Abstract
Background
Roux-en-Y gastric bypass increases circulating bile acid concentrations, known mediators of postprandial suppression of markers of bone resorption. Long-term data, however, indicate that Roux-en-Y gastric bypass confers an increased risk of bone loss on recipients.
Methods
Thirty-six obese individuals, median age 44 (26–64) with median body mass index at baseline of 42.5 (40.4–46) were studied before and 15 months after Roux-en-Y gastric bypass. After an overnight fast, patients received a 400 kcal mixed meal. Blood samples were collected premeal then at 30-min periods for 120 min. Pre and postmeal samples were analysed for total bile acids, parathyroid hormone and C-terminal telopeptide.
Results
Body weight loss post Roux-en-Y gastric bypass was associated with a median 4.9-fold increase in peak postprandial total bile acid concentration, and a median 2.4-fold increase in cumulative food evoked bile acid response. Median fasting parathyroid hormone, postprandial reduction in parathyroid hormone and total parathyroid hormone release over 120 min remained unchanged after surgery. After surgery, median fasting C-terminal telopeptide increased 2.3-fold, peak postprandial concentrations increased 3.8-fold and total release was increased 1.9-fold.
Conclusions
Fasting and postprandial total bile acids and C-terminal telopeptide are increased above reference range after Roux-en-Y gastric bypass. These changes occur in spite of improved vitamin D status with supplementation. These results suggest that post-Roux-en-Y gastric bypass increases in total bile acids do not effectively oppose an ongoing resorptive signal operative along the gut–bone axis. Serial measurement of C-terminal telopeptide may be of value as a risk marker for long-term skeletal pathology in patients post Roux-en-Y gastric bypass.
Introduction
Severe obesity is associated with a number of physical and biochemical disorders including low vitamin D status.1,2 Therefore, weight-loss per se may be anticipated to improve biochemical indicators of bone resorption including vitamin D. However, weight loss through bariatric surgery, including Roux-en-Y gastric bypass (RYGB) has exactly the opposite effect. RYGB is associated with long-term increases in bone resorption, which translate into an increased fracture risk for recipients. 3
Suppression of bone resorption in the postprandial period links micronutrient acquisition to the arrest of systemic liberation of calcium from mineralized bone and a switch to replenishment of calcium stores. Glucagon-like peptide 2 (GLP-2) has been implicated as a mediator of postprandial suppression of bone resorption. 4 A prospective study on normal healthy male volunteers revealed a significant negative correlation between percentage change of postmeal glycine-conjugated bile acids (BAs) and postprandial parathyroid hormone (PTH) concentration. 5 This suggests that total BAs may be important mediators of suppression of bone resorption in the fed state.
Bariatric surgery has been associated with elevated fasting and postprandial concentration of GLP-2 and circulating BAs6–9 but as stated is paradoxically associated with long-term increases in bone loss. 3 During long-term follow-up post-RYGB, this has been described to coincide with time-dependent progressive increases in fasting PTH concentration. 10 A relative de-repression of osteoclastic activity is evident post RYGB as indicated by increases in fasting C-terminal telopeptide (CTX). CTX is a peptide released into the bloodstream following enzymatic cleavage of the C-terminal non-collagenous domains of type I collagen by osteoclast-derived proteases. CTX release thus acts as a marker of bone resorption. 11 Vitamin D deficiency, secondary hyperparathyroidism and a failure to repress bone resorption in the postprandial period may all individually or in combination contribute to the causative sequence of events underpinning the pathogenesis of osteoporosis postbariatric surgery.12,13
In an attempt to delineate events occurring along the enteroendocrine-bone mineralization axis following RYGB, we aimed to describe the relative kinetics of CTX and PTH in the fasting state and postprandial period and examine their temporal correlation with circulating BAs in recipients of RYGB prior to and 15 months after surgery. Given the existing evidence base, we hypothesized that negative correlations between postprandial plasma total BAs concentration and PTH and CTX concentration would not be evident post RYGB. This would implicate dysfunction in normal gut-driven control of calcium homeostasis during the postprandial period as a driver of pathological increases in bone resorption following RYGB.
