Abstract
Background:
With a rising incidence of cerebrospinal fluid (CSF) leaks, endoscopic endonasal CSF leak repair is increasingly performed. Current approaches utilize a variety of materials including free mucosal grafts and vascularized flaps, but post-op leaks continue to be reported. Steroid-eluting bioabsorbable stents (SES) are used during functional endoscopic sinus surgery for chronic rhinosinusitis to reduce inflammation and scarring while maintaining patency of sinus ostia.
Objective:
The aim of this study is to assess the feasibility of SES as a graft/flap bolster for endoscopic endonasal CSF leak repair.
Methods:
This is a retrospective review of patients undergoing endoscopic endonasal CSF leak repair with SES placed as part of the bolster technique at a tertiary care center between January 2019 and May 2022. Age, sex, BMI, comorbid idiopathic intracranial hypertension, pathology, location of CSF leak, intraoperative CSF leak flow, reconstruction type, and presence of post-op CSF leak were recorded.
Results:
Twelve patients (mean age 52, median BMI 30.9, 58% female) had SES placement as part of the bolster technique. The most common pathology was meningoencephalocele (75%). Reconstruction was performed with either a free mucosal graft (6), or a flap (6). No post-op CSF leaks occurred at a reconstruction site with a stent, and no known complications were reported. All sinusotomies were patent at the last follow-up visit.
Conclusions:
SES placement as an adjunct to graft and/or flap bolster appears to be safe and feasible during anterior skull base reconstruction and CSF leak repair providing longer term structural support and preserving sinus drainage patency.
Keywords
Introduction
Cerebrospinal fluid (CSF) leaks of the anterior skull base can occur spontaneously or secondary to traumatic or iatrogenic insults. Common symptoms include CSF rhinorrhea and headache, along with a significantly increased risk of central nervous system (CNS) infection. 1 With increasing rates of obesity and idiopathic intracranial hypertension (IIH), the incidence of spontaneous CSF leaks is rising in the United States. 2 In addition, endonasal endoscopic surgery has become a common and safe approach for the resection of common anterior skull base tumors such as pituitary adenomas. 3 Thus, regardless of the inciting event, the number of patients requiring an operative repair of anterior CSF leaks is increasing, necessitating reliable, consistent repair strategies.
Current methods for repair of anterior CSF leaks include the use of biosynthetic xenografts, free autografts, vascularized local and regional flaps and free flaps. Studies have shown that vascularized flaps have the lowest rate of CSF leak recurrence.4 -6 The reconstruction is commonly packed against the bone or dura with no other mechanical support stabilizing it in place against gravity or sudden increases in CSF pressure (i.e. sneezing) as the mucosa heals in the perioperative period. 7 One potential source of decreased repair integrity is the reconstruction coming loose from the defect in the skull base following removal of packing material. Therefore, additional structural support of the graft/flap in the weeks following surgery might further decrease post-operative CSF leaks. Moreover, grafts, flaps, and scar tissue can lead to obstruction of the sinus drainage pathway with subsequent mucocele formation or sinusitis. 8
Steroid eluting bioabsorbable stents (SES) are frequently used during functional endoscopic sinus surgery (FESS) for chronic rhinosinusitis. Steroids in the stent help to reduce mucosal edema and synechiae formation and promote wound healing, while the stent itself provides support to maintain patency of sinus ostia. 9 A recent meta-analysis on the use of SES for chronic rhinosinusitis confirmed that SES reduce post-operative inflammation, recurrent polyposis, and the need for repeat intervention. 10
With their proven efficacy of promoting wound healing and structural support in FESS for chronic rhinosinusitis, SES could also play a role in the repair of anterior CSF leaks, specifically by helping secure the reconstructive tissue in the correct position and supporting efficient healing of the area while preserving the patency of the paranasal sinuses. Here, we assess the safety and feasibility of the use of SES as an adjunct to bolster grafts/flaps in the operative repair of anterior CSF leaks in a retrospective cohort study.
