Abstract
With an aging population, the demand for sensitive and specific biomarkers to assess bone turnover has surged. Bone turnover involves 2 key processes: bone formation, during which Type I procollagen is cleaved into Type I collagen and subsequently mineralized into bone, and bone resorption, during which Type I collagen is demineralized and degraded into peptides by cathepsin K. To identify biomarkers that accurately reflect these processes, extensive efforts have been made to characterize the peptides generated during both formation and resorption. Over the years, numerous biomarkers have been discovered for various disorders. However, despite their clinical utility, many of these markers lack specificity. This is due to factors such as the degradation of trimers into monomers, the coexistence of multiple peptide species arising from the unpredictable cleavage of Type I collagen/procollagen by cathepsin K and metalloproteinases, and the lack of assay standardization. Standardization is further hindered by the incomplete characterization of many of these peptides. For accurate assay development, a gold-standard technique like LC-MS/MS is essential, requiring full peptide characterization during method development. This review aims to present recent advances in the characterization of Type I collagen-derived peptides, providing a foundation for improved biomarker standardization and application in clinical practice.
Plain language summary
The aim of this review is to present the recent advances in the characterization of Type I collagen-derived markers. In this work, the current knowledge about the structure of all of the markers derived from Type I collagen, namely PINP, PICP, hydroxylysine, pyridinoline, CTX, NTX, 1α1 homotrimer and ICTP, is thoroughly described in parallel with the quantification methods and the challenges faced by these quantification methods, mainly immunoassays. The discussion focuses on the general challenges that need to be overcome to obtain a standardization of the different commercially available kits for the quantification of all these markers. These challenges encompass the characterization of the markers and the development of gold standard methods for their accurate quantification.
Keywords
Introduction
Collagen, as the primary structural protein in the animal kingdom, has been the subject of extensive research for many years due to its great abundance. In 1935, Clark et al provided the first description of the regular nature of collagen’s structure using X-ray diffraction techniques. 1 It was not until the mid-1950s that the triple helix structure of collagen was fully elucidated. Ramachandran and Kartha achieved this breakthrough by studying X-ray and infrared spectra, as well as the chemical composition of collagen derived from rat and kangaroo tail tendons.2,3 Subsequently, high-resolution studies, such as X-ray diffraction, confirmed the quaternary structure of collagen.4 -8 Concerning the primary structure, Berisio et al discovered a repetitive pattern in the amino acid sequence, where X – Y – Gly predominated, with X and Y primarily represented by proline and hydroxyproline residues. 9 The high abundance of glycine residues in collagen, 1 in 3 residues in the helical part, is crucial for the formation and stability of the triple helix.10,11 Both proline and hydroxyproline residues are rigid and aromatic, which restricts the rotation of the polypeptide backbones and enhances stability.12,13 Additionally, hydroxyproline contributes to helix stabilization through hydrogen bonding with water and by serving as a critical site for collagen’s intra- and inter-crosslinks.14 -16
To date, researchers have identified 28 Types of collagen and at least 46 polypeptide chains.17,18 Although there are some differences among species, collagens exhibit a high degree of conservation across all animals. 19 This conservation even extends through the millennia, as collagen has been detected in a 68-million-year-old Tyrannosaurus Rex.20,21
From all of these Types of collagens, Type I collagen, classified as a fibrillar collagen, represents not only the most abundant form of collagen in the human body (90% of its total collagen content 22 ) but is also the most prevalent protein of the body. It is predominantly found in various tissues such as bones, skin, teeth, organ capsules, tendons, cornea, and fascia. 22
Similar to other collagen Types, Type I collagen adopts a triple helical structure due to the X – Y – Gly repetitive pattern in its primary structure. 22 It consists of 2 α1 chains and 1 α2 chain, encoded by COL1A1 and COL1A2 genes, respectively, or it may be composed of 3 α1 chains depending on the variant. 23 The triple helix is flanked by telopeptides, which are non-helical regions of the molecule and lack the X – Y – Gly structure. This particular structure enables Type I collagen to form elongated fibrils. Once self-assembled, the chains of Type I collagen form fibrils with diameters ranging from 25 to 400 nm. However, factors such as pH, ionic strength, and temperature can influence the fibril formation process.