Subjects and methods
This prospective study was approved by the local ethics committee of the University of Gothenburg (Ref. 583-07) and was conducted according to the principles of the Helsinki declaration. All patients gave written informed consent. The study recruited 36 obese patients who underwent RYGB. Patient demographics were as follows: male/female 11/25, mean age 44 (range 26–64), mean BMI 42.5 kg/m2 (range 40.4–46). All patients were given 1 g calcium and 830 International Units (IU) of vitamin D supplement daily after the surgery. All patients had a 400 kcal test meal made up of 46.6 g of carbohydrate, 28.5 g of fat and 10.4 g of protein before and 15 months after RYGB following an overnight fast. A fasting sample was collected in EDTA tubes before the meal and over 30-min intervals for 180 min after consumption. 25(OH) D and VDBP were measured in fasting samples before and after RYGB. PTH and total BAs were assayed in fasting samples and at 30-min intervals to 180 minutes postmeal initiation. CTX was analysed in fasting samples and at 30, 60 and 120 min after meal initiation.
25-hydroxyvitamin D measurement
The Liaison® (DiaSorin Inc., USA) 25-hydroxyvitamin D assay is a direct competitive chemiluminescence immunoassay (CLIA). The minimal detectable concentration is 9.98 nmol/L. Intra-assay CVs were 7.45%, 4.51% and 5.49% at 14.8, 84.56 and 120.93 nmol/L, respectively. Inter-assay CVs were 13.31%, 4.80% and 6.26% at 14.85, 88.53 and 118.44 µg/L, respectively. Reference interval = 25–104 nmol/L.
VDBP measurement
Quantikine ELISA kit (R&D Systems®, USA) was used for the measurement of VDBP employing a sandwich enzyme immunoassay technique. The minimum detectable concentration of VDBP was 7.25 nmol/L. Intra-assay CVs were 5.7%, 5.8% and 6.2% at 64, 248.3 and 349.2 nmol/L, respectively. Inter-assay precisions were 5.1%, 6.0% and 7.4% at 102.6, 203.7 and 318.2 nmol/L, respectively. Assay was linear between 30.3 and 485 nmol/L.
PTH measurement
A two-site sandwich chemiluminescence immunoassay technique was used for the measurement of PTH on ADVIA Centaur® (SIEMENS Healthcare Diagnostic Ltd, UK). The limit of detection was 0.26 pmol/L. Intra-assay CVs were 5.8%, 7.1% and 1.7% at 1.95, 8.4 and 141.5 pmol/L, respectively. Inter-assay CVs were 9.9%, 2.8% and 6.4% at 3.3, 16.8 and 107 pmol/L, respectively. Reference interval = 1.06–7.4 pmol/L.
CTX measurement
CTX was measured using Serum CrossLaps® ELISA kit (IDS Ltd, UK) which is a two-site sandwich immunoassay. The limit of detection was 20 ng/L. Intra-assay coefficient of variations (CVs) were 3.0, 1.7 and 1.8% at 121, 444 and 1967 ng/L, respectively. Inter-assay CVs were 10.9, 9.7 and 2.5% at 121, 444 and 1967 ng/L, respectively. The assay was linear between 0 and 2.5 µg/L. Age-related reference range in male: 93–630 ng/L, premenopausal female: 25–573 ng/L and postmenopausal female: 104–1000 ng/L.
Total BAs measurement
Plasma total BAs were analysed using a high-performance liquid chromatography method (Jasco™, USA) coupled to tandem mass spectrometry (Applied Biosystems, Cheshire, UK) detection with a modified and extended methodology initially described by Tagliacozzi et al. 14 The method was linear between 0.1 and 10 µmol/L for all BAs with coefficients of variation (CV%) of 1.5–6.8% at the lower limit of quantitation (0.1 µmol/L). The inter-assay CV was 3.6–8.0%.
Statistical analysis
Statistical analysis was performed using GraphPad PRISM Version 6. Data were tested for normality using the Shapiro-Wilk test. Values were not normally distributed and are cited as median value with interquartile range (IQR). Fasting pre- and post-RYGB data were compared using Wilcoxon matched-pairs signed rank test. Two-way ANOVA was performed to assess impact of group and time on analytes across all time points following meal intake for total BAs and PTH. Delta changes (Δ) (difference in concentration between basal and maximal postmeal response) and area under the curve (AUC-180 min) were calculated for total BAs, CTX and PTH. Values of P ≤ 0.05 were considered as statistically significant.