Materials and Methods
Study Design and Subjects
This was a retrospective review of patients at a tertiary care center and was conducted with approval from the Duke University Medical Center institutional review board. All patients underwent an endoscopic endonasal anterior skull base reconstruction with placement of a steroid-eluting stent as part of a bolster technique between January 2019 through May 2022.
Surgical Technique
Endoscopic endonasal approach to the skull base was employed to repair anterior CSF leaks. A maxillary antrostomy, total ethmoidectomy, sphenoid, and frontal sinusotomy were performed. The middle turbinate was resected as indicated for access or per surgeon’s preference (RA, DJ). The tumor, if present, was resected using an endoscopic endonasal approach to the skull base.
The CSF leak and skull base bony defect were identified. When present, the meningoencephalocele was resected and the stump was cauterized using bipolar cautery. The dural defect was identified and measured. The surrounding bone was stripped from the sinus mucosa, the bony edges were smoothed using a drill or cutting instruments.
A xenograft was placed intracranially intradurally in an inlay fashion per surgeon’s preference. Then, a free intranasal mucosal graft, or a nasoseptal flap, or an anteriorly based middle turbinate flap were used as an onlay patch. No tissue sealant was used. A mometasone furoate biodegradable stent implant (PROPEL Contour® Sinus Implant, Intersect ENT, Menlo Park, CA) 11 was then positioned in the paranasal sinus recess up against the graft or flap to help secure its position. The graft/flap were found to be secured in an appropriate position, transmitting brain pulsation with no CSF leak spontaneously and with a Valsalva maneuver. Resorbable (SURGICEL®, Ethicon, Bridgewater, NJ; Gelfoam®, Pharmacia and Upjohn Company, Kalamazoo, MI; NasoPore®, Stryker, Kalamazoo, MI) and non-resorbable packing material (MEROCEL®, Medtronic, Minneapolis, MN) were used to further bolster the reconstruction in place. No biological glue was utilized during surgery.
Post-Operative Protocol
Postoperatively, patients were admitted for either overnight admission or longer based on the pathology and extent of surgery. No lumbar drains were used in any of the patients included in this cohort. CSF leak precautions were in place and included: no strenuous activity, sneezing and coughing with the mouth open, stool softeners as needed, no nose blowing, sinus rinses using saline irrigations at least every 8 hours. Antibiotics for gram positive coverage, typically cephalexin, were prescribed while the non-resorbable packing was in place. Patients with IIH were treated with Diamox 500 mg every 12 hours postoperatively and referred for a weight loss program if they had a BMI>30 kg/m2. Patients were seen 7 to 10 days postoperatively for non-resorbable packing removal and debridement, with long-term follow up at 1 month and 3 months.
Data Collection
Age, sex, BMI, pathology, presence of co-morbid IIH, location of CSF leak, intraoperative CSF leak flow, reconstruction type, location of SES placement, presence of post-operative CSF leak, and surgical site infection were recorded. A high-flow CSF leak was defined as a dural defect communicating with a cistern or ventricle, or a large dural defect >2 cm2.
Results
Twelve consecutive patients were identified as having a steroid-eluting stent placed as part of a bolster technique during endoscopic skull base reconstruction and CSF leak repair. Patient characteristics are shown in Tables 1 and 2. The median age in our cohort was 52 years (37-82 years) with 58% female patients and a median BMI of 30.9 (21.6-46.6).
Summary of Patient Characteristics.
