Abstract
Bone turnover markers of resorption and formation are released during the process of bone remodelling. These markers have been extensively studied in a number of therapeutic trials of osteoporosis during the past decade. This has led to better understanding of their physiology, clinical applications and possible ways to optimize analytical techniques. Bone markers can complement the results of bone mineral density in the management of osteoporosis, but their use in clinical practice is challenged by pre-analytical and analytical variability. This review will discuss different types of bone markers, their limitations, use in different metabolic bone diseases and current recommendations from the International Osteoporosis Foundation and the International Federation of Clinical Chemistry and Laboratory Medicine bone marker standards working group.
Introduction
Bone is a dynamic tissue consisting of cells and an extracellular matrix which contains organic (35%) and inorganic (65%) molecules. The organic matrix is composed predominantly of type I collagen (90%) with small amounts of non-collagenous proteins such as osteocalcin (OC), glycoproteins and proteoglycans. Inorganic calcium and phosphate form hydroxyapatite crystals which mineralize the organic matrix. 1 Three main types of bone cells are involved in the process of bone remodelling, namely osteoclasts, osteoblasts and osteocytes. Osteoblasts lay down and mineralize new bone matrix, while osteoclasts are responsible for bone resorption. Osteocytes mainly act as mechanosensors.1–3
Bone remodelling occurs in discrete foci of cells called basic multicellular units. It is a cyclical process essential for skeletal growth and comprises four phases: resorption, reversal, formation and a resting phase as shown in Figure 1. In a typical cycle, the resorption phase lasts about 7–10 days while formation takes up to 2–3 months.2–5 In the healthy young adult skeleton, bone turnover is in balance and no net loss of bone occurs.
6
The rate of bone remodelling is influenced by mechanical stress, systemic hormones and local factors (such as cytokines and growth factors).3,7 The changes in bone remodelling are reflected by bone markers, produced during different phases of remodelling.
Stages of bone remodelling.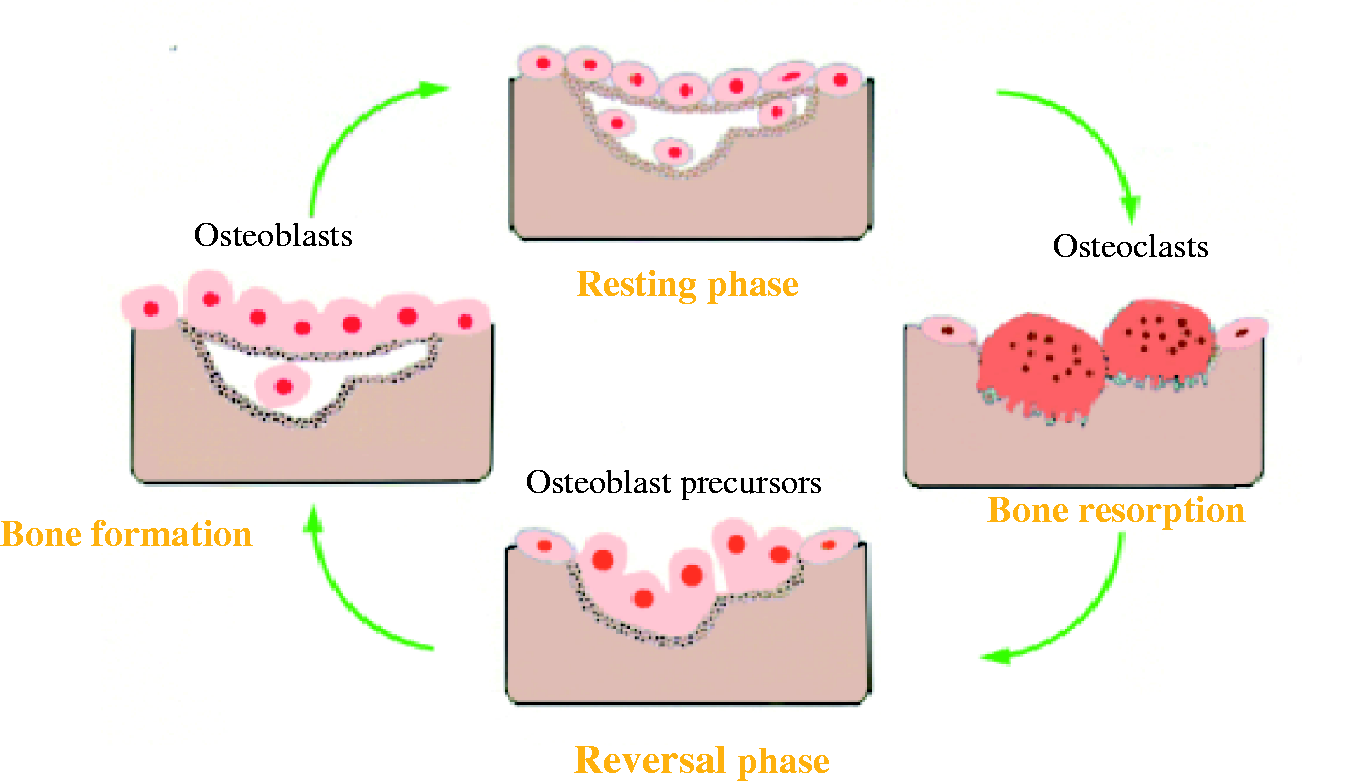
Bone turnover markers
Bone markers can be divided into formation and resorption markers, although some markers may reflect both activities. Bone formation markers are produced by osteoblastic cells or derived from procollagen metabolism, whereas resorption markers are the degradation products of osteoclasts or collagen degradation. 8 The ideal bone marker is specific to bone tissue, with an accurate and precise laboratory method available for its analysis.
