Abstract
Background
The mechanism surrounding bone suppression after a meal may involve several mediators, but is yet to be clarified. Bile acids (BA) function as signalling molecules in response to feeding, and may be directly involved in bone suppression acutely after a meal. The aim of this study was to test the hypothesis that BA are involved in the acute bone suppression observed after a meal.
Methods
A prospective study in which samples collected from volunteers fed a 400 Kcal test meal after an overnight fast were analysed for parathyroid hormone (PTH), BA, and carboxyterminal of type 1 collagen telopeptide (CTX). The study was carried out in 10 healthy male volunteers. Ethical approval was obtained from the Local Research and Ethics Committee at King's College Hospital.
Results
Total BA, glycine conjugated bile acids (GCBA), PTH and CTX showed a response to meal ingestion. There was a negative correlation between percentage change in PTH and CTX (R2 = −0.82, P = 0.004), and between PTH and GCBA (R2 = −0.39, P = 0.005).
Conclusion
This study demonstrated an association between GCBA and PTH suppression after a meal. The drop in PTH concentration after a meal may be responsible for the suppression of bone resorption as observed by the decrease in CTX concentration.
Introduction
Adequate nutritional intake is vital for bone health. It is established that after a meal bone turnover is acutely suppressed. This change occurs rapidly, and has been observed within minutes to hours after the intake of a mixed meal, calcium, glucose, protein or fat.1–3 This may be a protective mechanism to maximize skeletal strength in times of energy excess, and to maintain calcium homeostasis when nutritional intake is inadequate.1,4 The mechanism surrounding bone suppression after a meal may involve several mediators, as various factors are known to alter in plasma after feeding. The acute changes observed in bone turnover after a meal is not calcium dependent, as glucose has the same effect.2,5 Elnenaei et al. 6 have shown that the degree of suppression of bone turnover after feeding is similar with varying calorie intake.
Parathyroid hormone (PTH) may be involved in bone turnover after feeding, and functional PTH receptors have been identified on human osteoclasts. 7 Its secretion has been shown to decrease after glucose ingestion, 4 and in thyroparathyroidectomized rats fed small frequent feeds, the rhythm of bone resorption was unchanged suggesting a role for PTH in bone resorption after feeding. 8
Oral glucose ingestion led to a greater reduction in bone suppression compared to intravenous glucose. 5 This has led to the implication of gut hormones as possible mediators of the acute changes in bone resorption after a meal.2,4 Several gut hormones are released in the postprandial stage in response to food ingestion. Many of them including pancreatic hormones such as insulin have been shown to modulate bone turnover in vitro. 1 Octreotide, a somatostatin analogue that inhibits postprandial gastrointestinal and pancreatic hormones, has been shown to abolish the response in bone turnover to glucose ingestion, but octreotide by itself increased plasma PTH. 2
Calcium is the most important regulator of PTH. In vivo and in vitro studies have demonstrated a set-point for PTH release depending on a very narrow range of ionized calcium. 9 PTH release is regulated via the calcium sensing receptor (CaSR), a G-protein coupled receptor consisting of 1078 amino acids located in the chief cells of the parathyroid gland. 10 The CaSR has been identified in other tissues including the gastrointestinal tract where its role is not clearly defined. 11 Although calcium is the physiological ligand for this receptor, it can be activated by other cations and amino acids.10,12 Independent of plasma calcium, other modulators of PTH secretion include glucose and insulin.1,3
Bile acids (BA) are now recognized as signalling molecules during the feed/fast state, influencing different processes from lipid metabolism to glucose homeostasis and energy expenditure.13–16 They act as a ligand for a G-protein coupled receptor TGR5,14,16 and it may be possible that they are sensed by the CaSR.
We therefore hypothesize that the increase in BA after a meal may activate the CaSR thus suppressing PTH which in turn suppresses bone resorption. In this study, we looked at the relationship between postprandial BA, PTH and carboxyterminal of type 1 collagen telopeptide (CTX).
Subjects and methods
Subjects and sample collection
Ten healthy male volunteers participated in this study. The median (interquartile range (IQR)) age of the participants was 21 (20–21) years, with a body mass index of 22.9 (21.5–23.8) kg/m2. They fasted overnight from 23:00 before ingesting a 400 Kcal test meal made up of 46.6 g of carbohydrate, 28.5 g of fat and 10.4 g of protein at 09:00 the following morning. EDTA plasma and serum blood samples were then taken every 30 min from 0 to 120 min. Samples were stored at −80℃ until analysis.