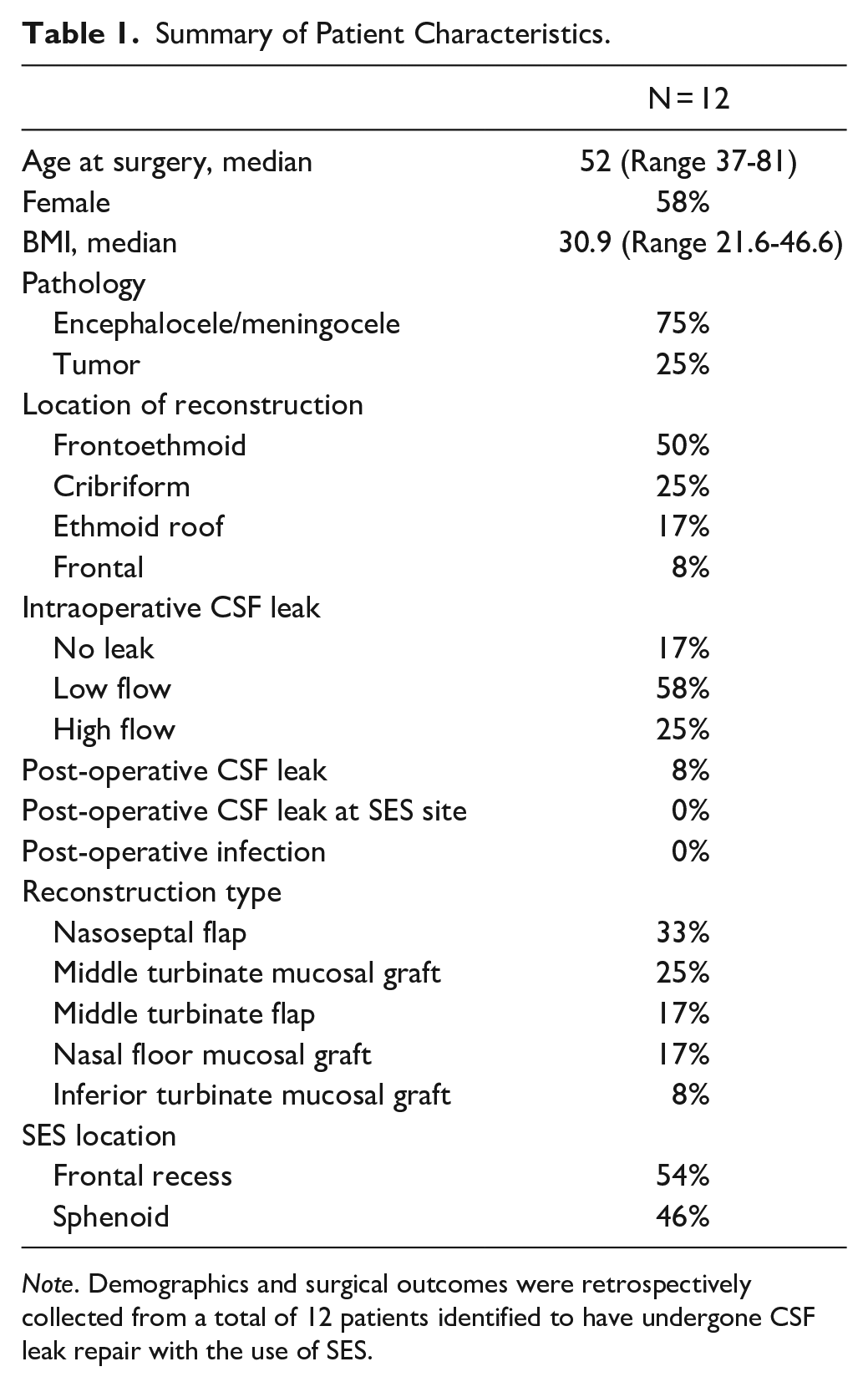
Note. Demographics and surgical outcomes were retrospectively collected from a total of 12 patients identified to have undergone CSF leak repair with the use of SES.
Operative Characteristics by Patient.
