Abstract
Introduction
Helicobacter pylori (H. pylori) infection affects approximately 50% of the global population 1 and has an impact on various gastric and extra-gastric diseases, such as chronic gastritis, gastroduodenal ulcer, iron-deficiency anemia, and gastric cancer.2–4 Eradicating H. pylori can significantly alleviate histologic gastritis, prevent progression to intestinal metaplasia, and reduce the incidence of gastric cancer.5,6
However, the effectiveness of H. pylori eradication has declined in recent years, while antibiotic resistance rates have continued to increase.7,8 Current guidelines suggested triple therapy with proton pump inhibitor (PPI), amoxicillin, and clarithromycin for 14 days as first-line eradication therapy in regions with low clarithromycin resistance (<15%).9–11 In China, quadruple therapy (two antibiotics + bismuth + PPI) for 14 days is recommended as the first-line treatment for H. pylori infection. 12 However, the complexity, duration, and side effects often reduce compliance, posing a significant challenge to their clinical effectiveness.
Achieving a near-neutral pH is crucial for the eradication of H. pylori.13,14 Vonoprazan, a new oral potassium-competitive acid blocker (P-CAB), exhibits a longer half-life and stronger gastric acid suppression than other PPIs.15–17 Therefore, vonoprazan is anticipated to be more effective in H. pylori eradication. Recently, several meta-analyses have assessed the efficacy and safety of vonoprazan-based treatment regimens.18–21 The meta-analysis by Zhou et al. 18 showed that 14-days vonoprazan-amoxicillin (VA) therapy was as effective as bismuth-containing quadruple therapy (BQT) in eliminating H. pylori, but with fewer adverse events (AEs). A network meta-analysis 19 found that 7-days vonoprazan-based triple regimen was the most effective in East Asia.
While numerous systematic reviews and meta-analyses have been conducted, these studies primarily compared eradication rates at identical treatment durations. Given vonoprazan’s superior acid suppression compared to PPIs, vonoprazan-based regimens might achieve effective eradication rates while shortening the treatment duration. Recently published RCTs22–27 investigated vonoprazan-based regimens with shorten treatment duration for first-line H. pylori eradication.
Therefore, in this study, we aimed to conduct a meta-analysis comparing the efficacy and safety of ten-day vonoprazan-based regimens with fourteen-day PPI-based regimens for H. pylori eradication.
Materials and methods
Search strategy and inclusion criteria
We conducted a PubMed search using keywords (“vonoprazan” or “VPZ” or “potassium-competitive acid blocker” or “P-CAB” or “takecab” or “TAK438” or “TAK-438”) AND (“Helicobacter pylori” or “H. pylori” or “HP”) for trials up to November 28, 2023. Search results were imported into reference management software (EndNote X6). Additionally, we explored completed but unpublished trials on relevant websites (https://www.clinicaltrials.gov) and examined reference lists of pertinent systematic reviews and included studies for comprehensive coverage.
Two investigators (WWG and QW) independently screened all publications, resolving any disagreements through consultation with a third investigator (LW). The specific inclusion criteria included: (a) articles reporting clinical trial results, (b) clinical trials comparing ten-day vonoprazan-based regimens with fourteen-day PPI-based regimens, (c) confirmed H. pylori infection by biopsy and/or urea breath test (UBT) prior to treatment, (d) eradication evaluation using accepted methods at least 4 weeks post-treatment. Exclusion criteria encompassed: single-arm studies, reviews, conference papers, and abstract-only publications.
Data extraction
Two reviewers independently extracted data from selected studies, with any discrepancies resolved by a third investigator. Extracted data included the first author’s name, publication year, study design, therapeutic regimens, eradication rates, sample size, intervention duration, test for confirming eradication, ethnicity, and H. pylori eradication rates as determined by intention-to-treat (ITT) and per-protocol (PP) analyses.
Quality assessment
The quality of RCTs was evaluated using the Cochrane Risk of Bias tool, assessing selection bias, performance bias, detection bias, attrition bias, reporting bias, and other potential biases. 28
Statistical analysis
Eradication and adverse reaction rates were analyzed using odds ratio (OR) with 95% CI. Q tests and I2 statistic assessed statistical heterogeneity. Heterogeneity was considered significant if the Q-test probability was less than 0.05 (p < .05). I2 scores of 0%–25% indicate negligible heterogeneity, 26%–50% low, 51%–75% moderate, and over 75% high heterogeneity. 29 We also did subgroup analyses to explore factors that might have an impact on the overall results and sources of heterogeneity. In the presence of significant heterogeneity (p < .1 or I2 > 50%), a random-effect model was employed; otherwise, a fixed-effect model was applied. Publication bias was assessed using a funnel plot, and all statistical analyses were performed with Review Manager (Revman Version 5.3).