The unique characteristics of Type I collagen have led to its extensive investigation in various fields, including tissue engineering, medical devices, pharmaceutical industry, cosmeceuticals, and the food industry.24 -29
Biomarkers Derived From Type I Collagen
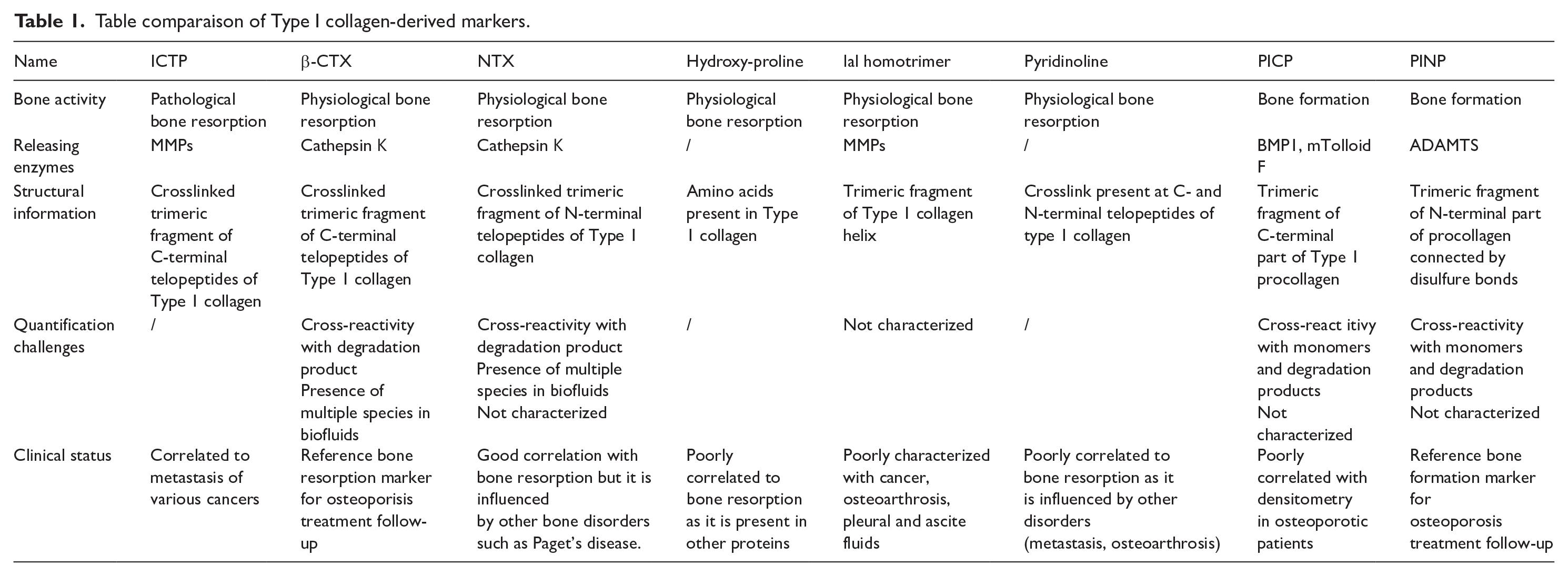
Given its widespread presence throughout the body, the digestion of Type I collagen and purification of its fragments has led to the identification of numerous biomarkers. These biomarkers have been utilized for diagnostic, prognostic, and follow-up purposes in various diseases. Figure 1 describes the release of these biomarkers from type I collagen during bone remodeling while Table 1 summerize the characteristics of each of them.

Summary of the Type I collagen-derived markers. PYR stands for pyridinoline, HYDROXY stands for hydroxyproline, BMP1 stands for bone morphogenic protein 1, mTolloid F stands for mammalian Tolloid F.
Table comparaison of Type I collagen-derived markers.