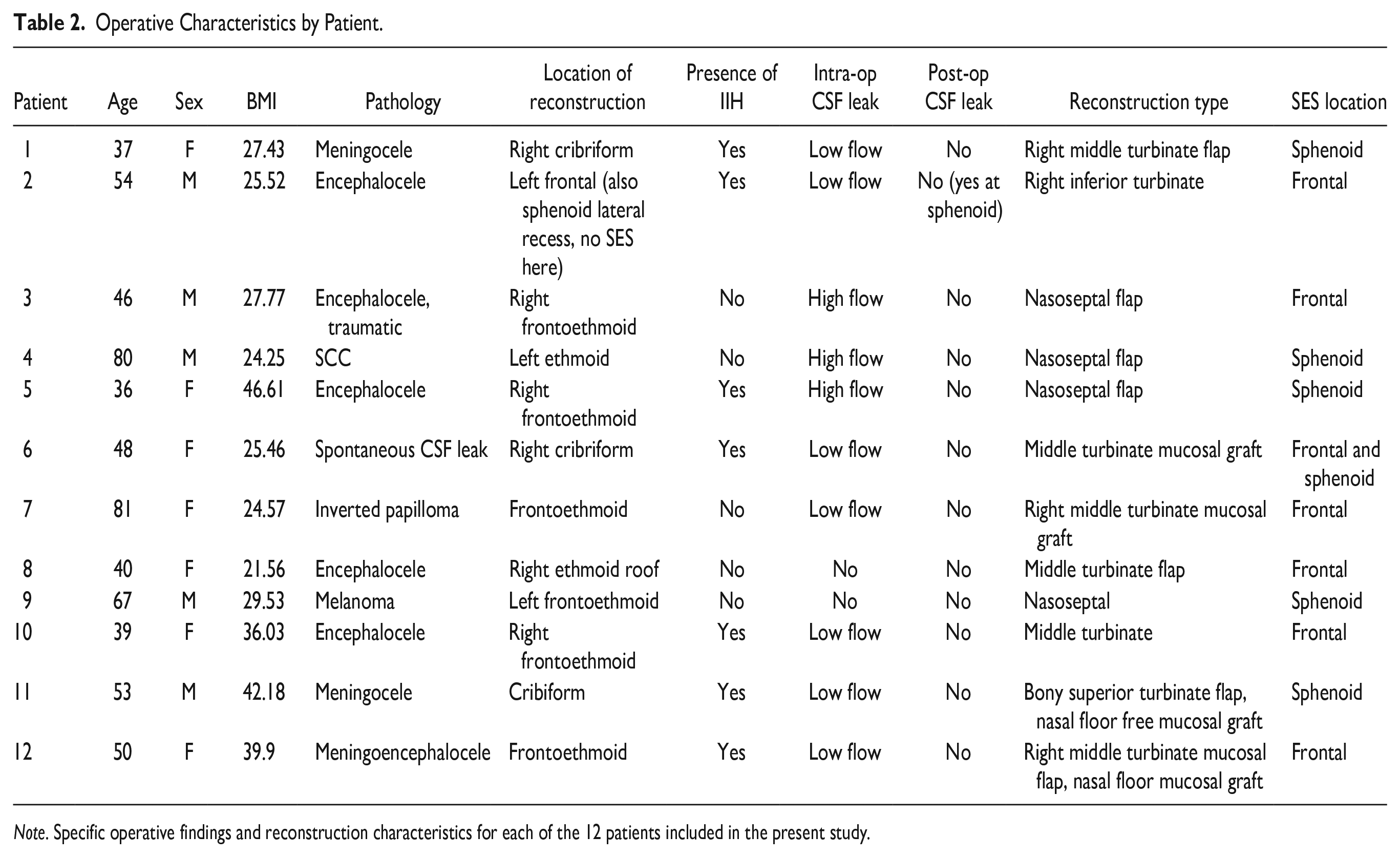
Note. Specific operative findings and reconstruction characteristics for each of the 12 patients included in the present study.
The most common pathology was encephalocele or meningocele (n = 9; 75%) followed by tumors (n = 3; 25%) (squamous cell carcinoma, mucosal melanoma and inverted papilloma of the skull base). Eight patients with meningoencephalocele and/or spontaneous CSF leaks had co-morbid IIH. The most common location of reconstruction was the frontoethmoid region (50%) followed by cribriform (25%), ethmoid roof (17%), and frontal sinus (8%) (Figures 1 and 2).
