Abstract
Background
Intact parathyroid hormone (iPTH) measurements are used to guide therapy in renal patients, but variability in results can occur depending on the assay used. This study has investigated iPTH assay variation in North West England and paired data with regional audit data to determine clinical relevance of assay variability.
Methods
Thirty-seven haemodialysis patients had blood taken (EDTA plasma, and serum), and samples were processed at 17 laboratories that analyse iPTH for North West dialysis patients. Correction factors were calculated and applied to the iPTH assay results to enable direct comparisons. These correction factors were also applied to Regional Audit data to determine if iPTH assay variability explains the variation in unit performance in achieving PTH targets.
Results
The iPTH results from the 37 patients were significantly different when either analysed by different assays and/or different laboratories (P < 0.001). The Abbott Architect method consistently produced the highest iPTH results. Of the 37 patients, between 49% and 65% would achieve the Kidney Disease: Improving Global Outcomes (KDIGO) iPTH target depending on the assay used. When results were adjusted using correction factors, 21% of the patients would require a change of management according to guidelines. Data from all haemodialysis units submitted for the regional audit were adjusted to the Roche assay and this led to a small change in achievement of KDIGO iPTH targets in individual units when compared to each other.
Conclusions
A combination of iPTH assay variability and diversity in clinical management leads to variation in achieving iPTH targets. Both need to be improved and/or standardized to improve patient care.
Keywords
Introduction
Chronic kidney disease (CKD) patients undergo regular intact parathyroid hormone (iPTH) measurements as part of their normal care. This measurement has been used to guide therapy in patients with CKD-mineral bone disorder (CKD-MBD) which is complex and can be difficult to treat. 1 Recent publications have noted that there is a wide variation in iPTH results depending on the assay used2–4 and that these differences may be influencing the clinical management of patients. Target concentrations of iPTH have been published for nephrologists to work towards; originally these were set targets such as Kidney Disease Outcomes Quality Initiative (KDOQI), 5 where a range of 150–300 pg/mL (15.9–31.8 pmol/L) was suggested. Subsequent guidelines suggest that the use of the upper limit of the reference range (ULN) of iPTH as provided by the local laboratory should be used to determine a target. UK Renal Association 6 have previously advised 2–4× ULN though this has been revised recently and is now consistent with the Kidney Disease: Improving Global Outcomes (KDIGO) 7 recommendation of 2–9× ULN as a target range for dialysis patients. Such recommendations incorrectly assume that all laboratories have local population defined reference ranges.
Some studies have shown that if the iPTH results are adjusted according to the assay method used, then this could lead to a more uniform approach to CKD management.2,3 This ‘correction’ could reduce misclassification and therefore sub-optimal treatment of dialysis patients. Correction factors are not used routinely in clinical practice, and in addition, sample type, plasma or serum can also influence the assay result.3,8 Almond et al. 2 have published recommended assay-specific target ranges which were calculated from a large renal population in Scotland and in this study we aim to compare our data to their recommendations. In practice, clinical audit is used to monitor performance in CKD-MBD management and to compare this with other units. When auditing iPTH performance, sample type and the specific assay used by a given laboratory are rarely taken into consideration, but the evidence base and logic suggests that they should be.
The aims of this study were to investigate:
the iPTH assay variability in the North West of England and determine if variability would affect clinical management; if conversion factors could be established to align the different assay methods used in the North West and whether these would be consistent with other published conversion factors; if the variation in iPTH targets achieved in regional audit data could be attributed to the iPTH assay used and whether, if corrected, this would alter performance of individual renal units.
Materials and methods
Summary of the laboratories, their assays and the associated dialysis units which utilize them for iPTH assay measurements.
