Abstract
Background
Urinary N 1 ,N12-diacetylspermine (DiAcSpm) is a novel tumour marker that can be used to detect early cancers. In this study, we examined whether spot urine samples could represent the daily excretion of DiAcSpm after creatinine normalization and which factors should be taken into account in determining reference values for this biomarker.
Methods
We collected the following urine samples: (1) samples from seven healthy volunteers collected on each day of two 2-day sessions to examine the circadian variation of DiAcSpm excretion; (2) samples from 3952 male and 1782 female volunteers to estimate the DiAcSpm concentrations in apparently healthy adults and (3) samples from 16 female volunteers collected every morning over a 3-month period to examine the menstruation-related variation in DiAcSpm excretion. The DiAcSpm concentrations were determined by enzyme-linked immunosorbent assay or a colloidal gold aggregation procedure using DiAcSpm-specific antibodies.
Results
(1) The circadian variation of DiAcSpm in the urine was greatly diminished after creatinine normalization. (2) DiAcSpm was higher in females than in males, and the creatinine-normalized medians (95th percentile) of the urinary DiAcSpm concentrations were 149 (305) and 100 (192) nmol/g creatinine for females and males, respectively. (3) The mean concentrations of urinary DiAcSpm were lower after menstruation than before menstruation by approximately 30 nmol/g creatinine.
Conclusion
Spot urine samples obtained at any time of a day may be used to estimate the daily excretion of DiAcSpm in nmol DiAcSpm per gram creatinine. Sex, age and menstrual condition should be considered when determining the reference values for urinary DiAcSpm.
Introduction
N 1 ,N12-diacetylspermine (DiAcSpm) is a minor constituent of the total polyamine species in human urine. We discovered that DiAcSpm is excreted in the urine of healthy persons in small but definite quantities with little individual variation. 1 Subsequent studies revealed that DiAcSpm is significantly and frequently elevated in patients with various cancers, including prostate cancer, colorectal cancer and breast cancer.2–4 Increased excretion of polyamines in the urine of cancer patients has been widely recognized.5–7 However, extensive analysis of the major components of human urinary polyamines, specifically the monoacetylated derivatives of putrescine and spermidine, once led to a negative evaluation of these major urinary polyamine components as useful tumour markers because of the high frequency of false-positives and false-negatives with these monoacetyl polyamines.8,9 In contrast, increased DiAcSpm concentrations are more frequently associated with malignancies than are other conventional tumour markers.4,10
One of the prominent characteristics of urinary DiAcSpm is that it can be used to detect cancers, such as colon and breast cancers, at early clinical stages. For example, using a tentative cut-off value based on measurements from 52 apparently healthy volunteers, urinary DiAcSpm concentrations were found to be above the cut-off in 60% of stage Tis and stage I colon cancer patients, whereas only 10% and 5% of colon cancer patients at these early stages were CEA- and CA19-9-positive, respectively. 3 This observation implies that DiAcSpm may serve as a novel tumour marker, although other monoacetyl polyamines, which constitute a major portion of the total human urinary polyamine species, do not. Established tumour markers usually have limited sensitivity for detecting early stage cancers, and a novel marker that enables the detection of early stage cancers would greatly contribute towards reducing the number of fatal cases of cancer through early detection and timely treatment. DiAcSpm is a promising candidate for such a marker.
Urine samples are easily and noninvasively obtained from individuals and, therefore, are suitable test specimens for cancer screening purposes, as long as readily available spot urine samples can be used to assess the DiAcSpm concentration for each individual. Therefore, it is important to carefully examine whether it is appropriate to estimate the quantity of daily DiAcSpm excretion from spot urine samples by creatinine normalization. For this reason, in the first half of this study, we sought to determine the validity of creatinine normalization.
As a small quantity of DiAcSpm is excreted in the urine of healthy persons as stated above, detailed information on normal DiAcSpm concentrations is indispensable for distinguishing people with abnormally high urinary DiAcSpm concentrations from those with normal concentrations. The second half of this study was devoted to estimating the normal urinary DiAcSpm concentrations in healthy individuals. We examined whether the urinary DiAcSpm concentrations in healthy individuals were significantly different between males and females and whether the concentrations varied throughout the menstrual cycle in females.
Materials and methods
Urine samples were obtained from the volunteers described below with written consent. We received prior approval to use these samples from the ethical committees of the Tokyo Metropolitan Cancer and Infectious Diseases Center, Komagome Hospital, Nagano Prefectural Suzaka Hospital and the Tokyo Metropolitan Institute of Medical Science.