Results
Body weight, vitamin D status and BA concentration
Following RYGB, the BMI decreased from 42.5 (40.4–46) kg/m2 to 27.7 (25.1–32.1) kg/m2 (P < 0.001). Fasting 25(OH)-D increased from 38.9 nmol/L (11.5–104.) to 58.4 nmol/L 23.4 µg/L (20–93.1) after RYGB (P < 0.001). Fasting VDBP concentrations did not change (P = 0.13). 25(OH) D/VDBP increased from 96.2 (69.1–138.1) to 153.6 (11.4–186.4) after RYGB (P < 0.01).
In 27 out of 36 (75%) of samples analysed and compared as paired repeated measures, the postoperative fasting total BAs value was higher than that obtained before surgery. A trend for fasting total BAs to increase post RYGB was observed with median values rising from 2.58 µmol/L (1.12–3.69) to 3.19 µmol/L (1.52–6.06) (P = 0.08 Mann Whitney U-test). Equivalent parametric testing by paired t-test results in a significant increase post-RYGB (P = 0.03). A significant time (P < 0.01) and treatment effect (P < 0.01) was observed for total BA response over the course of the meal challenge. Peak delta total BAs response increased from 1.93 µmol/L (0.87–22.03) to 9.36 µmol/L (1.69–57.03) after RYGB (P < 0.01). The AUC for postprandial BAs was increased from 14.74 µmol/30 min/L (10.17–24.40) to 36 µmol/30 min/L (22.31–62.43) after surgery (P < 0.01) (Figure 1).