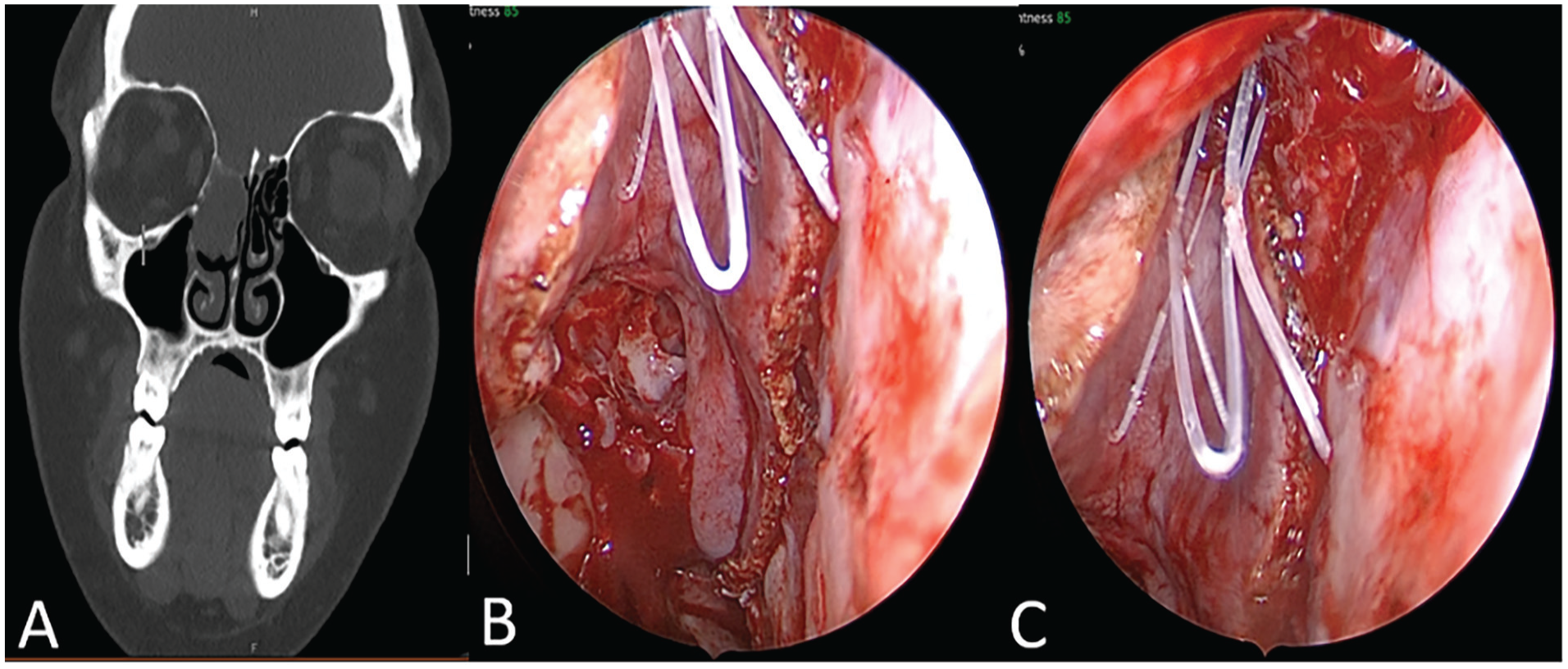
(A) Coronal sinus computed tomography of a patient with a large right frontoethmoid meningoencephalocele. (B and C) Intraoperative endoscopic images of a right frontoethmoid defect reconstructed with a right nasoseptal flap. A PROPEL stent was placed as a bolster for the flap in the frontal recess, maintaining its pre-designed shape. Images show the flap, donor site, and SES in situ.