The urine samples that were used for determining the validity of comparing the quantity of DiAcSpm excreted in nmol/g creatinine were obtained from seven healthy volunteers (three males and four females, aged 21–25 years old). Sampling was performed in two sessions that were separated by a period of three months, each consisting of two consecutive days. For each day of the session, urine samples were collected, and their volumes were recorded. A 10-mL aliquot of each urine sample was set aside to serve as a spot urine sample, and the rest was pooled to serve as the 24-hour urine sample for the day. The spot urine samples were used to examine the circadian variation of DiAcSpm and creatinine excretion and the validity of creatinine normalization.
The DiAcSpm concentrations in healthy adults were estimated using the spot urine samples obtained from 3952 male and 1782 female volunteers among the employees of the Tokyo Metropolitan Government at the time of their annual health examination. Pregnant females were excluded from the analysis.
We also collected urine samples from 16 female volunteers aged 18–39 years old among the students of the Nagano Prefectural School of Nursing every morning for three months to examine menstruation-related variation in DiAcSpm excretion.
The urine samples were supplemented with 3 mmol/L NaN3 and stored frozen at −20℃ until use. DiAcSpm was measured using an enzyme-linked immunosorbent assay (ELISA)3,12 or a colloidal gold aggregation procedure on a JCM BM-6010 automatic biochemical analyzer (JEOL, Tokyo, Japan), 11 as specified in the appropriate figure legends. ELISA was performed using DiAcSpm-specific antibodies that were prepared through several steps of affinity-based purification to reduce cross-reactivity with N1-acetylspermidine to a level as low as 0.03%, which has been shown to minimize N1-acetylspermidine interference in the measurement of DiAcSpm by ELISA. 12 The colloidal gold aggregation procedure is based on the specific binding of a bovine serum albumin (BSA)–acetylspermine conjugate, which is a DiAcSpm mimic, and a stable red-purple solution of colloidal gold–antibody complexes. The BSA-acetylspermine conjugate induces a colour change from red-purple to grey by causing the aggregation of colloidal gold particles. When a urinary sample containing DiAcSpm is added to the system, DiAcSpm competitively binds to the colloidal gold–antibody complexes and suppresses the colour change caused by aggregation because it is a monovalent antigen that cannot cross-link multiple gold particles. The concentration of DiAcSpm in a urinary sample can be determined by measuring this colour change. The reagent Auto DiAcSpm® (Alfresa Pharma Co., Osaka, Japan), which is intended for use in automatic clinical analyzers, is developed. The analytical values of DiAcSpm determined by the ELISA and colloidal gold aggregation procedures have been shown to closely correlate. 11 The DiAcSpm that was used was a kind gift from Dr A Shirahata of Josai University (Saitama, Japan).
The creatinine levels were measured enzymatically using the NESCAUTO® VLII CRE reagent (Alfresa Pharma Co., Osaka, Japan) on a JCM BM-6010 automatic biochemical analyzer (JEOL, Tokyo, Japan).
Results
Variation in the amount of DiAcSpm and creatinine in spot urine samples
To determine whether daily DiAcSpm excretion in the urine may be approximated by the amount of creatinine-normalized DiAcSpm in spot urine samples expressed in nmol per gram creatinine, we examined the circadian variation of DiAcSpm and creatinine in voluntarily provided spot urine samples.
Figure 1 shows a typical example of the temporal variation in the amount of DiAcSpm and creatinine excreted in each urine sample collected. Both the net quantity and the concentration (not shown) of these components in the spot urine samples varied considerably and irregularly between samples, but the variation in the amounts of these two components tended to parallel each other. Consequently, dividing the amount of DiAcSpm by the amount of creatinine excreted greatly diminished the variation observed in the spot urine samples, and the data points were distributed more closely around the total amount of DiAcSpm divided by the total amount of creatinine excreted. The values of average, standard deviation and coefficients of variation for the concentrations of DiAcSpm in the spot urine samples, which are shown in Figure 1, were 29.2 and 9.9 nmol and 34.0%, respectively, and those of creatinine were 193 and 60.1 mg and 31.2%, respectively. The corresponding values for the DiAcSpm concentration in the spot urine samples which are shown in Figure 1 were 213 and 128 nmol and 62.0%, respectively (data not shown). As to the DiAcSpm/creatinine values, average, standard deviation and coefficient of variation were 153 and 22.1 nmol/g creatinine and14.5%, respectively.