Assessment of fasting (a), postprandial changes (b) and AUC (c) for total bile acids before and after RYGB *p < 0.01 post vs. pre.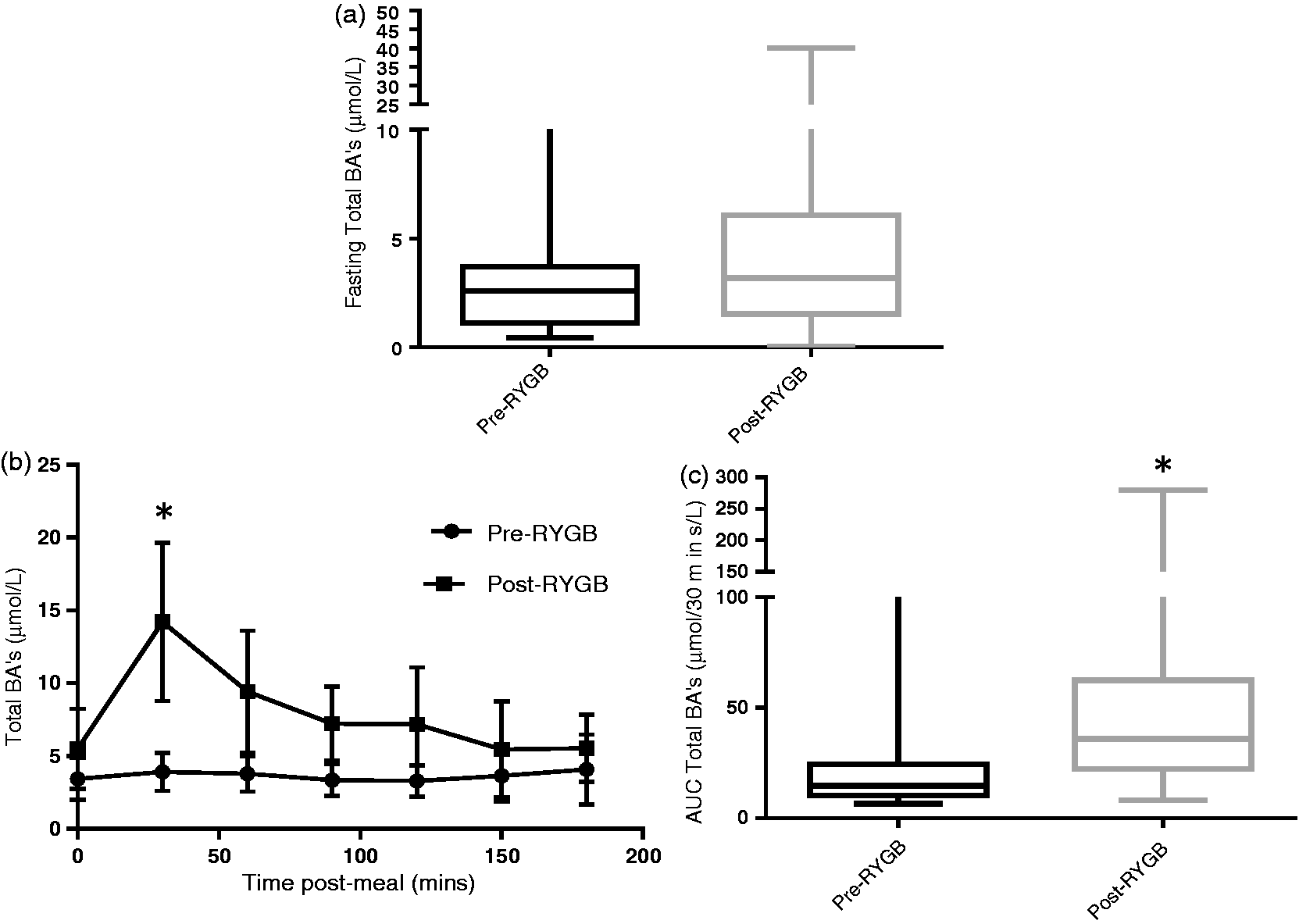
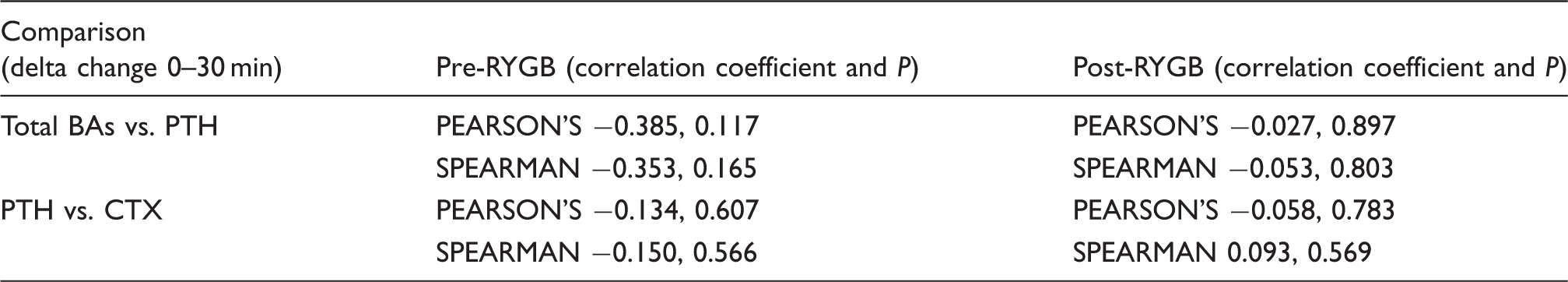
Five out of 36 patients had fasting PTH concentrations more than twice of the upper limit of the reference interval (all PTH > 12.7 pmol/L and four out of five had vitamin D < 49.9 nmol/L). We assumed the possible presence of primary and secondary hyperparathyroidism in these patients and therefore we excluded them from the analysis. Fasting PTH concentration showed a trend towards decline from baseline 5.4 pmol/L (3.7–7.2) to 4.5 pmol/L (2.9–6.0) after surgery (P = 0.06). Two-way ANOVA showed that there was a significant time dependent decrease in in PTH concentration at 30 min postprandially. The AUC for postprandial PTH measured at 60-min intervals for 2 h was unchanged between before 9.4 pmol/h/L (6.8–13.3) and after surgery 10.1 pmol/h/L (7.7–11.9) (P = 0.55) (Figure 2). Table 1 demonstrates that there is a negative correlation between change in total BAs and change in PTH in the postprandial period before surgery, although this does not reach statistical significance. There is no suggestion of such an association in the postprandial period after surgery at which point elevations in total BAs and decrements in PTH appear uncoupled (Table 1). Changes in PTH and CTX are not correlated (Table 1).