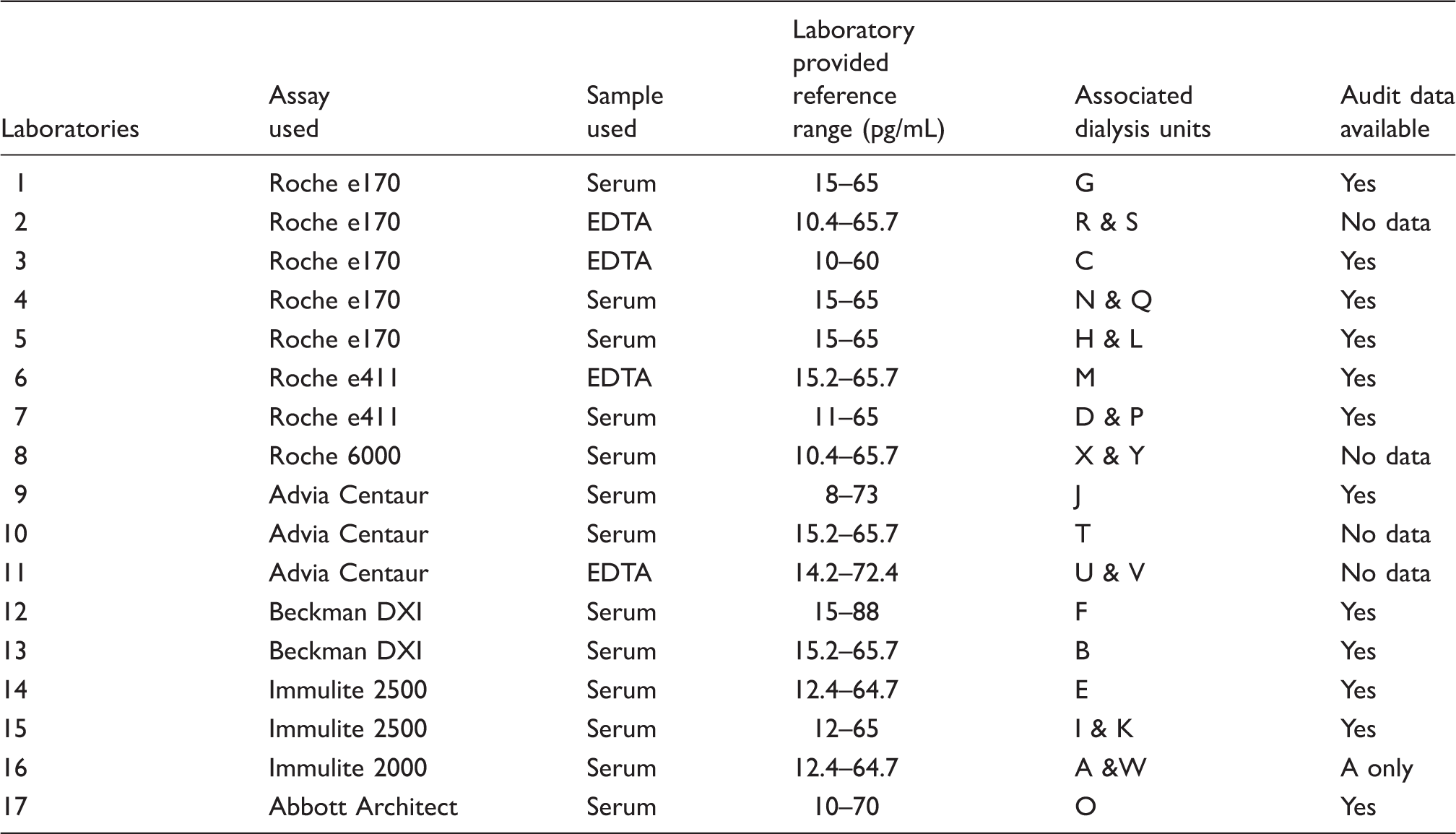
During a single working day, all 37 patients had pre-dialysis blood taken in five different blood collection bottles (Becton Dickinson: Serum Separation Tube (SST) and EDTA; Sarstedt: 2 SST and 1 EDTA). No phenotypic data were collected. Prior to blood collection, each laboratory stated the sample (e.g. EDTA or SST) and bottle type (e.g. BD, Sarstedt, Greiner) used routinely within their laboratory for iPTH measurement. Within 3 h of the blood draw, samples were centrifuged (10 min at 1400 g using a Hettich Rotina 420R centrifuge). All patient samples were divided into anonymised 0.5 mL aliquots. All patients provided enough blood for at least one sample to be sent to all laboratories; where possible two samples per patient were sent. Samples were frozen at −80 ℃ overnight and distributed to all 17 laboratories on dry ice the following day via same day courier. These samples were stored frozen overnight at each centre and analysed the following day (i.e. 48 h after collection). One laboratory analysed the samples on day 6. This was in line with their standard operating procedure. All samples were defrosted on site and processed within 1 h (normal practice). All iPTH results have been reported as pg/mL. Where laboratories reported iPTH as pmol/L this was divided by a conversion factor of 0.106 to convert from pmol/L to pg/mL. 9
Almond et al. 2 recently suggested the use of differing iPTH targets according to the iPTH assay used; these were devised from correction factors developed from their data in Scotland. We used our 37 patients’ iPTH results with a view to validate these targets in a post hoc analysis. The number of patients achieving 2–4× and 2–9× times ULN targets were calculated using their laboratory-provided reference ranges and the ranges suggested by Almond et al. 2
North West regional audit data were collected for November 2009; in this study, we have only used submitted iPTH data for haemodialysis patients in the North West. The audit used the most recent iPTH within the last six months.
All biochemistry laboratories were approached for approval to access UK National External Quality Assessment Service (NEQAS Edinburgh) data. Average bias and variance for iPTH measurement over the last year was obtained for 15 of the 17 laboratories and for the remaining laboratories the national average UK NEQAS data for their respective methods were used for the month in which bloods were drawn.