This study was performed according to the principles of the Declaration of Helsinki. The Local Research and Ethics Committee at King's College Hospital approved the study (reference number: 05/Q0703/4), and written informed consent was obtained from the volunteers.
Methods
PTH (second generation assay) was measured by ADVIA Centaur analyser (Siemens Healthcare Diagnostics, Frimley, UK). The detection limit of the assay was 10 ng/L. The intra-assay coefficient of variation (CV) was 5.2% at 40.4 ng/L, 3.4% at 223.8 ng/L and 3.5% at 859.3 ng/L. Carboxyterminal telopeptide of type 1 collagen (CTX) was measured by ELISA (Immunodiagnosticsystems Ltd, Boldon, Tyne & Wear, UK) on an automated ELISA system (Grifols Triturus, Cambridge, UK). The intra-assay CV was 3% at 0.12 µg/L, 1.7% at 0.44 µg/L, and 1.8% at 1.97 µg/L. BA fractions (primary BA, secondary BA and their glycine and taurine conjugates) in plasma were measured using high-pressure liquid chromatography (Jasco™, Dunmow, Essex, UK) with tandem mass spectrometry (Applied Biosystems, Cheshire, UK) as previously described. 17 The method was linear between 0.1 and 10 µmol/L for all the BA and their conjugates except for taurine BA conjugates which were below the limit of detection. The CV was 1.5–6.8% at the lower limit of quantitation (0.1 μmol/L).
Statistical analysis