Results
Study selection and characteristics
Our search yielded 287 potentially relevant articles. After screening titles and abstracts, 9 studies were selected for detailed evaluation. Finally, 4 studies24–27 met the criteria for our final data analysis (Figure 1). Two studies30,31 were excluded due to the absence of a suitable control group, and three22,23,32 for an inappropriate study duration. The analysis included 1560 patients and all were conducted in China. The basic characteristics of these studies are presented in Table 1. Flow chart demonstrating process of study selection. Characteristics of included studies. VON, Vonoprazan; ESO, Esomeprazole; RAB, Rabeprazole; OME, omeprazole; BIS, bismuth; TIN, tinidazole; AMO, amoxicillin; CLA, clarithromycin; FUR, furazolidone; ITT, intention-to-treat; PP, per protocol; UBT, urea breath test; qd, once daily, bid, twice daily; tid, three times a day.

Quality assessment
The quality assessment, detailed in Figure S1, identified blinding of participants and personnel as the primary potential bias source.
Overall H. pylori eradication rate
All four RCTs provided data on H. pylori eradication rates. In the ITT analysis, the eradication rate with ten-day vonoprazan-based therapy was 692/780 (88.7%) compared to 647/780 (82.9%) in the fourteen -day PPI-based group. The OR was 1.53 (95% CI = 0.85–2.75, I2 = 69%) (Figure 2), with no significant difference (p = .16) observed between the two groups. The funnel plot, shown in Figure S2, revealed no asymmetry. Subsequently, PP analysis indicated that the eradication rate was 681/742 (91.8%) for the ten-day vonoprazan-based therapy, and 631/727 (86.8%) for the fourteen-day PPI-based group. The eradication rate for the vonoprazan-based therapy was higher than that of PPI-based regimen significantly (OR [95% CI] = 1.72 [1.23–2.42], I2 = 20%, p = .002; Figure S3). Forest plot of eradication rate of ten-day vonoprazan-based regimen versus fourteen-day PPI-based regimen by ITT analysis.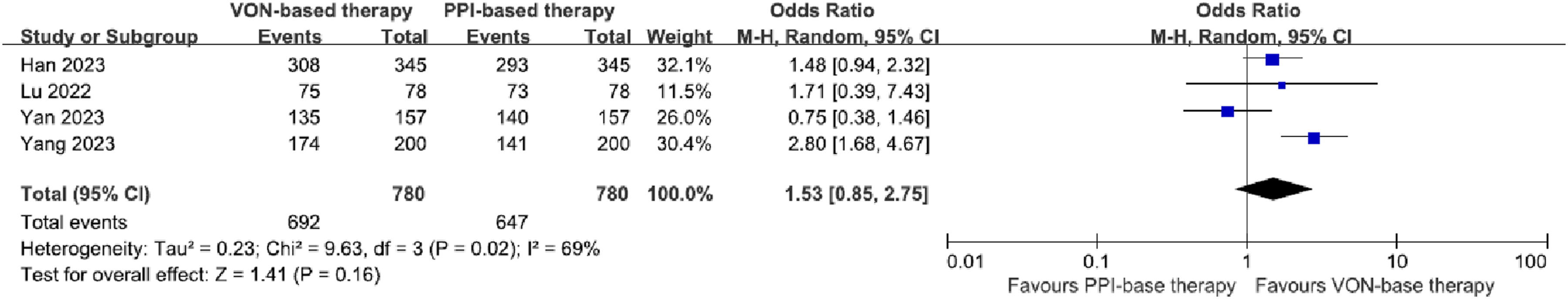
Subgroup
Results of subgroup analyses of eradication rate based on ITT data.

AEs
All included studies reported AEs. The incidence rates of AEs in the ITT analysis were 87/780 (11.2%) in the ten-day vonoprazan-based therapy versus 137/779 (17.6%) in the fourteen-day PPI-based group. There were on significant differences between the two groups (OR 0.66, 95% CI: 0.33–1.31, I2 = 78%, p = .24; Figure 3). Forest plot of AEs between ten-day Vonoprazan-based regimen versus fourteen-day PPI-based regimen.
Discussions
Identifying optimal regimens for H. pylori treatment is challenging due to increasing antibiotic resistance, particularly to clarithromycin. In China, standard PPI-based quadruple therapy with a 14-days duration has shown continued efficacy. 33 However, a shorter treatment duration could potentially reduce AEs and improve patient compliance.