Biomarkers of Bone Turnover in Physiological Context
Bone consist of approximately 90% Type I collagen. 30 The collagen present in bone undergoes mineralization to provide stiffness and mechanical resistance. 31 The integrity of bone tissue is maintained through a process known as bone remodeling. 32 During bone remodeling, osteoclasts first acidify the surrounding environment to demineralize the extracellular matrix. They then release proteases, including cathepsin K, to digest collagen. 33 This process, referred to as bone resorption, helps reshape old bone tissue and address potential micro-fissures, preparing the ground for new bone tissue formation. 34 To generate new bone tissue, osteoblasts secrete collagen and other molecules into the extracellular matrix. As the newly formed matrix matures, osteoblasts become embedded within it and differentiate into osteocytes. Osteocytes play a crucial role in regulating osteoclast activity in response to mechanical stimuli applied to the bone tissue. The extracellular matrix is subsequently mineralized, completing the formation of new bone tissue. The cycle of bone resorption and formation is tightly regulated by various factors.
The balance between bone formation and resorption can be disrupted by many factors, including age, medication use, and nutritional deficiencies. One prominent example of this imbalance is osteoporosis, the most prevalent skeletal disorder. In individuals with osteoporosis, bone resorption surpasses bone formation, resulting in reduced bone mass and compromised bone architecture. While bone resorption is a natural process, it occurs at an accelerated pace in individuals with osteoporosis.
Several biomarkers derived from Type I collagen have been developed to assess bone remodeling. These markers can be classified into 2 main categories: bone formation markers and bone resorption markers.
Bone Formation Biomarkers
Bone formation biomarkers are derived from the synthesis of Type I collagen by osteoblasts. Initially, Type I collagen is synthesized as pre-procollagen by osteoblasts. 35 Subsequently, it assembles into a triple helix structure called pro-collagen within the endoplasmic reticulum.36,37 Procollagen, containing 2 pro-peptides known as the amino-terminal propeptide of procollagen Type I (PINP) and the carboxy-terminal propeptide of procollagen Type I (PICP), serves to prevent premature fibril formation within the cell. 38 These propeptides are subsequently cleaved by procollagen C-proteases (BMP1, mTolloid F) and ADAMTS (a disintegrin and metalloproteinase with a thrombospondin Type 1 motif) family proteases to form mature collagen fibers. 39 PICP and PINP, as bone formation biomarkers, can be measured in biological fluids. 40 Once the procollagen is cleaved, collagen fibers form large collagen fibrils, which are later crosslinked and mineralized. The propeptides are released into the bloodstream and can be eliminated in the urine or degraded by the liver.
PICP, a 100 kDa glycoprotein, is composed of 3 subunits connected by disulfide bonds, resulting in its globular structure.38,41 It undergoes glycosylation with high-mannose Type oligosaccharides. 41 Following bone formation, PICP enters the bloodstream and is eventually cleared by the liver due to its oligosaccharide structure, which allows it to be scavenged by mannose receptors present on the liver surface. Its large size suggests that it is not cleared by the kidneys. 41
Unfortunately, studies have shown that serum PICP poorly correlates with bone formation in patients with vertebral osteoporosis and does not correlate with the rate of bone loss measured by densitometry in osteoporotic patients. It is also highly influenced by bone metabolic diseases such as Paget’s disease, with 2-6-fold increased levels observed in affected patients compared to healthy individuals. Due to these limitations, PICP was not considered a reference bone formation marker.42 -44
In contrast, PINP is currently recognized as a reference biomarker for osteoporosis treatment follow-up by the International Federation of Clinical Chemistry and Laboratory Medicine (IFCC), the International Osteoporosis Foundation (IOF), and the National Bone Health Alliance.45,46 Similar to PICP, PINP consists of three subunits; however, in PINP, the subunits are linked by non-covalent bonds.47,48 Indeed, the three subunits are held together by a collagenous domain in the middle of the propeptide. The accurate mass of PINP is not yet known, but it has been reported as 35, 41 55, 35 and 70 kDa 36 in different studies. Its exact structure is not well understood, but it is known to be phosphorylated at serine residues and contains collagenous regions. 41 Unlike PICP, PINP has an elongated structure instead of being globular. After its release during bone formation, PINP enters the bloodstream and is cleared by the liver after endocytosis by scavenger receptors.