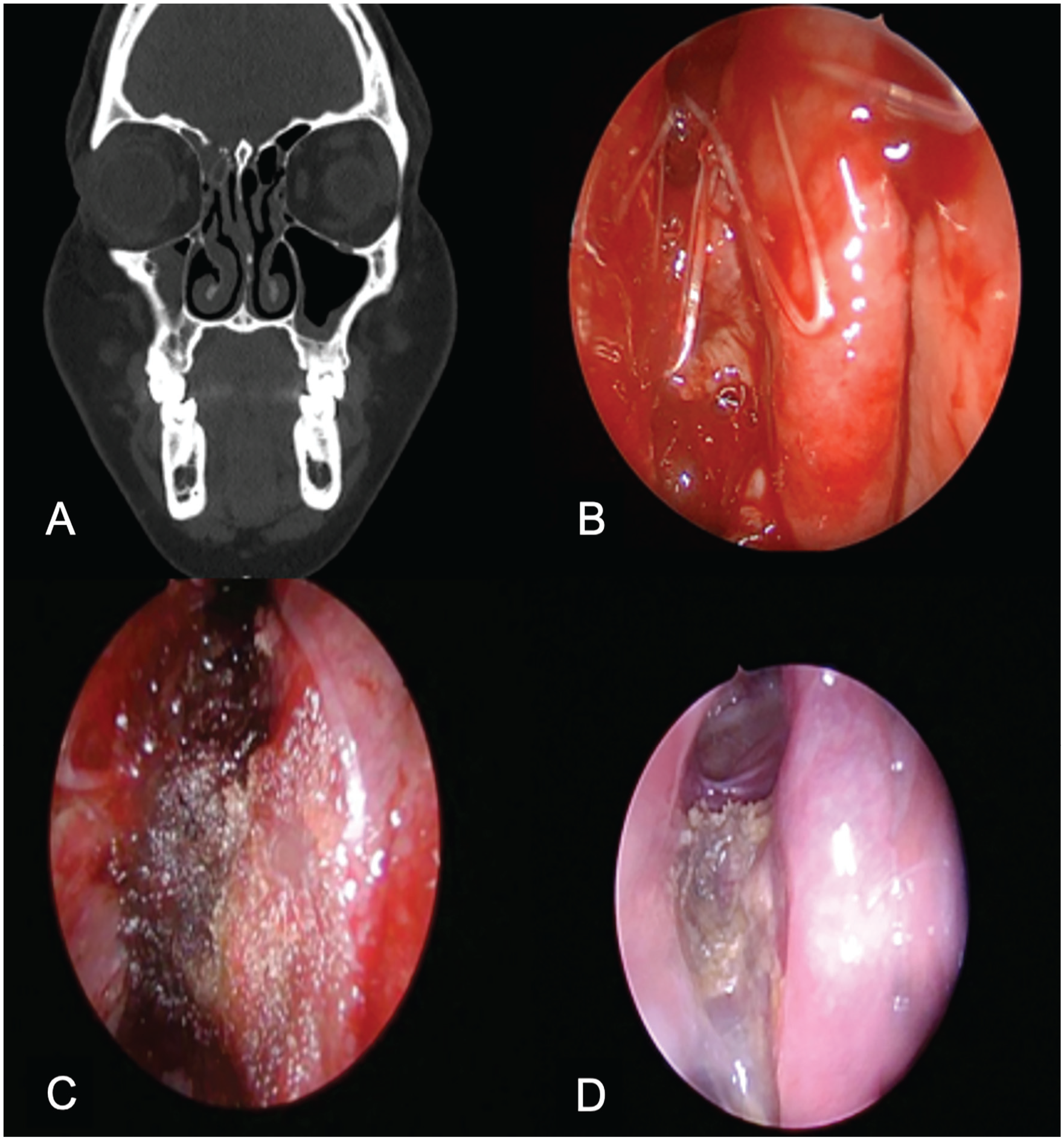
(A) Computed tomography, coronal section of a patient with a right fronto-ethmoid meningoencephalocele showing a bony defect in the right fovea ethmoidalis. (B) Intraoperative endoscopic picture showing the SES in the right frontal recess, bolstering an anteriorly based middle turbinate flap used for the onlay reconstruction. (C) Postoperative day 7 endoscopic image, following removal of the non-resorbable packing showing the SES within the frontal sinusotomy and the prongs bolstering the middle turbinate flap. (D) Postoperative day 60 endoscopic image, showing a widely patent frontal recess, and pink flap with some residual crusting.