Variations in DiAcSpm, creatinine and the DiAcSpm vs. creatinine ratio in randomly excreted urine samples.
In Figure 2, the amount of DiAcSpm excreted in each spot urine sample obtained from each person ((a)–(g)) was plotted against the amount of creatinine in the same spot urine sample. In general, the amounts of DiAcSpm and creatinine in the urine samples correlated well, with correlation coefficients ranging from 0.73 to 0.94. The data points for Subject (g) (Figure 2(g)) were scattered, with a correlation coefficient of 0.73, but a close examination of these data points revealed that this was due to large variations in the amount of daily excretion between the two sessions and between days 1 and 2 of session II (Figure 2(g)*). The large daily variation with this particular subject (female, 21 years old) may be related to the menstrual cycle-dependent variation of DiAcSpm excretion which is described below. However, we are not able to trace the menstrual condition of this subject because we were not aware of the menstrual cycle-dependent variation of DiAcSpm at the time when the samples were collected from Subject (g).
Correlation between the amounts of DiAcSpm and creatinine excreted in the spot urine samples of each volunteer.
The data points obtained with the early morning urine samples on each day of the two sessions (open symbols in Figure 2) are, on the whole, scattered similarly with other data points around the regression lines whose slopes represent the mean daily excretion in nmol DiAcSpm per gram creatinine. These findings indicate that any randomly excreted spot urine sample can be used to obtain a reasonable estimate of daily DiAcSpm excretion in nmol DiAcSpm per gram creatinine.
DiAcSpm in urine samples obtained from healthy adults
Urine samples from apparently healthy adults, which were obtained from volunteers among the employees of the Tokyo Metropolitan Government at their annual health examination, were analysed for creatinine-normalized DiAcSpm. The distribution of their DiAcSpm values is shown in a histogram (Figure 3). We noted that the urinary DiAcSpm concentration differed between males and females; the average, median and 95th percentile values for the females were approximately 1.5-fold higher than for the males. The median values were 100 and 149 nmol/g creatinine for the males and females, respectively, and the 95th percentiles were 192 and 305 nmol/g creatinine for the males and females, respectively.
Urinary DiAcSpm concentrations in healthy males and females.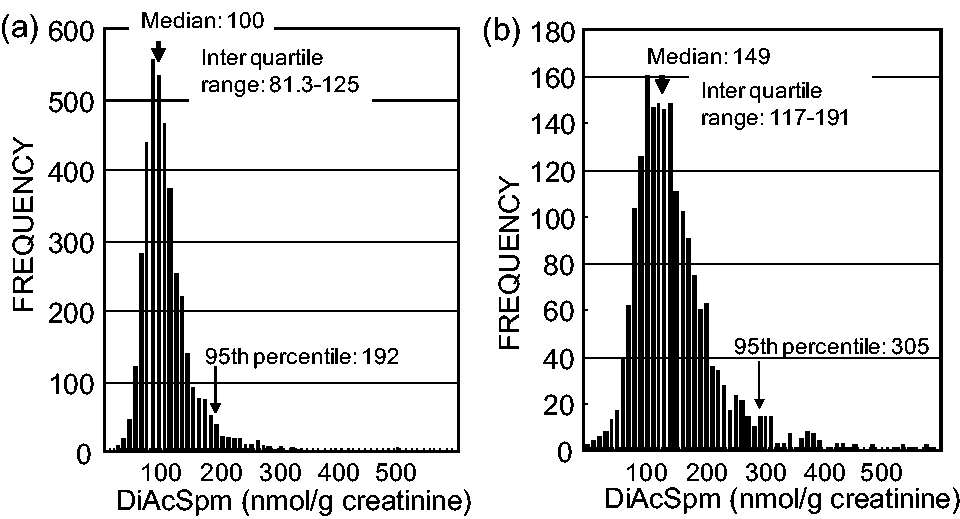
As judged by the shape of the histogram, the males constituted a homogeneous population with respect to their urinary DiAcSpm concentrations (Figure 3(a)) because the frequency of subjects rapidly declined on both sides of a sharp peak, giving a single modal value. In contrast, the females were suspected to be heterogeneous in this respect, consisting of more than one group. Each group of females seemed to distribute around different modal values because their DiAcSpm concentrations were distributed over a broader range compared with those of the males (Figure 3(b)).