Statistics
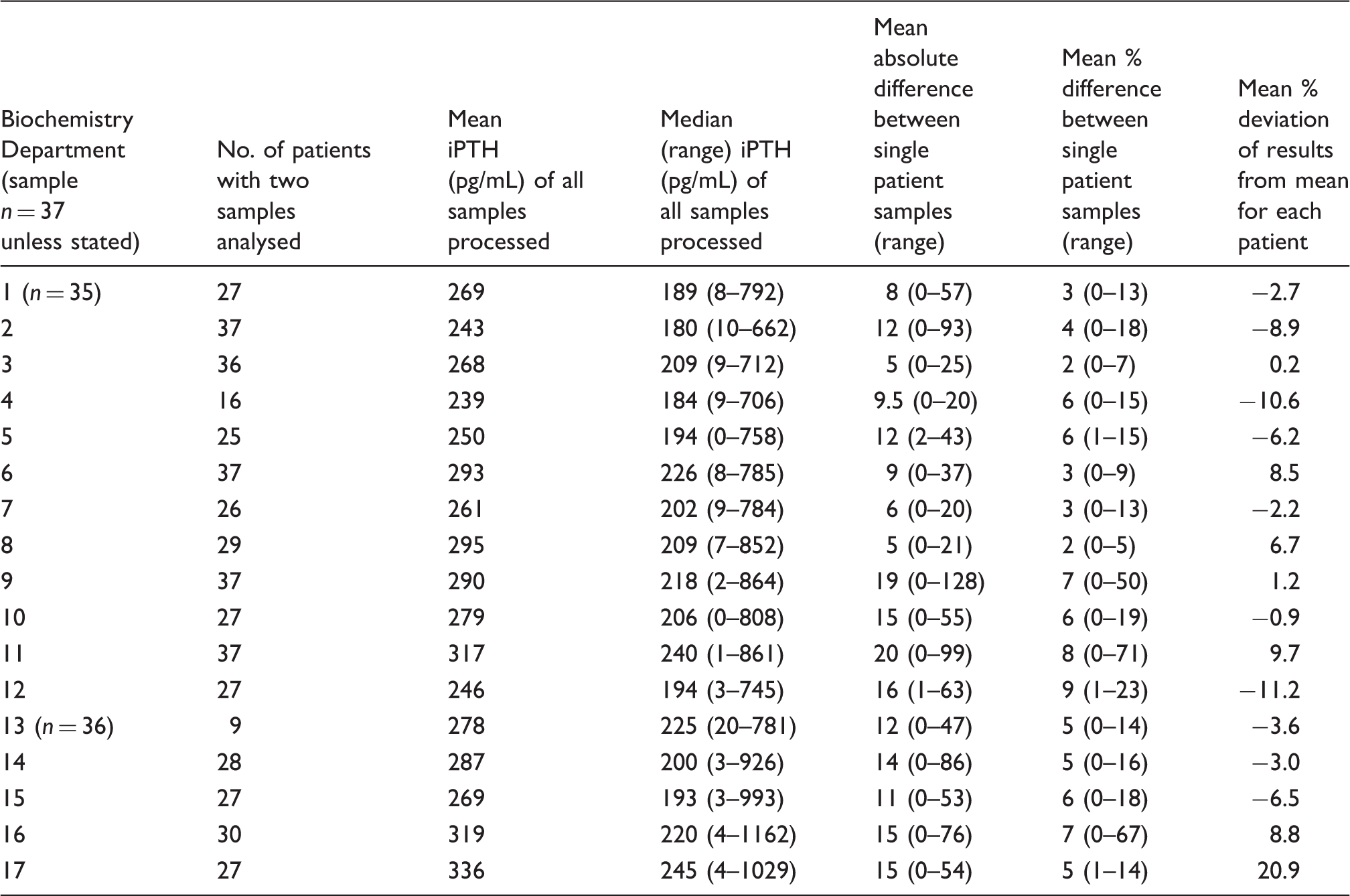
The mean iPTH value produced for each centre was the mean of all averaged samples. Where only one sample was available, then this result was used. The absolute difference shown is the mean absolute difference between the two results, where two samples were available for analysis. Percentage difference is the absolute difference as a percentage of the mean.