Controlling intragastric pH is essential for effective H. pylori eradication. H. pylori grows at a pH range between 6 and 7 and is sensitive to antibiotics. 34 Compared to PPIs, vonoprazan has shown to offer faster, stronger, and more sustained acid suppression.35,36 To date, several meta-analyses18,37 have suggested that Vonoprazan-based therapy is effective for H. pylori eradication. A meta-analysis 37 of 15 studies showed that vonoprazan-based triple therapy outperformed conventional PPI-based triple therapy in eradicating H. pylori. Another meta-analysis 18 included six RCTs indicated that VA therapy was just as successful as BQT in a 14-days first-line therapy, yet it has fewer adverse events. In Japan, vonoprazan is used in a 7-days first-line therapy along with amoxicillin and clarithromycin or metronidazole, yielding favorable results.38–40 The Japanese guidelines 41 recommend a 1-week triple therapy for first-line H. pylori eradication, including two antibiotics with a P-CAB. However, there has been absent of meta-analyses comparing the eradication rates between vonoprazan-based regimens with shorter treatment duration and fourteen-day PPI-based regimens.
In this study, we demonstrated that a ten-day vonoprazan-based therapy was as effective as a fourteen-day conventional PPI-based therapy in treatment-naive H. pylori patients, with an overall eradication rate of 88.7% versus 82.9%. However, the PP analysis revealed that the eradication rate with the vonoprazan-based therapy (91.8%) was significantly higher than that of the control therapy (86.8%).
To evaluate the impact of different treatment durations of vonoprazan-based therapy, we performed a subgroup analysis. The results indicated that compared to the fourteen-day conventional PPI-based therapy, the eradication rate was higher in vonoprazan-based therapy no matter that the number of drugs in two group was same (90.5% vs 86.5%) or different (86.6% vs 78.7%), but these differences were not statistically significant. Variation in drug numbers was also a major contributor to heterogeneity (I2 = 89%) (Table 2). Similar trends were observed in a subgroup analysis by the type of PPI in control group. In comparing different study designs, eradication rates were comparable between the ten-day vonoprazan-based regimen and fourteen-day PPI-based therapy in multicenter studies (88.2% vs 86.3%, p = .78), while the former showed higher eradication rates in single-center studies (89.6% vs 77.0%, p = .0001). It is important to note that variations in study design outcomes might stem from the limited number of studies and sample sizes.
Safety of the eradication regimens is another crucial aspect. Our findings indicated that the incidence of AEs in vonoprazan-based therapy was lower than in the PPI-based group (11.2% vs 17.6%), but this difference was not statistically significant (p = .24). Despite the shorter treatment duration of vonoprazan-based therapy, an increased frequency of amoxicillin administration was noted in three studies,24,26,27 which might explain the lack of significant difference in AEs between the two groups.
However, our meta-analysis had several limitations. First, although the included RCTs were of high quality, statistical heterogeneity was present. This may have stemmed from differences in study design and the number of drugs in each group. Second, all studies were conducted in China, necessitating further research to determine if these conclusions are applicable to other global populations. Third, variations in the types and dosages of antibiotics, and the number of drugs used across the studies, added complexity and challenges to deriving a singular effect in sub-analysis of the data. Fourth, since only treatment-naive H. pylori patients were enrolled, the findings might not extend to patients undergoing rescue treatment. Future international and multicenter studies are essential to arrive at a definitive conclusion.
Conclusions
In conclusion, our meta-analysis suggests that the efficacy and safety of ten-day vonoprazan-based therapy are comparable to those of fourteen-day conventional PPI-based therapy. However, given the small number and moderate heterogeneity of the included studies, along with the lack of data for other populations, these findings should be interpreted with caution. Additional researches are required for confirmation.
Supplemental Material
Supplemental Material - Ten-day vonoprazan-based versus fourteen-day proton pump inhibitor-based therapy for first-line Helicobacter pylori eradication in China: A meta-analysis of randomized controlled trials
Supplemental Material for Ten-day vonoprazan-based versus fourteen-day proton pump inhibitor-based therapy for first-line Helicobacter pylori eradication in China: A meta-analysis of randomized controlled trials by Wenwen Gao, Qian Wang, Xiang Zhang and Lu Wang in International Journal of Immunopathology and Pharmacology.
Footnotes
Author contributions
WWG ang LW designed and supervised the project. WWG, QW and XZ prepared the manuscript and analyzed the data.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by Natural Science Foundation of Shandong (ZR202210300051), Jinan Science and Technology Bureau (#202134050) and Youth Science Foundation of Shandong First Medical University (202201-136).
Supplemental Material
Supplemental material for this article is available online. Additional supporting information may be found online in the Supporting Information section at the end of the article.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