Statistical analysis was performed using Microsoft excel software (2010, Microsoft corporation, Redmond, WA, USA) and Analyse-It® version 2.21(Leeds, UK). The Shapiro-Wilk W test was used to assess the distribution of the data. As the data were non-parametric, Kruskal-Wallis analysis of variance (ANOVA) test was used to determine whether differences existed across all the time points for CTX, PTH, total BA and glycine conjugated bile acids (GCBA). Where there was a difference, the Mann-Whitney pair-wise test was used. Spearman's correlation was performed to check for correlation. A P-value of < 0.05 was considered significant. Data are reported as median and IQR.
Results
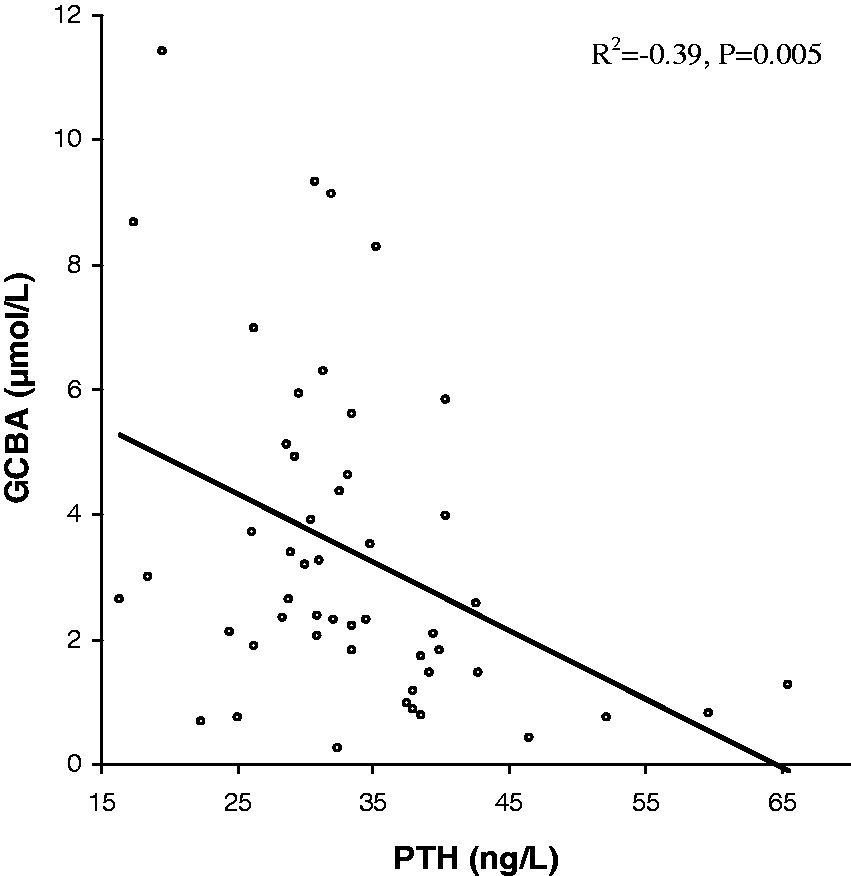
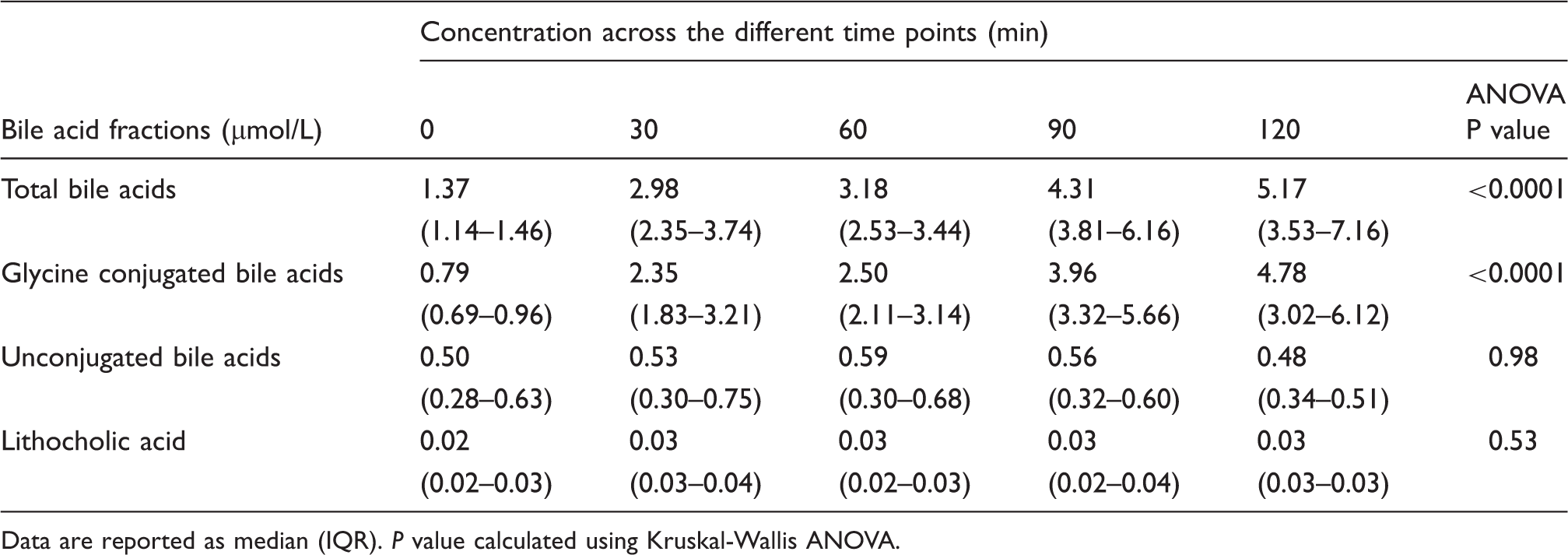
Following ingestion of a 400 Kcal test meal after an overnight fast, the trend in the response to the meal was demonstrated graphically from time 0 to 120 min (Figure 1). There was a decrease in CTX, with a median maximum percentage fall of 46 (40.5–47.8)% at 90 min. There was also a drop in PTH, with a median maximum percentage fall of 30.5 (27.3–34)% noted at 30 min. Calcium was within the reference range, and there was no change in response to meal between 0 and 120 min. An immediate steady rise was seen in total BA and GCBA following the meal. No change was seen in unconjugated BA and lithocholic acid (LCA) during the study period (Table 1). Glycine conjugated BA, which constituted the majority of the total BA (78%), showed an immediate steady increase with time following a meal. The concentration of taurine-conjugated BA was negligible as the method used was not sensitive enough for measuring this fraction of BA. There was a correlation between the postprandial rise in GCBA (and total BA), and the drop in PTH (Figure 2).