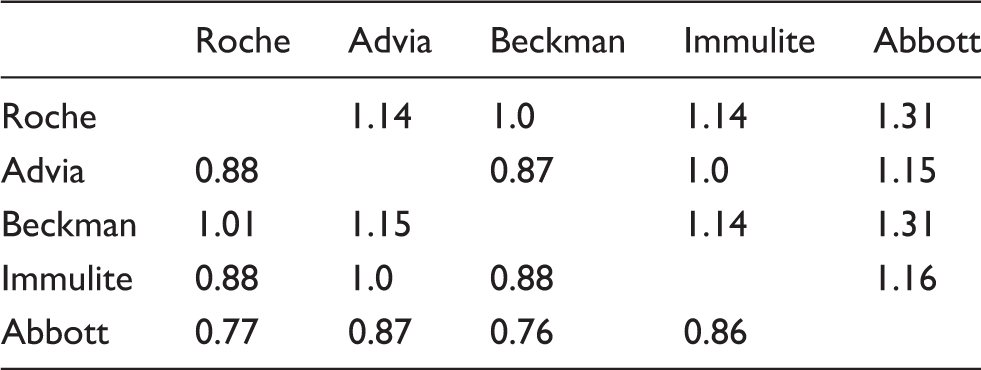
To assess inter-assay variability, the Friedman test was used as it does not assume normal distribution. This was performed for all laboratories and also between laboratories using the same assay and method. To generate correction factors, a graph was plotted of all results between pairs of laboratories. Outliers were excluded and a reference line was generated using SPSS 16.0 software. The gradient of the line was used to determine a correction factor for each laboratory combination. A Spearman rank correlation test was performed to determine the correlation of iPTH results between all laboratories.
Using the 37 patients’ results, the percentage of samples that achieved the KDIGO was calculated for each laboratory. Chi-square was used to determine if there was a significant difference in achieving targets. All samples were then corrected to laboratory 3 Roche e170 assay using the correction factor generated. This was chosen as it is thought to produce similar results to the, now unavailable, Nichols Allegro assay which was used to develop the KDOQI guidelines.5,10 The number of patients who, after correction, changed classification according to targets was calculated and the significance was tested using Chi-square. When corrected an upper limit of iPTH, reference range was set at 65 pg/mL (mean ULN of all Roche assays) to calculate achievement of targets.
For the post hoc analysis using the target ranges suggested by Almond et al., significance was assessed using the Wilcoxon Rank Test.
Percentage bias using the all laboratory trimmed mean (ALTM) for the study patients was calculated using the equation: (result-ALTM)/ALTM × 100. The percentage of regional audit data within UK Renal association guidelines as per audit protocol 2009 (2–4× ULN) was determined for each dialysis unit. The correlation between the median iPTH for each dialysis unit and the bias from both this study and UKNEQAS was analysed using the Spearman rank correlation. The audit data were corrected to the Laboratory 3 Roche e170 assay and a Wilcoxon Signed Rank test was performed to assess difference. Median iPTH from both original audit data and post correction was compared to see if this altered the clinical picture.
Results
Detailed results on 37 patient iPTH measurements per laboratory.
Assay correction factors calculated for the North West Region using Salford sample population.
The percentage of the 37 patients reaching the KDIGO ‘target’ was examined for each laboratory. There was a 16% difference (18 vs. 24 patients) in the number of patients achieving the KDIGO target depending on where their samples were analysed. Further differences were seen in patients below target (nine vs. 16 patients, 19%) though the highest variation (zero vs. nine patients, 24%) was seen in patients who were above target. There was some variability in iPTH reference ranges but many laboratories had similar ranges despite being ‘locally derived’. When we applied the same reference range across the region (ULN 65 pg/mL), the variation in patients achieving target was reduced to 10% with only a 19% variation seen above target.
To review assay variation, correlation and Bland–Altman plots were obtained and are shown in Figure 1. Figure 1(a) and (b) shows the relationship between laboratory 2 (Roche e170) and laboratory 17 (Abbott Architect). It is clear to see that the Abbott Architect assay consistently reads higher than the Roche e170 assay despite being well correlated (R2 = 0.982, P < 0.001). Figure 1(c) and (d) compares two laboratories (Roche e170) one of which uses EDTA, the other serum samples. The assays are in agreement and are well correlated (R2 = 0.971 P < 0.001); however, despite using the same assay, the EDTA plasma samples produce persistently higher iPTH results.