Although PICP and PINP originate from type I procollagen—a protein secreted by multiple tissues—no studies have reported the presence of PICP or PINP derived from non-bone tissues. Given that approximately 90% of type I collagen is found in bone, the likelihood that PINP and PICP suffer from a lack of bone specificity remains low or not significant.
Bone Resorption Biomarkers
As mentioned earlier, during bone resorption, osteoclasts secrete cathepsin K to digest collagen after demineralization. This digestion process leads to the complete dismantling of the 3 chains of collagen, resulting in the production of various collagen-derived peptides. Following the digestion of Type I collagen by cathepsin K, these peptides are released into the bloodstream before being eliminated in the urine or degraded by the liver.
In the early 1960s, hydroxyproline was the first molecule investigated as a bone resorption marker. 41 Its high specificity to Type I collagen in bones, due to the hydroxylation of proline residues, led to the belief that bone resorption would release significant amounts of this residue 49 and indeed, hydroxyproline was extensively used as a marker for assessing bone resorption. However, limitations were identified, including the fact that other proteins, such as the C1q component of complement, may release hydroxyproline and that it can be absorbed from the diet. 41 To overcome these limitations, attention shifted to the measurement of free pyridinoline crosslinks.
Given the high presence of pyridinoline crosslinks in Type I collagen of bone, they were investigated as potential bone resorption biomarkers to replace hydroxyproline. The free pyridinoline crosslink was first characterized in bovine Achilles tendon collagen by Fujimoto in 1977, and its intrinsic fluorescence allowed for its discovery. 50 The structure of pyridinoline was further confirmed through NMR analysis, and its molecular structure is depicted in Figure 2. Additional confirmations of the pyridinoline structure were obtained through fast atom bombardment mass spectrometry, electrophoresis, and ultraviolet spectroscopy. 51

Chemical structure of pyridinoline and deoxypyridinoline extracted from pubchem.
Unfortunately, although free pyridinoline was correlated with bone resorption, 52 it was found to be influenced by other conditions such as metastasis, 53 osteoarthritis 54 and rheumatoid arthritis. 55 Therefore, the development of new bone resorption biomarkers became necessary.
In the early 1990s, 2 crosslinked telopeptides were investigated as bone resorption markers. Since Type I collagen is widely distributed in the body, only crosslinked peptides can be considered valid biomarkers for assessing bone resorption.
C-Terminal Crosslinked Telopeptide of Type I Collagen (CTX) and N-Terminal Crosslinked Telopeptide of Type I Collagen (NTX) are degradation products of Type I collagen, crosslinked by pyridinoline—a bone-specific crosslink that ensures the specificity of these markers to bone tissue. These fragments are generated during collagen degradation by cathepsin K. 41 In 1996, Hanson et al described the site specificity of pyrrole and pyridinoline crosslinks in human bone. They produced bone powder from human bone and digested it using bacterial collagenase. The different compounds were then separated through multiple rounds of reversed-phase preparative LC and HPLC. Fractions were assessed for their content of crosslinked peptides using a custom immunoassay previously described in 1992. 56 The compounds in concentrated fractions were N-sequenced, and their masses were determined using electrospray ionization mass spectrometry. Several crosslinked peptides were identified, corresponding to N-terminal pyridinoline crosslinks and C-terminal pyridinoline crosslinks. Regarding CTX, the crosslink is formed by the condensation of 2 lysine residues at position 1208 in the α1 chain and 1 lysine residue at position 265 in the α1 chain or 1 lysine residue at position 1023 in the α2 chain. In 2024, the full structures of multiple species of CTX extracted from human plasma and serum were characterized using a combination of preparative LC, affinity chromatography and high-resolution mass spectrometry (HRMS). 57 The latter work highlighted the complexity of CTX mainly due to the nature of cathepsin K digestion during bone resorption 58 and metalloproteinases (MMPs) digestion of CTX species in the bloodstream. Such complexity may provide a solid explanation for the lack of harmonization observed in the CTX immunoassays. 