Changes in plasma Calcium (a), CTX (b), PTH (c), total BA (d) and total GCBA (e) (median and interquartile ranges) following a 400 kcal test meal in 10 healthy subjects. Spearman's correlation showing a negative linear association between PTH and GCBA response to a 400 Kcal meal intake (R2 = −0.39, P = 0.005). Concentration of the bile acid fractions across the different time points following meal intake. Data are reported as median (IQR). P value calculated using Kruskal-Wallis ANOVA.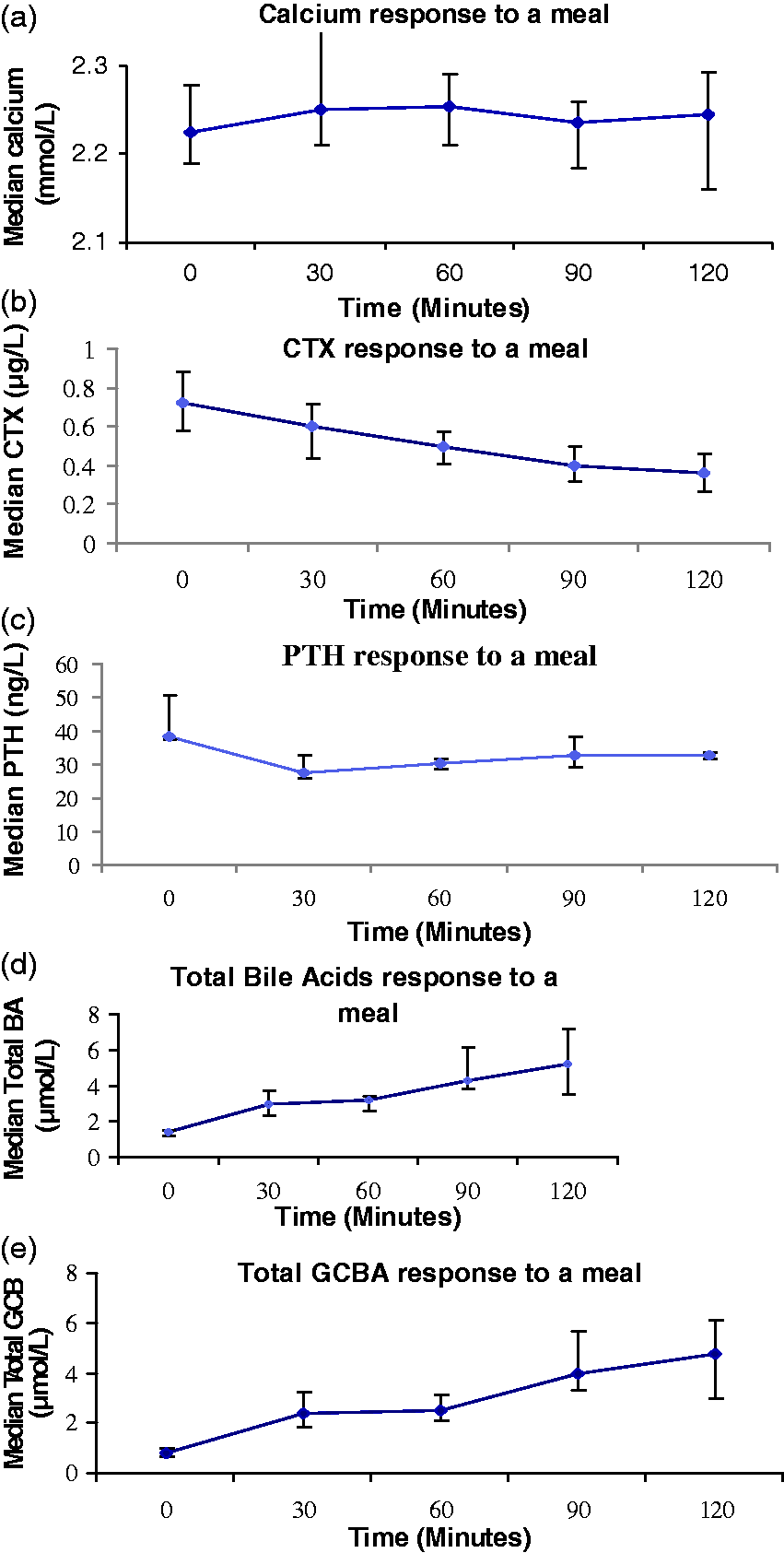
To determine whether there was a relationship between the drop in PTH and CTX observed after a meal, we sought a correlation between the maximum percentage drop in PTH which was observed between 0 to 30 min and the maximum percentage drop in CTX observed between 0 and 90 min. There was a negative correlation between the maximum percentage drop in PTH and CTX (R2 = −0.82, P = 0.004), Figure 3.