Comparison of assays and sample types using correlation and Bland–Altman plots. (a) and (b) compare the Roche e170 and the Abbott Architect assay. (c) and (d) compare the Roche e170 assay using EDTA plasma and serum.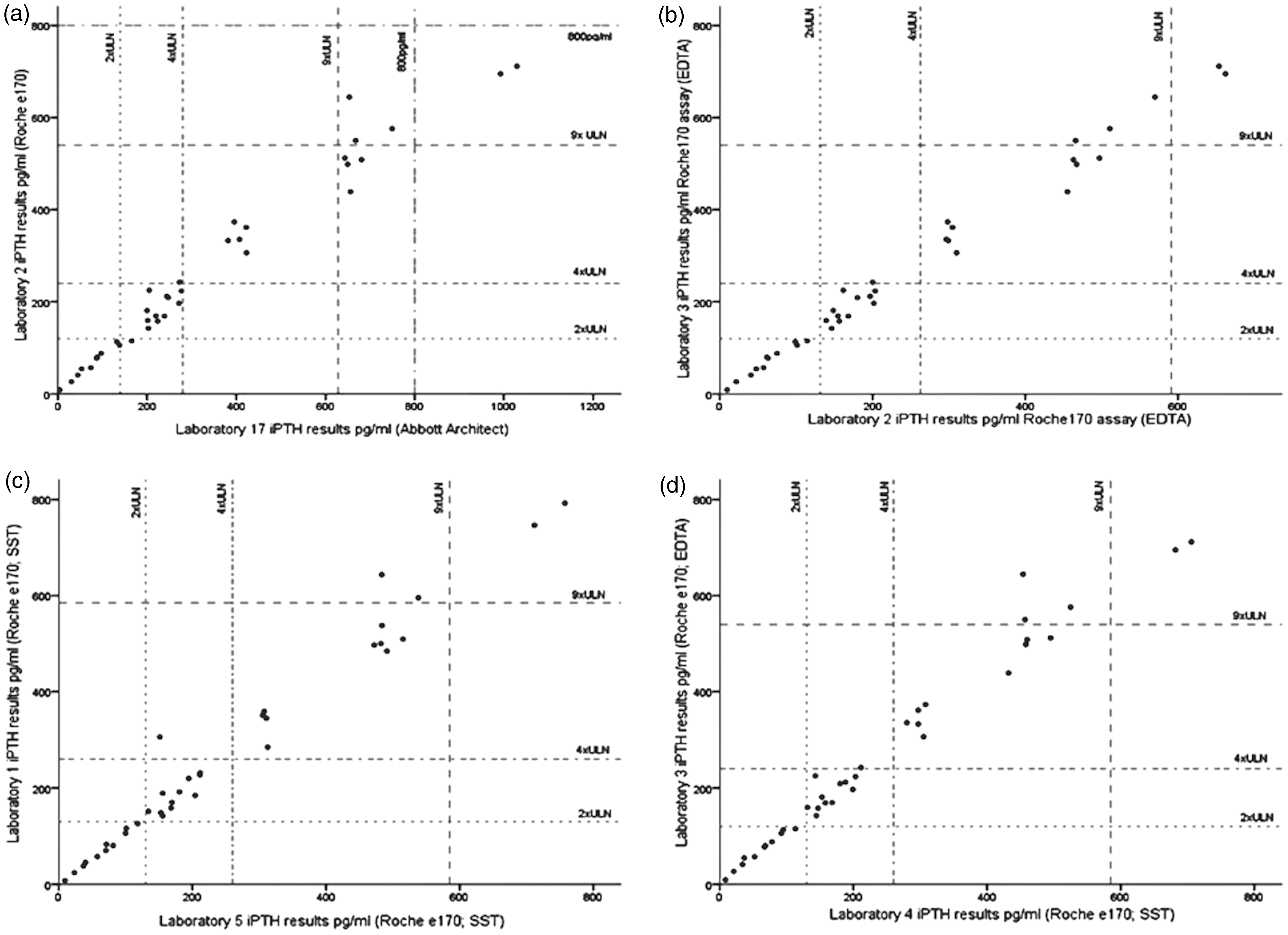
Using the correction factors, all previously generated 37 patients’ results were corrected to Laboratory 3. When corrected, 21% patients would have undergone different clinical management if KDIGO guidelines applied (P < 0.001). This again suggests that clinical decision making is currently influenced according to which laboratory performs the analysis. The variation in achieving KDIGO targets is reduced from 16% to 8% after correction factors are applied. Patients who were classified as above target were reduced from 24% to 13%.
Number of patients within each classification according to 2–4 × ULN and 2–9 × ULN.