Bone formation markers
Procollagen type 1 extension peptides
Type 1 collagen is derived from its precursor, procollagen. The procollagen molecule contains amino- and carboxy-terminal extensions, which are removed enzymatically during extracellular processing, resulting in the production of type 1 collagen. This cleavage results in the release of procollagen type 1 N-terminal propeptide (P1NP) and procollagen type 1 C-terminal propeptide (P1CP).2,9 Although the majority of P1NP and P1CP is produced primarily from bone, a small amount may originate from skin, cartilage and tendon as type 1 collagen matrix is also present in these tissues.
P1NP can be present in the circulation as four different forms: an intact trimeric form, a dimeric form, a monomeric form and a fragmented form. The original intact trimeric form is unstable and degrades to more stable monomers and fragments at body temperature.10–12 On the other hand, P1CP is a single protein containing mannose-rich carbohydrate side chains inserted post-translationally.
P1NP is cleared through a scavenger system, and P1CP is cleared through mannose receptors of liver endothelial cells.8,13
P1CP is stable at room temperature for 15 days,14,15 and P1NP is stable for five days at both room temperature and at 4℃. 16
Both P1CP and P1NP can be measured in serum. The analysis of P1NP is more extensively described in the literature than that of P1CP. There are two commercially available assays for analysis of serum P1NP. The manual Orion radioimmunoassay (RIA) detects only the trimeric form (intact P1NP), whereas the high throughput automated Roche Elecsys 2010 analyser recognizes both the trimeric form and thermal degradation forms of P1NP except for fragments (total P1NP). Garnero et al. evaluated the performance of the fully automated Roche Elecys analyser versus the manual competitive RIA method (Uniq™ P1NP Orion Diagnostica) by measuring P1NP in 59 healthy pre- and postmenopausal women. The serum P1NP concentrations obtained from the automated analyser were found to be significantly higher than those measured by manual RIA which can be explained by the fact that manual RIA only detects the intact trimeric form and underestimates P1NP concentration. In addition, the automated Roche Elecys is reported to have a greater sample throughput with better analytical reproducibility than the manual RIA. 16
Procollagen peptide concentrations are reduced by antiresorptive treatments (such as oestrogen and bisphosphonates) and are increased by the anabolic agents such as teriparatide (human recombinant parathyroid hormone).17,18
Bone-specific alkaline phosphatase
Alkaline phosphatase (ALP) is a membrane-bound enzyme and is present in almost all body tissues. There are four isoenzymes of ALP in the circulation: intestinal, placental, germ cells and a non-specific form. The tissue non-specific ALP gene encodes kidney, liver and bone isoforms of ALP. During childhood and adolescence, the bone isoform predominates whereas in healthy adults, liver and bone isoforms contribute 95% of total ALP and are present in almost equal proportions. 2 Bone-specific alkaline phosphatase (BALP) is produced by osteoblasts and its production is correlated positively with bone formation rate as measured by histomorphometry. 10
BALP is measured in serum and many methods have been developed to differentiate BALP from the liver isoform. The simplest method is the heat inactivation of BALP, but the rate of heating has been difficult to standardize. Other reported methods include electrophoresis, wheat germ lectin inactivation, neuraminidase inhibition, and immunoradiometric, immunoassays and high-performance liquid chromatography (HPLC)-based methods.2,19–21 Among these, immunoassays provide better assay reproducibility and precision than other methods but cross-reactivity with the liver isoform is still an issue with all types of immunoassays (∼15% with BALP Ostase immunoassay, ∼3–10% with Alkphase-B™ enzyme immunoassay). Thus, results must be interpreted with caution in patients with liver disease. BALP immunoassays generally have greater sensitivity and specificity for bone formation than total ALP.22,23
Osteocalcin
Osteocalcin (OC) is a small non-collagenous protein synthesized by osteoblasts. Its synthesis is stimulated by 1,25-dihydroxyvitamin D. OC contains three glutamic acid residues, which are converted to gamma-carboxyglutamate (gla) by vitamin K-dependent post-translational carboxylation.2,17 The carboxylation process leads to a conformational change in protein which allows the binding of OC with hydroxyapatite and mineralization in the bone matrix. There are also non-carboxylated or undercarboxylated forms of OC released into the circulation, 24 but its significance in postmenopausal osteoporosis fracture prediction is less than intact OC.
OC in the circulation is short-lived (half life ∼5 min) and is rapidly degraded into fragments. Thus, the circulating OC contains intact OC, large (1–43) N-terminal/mid-region fragments and small (20–40) mid C-terminal fragments. The smaller OC fragments are thought to be derivatives of bone resorption. OC is metabolized by the liver and kidneys. Due to its renal clearance, higher concentrations are observed in renal failure.2,25,26
The majority of assays measure OC in serum, but some methods use lithium heparin plasma. A previous study has shown that OC values can be affected by the presence of anticoagulants in plasma, especially oxalate and fluoride.
27
Samples should be collected on ice and separated immediately as OC is highly unstable
Available methods include RIA, enzyme-linked immunosorbent assay (ELISA) or chemiluminesence immunoassays. 26 Due to the heterogeneity of OC in the circulation, varying results are observed when different assays are used. Assays that detect the intact OC and large N-terminal fragment of OC are the most reproducible.15,20,29
Despite being a sensitive marker of bone formation, the use of OC measurements in clinical practice is limited by assay variability, sample instability and high biological variability.
