussed in greater detail below.
Treatment of hypomagnesaemia
Patients with symptomatic hypomagnesaemia should be treated with intravenous magnesium, reserving oral replacement for asymptomatic patients. General guidance is provided below, but local guidelines are likely to exist and should be referred to when treating a patient with hypomagnesaemia. The most common formulation of intravenous magnesium is magnesium sulphate (MgSO4). One gram of magnesium sulphate contains 100 mg of elemental magnesium, equivalent to approximately 4 mmol of magnesium. Magnesium sulphate injection is available as 10, 20 and 50% preparations; for intravenous use it should be diluted with sodium chloride 0.9 or 5% glucose to a concentration of 20% magnesium or less. The 50% solution contains 0.5 g magnesium sulphate per mL (2 mmol/mL). No trials have been performed to determine the optimal regimen for magnesium replacement, but consensus statements suggest administration of 8 to 12 g of magnesium sulphate in the first 24 h followed by 4 to 6 g/day for three or four days to replete body stores. 9 Maximum infusion rates should not exceed 2 g/h. The aim should be to maintain serum magnesium concentrations above 0.4 mmol/L. Patients with renal insufficiency should receive 25 to 50% of the initial dose recommended for patients with normal kidney function. 4 ECG monitoring is recommended with high doses and in the elderly.
Intramuscular administration is also possible, but is painful and should be reserved as a last resort for patients with no intravenous access. Intramuscular administration of the undiluted 50% solution results in therapeutic serum concentrations in 60 min. One to two grams of magnesium sulphate (2–4 mL of the 50% solution) can be injected intramuscularly every 6 h for 24 h (4 doses in total).
Oral magnesium salts can be used to supplement body magnesium, but they are generally not well absorbed from the gastrointestinal tract, hence the use of magnesium sulphate as an osmotic laxative. Magnesium glycerophosphate, oxide, citrate, phosphate, aspartate, hydroxide and lactate are available, but are unlicensed in the UK for treating hypomagnesaemia. These preparations provide 2.5–4.0 mmol (60–96 mg) of magnesium per tablet. Six to eight tablets should be taken daily in divided doses for severe magnesium depletion. Two to four tablets may be sufficient for mild, asymptomatic disease. 22 If one particular magnesium salt is ineffective or not tolerated, another should be considered. 36 Patients with chronic renal magnesium wasting or diuretic-induced hypomagnesaemia who cannot discontinue diuretic therapy may benefit from the addition of a potassium-sparing diuretic such as amiloride or triamterene, as they lower magnesium excretion by increasing re-absorption in the collecting tubule. 22
PPI-induced hypomagnesaemia
PPI drugs inhibit gastric acid secretion and are widely used for the treatment and prevention of dyspeptic symptoms. Since 2006, a number of cases of severe hypomagnesaemia have been reported in long-term PPI users, with serum magnesium concentrations returning to normal when PPI therapy was discontinued.37–40 Epstein et al. 37 described two patients on PPI therapy who presented with carpopedal spasm in association with severe hypomagnesaemia and hypocalcaemia, but interestingly without an appropriate increase in the concentration of PTH. Urinary magnesium concentration was low in both patients, but concentrations of magnesium in both urine and serum rose rapidly to within reference range when the PPI drug was stopped.
The exact mechanism by which PPI drugs cause hypomagnesaemia has not yet been elucidated, but low 24-h urine magnesium excretion has been consistently demonstrated, suggesting it is not mediated via excess renal loss.38,40 Cundy and Dissanayake. 38 performed magnesium infusion studies on two patients with PPI-induced hypomagnesaemia to assess both renal tubular handling of magnesium and the retention of a parenteral magnesium load. Both patients were severely hypomagnesaemic at the time of testing, with low 24-h urine magnesium excretion. Plasma PTH concentration was detectable in both subjects, but inappropriately low given that both were hypocalcaemic. As the plasma magnesium concentration increased during an infusion test, PTH also increased until supranormal plasma magnesium concentrations were obtained, after which PTH fell. There was no evidence of renal magnesium wasting, with urine excretion of magnesium remaining extremely low until the ultrafilterable magnesium exceeded 1.0 mmol/L, and both subjects retaining around 75% of the parenteral magnesium load. In the described cases, hypomagnesaemia was partially corrected by high dose oral magnesium supplements, suggesting the passive intestinal magnesium transport pathway was intact; it was therefore hypothesized that PPI drugs may interfere with the active transport pathway across the intestinal wall via disruption of TRPM6 channel function. 38 One suggestion is that PPI-induced hypochlorhydria could lead to impaired solubilization and absorption of magnesium ions in the small intestine, eventually leading to depletion of total body magnesium stores. 41 However, short-term studies have shown no detectable effect of PPI drugs on magnesium absorption. 42 Alternatively, variant alleles of TRPM6/TRPM7 may predispose susceptible patients to the side effect of PPI drugs. 39 Such variants may be associated with subtle intestinal malabsorption or persistent magnesium leak through the kidneys, causing depletion of magnesium stores which may be further aggravated by the use of PPI drugs. 43
Hypomagnesaemia due to PPI therapy is thought to take years to develop, and oral magnesium supplements are only partially effective at correcting the hypomagnesaemia whilst PPI therapy is maintained. 40 Hypomagnesaemia is often associated with other electrolyte abnormalities such as hypocalcaemia and hypokalaemia, possibly exacerbated by concomitant diuretic therapy. However, stopping diuretics does not correct the hypomagnesaemia if patients remain on PPI therapy, whereas hypomagnesaemia resolves in those who remain on a diuretic when PPI therapy is discontinued. 40 Symptoms can include muscle weakness, muscle cramps, tetany, seizures and cardiac arrhythmias. In many of the reported cases, hypomagnesaemia resulted in functional hypoparathyroidism, with inappropriately low PTH concentrations, and some of the patients’ symptoms may have been due to hypocalcaemia rather than hypomagnesaemia.37,40,44
In a closely monitored PPI withdrawal study, Mackay and Bladon 40 demonstrated rapid resolution of hypomagnesaemia within two weeks of the PPI being discontinued. However, of a total of 10 patients, six complained of reflux dyspepsia which was not controlled on alternative medication and requested to go back on to PPI therapy. The introduction of pantoprazole, the least potent PPI, combined with oral magnesium supplements where necessary, resulted in acceptable control of reflux symptoms without hypomagnesaemia in most of these patients. Given the variations in the degree of susceptibility to PPI-induced hypomagnesaemia and the response to treatment, it has been suggested that genetic factors may play a significant role in the development of this condition. 40
Hypermagnesaemia
The kidney has a large capacity for magnesium excretion, and is capable of increasing fractional excretion to nearly 100% when the renal magnesium threshold is exceeded. Clinically significant hypermagnesaemia is therefore uncommon in patients with normal renal function, generally occurring in the setting of renal insufficiency and excessive magnesium intake. 14 The prevalence of hypermagnesaemia overall ranges from 5.7 to 9.3% in hospitalized patients, but in many cases it does not manifest clinically.27,45
Clinical manifestations of hypermagnesaemia
Symptoms and signs of magnesium toxicity do not generally manifest until serum concentrations exceed 2.0 mmol/L.