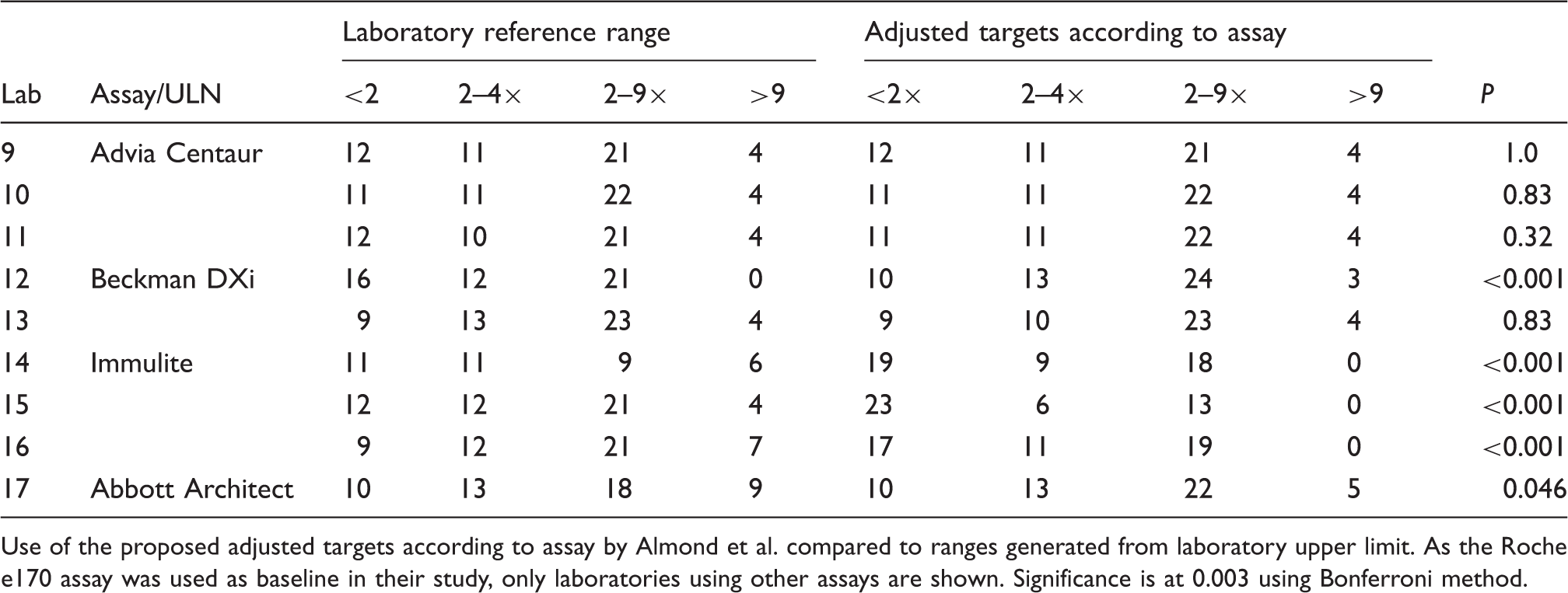
Use of the proposed adjusted targets according to assay by Almond et al. compared to ranges generated from laboratory upper limit. As the Roche e170 assay was used as baseline in their study, only laboratories using other assays are shown. Significance is at 0.003 using Bonferroni method.
The North West regional audit gathered data from 1703 patients in 17 dialysis units which included four hubs and 13 satellite units. A further seven dialysis units did not submit audit data and were therefore excluded from this study. The percentage of haemodialysis patients achieving the UKRA iPTH target (2–4× ULN in 2009) was variable across the region (17–44%, data not shown). We used the Spearman Rank correlation to determine if each dialysis unit’s achievement of iPTH targets was related to the assay used, taking into consideration the bias (percentage variation from the mean) and no association was seen (NEQAS P = 0.08, study P = 0.7). This suggests that, although variable results are seen, they are not solely due to different assays. To further investigate the relevance of iPTH audit data, we used the correction factors generated and adjusted all results to Laboratory 3 (Roche e170 assay). This should remove a large proportion of the assay effect. In Figure 2, we show the median iPTH for each dialysis unit before and where required, after correction. The difference is statistically significant (P = 0.04) though clinically it is less relevant with similar results across the region pre- and post-correction.
Median iPTH values from the North West Regional Audit. Variation of results when adjusted to Roche assay.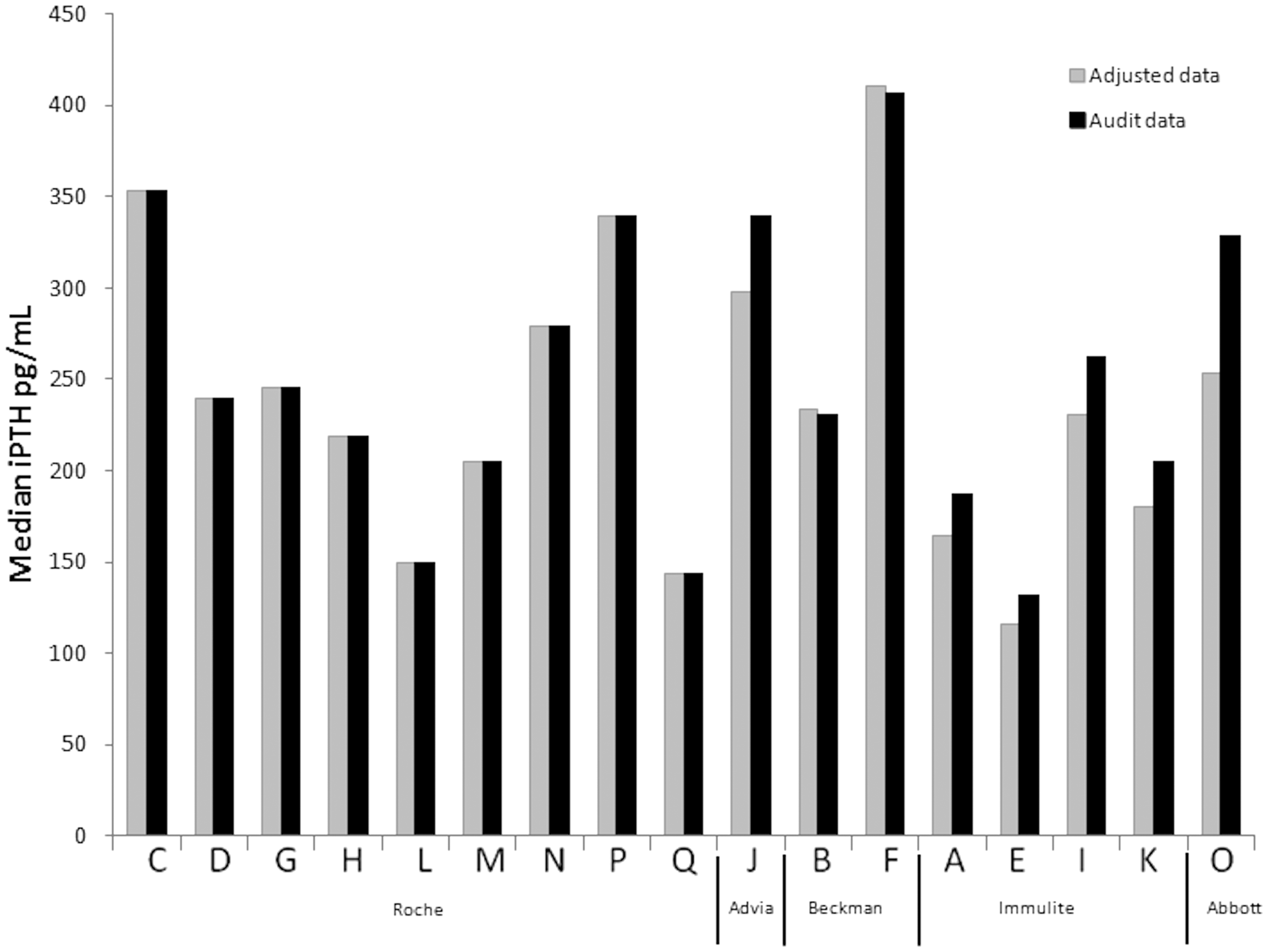
Discussion
Our study has confirmed the significant variability between iPTH assay methods and supports conclusions of previous studies.2,3,10 There were marked differences between laboratory results as shown in Table 2, and these were significant even when the same assay was used (P < 0.001); this led to differing patient classification in relation to international PTH guidelines. The difficulties in generating correction factors for general use were also highlighted when we applied other published factors to our sample population. Our study was designed to enable better comparison of local clinical audit data. To do this, we required samples to be processed as close to clinical practice as possible. This variation also highlighted the importance of using appropriate samples for different methods. 11