Bone resorption markers
Tartrate-resistant acid phosphatase
Tartrate-resistant acid phosphatase (TRACP) is a member of a heterogeneous group of lysosomal enzymes. There are two forms of TRACP in the circulation: TRACP5a and TRACP5b. These two isoforms are structurally and antigenically the same, but they have a different pH optimum and different carbohydrate contents, 5a but not 5b containing sialic acid. TRACP5b is derived from osteoclasts, and TRACP5a is found in activated macrophages.2,15,20 During bone resorption, osteoclasts secrete TRACP5b which produces reactive oxygen species to digest bone degradation products in the microenvironment of the bone matrix. Therefore, it is used as an index of osteoclast activity and numbers. 30 Once released into the circulation, it is hydrolysed by proteases and forms fragments that are metabolized in the liver and are excreted in the urine. TRACP5b is not affected by renal function or dietary intake.30,31
TRACP5b is stable in serum samples with approximately 90% detectable after three days of storage at 4℃.
TRACP5b can be measured in the serum by enzymatic and immunoassay techniques. However, enzymatic assays are non-specific because serum contains other forms of TRACP enzymes derived from erythrocytes and platelets. In addition, bilirubin interferes with the end product of hydrolysis in spectrophotometric assays.2,15,30 The immunoassay that uses an antibody specific for TRACP isoform 5b detects active TRACP5b but not fragment forms.30,31 It has been extensively studied and shown to have good specificity, sensitivity, an acceptable intra- and inter-assay coefficient of variation (CV) and good correlations with other resorption markers. 32
Collagen cross-link molecules
Pyridinoline (PYD) and deoxypyridinoline (DPD) are molecules which mechanically stabilize collagen molecule by cross-linking between individual collagen peptides (Figure 2). PYD is found in cartilage, bone, ligaments and blood vessels, whereas DPD is almost solely found in bone and dentin. Thus, DPD is a more specific and sensitive marker than PYD.2,33 Collagen cross-links are formed during the extracellular maturation of collagen and are released into the circulation from degradation of mature collagens. PYD and DPD are excreted in the urine in free (40%) and peptide-bound (60%) forms.
Mature collagen fibrils with N-terminal and C-terminal telopeptides and pyridinium crosslinks linking between adjacent fibrils.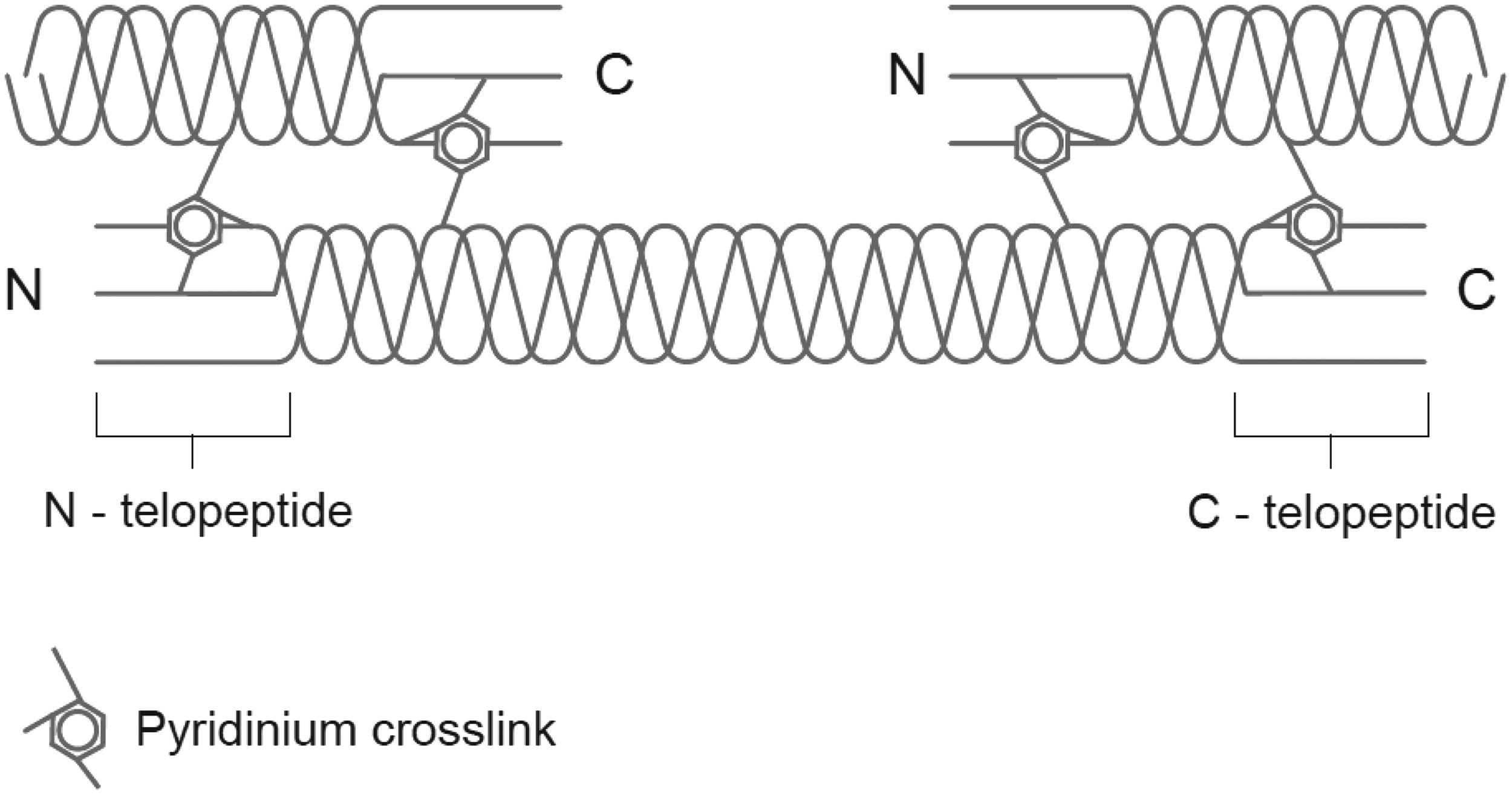
Cross-links can be measured in both timed and random urine samples, but early morning spot urine is a preferred sample due to diurnal variation of the cross-links. Although cross-links are not affected by dietary intake, exposure of urine samples to UV light will lower DPD and PYD concentrations. 34
Previously, PYD and DPD were measured by a reverse phase HPLC technique, but more recently, automated immunoassays detecting both free and peptide forms or free forms alone have been developed. Increased production of collagen cross-links is seen in metabolic bone diseases associated with increased bone turnover. 35
Cross-linked telopeptides of collagen I