Spearman's correlation showing a negative correlation between maximum percentage change in PTH and CTX following a 400 Kcal meal intake (R2 = −0.82, P = 0.004).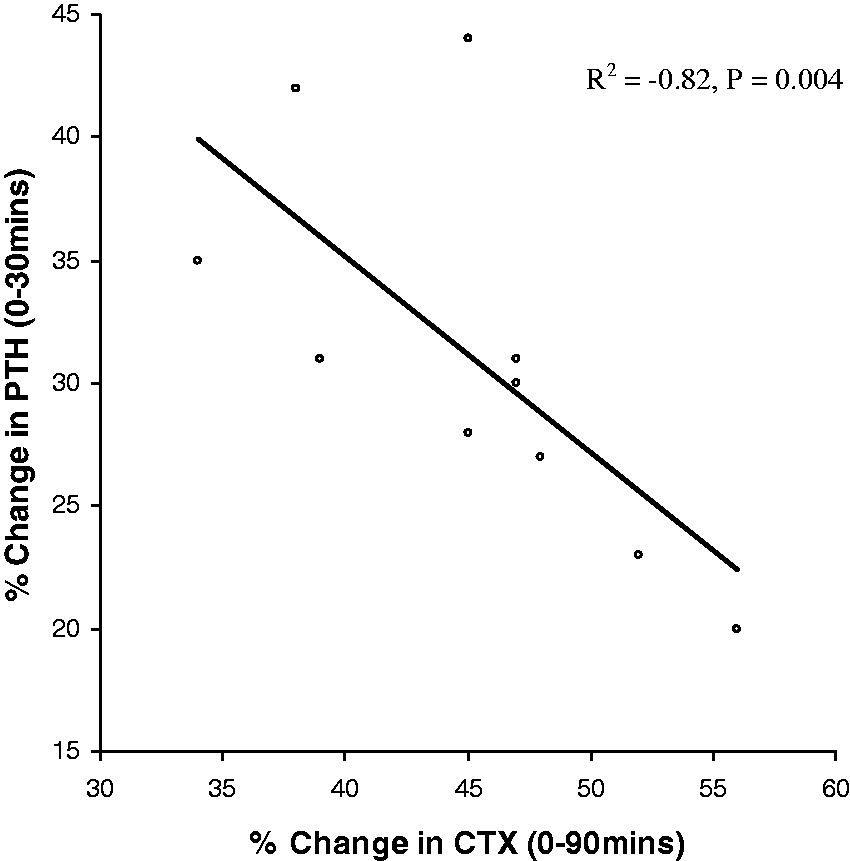
Mann-Whitney U test showing significant differences across the different time points after a meal compared with time 0 min for the different analytes.
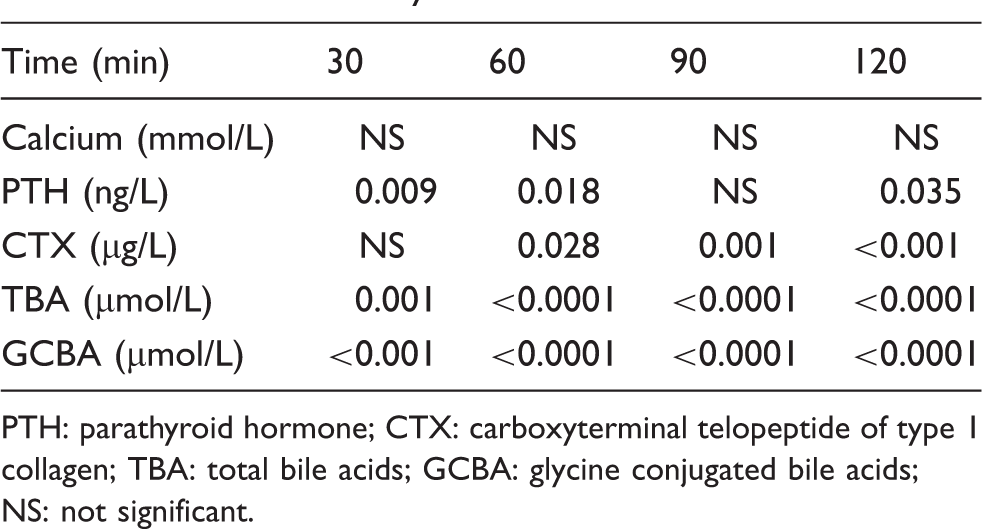
PTH: parathyroid hormone; CTX: carboxyterminal telopeptide of type 1 collagen; TBA: total bile acids; GCBA: glycine conjugated bile acids; NS: not significant.