Comparison of urinary DiAcSpm concentrations in females younger than 50 years old and females who were 50 years of age and older
To determine whether the broader distribution of the DiAcSpm concentrations in the female urine samples was due to age-dependent variation in DiAcSpm excretion, the female volunteers were divided into the following age groups: 20–29, 30–39, 40–49 and 50 and older. The distribution of DiAcSpm within each age group is presented as a histogram in Figure 4. The median DiAcSpm values for 20–29, 30–39, 40–49 and 50 and older age groups were 154, 156, 148 and 128 nmol/g creatinine, respectively. The median values were significantly different between 50 and older age group and either of 20–29, 30–29 and 40–49 age groups (P = 0.012, 0.0007 and 0.018, respectively; 2 × 2 χ2-test), but not significantly different between any other age groups. The DiAcSpm concentrations of male volunteers were not significantly different between age groups (data not shown). Not only the median DiAcSpm values were significantly lower in 50 and older age group females compared with those in other age groups, but the shape of the histograms also seems different, though not statistically convincing, between 50 and older and other age groups. The distributions of the DiAcSpm values did not seem much different between the age groups for values lower than 140 nmol/g creatinine. However, above this value, the frequency of subjects rapidly decreased in the 50 and older age group (Figure 4(d)), whereas an additional peak was observed for individuals in their 30 s and 40 s, and a broad shoulder was evident for those in their 20 s (Figure 4(a)–(c)). This is consistent with the idea that mainly post-menopausal females in the 50 and older age group constitute a homogeneous population with respect to their urinary DiAcSpm concentrations, whereas mainly pre-menopausal females in the below 50 age groups may consist of heterogeneous populations with different average DiAcSpm concentrations. If a menstrual cycle consists of high and low DiAcSpm phases, then the resulting histogram would show dual peaks or, at least, a broader peak compared with that expected for a homogeneous population.
Urinary DiAcSpm values in females from different age groups.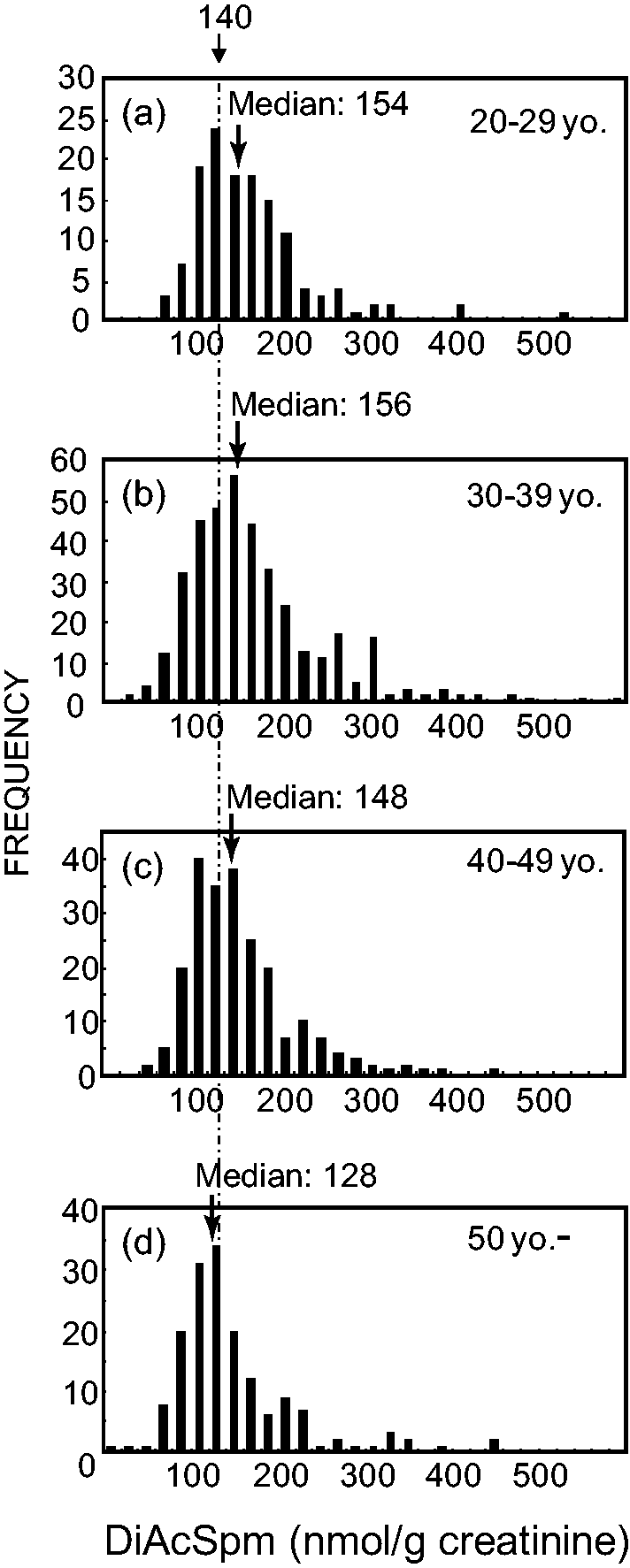
Alteration of the urinary DiAcSpm concentration during the menstrual cycle
To examine whether DiAcSpm excretion varies during the menstrual cycle, we collected morning urine samples from 16 volunteers aged 18–39 years old every day for 90 days (3–4 menstrual cycles). The volunteers also recorded their basal body temperature every morning in parallel with urine collection. A typical example of the menstrual cycle-dependent variations observed with respect to DiAcSpm excretion in urine and basal body temperature is shown in Figure 5. DiAcSpm excretion tended to increase before menstruation and then remain at a relatively low concentration after menstruation. This tendency was consistent among the volunteers, although the daily variation in DiAcSpm excretion occasionally obscured the results. To confirm this tendency, we measured the urinary DiAcSpm concentrations three days before the onset and three days after the end of menstruation for the 16 volunteers. The distribution of DiAcSpm values before and after menstruation is shown in histograms (Figure 6(a) and (b)), and the cumulative frequency distribution is depicted in Figure 6(c). These figures clearly show that lower urinary DiAcSpm values were more frequent among the urine samples collected immediately after the end of menstruation than before its onset. The mean urinary DiAcSpm concentrations (155 and 185 nmol/g creatinine after and before the onset of menstruation, respectively) as well as the median values (149 and 176 nmol/g creatinine after and before the onset of menstruation, respectively) were approximately 30 nmol/g creatinine lower after menstruation than before, whereas the 95th percentile was approximately 50 nmol/g creatinine lower after menstruation than before. The mean values before and after menstruation were significantly different (P = 0.0002; unpaired t-test). The median values were also significantly different (P = 0.002; χ2-test) before and after menstruation.
Variation in urinary DiAcSpm concentrations during the menstrual cycle. DiAcSpm excretion in urine collected before the onset and after the end of each menstruation period.
Discussion
In previous studies, we determined the concentration of DiAcSpm in randomly excreted spot urine samples, and the amount of DiAcSpm excreted in the urine was compared in nmol per gram creatinine as an approximation of the daily DiAcSpm excretion in the urine. Shinpo et al. 13 previously reported that the amounts of putrescine and spermidine (mg per gram creatinine), which are mainly derived from acetylputrescine and acetylspermidines, in acid-hydrolyzed morning urine samples are similar to the corresponding values in acid-hydrolyzed 24-hour urine samples. The fact that the urinary DiAcSpm concentration can serve as a sensitive measure of early cancer detection 3 implies that creatinine normalization is useful and appropriate for estimating the amount of daily DiAcSpm excretion from spot urine samples, but this has not been confirmed for this particular polyamine species.
The present study clearly indicates for the first time that the amount of DiAcSpm excreted in 24-hour urine samples and expressed in nmol/g creatinine may be approximated using the corresponding values obtained from randomly collected spot urine samples. It has been suggested that the values obtained from early morning urine represent a better approximation of the 24-hour average of a urinary component in the amount per gram creatinine than those obtained with randomly excreted spot urine samples collected over one day. In fact, daily albumin excretion was reported to be better approximated with early morning urine. 14 However, it can be seen in Figure 2 that some data points obtained with the early morning urine are positioned close to the regression lines with slopes representing the mean daily excretion, but others deviate considerably from the regression line. In this respect, we did not see any distinction between the analytical data obtained with the early morning urine samples and randomly excreted urine samples. The present results therefore indicate that any randomly excreted urine sample may be used to estimate the daily DiAcSpm excretion in the urine. Once validated, this method will greatly simplify sample collection, and together with the noninvasive nature of urine sampling process, this method will help to extend the use of DiAcSpm in cancer screening.