Telopeptides of type 1 collagen are the most extensively studied and used bone resorption markers. There are two forms depending on the cross-link forming site with collagen: the N-terminal telopeptide (NTX) and C-terminal telopeptide (CTX), which are released during collagen degradation (Figure 2). CTX undergoes post-translational modifications known as racemization and isomerization. These processes give rise to four isomers of CTX: the native form (á-L) and three age-related forms which are an isomerized form (â-L), a racemized form (á-D) and an isomerized/racemized (â-D) form. The ratio of these isomers is informative since an increased ratio of áCTX/âCTX is associated with accelerated bone turnover. This has been described in patients with Paget’s disease or bone metastases and in postmenopausal women with rapid bone loss.36–38
Both NTX and CTX are excreted in the urine and can be measured in 24-h urine samples, in early morning spot urine samples and in serum. The timing of the sample collection is critical due to the marked circadian variation which is more pronounced for CTX. Previous work has shown that fasting significantly reduces circadian variations. Thus, fasting morning samples are required for optimal clinical use.39,40
Available analytical methods include ELISA, RIA and electrochemiluminescence assay. An automated immunoassay using antibodies specific for â-CTX fragments in serum was reported to be a precise, high throughput assay after measuring serum CTX in 728 healthy women as the results obtained correlated with bone loss and fracture risk. 41 The clear advantage of using a serum marker over a urine marker is that it offers better reproducibility because it avoids the need to correct for creatinine excretion which adds variability to the result.
Hydroxyproline
Hydroxyproline (OHP) is an amino acid that is derived from the post-translational hydroxylation of proline. It contributes about 13–14% of the amino acid content of collagen, and hence, it is found in collagen-containing tissues other than bone such as cartilage and skin. OHP in the circulation can also be derived from dietary intake and the C1Q component of complement.
Although it is used principally as a resorption marker, OHP is released into the circulation during bone resorption from collagen degradation as well as during bone formation (∼10%) from newly synthesized procollagen peptides. Therefore, OHP is neither specific to bone tissue nor to the resorption process.
About 10% of the OHP released during the collagen breakdown is in the form of small polypeptide chains that are small enough to be filtered and excreted through the kidney without further metabolism. The remaining 90% of the liberated OHP is degraded to free amino acid form which is filtered freely by the glomerulus and reabsorbed in the tubules. It is finally metabolized by the liver to urea and carbon dioxide.2,42
OHP is measured in urine by colorimetric or HPLC methods. Its use has been superseded by more sensitive and specific markers.8,20,43
Hydroxylysine-glycosides
Collagen contains hydroxylysine which is an amino acid formed by post-translational modification of lysine. There are two forms of hydroxylysine, namely galactosyl hydroxylysine (GHL) and glucosyl-galactosyl-hydroxylysine (GGHL). GHL is specific for bone, while GGHL is present in skin and complement molecules. 2 Hydroxylysine is released into the circulation during collagen degradation and is excreted in the urine.
Unlike OHP, hydroxylysine is not influenced by dietary intake and its glycosides are only derived from bone resorption. Therefore, it is regarded as a better marker than OHP.