Discussion
We have demonstrated for the first time, as far as we are aware, an association between post-meal PTH drop and post-meal rise in GCBA. This study also confirms previous observations of a drop in CTX after a meal. 18 A study showed the acute changes in bone resorption after a meal to be completely reversed by octreotide. 2 However, octreotide on its own raised PTH, 2 and therefore made this study difficult to interpret.
It has been shown that the decrease in bone resorption after a meal is similar with varying calorie content between 250 and 3000 kcal, 6 and the suppression in bone resorption is not calcium dependent because glucose also inhibits bone resorption.2,5 Some studies have demonstrated acute changes in PTH concentration in response to glucose, but no changes in ionized calcium.1,2
In the parathyroid glands, the CaSR inhibits PTH release upon activation by an increase in extracellular calcium concentration. Conversely, a decrease in extracellular calcium inactivates the CaSR stimulating PTH release. 10 There is a set-point for PTH release, which is defined as the ionized calcium concentration at which PTH values are midway between the maximum concentration achieved at low calcium concentrations and the minimum concentration obtained at high calcium concentrations. 9 In a normal human parathyroid cell, the set-point for PTH secretion is approximately 1.0 mmol/L of ionized calcium. 19 This set-point allows robust regulation of calcium within a narrow physiological range. 10 Therefore, it means that with small changes in ionized calcium concentration, PTH concentration may remain unchanged. Other agonists apart from calcium are known to activate the CaSR10,12; hence, a similar effect can be expected in terms of PTH suppression.
BA concentrations vary during the day following a pattern dictated by feeding. They act as signalling molecules to peripheral tissues informing them about meal intake and energy availability. 13 These effects are mediated via the TGR5 receptor. TGR5 like the CaSR is a G-protein coupled receptor which is widely expressed in various tissues in the body, mostly the gastrointestinal tract, adipose tissue and the immune system. 16 BA may also act on the CaSR, causing suppression of PTH release. In this study, at 30 min after meal ingestion when the maximum suppression of PTH occurred, we did not expect to get any further response of PTH to BA because it is not dose dependent as described by the set-point of PTH. Therefore, after a meal the immediate rise in GCBA may inhibit PTH acutely, and any further increase in GCBA will not affect the PTH concentration. The secondary BA lithocholic acid (LCA) has been suggested to act on bone. 20 In vitamin D-deficient male rats fed different doses of LCA, serum calcium concentrations where shown to increase in a dose-dependent manner. 20 However, in this study we were unable to show a response of LCA to a meal.
In this preliminary study we have shown an association between BA and post-meal bone suppression. Therefore, it is possible that BA may in part be responsible for PTH suppression, which may be responsible for bone suppression. It must be emphasized that although we have shown an association between BA and post-meal bone suppression, it does not necessarily imply cause and effect. Further work is required in a larger population involving both sexes, and different age groups to prove this. Also, in order to test the hypothesis further, the direct effects of BA on osteoclast activity should be assessed in vitro.
In conclusion, this study is consistent with the hypothesis that bile acids may be one of the acute mediators of nutrient-induced bone suppression.
Footnotes
Acknowledgements
We would like to thank Tracy Dew and Rebecca Musto who supervised the carboxyterminal of type 1 collagen telopeptide assay.
Declaration of conflicting interests
None.
Funding
Department of Clinical Biochemistry, King's College Hospital.
Ethical approval
Local Research and Ethics Committee at King's College Hospital (Ethics reference number: 05/Q0703/4).
Guarantor
ME.
Contributorship
JA conceived the study and researched the literature with ME. JA and RV were involved in patient recruitment and ethical approval for this study. ME and RV did the practical work. RS and RV were involved in data analysis. ME wrote the first draft and all authors reviewed and edited the manuscript.