We previously examined polyamine uptake from the apical surface of renal proximal tubule-derived cultured cells. Using this simple model of reabsorption from the renal brush border, we demonstrated that spermine and monoacetylspermine are efficiently transported across the apical membranes, whereas DiAcSpm is not taken up by the free and monoacetyl polyamine transport system. 15 Because of the absence of a reabsorption route for DiAcSpm, the glomerular clearance of DiAcSpm must be as high as that of creatinine. This result provides a reasonable explanation for the fact that circadian variation in DiAcSpm and creatinine parallel each other. The absence of a reabsorption route for DiAcSpm also implies that DiAcSpm that is produced in early cancer tissues 16 and excreted in the circulation is recovered in the urine without significant loss, which may explain why DiAcSpm serves as a useful tumour marker that is highly sensitive for early stage colorectal and breast cancers. 3
The urinary DiAcSpm concentrations were found to differ between males and females in this study. The average, median and 95th percentile values for females were approximately 1.5-fold higher than those for males. Urine creatinine is primarily derived from creatine and phosphocreatine in the muscle through nonenzymatic processes. On average, females have lower muscle mass than males, leading to lower 24-hour excretion concentrations of creatinine in the urine. The fact that the creatinine-normalized urinary DiAcSpm values were higher for females may be explained by the lower creatinine concentrations in females. In fact, the 24-hour excretion of creatinine in female urine is reported to be approximately 2/3 of that in male urine.17,18 The urine C-peptide–creatinine ratio was also recently reported to be 1.5-fold higher in females than in males. 19
Age-related variation in DiAcSpm excretion among females was also examined, and we found that females under 50 years old (who are primarily pre-menopause) contained a larger proportion of a subpopulation with DiAcSpm concentrations higher than 140 nmol/g creatinine compared with those 50 years of age and older (who are likely post-menopause). This result suggests that the distribution of urinary DiAcSpm values differs between females before and after menopause. If we assume that the DiAcSpm concentration varies during the menstrual cycle, the age-related differences in the distribution of urinary DiAcSpm may occur because the menstrual cycles of the subjects were not synchronized at the time of their health examination, and thus, the urine samples were obtained from every phase of the menstrual cycle. This explanation is supported by the observation that the urinary DiAcSpm concentrations were significantly lower after menstruation than before. The sharp decline in the frequency of subjects who were 50 years of age and older with DiAcSpm concentrations above a single modal value was similar to males, which is consistent with the idea that hormonal conditions affect the production and/or excretion of DiAcSpm in the urine.
In conclusion, in this study, we demonstrated that variation in the amounts of DiAcSpm and creatinine excreted in spot urine samples on the same day tends to parallel each other. Therefore, creatinine normalization is useful for estimating daily DiAcSpm excretion in nmol DiAcSpm per gram creatinine based on the amount of DiAcSpm and creatinine in arbitrary spot urine samples. Subsequent analysis indicated that creatinine-normalized urinary DiAcSpm concentrations are higher in females than in males, and the 95th percentile of urinary DiAcSpm were 192 and 305 nmol/g creatinine for 3952 adult males and 1782 adult females aged 20–65 years old, respectively. Furthermore, we demonstrated that in females, urinary DiAcSpm concentrations vary during the menstrual cycle, increasing before the onset of menstruation and decreasing after the end of menstruation. These findings underscore the necessity for taking sex, age and menstrual condition into consideration when determining urinary DiAcSpm reference values.
Footnotes
Acknowledgements
We would like to thank Alfresa Pharma Co. for providing the AutoDiAcSpm® reagent at a reduced cost.
Declaration of conflicting interests
There are no conflicts of interest to be declared.
Funding
This work was supported in part by a Grant-in-Aid for Scientific Research (C) from the Japan Society for the Promotion of Science (#21590639).
Ethical approval
This study was approved by the ethical committees of the Tokyo Metropolitan Cancer and Infectious Diseases Center, Komagome Hospital (date of approval: 15 June 2010), Nagano Prefectural Suzaka Hospital (date of approval: 20 July 2011) and Tokyo Metropolitan Institute of Medical Science (date of approval: 5 July 2010).
Guarantor
MK.
Contributorship
KH was involved in study concept and design, acquisition, analysis and interpretation of data and writing of the first draft of the manuscript; KS, MS and KT were involved in study concept and design and critical revision of manuscript for scientific content; NF, FS, ET, SM and KI performed acquisition, analysis and interpretation of data; MK was involved in study concept and design, analysis and interpretation of data and critical revision of manuscript for scientific content. All authors reviewed and edited the manuscript and approved the final version of the manuscript.