Hydroxylysine is measured in urine by HPLC, but its use has not been extensively studied in relation to fracture risk prediction or monitoring treatment. Its use in clinical practice is limited due to lack of availability of routine methods for its measurement. 42
Bone sialoprotein
Bone sialoprotein (BSP) is a phosphorylated non-collagenous glycoprotein of bone matrix. It is found only in mineralized tissues such as bone, dentin and hypertrophic cartilage. It is produced by osteoblasts, odontoblasts and osteoclasts. BSP is thought to be important for cell-matrix adhesion processes and appears to stimulate osteoclast-mediated bone resorption. 44
BSP is measured in serum by RIA or immunoassay. The latter uses polyclonal antibodies, and previous work has shown that not all bioavailable BSP is detected by this assay. 43
Similar reductions in serum concentrations are seen to those observed with other markers of resorption such as cross-links and telopeptides, in response to antiresorptive therapy. 45
Cathepsin K
Cathepsins are members of the cysteine protease family, and 11 isoforms have been identified. Cathepsin K, in particular, is expressed at the ruffled border of actively resorbing osteoclasts. 46 Osteoclasts secrete cathepsin K into bone resorption lacunae for degradation of bone matrix proteins including type I collagen, osteopontin and osteonectin, and hence, cathepsin K plays an important role in bone resorption. It is produced in a precursor form containing 329 amino acids, which is later cleaved to 215 amino acids to yield the active form. Cathepsin K inhibitors are currently being evaluated as a potential treatment option for osteoporosis. 47
Cathepsin K is measured in serum by ELISA. Although it is potentially a useful marker of bone resorption, being expressed and secreted by osteoclasts, further studies are required for commercial use. 12
Osteoprotegerin and receptor activator for nuclear factor kappa-B ligand
Osteoprotegerin (OPG) is a soluble member of the tumour necrosis factor-á (TNF-á) superfamily and acts as a decoy factor for receptor activator for nuclear factor kappa-B ligand (RANKL), inhibiting osteoclast formation and activity. RANKL is a member of TNF receptor superfamily and is produced by osteocytes, preosteoblasts and endothelial cells. RANKL binds with its receptor, RANK, in the presence of macrophage colony-stimulating factor, resulting in increased osteoclast formation and activity. RANK is expressed by osteoclast precursors. 48
OPG can be measured in serum, plasma EDTA, citrate and heparin samples. RANKL can be measured in serum or plasma, but its stability in lithium heparin plasma is uncertain. There are commercially available sandwich ELISA assays for analysing OPG and RANKL, using a monoclonal capture and polyclonal detection antibodies.
A significant decrease in OPG and RANKL concentrations was described previously after storing for six months at −70℃, and it is suggested that samples are either analysed immediately or stored at −70℃ for short term. Investigators are recommended to perform their own sample stability assessment. 49
The use of serum RANKL and OPG in fracture risk prediction and monitoring the response to osteoporosis treatment has been investigated, but results have been inconsistent. Thus, further improvement in assay methods and better understanding of the causes of variability is still needed. 49
Lists of bone markers and required types of samples.
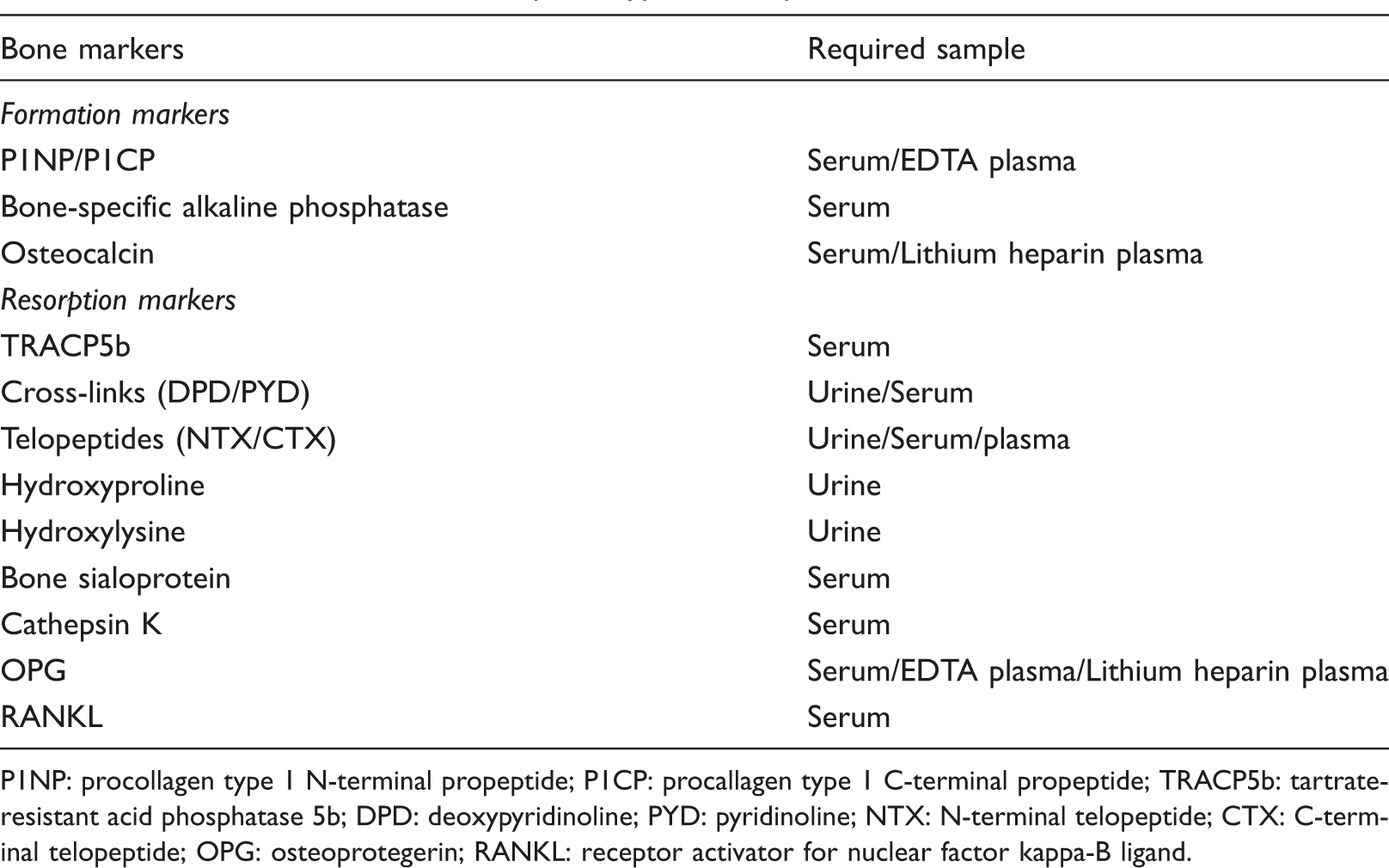
P1NP: procollagen type 1 N-terminal propeptide; P1CP: procallagen type 1 C-terminal propeptide; TRACP5b: tartrate-resistant acid phosphatase 5b; DPD: deoxypyridinoline; PYD: pyridinoline; NTX: N-terminal telopeptide; CTX: C-terminal telopeptide; OPG: osteoprotegerin; RANKL: receptor activator for nuclear factor kappa-B ligand.