Neuromuscular manifestations
Magnesium prevents the release of presynaptic acetylcholine from both sympathetic and neuromuscular junctions, resulting in some of the earliest and most commonly seen features of hypermagnesaemia, including loss of deep tendon reflexes, flaccid paralysis, lethargy and confusion.6,9 Other neurological manifestations include ileus, urinary retention, depressed respiration and coma.
Cardiovascular manifestations
Early cardiac manifestations of hypermagnesaemia include bradycardia and hypotension. Magnesium concentrations greater than 3.0 mmol/L cause PR interval prolongation, increased QRS duration and prolonged QT interval.6,9,46 Extremely high magnesium concentrations can result in complete heart block or cardiac arrest.
Other manifestations
Non-specific manifestations of hypermagnesaemia include nausea, vomiting and cutaneous flushing. Magnesium intoxication can cause hypocalcaemia, most commonly reported in patients receiving magnesium therapy for pregnancy-induced hypertension. 47 Inhibition of PTH secretion has been proposed as the cause, although elevated PTH was found in some cases. 48 Magnesium intoxication can also be associated with hyperkalaemia, possibly caused by decreased renal potassium clearance or suppression of plasma renin activity.49,50
Causes of hypermagnesaemia
Renal failure
The ability of the kidneys to maintain magnesium balance by increasing the fractional excretion of excess magnesium means hypermagnesaemia generally does not occur unless the GFR falls below 30 mL/min. 9 However, even in advanced renal insufficiency, significant hypermagnesaemia is rare unless magnesium-containing medications are used.
Increased intake
Symptomatic hypermagnesaemia in patients receiving exogenous magnesium occurs most often in the presence of renal impairment. However, intravenous magnesium infusions can result in hypermagnesaemia even in the presence of normal kidney function. The typical setting is treatment of preterm labour or pre-eclampsia/eclampsia, with most patients having good outcomes despite occasionally very high magnesium concentrations, in one case >15 mmol/L. 51 Reported causes of hypermagnesaemia due to oral ingestion of magnesium include antacids, laxative abuse, magnesium cathartics used to treat overdoses and accidental ingestion of Epsom salts.6,9
Miscellaneous causes
Mild hypermagnesaemia has been reported in patients on lithium therapy, and those suffering from hypothyroidism, Addison’s disease or milk alkali syndrome.6,14 Near drowning in the Dead Sea, which has no draining rivers and only loses water through evaporation, is associated with hypermagnesaemia due to aspiration or ingestion of the magnesium-rich seawater. 52
Treatment of hypermagnesaemia
Hypermagnesaemia can be prevented by avoiding exogenous magnesium preparations in patients with renal impairment. In patients with mild hypermagnesaemia and intact renal function, simply withdrawing magnesium therapy is often sufficient to restore normal magnesium concentrations. In symptomatic hypermagnesaemia, administration of calcium, usually at a dose of 100 to 200 mg intravenously over 5 to 10 min, antagonizes the toxic effects of magnesium. 9
In patients with significant renal dysfunction, peritoneal dialysis or haemodialysis using fluid with low magnesium concentration will rapidly and effectively lower serum magnesium concentrations.6,9
Conclusion
Magnesium is essential for a wide variety of metabolically important reactions in the body. Disorders of magnesium metabolism are common in hospital patients, with hypomagnesaemia occurring more frequently than hypermagnesaemia. Although asymptomatic in many cases, both states can have significant cardiovascular, neuromuscular and metabolic manifestations. Management involves addressing the underlying cause, magnesium replacement in case of hypomagnesaemia and in severe hypermagnesaemia intravenous calcium administration and dialysis in patients with renal impairment.
Footnotes
Acknowledgements
This article was prepared at the invitation of the Clinical Sciences Reviews Committee of the Association of Clinical Biochemistry.
Declarations of conflicting interests
None declared.
Funding
This research received no specific grant from any funding agency in the public, commercial, or not-for-profit sectors.
Ethical approval
Not applicable.
Guarantor
NJLG.
Contributorship
Both authors contributed equally.