(a) Assessment of 30-min postprandial PTH response before and after RYGB (b), AUD over 120 min measured at 60-min intervals *p < 0.050 vs. 30 min. Correlation analyses of delta changes in circulating Total BAs, PTH and CTX 30 min after meal initiation pre- and post-RYGB.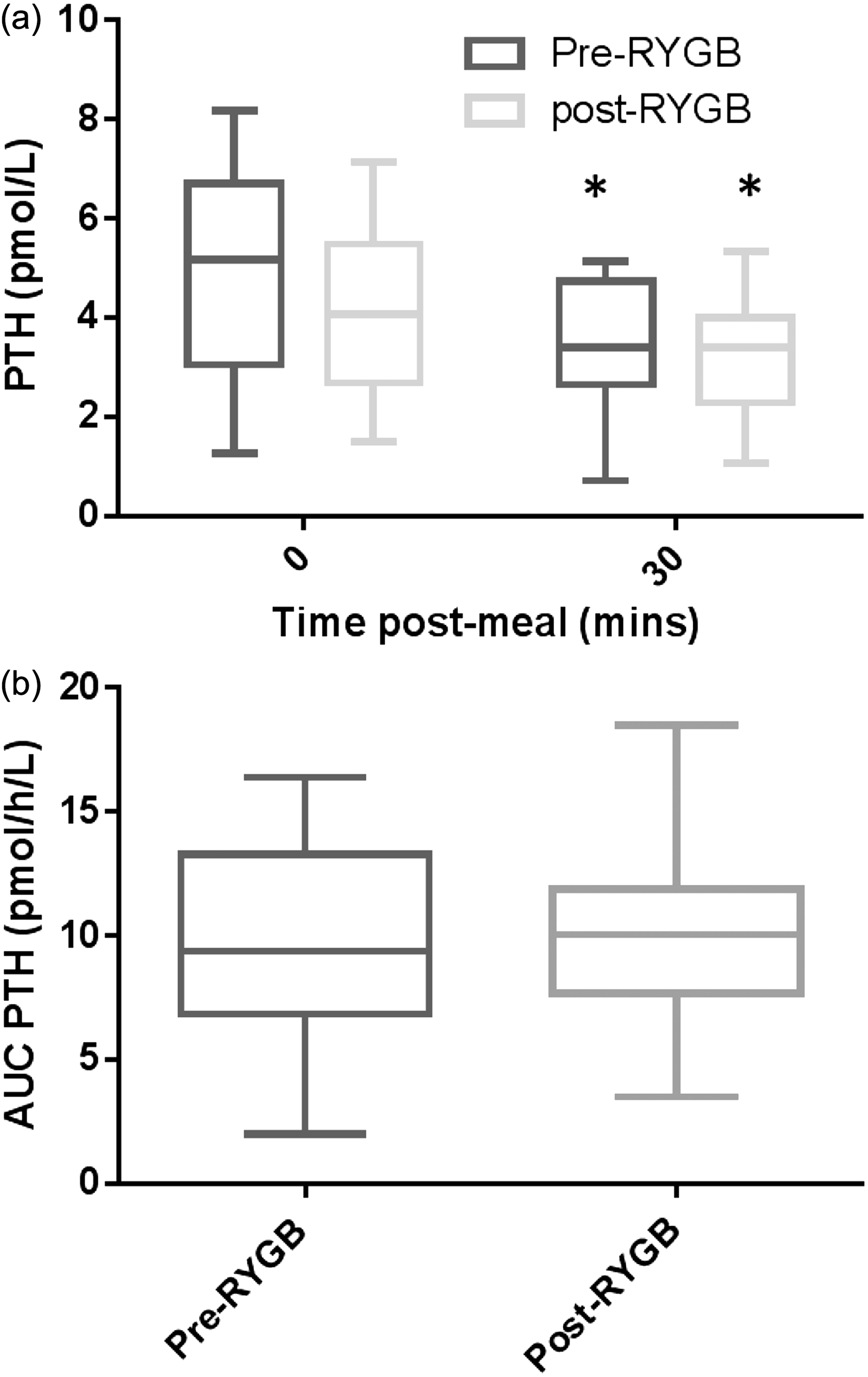
Fasting CTX increased 2–3-fold from 330 ng/L (220–510) to 770 ng/L (60–910) 6 (0.60–0.91) (P < 0.01). A total of 19 patients had complete data for postprandial CTX. A significant time (P < 0.01) and treatment effect (P < 0.01) was observed for CTX response over the course of the meal challenge. Peak delta CTX response increased from 90 ng/L (40–140) to 312 ng/L (100–770) after RYGB (P < 0.01). The AUC for postprandial CTX increased from 1650 ng/30 min/L (1238–2026) to 3280 ng/30 min/L (4308–44,605) after surgery (P < 0.01) (Figure 3), and this occurred in the context of a reduction in CTX between 0 and 30 min in 75% of participants (full data at 0 and 30 min available for n = 32). A weak non-significant positive correlation for CTX and total BAs before surgery (Pearson’s R2 0.313, P = 0.07 and Spearman Rank coefficient 0.291 P = 0.10) was obtained presurgery, but this was not apparent after surgery.