59 For NTX, the crosslink seems to be formed by 2 lysine residues at position 170 of the α1 chain and 1 lysine residue at position 1108 of the α1 chain or at position 177 of the α2 chain. This work paved the way for further studies on NTX. Immunoassays were quickly developed to assess the utility of these molecules as bone resorption biomarkers.60 -65 CTX and NTX provided interesting results for the monitoring of osteoporosis pharmaceutical treatments. In 1997, Fledelius et al hypothesized that CTX, containing an aspartic acid-glycine sequence, may undergo β-isomerization, resulting in a perturbation of the peptide backbone. 66 β-Isomerization is associated with protein aging.67 -69 Based on this hypothesis, Fledelius et al generated antibodies against the peptide EKAHDβGGR and compared the concentrations obtained using their custom ELISA with the concentrations obtained using a commercially available kit targeting EKAHDGGR. Similar results were obtained by both immunoassays in human urine samples. 70 It was concluded that a significant portion of CTX present in biological fluids corresponds to β-isomerized C-terminal crosslinked telopeptide of Type I collagen (β-CTX). This finding aligned with the fact that bone resorption primarily affects old bone tissues and thus, β-isomerized peptides. To this day, β-CTX is defined as a reference biomarker for osteoporosis treatment follow-up, by the IFCC and the IOF.45,46 Both PINP and β-CTX are currently incorporated into an algorithm to evaluate treatment response in osteoporotic patients. In these patients, blood samples are collected right before the treatment and 3 months after the beginning of the treatment. A significant decrease in CTX and PINP levels after 3 months, followed by sustained low levels throughout the course of treatment, indicates that the therapy is effective and that the patient is adhering to the prescribed regimen. This assessment is crucial for reducing the risk of fractures in osteoporotic patients and preventing the long-term complications associated with them.
Biomarkers of Bone Turnover in Pathological Context
Pathological bone turnover can be observed in various conditions, ranging from bone metastasis to inheritable disorders. In cases of pathological bone turnover, bone resorption may occur in newly formed bone, leading to bone fragility as seen in Paget’s disease 43 or may involve different enzymes such as matrix metalloproteases (MMPs) in the context of bone metastasis, inflammatory disease, or autoimmune disorders.
Paget’s disease is characterized by an increased number, size, and activity of osteoclasts, the cells responsible for bone resorption. This increased rate of bone resorption results in bone deformities, fractures, and pain. 71 In Paget’s disease, both newly formed and old bone tissues are resorbed due to the anarchical nature of resorption. Consequently, the digestion of Type I collagen by cathepsin K releases both isomerized β-CTX and non-isomerized α-CTX. 72 Since α-CTX is specific to the resorption of newly formed bones, it has been investigated for assessing bone turnover in patients with Paget’s disease 73 and for monitoring the response to different treatments for the disease.72,74 Currently, a manual ELISA and an automated Electrochemiluminescent immunoassay (ECLIA) developed by ImmunoDiagnostic Systems Holdings PLC are commercially available for measuring α-CTX in urine.
1α1 homotrimer collagen is a poorly characterized marker associated with cancer, osteoarthritis, and the presence of pleural and ascites fluids. 41 It is described as a degradation product of bone Type I collagen digestion by MMPs, which play a role in pathological bone resorption. 1α1 homotrimer collagen may be produced in the vicinity of cancerous cells due to the increased expression of MMPs by these cells. 75 MMPs are released by cancerous cells to promote evasion by digesting Type I collagen in the extracellular matrix. Inflamed tissues, such as those affected by osteoarthritis, also exhibit overexpression of MMPs, leading to the formation of homotrimers of α1 chains by osteoblasts. These homotrimers are subsequently digested by MMPs, resulting in elevated levels of 1α1 homotrimer collagen. 76
Trivalently crosslinked C-telopeptide of the α1-chain of Type I collagen (ICTP) is a crosslinked peptide that is very similar to CTX as they both originate from the same region of Type I collagen. Indeed, the sequence of CTX is entirely encompassed within the ICTP sequence as they are released through 2 different enzymatic pathways involving different enzymes. 77 ICTP is produced during pathological bone resorption by MMPs, whereas CTX is released during physiological resorption by cathepsin K. MMPs are known to be overexpressed in bone resorption occurring in bone metastases.78 -80 ICTP was first characterized by Eriksen et al in 2004, and its molecular structure is shown in Figure 3. 81

Molecular structure of ICTP in which the lysine residues in gray form a pyridinoline crosslink. U stands for hydroxyproline.