Utilization of conversion factors to harmonize the results from different assays has been investigated previously.2,12 Such studies have used the Roche Elecsys assay as the standard as this had been shown to correlate well with the Nichols Allegro assay 10 which was used to develop the KDOQI 5 guidelines. We determined that a conversion factor of 1.3 was needed between the Roche Elecsys and Abbott Architect methods, and this is consistent with other studies. 12 Almond et al. 2 have published data from Scotland emphasizing iPTH measurement variability in haemodialysis patients. Almond's study and our study both show significant variation in iPTH results depending on the assay used. However, the suggested correction/relationship between assays differ. The main difference between studies was seen with the Immulite assay. Almond et al. used EDTA samples for all analyses, whereas all the laboratories using this assay in the North West used SST samples. EDTA samples have previously been shown to produce higher iPTH3,8 results than serum samples processed in the same way and this may account for the difference. This highlights the difficulty in adjusting assays in different laboratories, where samples are analysed under different conditions. Unfortunately, due to limitations, our study did not have the ability to analyse results with both serum and EDTA in all assays. It should be recommended that all clustered laboratories, such as the North West, or indeed all laboratories across the UK should use a single agreed sample type, i.e. EDTA due to sample stability.
The variation of iPTH results seen with different assays has clinically significant implications, given the expense and difficulty in managing secondary hyperparathyroidism in renal patients. When results were corrected to one assay method, 21% of the patients would have required a clinical management change based upon the corrected result. However, the North West region audit data also emphasized the importance of the clinician in the management of CKD-MBD. The 2008 UK Renal Registry data 13 showed a 17% difference in achievement of renal association iPTH targets between dialysis units and given the significant variability in assay performance, it is important to take this into consideration when reviewing audit data. However, this study has shown that assay variability, though important, may not account for all of the variation observed in an audit. Dialysis units that have a low percentage within the iPTH target probably have room for improvement at a clinical level.