Variability
Like any other biological markers, sources of variability can be divided into pre-analytical, analytical and post-analytical. In general, bone formation markers are less variable than resorption markers. Urinary markers show more variability than serum markers.
Pre-analytical variability
This can be divided into technical and biological factors.
Technical factors
Sources of variability in bone markers.

Biological factors
Endogenous
Exogenous
Analytical variability
The analytical variability of bone markers has greatly improved with the development of fully automated platforms. However, inter-laboratory variation on the same sample, even using the same method, can be substantial. The recorded inter-laboratory CVs are 39% for NTX, 30% for PYDs and 68–71% for OC. 67 This is due to complex, multiple factors such as the variety of available immunoassays using different epitopes and reagents, instability of calibrants and variation between manufactured quality control materials. Thus, it is mandatory to provide information to clinicians that bone marker results may not be comparable between different laboratories. 67
Ideally, a laboratory should derive its own reference ranges for each specific group of patients to accommodate age, gender and ethnicity variations.
Bone markers in clinical practice
Osteoporosis
Osteoporosis is characterized by low bone mass and increased risk of fractures. It is estimated that one in two women and one in five men over the age of 50 in the UK will sustain a fracture as a result of osteoporosis. The diagnosis of osteoporosis can be made by bone mineral density (BMD) measurement using dual-energy X-ray absorptiometry (DXA). BMD is usually measured in the lumbar spine and the hip and occasionally in the forearm. It is expressed in g/cm2, but values differ between machines and manufacturers. Therefore, T-scores are used, defined as the number of standard deviations from peak bone mass of healthy young adults. The World Health Organisation (WHO) criterion for diagnosing osteoporosis in postmenopausal women is a T-score ≤ −2.5. 68 However, there is a considerable overlap in BMD values between fracture and non-fracture groups, highlighting the role of other clinical risk factors for development of fractures. 69 A fracture risk assessment tool (FRAX) has been now developed, supported by the WHO that uses clinical risk factors (for example, age, gender, family history of osteoporosis and postmenopausal status) and hip BMD to calculate 10-year probability of major osteoporotic fractures and of hip fractures and to identify patients requiring treatement. 70 However, it is not certain if FRAX could also be applied in patients who are already on osteoporosis treatment. A recent study on 35,764 women (≥50 years) by Leslie et al. 71 suggested that FRAX may have a role in patients who are on current treatment as well as who have past treatments, to decide the need for continued therapy.
Whereas DXA scans provide information on BMD, bone markers, in contrast, indicate the rate of bone remodelling. Bone markers are currently not recommended for use in the diagnosis of osteoporosis. However, they have a potential role in the monitoring of treatment as the changes that occur are more rapid, larger and more closely related to fracture reduction when compared to BMD changes.
Prediction of bone loss
Accelerated bone turnover is associated with higher risk of fracture independent of age, gender or underlying diseases. Previous studies have demonstrated that increased concentrations of bone markers in postmenopausal women are associated with more rapid bone loss. Bone loss at different sites including the forearm, spine and hip has been studied using a variety of markers. Overall, resorption markers were shown to be more strongly predictive of the rate of bone loss than formation markers.72–75
The relationship between rate of bone loss at the hip and markers of resorption and formation in men >65years of age was recently studied using P1NP, β-CTX and TRACP5b. It was found that men in the highest quartile of P1NP showed greater hip bone loss, but no significant association with β-CTX and TRACP5b was established. 76
Prediction of fractures
Large population-based studies have demonstrated that high bone turnover is associated with increased risk of osteoporotic fractures and that bone markers may be useful in prediction of fractures independent of BMD. The association between fracture risk and bone formation markers is weaker than that for bone resorption markers. Three studies (Rotterdam study, EPIDOS and OFELY) have established that high levels of resorption markers were associated with increased risk of hip, vertebral and non-vertebral fractures in healthy postmenopausal women. In the prospective EPIDOS study,77,78 no significant association was found between OC, BALP and urine NTX and fracture, but increased CTX and free DPD were significantly correlated with fracture risk in elderly postmenopausal women. In addition, a greater risk of hip fractures was seen in postmenopausal women with low BMD and high resorption markers than those with high resorption markers alone or low BMD alone.36,74,78,79
The association between bone markers and fracture risk in men is less well studied. Increased bone resorption was also found to be associated with increased risk of fracture, independent of BMD, in elderly men in the Dubbo Osteoporosis Epidemiology Study in Australia. 80 Although these findings from population-based studies may not be directly transferable to individual cases, there is some evidence to suggest that bone turnover markers may improve fracture risk assessment when used in conjunction with BMD and other clinical risk factors in both men and postmenopausal women.