To further investigate ICTP, bone samples from human femoral heads were subjected to a series of preparation steps. The bones were demineralized, powdered, and defatted before being resuspended in phosphate-buffered saline (PBS). The proteins present in the samples were denatured by heat treatment and then digested using the enzyme trypsin. The resulting components were separated using multiple rounds of preparative LC and polyacrylamide gel electrophoresis (PAGE). 81 ICTP was subsequently characterized using several methods, including matrix-assisted laser desorption/ionization time-of-flight (MALDI-TOF) mass spectrometry, N-terminal sequencing, and amino acid analysis, which were considered gold standard methods for characterizing proteins and peptides at that time.82 -85
ICTP was primarily investigated as a marker for bone metastases. It was found to be associated with bone metastases from various cancers, including prostate cancer, 86 breast cancer, 87 lung cancer 88 , and multiple myeloma.89,90
Current Quantification Methods and Faced Challenges
Currently, various quantification methods are available in clinical laboratories for the measurement of proteins and peptides. In most cases, protein quantification relies on automated immunoassays due to their high sensitivity, throughput, and ease of use, as they do not require extensive development or specific qualification. However, immunoassays are known to suffer from cross-reactivity, leading to reduced specificity. This issue is particularly problematic in protein quantification for several reasons.
First, many proteins share significant sequence homology, particularly those derived from the same precursor protein, family, or gene, increasing the likelihood of antibody cross-reactivity. Second, proteins undergo degradation in the circulation and after sample collection, leading to the formation of degradation products that may cross-react with the original protein. This issue is especially pronounced in patients with chronic kidney disease, who have impaired renal clearance of peptides. Third, many proteins undergo post-translational modifications (PTMs), such as phosphorylation, which can alter their biological activity. Immunoassays may not distinguish between active and inactive forms, potentially complicating clinical interpretation.
To address these limitations, liquid chromatography coupled to tandem mass spectrometry (LC-MS/MS) has been developed for protein and peptide quantification in clinical laboratories. LC-MS/MS offers significantly higher specificity than immunoassays and achieves comparable sensitivity, particularly for peptides. However, these methods require extensive sample preparation, which is challenging to automate, and specialized training for operating the mass spectrometer and processing results. Additionally, developing and validating in-house LC-MS/MS assays requires highly skilled scientists, making widespread implementation in clinical settings more complex.
As mentioned earlier, the first characterized marker derived from Type I collagen was hydroxyproline. The initial assay for this marker, developed by Rogers et al in 1954, utilized liquid chromatography (LC) and spectrophotometry for the detection of gelatin in body fluids. 91 Over the subsequent 25 years, additional spectrophotometry-based and nuclear magnetic resonance (NMR)-based methods for hydroxyproline quantification were developed.92 -95
The second method for quantifying a Type I collagen-derived marker was developed by Fujimoto et al in 1978, targeting pyridinoline in tissues. Pyridinoline was extracted from various tissues, including rat tendons and bones, using a saline solution at 4°C. The mixture components were then isolated through successive rounds of preparative reversed-phase LC, and pyridinoline was quantified using a fluorescence detector. Over the years, pyridinoline quantification was extended to various matrices, including urine,96 -98 ligaments, 99 bone powder100,101 and other tissues, 102 utilizing high-performance liquid chromatography (HPLC) coupled with fluorescence detection. Competitive enzyme-linked immunosorbent assays (ELISAs) for pyridinoline determination were also developed, with Robins et al introducing assays for urinary deoxypyridinoline in 1984 103 and pyridinoline in urine in 1986. 104 Additionally, Meddah et al in 1999 developed a method combining HPLC, gel filtration, and NMR analysis to quantify pyridinoline in bone powder. 105 However, no immunoassay or mass spectrometry method for these markers were developed during this period.