Assessment of fasting (a), peak delta post-prandial changes (b) and AUC (c) for C-terminal telopeptide (CTX) before and after RYGB *p < 0.01 post vs. pre.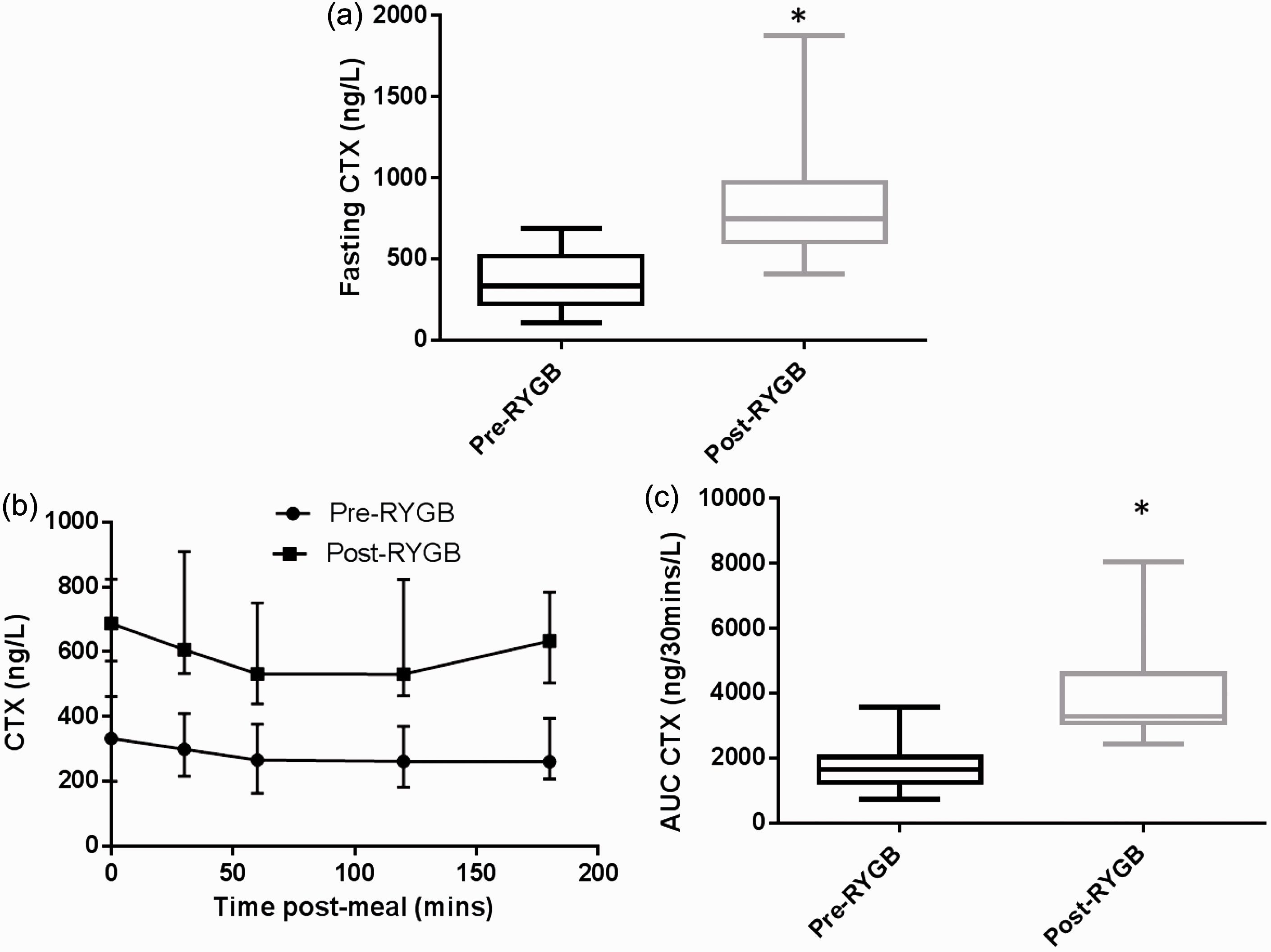
Discussion
BAs have been described to be involved in osteogenesis and suppression of bone resorption. It therefore seemed counter-intuitive that a BA enhancing intervention such as RYGB should be associated with bone loss, although this phenomenon is well described. This prompted us to study in tandem the effect of RYGB on total BAs changes and how these related to CTX kinetics in the fasted and postprandial state. We hypothesized that postprandial CTX changes after RYGB would occur independently of total BAs changes.