Selection of treatments
Therapeutic options for the treatment of osteoporosis have greatly advanced over the last decade with the development of antiresorptive agents (e.g. bisphosphonates) and anabolic agents (human recombinant parathyroid hormone, PTH, peptides). In theory, it might be desirable to use antiresorptive drugs for patients with high bone turnover and anabolic agents for those with low bone turnover. This concept has not been widely studied, but if shown to be valid, it might be useful for selecting treatment options in clinical practice.
Monitoring of treatment efficacy and compliance to treatment
Although BMD is used to diagnose osteoporosis, it has limitations in the monitoring of treatment efficacy. First, changes in BMD take longer to occur than bone markers. Second, interpretation of follow-up DXA scans requires documentation of the reproducibility of measurements and estimation of the least significant change (LSC). Last, changes in BMD explain only a small proportion of the fracture reduction in treated patients.81,82
Bone turnover markers change in response to antiresorptive agents as early as two weeks and reach a plateau within 3–6 months. Treatment with oestrogen, raloxifene, bisphosphonates and denosumab (a humanized monoclonal antibody to RANKL) are associated with a decrease in bone resorption (CTX, TRACP5b, DPD) and formation markers (P1NP and OC), whereas human recombinant PTH treatment is associated with an early increase (within two days of starting treatment) in bone formation markers (P1NP) followed by an increase in bone resorption markers.30,81,83–87 P1NP is the most sensitive marker of response to teriparatide treatment.
At present, there are no consistent recommendations for the calculation LSC for bone markers. The LSC for bone turnover markers is greater than for BMD, but on the other hand, bone turnover changes are larger. A common aim of antiresorptive therapy in the treatment of postmenopausal osteoporosis is to achieve bone marker concentrations within the premenopausal range. The extent and direction of bone marker changes depend on the therapeutic agent, dose and mode of administration and the sensitivity of the marker.75,81,88 Telopeptides were reduced up to 50–70% after 12 weeks of bisphosphonate therapy in contrast to only 20% reduction with TRACP5b.32,89 It was also shown that women on hormone replacement therapy (HRT) who had the highest quartile of percentage change from baseline to six months in NTX, BALP and DPD had the greatest change in spine BMD after one year in the study where early postmenopausal women were randomized to HRT or calcium supplements for one year. 75
Ensuring adherence to treatment for osteoporosis is a major challenge. A significant fall in bone markers after starting a treatment indicates that medication is being taken regularly and is absorbed properly. However, to what extent the measurement of bone markers and subsequent feedback to patients improve the compliance is uncertain. In one study, persistence with risedronate therapy was greater in patients provided with feedback about a good NTX response (>30% decline from baseline) but actually declined in patients who were given feedback about a poor NTX response.90,91
Other metabolic bone disorders
Paget’s disease
Paget’s disease is the second most common metabolic bone disorder in the UK. Its prevalence is about 1–2% in people older than 55 years but may increase up to 8% in men and 5% in women above 80 years of age. 92 It is a chronic bone disease characterized by a marked increase in osteoclastic bone resorption followed by abnormal, excessive bone formation, resulting in enlarged and deformed bones. It can affect one (monostotic) or multiple (polyostotic) bones. The spine, sternum, skull, pelvis, hip, upper arm, tibia and ribs are commonly affected by Paget’s disease. Symptoms and signs include bone pain, deformity, neurological complications and fractures, but the majority of patients with Paget’s disease are asymptomatic and are often diagnosed incidentally by the finding of a raised serum ALP during investigation for unrelated disease conditions. The diagnosis can be further confirmed by performing plain X-rays of affected bones and isotope bone scanning.93,94
Since Paget’s disease is associated with increased bone turnover, bone markers are helpful in assessing disease activity. The type and extent of bone marker changes depend on the extent and severity of disease and level may be normal in monostotic disease. Bone formation markers (total ALP, BALP, P1NP,OHP) and resorption markers (urine NTX, serum CTX, urine CTX) are significantly higher in untreated Paget’s patients than in healthy individuals.95,96 In a comparative study on the biological variation of bone markers in Paget’s disease, serum markers showed less variation than the urine markers. This study reported that serum BAP and P1NP were the most sensitive markers in monitoring disease progress in Paget’s disease. 95 However, a later review in 2002 by Selby et al. has suggested using serum total ALP as the standard marker, since this has an analytical sensitivity of 74% and is more readily available in routine practice. Although Paget’s disease is primarily a disorder of osteoclasts, the analytical sensitivity of resorption markers is less satisfactory than that of formation markers in monitoring disease activity. 97
Paget’s disease is most commonly treated with bisphosphonates, which reduce osteoclastic bone resorption. The main aim of treating Paget’s disease is to relieve symptoms such as pain and to prevent complications. The response to treatment can be monitored by improvement in symptoms, bone markers or radiological appearances. The changes in bone markers can be observed rapidly and are easily detected. Bone markers (total ALP, BALP, P1NP, serum CTX, OHP, urine NTX and urine CTX) were studied in a group of patients being treated with tiludronate for three months. Mean values of all markers decreased after treatment, but BALP and P1NP were the most sensitive markers for monitoring changes, followed by total ALP and urine NTX. OHP, serum and urine CTX had lower sensitivity to detect changes following treatment. 93 Bone markers should be assessed six months after starting bisphosphonate therapy, when the nadir level is seen. BALP changes earlier than total alkaline phosphatase when the disease relapses. 96 In summary, BALP and P1NP are more sensitive and specific than total ALP, but total ALP is readily available which makes it more appropriate for routine use. BAP and P1NP measurements should be reserved for those with liver disease and for some cases with monostotic disease when interpretation of total ALP may be problematic. 97
Metastatic bone diseases
Bone metastases are seen in a variety of cancers; up to 73% in breast cancer (BC), 68% in prostate cancer and nearly 100% in myeloma patients. 98 Common sites of metastases include the vertebral column, skull, ribs and the proximal ends of the long bones, which have a rich blood supply. Bone lesions can be osteolytic, osteoblastic or mixed. 99 BC and multiple myeloma typically give rise to lytic lesions as opposed to the osteoblastic lesions seen in advanced prostatic disease. These skeletal-related events (SREs) are associated with high mortality and morbidity. They can present as bone pain, pathological non-vertebral fractures, vertebral fractures and spinal cord compression. The purpose of treating metastatic bone disease is to reduce the incidence of SREs and to improve quality of life.100,101
Bone turnover markers change as a result of the alterations in bone remodelling induced by cancers. As assays for the markers of bone turnover have become more accessible and less expensive, there has been increasing interest in the use of these markers in the diagnosis, prognosis and prediction of the clinical outcome in metastatic bone disease.