To date, only hydroxyproline and pyridinoline—both fully characterized small molecules—have been quantified using NMR-based methods that align with the criteria for gold-standard small molecule analysis. 106 However, their limited specificity has significantly reduced their clinical utility.
Current markers, predominantly trivalently crosslinked peptides, are measured exclusively using immunoassays. However, the development of these assays has been hindered by challenges inherent to the nature of these markers. Two primary issues remain unresolved. First, these markers are enzymatically cleaved from Type I collagen by proteases with unpredictable digestion patterns, such as cathepsin K. This results in multiple coexisting peptide species in the bloodstream, necessitating antibodies that can specifically target a region of the trimeric peptide to recognize all species. Second, these markers are trimeric in nature, comprising bonded fragments of the 3 Type I collagen chains. Upon release into circulation, the trimers can disaggregate into monomers depending on the stability of the bonds linking them. Consequently, antibodies must be designed to target the trimeric form exclusively to avoid cross-reactivity with monomers. 107 In this context, sandwich immunoassays offer a potential advantage by employing 2 antibodies: a capture antibody that binds a stable epitope on the trimeric peptide and a detection antibody that targets another epitope, ensuring high specificity. This dual-recognition approach could enhance assay sensitivity, minimize interference from monomers or partially degraded fragments, and improve reproducibility across different sample conditions. However, the feasibility of developing such assays depends on identifying epitopes that remain structurally accessible and unique to the intact trimeric form.
Despite these challenges, numerous immunoassays have been developed for Type I collagen markers. For bone formation markers, PICP was initially quantified using a radioimmunoassay (RIA) with rabbit antibodies targeting the propeptides produced by human fibroblast cell culture.108 -110 More specific antibodies were later generated by isolating PICP from fibroblast culture medium and digesting it with bacterial collagenase to obtain the antigen. 111 Over time, additional immunoassays for PICP quantification in biological fluids have been developed, but no reference method has been established.112 -115
For PINP, its non-covalent bonding can lead to disaggregation in the bloodstream, producing “PINP monomers”.38,107,116 Numerous commercially available assays exist for PINP quantification, measuring either intact PINP or total PINP. Intact PINP refers to the full-length, unaltered form, whereas total PINP includes intact PINP and associated fragments. While studies indicate that intact and total PINP assays yield comparable results in most cases, discrepancies can arise in conditions such as chronic kidney disease. These differences are likely due to the kidneys clearing monomers and Kupffer cells in the liver clearing intact PINP. 56 Intact PINP assays include the RIA by Orion Diagnostica (Espoo, Finland) and an automated chemiluminescent immunoassay (CLIA) for the iSYS system by ImmunoDiagnostic Systems Holdings PLC (Boldon, UK). Total PINP can be measured using the Electrochemiluminescent Immunoassay (ECLIA) for Maglumi by Shenzhen New Industries Biomedical Engineering Co., Ltd. (Shenzhen, China), and an ELISA by Uscn Life Science Inc. (China). Custom immunoassays have also been developed for studying PINP in diseases such as osteoporosis and inherited bone metabolism disorders.43,117 -119
For bone resorption markers, 4 immunoassays are currently commercially available: (1) a manual ELISA (ImmunoDiagnostic Systems Holdings PLC), (2) an automated CLIA for the IDS-iSYS immunoanalyzer (ImmunoDiagnostic Systems Holdings PLC), (3) the Roche Cobas B-Crosslaps CTX ECLIA for the Roche Diagnostics Elecsys 2010 and E170 analyzers (Roche Diagnostic GmbH), and (4) the CTX CLIA for the Maglumi System SNIBE. 120 Additionally, ICTP quantification has been made possible through a RIA developed by the research team of Sassi et al. 121
As the number of commercially available immunoassay kits has increased, the lack of harmonization between different kits has become evident, 122 particularly for CTX and PINP, which are the recommended markers of the IOF and IFCC. Studies have shown that results for the same sample can vary depending on the kit used, leading to confusion during patient follow-ups and reducing clinicians’ confidence in these markers.