This study was designed to be applicable to the regional audit performed in the North West and this is both a strength and a limitation. This allowed us to determine clinician importance in target achievement, but given the previously published variation of iPTH results with differing sample types3,8 and the assay variability, the conversion factors we generated may not be applicable to all populations. For some of the assays, only one laboratory used the method so the applicability of these data to generate conversion factors is also limited. Most patients had two samples analysed at each laboratory, but even this led to a wide variation in results produced in the same patient at the same laboratory using the same method. Gardham et al. 14 recently published data to suggest that iPTH would need to be measured 26 times to provide 95% confidence of an accurate iPTH estimation and this further highlights the problems we have in altering patients’ clinical management based on iPTH results.
There is still some confusion about which PTH ‘peptides’ are measured by each assay and whether peptide fragments such as PTH (7–84) are clinically significant. 15 The PTH (7–84) fragment (and other carboxy terminal peptides) have been shown to have some antagonistic effects to PTH (1–84) and they accumulate as renal function declines.16–19 A third generation assay is available, but this is not widely utilized. This assay has an antibody directed to PTH (1–4) as well as PTH (53–84) and does not cross-react with PTH (7–84). However, the studies that have examined the association between PTH measured by third generation assays and bone biopsy data20–24 have yielded conflicting results. The lack of consistency may be explained by skeletal resistance to PTH (1–84) that develops in patients with renal disease 25 though variation in PTH ‘peptide’ concentrations may also have a role and further assay development is on-going. Despite the variation recognized in iPTH assays, there is increasing evidence regarding the association of low and high iPTH concentrations and increased mortality26,27 and it has been suggested that a narrower target for iPTH is required. These latter studies incorporated large numbers of patients and this may have attenuated the confounding impact of the variability of the iPTH assays onto the final outcome. Further bone biopsy studies will be needed in the future in order to improve the validity of iPTH assays and to identify accurate target ranges that are applicable at different levels of CKD.
Overall, our study highlights the need for development of better markers of bone turnover. An important aspect of current assay development is standardization of all PTH assays using a recognized international material; 28 once available, standardization of assays should reduce inter and intra assay variation and will hopefully lead to more focussed clinical management.
In conclusion, the iPTH assays used in the different laboratories in the North West region of the UK are significantly variable and this variability contributes to a misclassification of patients when considered according to current iPTH guidelines. However, it should not be overlooked that clinician management is a major factor contributing to non-achievement of target iPTH values in their patients. This is achieved in many haemodialysis units with the education, medications and assays currently available and shows that there is room for improvement while the standardization of iPTH assays continues.
Footnotes
Acknowledgments
North West Regional Audit Team and all the biochemistry departments of the hospitals listed below:
Aintree Hospital (Peter Thompson and David Wile); Arrowe Park Hospital (Duncan Neithercut); Central Manchester and Manchester Children’s Hospital Trust (Adrian Holt); Countess of Chester Hospital (Emma Lewis); Leighton Hospital (Marten Davies); Royal Blackburn Hospital (Terry Dyer and Katherine Brownbill); Royal Bolton Hospital (Carolyn Williams); Royal Lancaster Infirmary (Richard Neary); Royal Liverpool University Hospital (Prof WD Fraser); Royal Oldham Hospital (Neil McAuley and Keith Wiener); Royal Preston Hospital (Martin Myers); Salford Royal NHS Foundation Trust (Mark Guy); Tameside General Hospital (Tony Tetlow); University of South Manchester Hospital (Helen Sumner); Victoria Hospital, Blackpool (Steve Butler); Warrington Hospital (Alison Davis); Whiston Hospital, St Helens and Knowsley Teaching Hospitals NHS trust (Mohammed Al-Jubouri).
Declaration of conflicting interests
PAK does consultancy for Shire, Amgen and Sanofi (Genzyme) and has received research grants and honorariums from the same companies. WDF has been paid for developing and delivering educational presentations for Siemens and received honorariums from Roche, Siemens and Abbott. HE has received honorariums from Shire and Amgen. AJH none declared.
Funding
This research was funded by endowment funds. This research received no specific grant from any funding agency in the public, commercial, or not-for-profit sectors.
Ethical approval
This research did not require ethical approval and was discussed with the lead of the Salford and Trafford LREC committee. R&D approval was gained.
Guarantor
PAK.
Contributorship
HE designed the study and researched literature; PAK, AJH and WDF were involved in protocol development. JEH and RLO were involved in study design and sample handling, processing and logistical organization. All authors reviewed and edited the manuscript and have approved the final version.