Obesity is known to cause a blunted postprandial BA excursion 15 ; hence, it is consistent that the results of the presurgical test here do not mimic the 3-fold magnitude increase observed in healthy controls in our previous paper. We demonstrate an increase in fasting total BAs and postprandial peak of delta total BA response post-RYGB in line with previous studies.
The AUC for postprandial PTH remained unchanged post-RYGB compared with pre-RYGB values. As far as we are aware, we have for the first time demonstrated an increased postprandial CTX response in a group of patients on vitamin D supplement after RYGB as a cardinal sign of bone resorption. No correlation was obtained for CTX and total BAs post-RYGB in our study.
The postprandial kinetics of changes in CTX concentration implies a role for a gut factor, directly or indirectly in enhancement of bone resorptive activity. Gastric inhibitory peptide (GIP) is secreted from endocrine K cells in response to food digestion. It has been suggested that GIP is a bone-forming factor that is reduced post-RYGB as a consequence of duodenal exclusion. 16
Other factors have been implicated to affect bone turnover after RYGB. Adiponectin produced by fat cells is a polypeptide hormone which regulates glucose and fatty acid metabolism. Plasma adiponectin concentration is low in obese patients and increases after RYGB.14,17 It has been proposed that increased plasma adiponectin concentrations stimulate bone resorption following bariatric surgery. 18 Sclerostin a key regulator of bone metabolism is produced by osteocytes and acts as an inhibitor of bone formation. A study on 52 premenopausal women over two years demonstrated that sclerostin concentration increase after bariatric surgery and remains elevated during the entire study period. 19 It is possible that increased basal and postmeal bone resorption can be partly related to increased adiponectin and sclersotin concentrations or reduction in GIP after RYGB.
Calcium malabsorption may persist after RYGB, and can be unrelated to vitamin D status. 15 All our patients were on vitamin D supplements, and we excluded patients with secondary hyperparathyroidism as defined by a serum PTH concentrations two standard deviations above the mean. Vitamin D deficiency and secondary hyperparathyroidism have been suggested as responsible factors for increased CTX, a bone resorption marker, post-RYGB. 2 However, the results from our study suggest that deactivation of parathyroid responses by vitamin D replacement does not seem to prevent ongoing postprandial bone resorptive reaction post-RYGB.
Our study is limited by the lack of evidence of bone mineral density changes in these specific patients, although the changes after RYGB are well described.20,21 Basal and postprandial changes of calcium and magnesium concentrations were not measured as samples were collected in EDTA tube, which chelates these elements. We were also unable to cross-reference values for each analyte in all patients across all time points, and this may have restricted statistical power in correlation analyses. Any future work needs to include postprandial calcium concentration in order to evaluate the correlation with postprandial changes of PTH observed. There are also potential confounders arising from menopausal status and the use of medications that influence mineral metabolism which have not been captured in this study and should be obtained for future studies.
In conclusion, we have shown for the first time that CTX is elevated and remains so in the postprandial period after RYGB in this group of patients on vitamin D supplements. A rise of CTX in the presence of normal PTH and vitamin D concentration possibly due to the presence (adiponectin) or absence (e.g. GIP) of postpostprandial signal modulating osteoclast function has credibility as a working hypothesis. Future work should seek to identify this gut factor and correlate its presence or absence across different bariatric procedures with cognate prevalence rates of osteoporosis.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
ClR is funded by Science Foundation Ireland (ref 12/YI/B2480).
Ethical approval
This study was approved by the local ethics committee of the University of Gothenburg (Ref. 583-07) and was conducted according to the principles of the Helsinki declaration. All patients gave written informed consent.
Guarantor
CL.
Contributorship
MW, LF and TO were involved in conceiving the clinical study design, patient recruitment and ethical approval of the study. JA, RS and CL conceived the bone marker study design. NM did literature review, sample analysis and statistical analysis. JA was involved in literature search and writing the manuscript. GC and TD were involved in analytical work. JA RS, RV NGD and GC were involved in statistical analysis of the data. NM wrote the first draft of the manuscript and CL, JA, RV NGD and RS read and edited the manuscript. All authors approved the final version for submission.