Path Toward Standardization
The aging population has heightened concerns about connective tissue disorders, particularly osteoporosis. This underscores the urgent need for reliable biomarkers to assess bone turnover for diagnosis and follow-up in such conditions. Consequently, degradation products of Type I procollagen and collagen in plasma, serum, and urine have been explored as potential markers of bone formation and resorption. From these investigations, several Type I collagen-derived markers, such as CTX and PINP, have been identified, leading to the development of immunoassays. Unfortunately, as said above, these immunoassays tend to lack harmonization.
To address the lack of harmonization in immunoassays, 2 distinct approaches can be pursued: harmonization and standardization. The key distinction between these approaches lies in their requirements. Harmonization does not rely on the availability of pure and homogeneous primary reference materials or primary reference measurement procedures, whereas standardization does. 123
For example, a multicenter study conducted in 2019 by the IFCC-IOF Joint Committee for Bone Metabolism demonstrated the feasibility of harmonizing PINP assays. This was achieved by applying a correction factor or appropriately assigning calibrator values. 124 A similar multicenter study was later conducted by the same committee for CTX assays. However, this study revealed significant within- and between-assay variability in β-CTX measurements, particularly in serum, rendering harmonization impracticable. 120 In such cases, standardization becomes the preferred approach. Unlike harmonization, standardization requires the development and validation of a reference method, which depends on the comprehensive characterization of the markers to produce traceable standards. Unfortunately, the incomplete structural characterization of Type I collagen-derived markers has significantly hindered progress in this area.
Traceability, a cornerstone of metrology, relies on well-defined reference materials that provide an unbroken chain of comparisons to recognized standards, ensuring the accuracy and reproducibility of biomarker quantification.125 -127 To overcome these limitations, it is crucial to deepen our understanding of the structure of Type I collagen-derived markers. Detailed structural insights would enable the development of gold-standard reference methods, facilitating accurate and specific quantification. HRMS has already been instrumental in identifying various Type I collagen peptides, such as ICTP 121 and CTX, 57 paving the way for the development of LC-MS/MS methods. HRMS may also be used to identify post-translational modifications, degradation patterns, and isoforms that contribute to variability in immunoassays. Advances in HRMS-based technics can thus improve marker specificity, facilitating the development of more robust immunoassays or reference LC-MS/MS methods.
Conclusion
In conclusion, advancing the characterization of Type I collagen-derived markers like CTX is critical for their effective application as bone resorption and formation markers. Once their structures are well-characterized, gold-standard methods must be established to standardize immunoassays. This standardization will ensure consistent results across different kits, increasing confidence in these biomarkers and reducing uncertainty in the follow-up and management of patients with bone turnover disorders.
Footnotes
List of abbreviations
α-CTX α-isomerized C-terminal crosslinked telopeptide of Type I collagen
ADAMTS a-disintegrin and metalloproteinase with a-thrombospondin Type 1 motif
β-CTX β-isomerized C-terminal crosslinked telopeptide of Type I collagen
CLIA Chemiluminescent immunoassay
CTX C-terminal crosslinked telopeptide of Type I collagen
ECLIA Electrochemiluminescent immunoassay
ELISA Enzyme-Linked Immunosorbent Assay
HPLC High-performance liquid chromatography
HRMS High-resolution mass spectrometry
ICTP C-telopeptide of the α1-chain of Type I collagen
IFCC International Federation of Clinical Chemistry
IOF International Osteoporosis Foundation
LC Liquid chromatography
MALDI-TOF Matrix-assisted laser desorption/ionization time-of-flight
MMPs Matrix metalloproteinases
NTX N-terminal crosslinked telopeptide of Type I collagen
PAGE Polyacrylamide gel electrophoresis
PBS Phosphate-buffered saline
PICP Carboxy-terminal propeptide of procollagen Type I
PINP Amino-terminal propeptide of procollagen Type I
RIA Radio-immunoassay
Author Contributions
JD wrote the manuscript. All authors contributed to the bibliographic research, read and approved the final manuscript.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