Many studies have shown that bone turnover markers, especially resorption markers, are raised in the majority of metastatic bone diseases102–104 and are usually higher than levels found in postmenopausal osteoporosis. 95 However, their use in the diagnosis of bone metastases is not yet fully validated. Lipton et al. investigated retrospectively the association of normalization of urine NTX after zoledronic acid treatment and increased survival in patients with metastatic BC, hormone-resistant prostate cancer (HRPC), non-small cell lung cancer (NSCLC) and other solid tumours (OST). After three months of zoledronic acid treatment, urine NTX concentrations reverted to normal in those with baseline elevated NTX (BC-81%; HRPC-70%; NSCLC/OST-81%). Reduction in SREs and death rates was seen in patients whose urine NTX reverted to normal versus persistently elevated urinary NTX. 105 Therefore, normalization of bone turnover markers seems a reasonable target of treatment with antiresorptive agents and is used as a surrogate end point in the development of drug therapies. However, it is important to recognize that these associations between bone markers and outcomes of SREs have predominantly been derived from retrospective analyses. A number of ongoing prospective trials, OPTIMIZE 2 (a prospective, randomized, double-blind, multi-centre, two-arm trial of the continued efficacy and safety of zoledronic acid [every four weeks versus every 12 weeks] in patients with metastatic BC) and the BISMARK (cost-effective use of bisphosphonates in metastatic bone disease: bone marker-directed zoledronic acid therapy compared with a standard schedule), should offer a greater insight into their diagnostic and prognostic values. Lastly and more importantly, variability of markers should receive specific attention in bone metastases as there could be multiple confounding factors (tumour burden, malnutrition, chemotherapy, radiotherapy, immobility) causing variations in their concentrations.
Overall, bone markers may have a role in monitoring effective treatment of antiresorptive therapy, but currently available data do not support their use as diagnostic or prognostic indicators in bone metastases.
International Osteoporosis Foundation–International Federation of Clinical Chemistry and Laboratory Medicine bone marker standards working group
Although bone markers have considerable potential to be used in the management of osteoporosis, their use is greatly hindered by biological and analytical variability. To address this, the International Osteoporosis Foundation (IOF) and International Federation of Clinical Chemistry and Laboratory Medicine (IFCC) formed a joint working group to standardize bone markers. The aim of the group was to produce recommendations on the applications of bone turnover markers in fracture risk prediction and monitoring of osteoporosis treatment based on current available evidence. It was concluded that available data on the same bone marker were limited to comparison of treatment outcomes. The position paper published in 2011106 recommended the use of one formation marker (P1NP) and one resorption marker (CTX) in future clinical trials and observational studies to strengthen the knowledge of applications of bone markers in clinical practice.
Recommendations
The following steps should be considered to minimize variability of bone markers and hence, to provide better results.
Pre-analytical: Fasting morning serum or urine samples should be collected over 2–3 days and possibly at the same time of the day. Information on appropriate sample handling and protection from light must be made available to staff and physicians. Analytical: Implementation of IOF-IFCC’s recommendation on the use of P1NP and CTX as reference standard markers in future clinical studies should provide better understanding of the performance of different assays in different populations. It is also mandatory for laboratories to participate in the external quality control schemes to minimize inter-laboratory variations. Further work is needed to develop standard materials and reference laboratory networks. Post-analytical: Laboratories should derive and validate appropriate reference ranges for relevant population groups to enable correct interpretation of values. By providing more precise and consistent results, the utility of these markers in an individual patient setting would be improved. Finally, a consensus guideline on indications, choice of markers and the frequency of testing would promote uniform practice across the health service.
Conclusion
From the molecular level to clinical trials, the ability of bone markers to reflect bone remodelling in physiological and pathological states has been demonstrated. Bone markers have the potential to serve as useful tools in the monitoring of osteoporosis therapy to identify non-responders and may also have a role in fracture prediction. Other applications in bone disease also require further validation.
Footnotes
Acknowledgements
This article was prepared at the invitation of the Clinical Sciences Reviews Committee of the Association of Clinical Biochemistry.
Declaration of conflicting interests
None declared.
Funding
This research received no specific grant from any funding agency in the public, commercial, or not-for-profit sectors.
Ethical approval
Not applicable.
Guarantor
TTH.
Contributorship
TTH researched the literature and wrote the first draft of the manuscript. JEC reviewed and edited the first draft. All authors reviewed and edited the manuscript and approved the final version of the manuscript.