High flow leaks at the time of reconstruction were recorded in 25% of patients. Two patients had a skull base defect with previous history of meningitis or a positive beta-2-transferin test, with no intraoperative active CSF leak at the defect site.
Onlay reconstruction was performed with either a nasal floor mucosal graft (n = 2), middle turbinate free mucosal graft (n = 3), inferior turbinate free mucosal graft (n = 1), anteriorly based middle turbinate flap (n = 2), or nasoseptal flap (n = 4).
One patient with comorbid IIH had a post-operative CSF leak at a secondary site (not bolstered with a stent). No post-operative CSF leaks occurred at a reconstruction site where an SES was used. No post-operative surgical site infections were observed. No other known adverse events were related to stent placement. Stents were left in place to disintegrate spontaneously within the paranasal sinus opening.
The SES were placed within the frontal recess in 7 cases and the sphenoid sinusotomy in 6 cases. One patient had 2 SES placed, 1 in the frontal recess and another in the sphenoidotomy. In all cases, the implant prongs were coming out from the frontal sinusotomy or the sphenoid sinusotomy and were abutting the onlay reconstruction against the anterior skull base. All patients had patent sinusotomies on endoscopic exam at their last follow up visit. The median duration of follow-up was 7.1 months (range, 1-21 months)
Discussion
This retrospective study evaluated the use of SES to bolster the reconstructive tissue in the surgical repair of anterior skull base CSF leaks and skull base reconstruction while preserving the patency of the paranasal sinuses. We asked whether this novel use of SES is safe and effective in a small cohort of patients. This is the first study to evaluate a potential role for the use of SES in anterior skull base reconstruction and CSF leak repairs.
We show that the use of SES as a bolster of free mucosal graft or a nasal flap is safe in patients, with no adverse events, including infection, associated with the stent. This proved true across patients with a diverse set of inciting conditions requiring different reconstruction locations along the anterior skull base. We also show that the use of SES is feasible in a variety of reconstructive techniques including free mucosal grafts, turbinate flaps, and nasoseptal flaps. While 1 patient had a post-operative CSF leak, this occurred at a secondary site not bolstered by SES and in the setting of comorbid IIH. Therefore, this likely did not represent a complication secondary to stent use.
Prior to the use of SES during FESS for chronic rhinosinusitis, a variety of packing techniques and stents were commonly used to help maintain sinus patency. 12 However, these often required either topical or oral steroids and postoperative interventions such as removal of the packing and lysis of adhesions. Since then, the benefits of using SES for chronic rhinosinusitis have been evaluated in blinded multi-institutional clinical trials.13,14 Specifically, patients who had SES placed during surgery required a significantly lower number of post-operative interventions, had a decreased amount of frank polyposis, and less frequently required oral steroids as compared to patients who received a stent that did not release steroids. 14 While we evaluated the use of SES in the repair of anterior CSF leaks and not chronic rhinosinusitis, it is clear from the chronic rhinosinusitis literature that SES can contribute to improved healing of the nasal mucosa in the post-operative period. In our study, the steroid released by these stents did not appear to affect the healing process for the graft or flap to take at the recipient site.
Additionally, the favorable, pre-designed shape of the SES and its elastic property makes it suitable for proper placement in the paranasal sinusotomies or recess during surgery thus preventing obstruction of the sinus by the tissue used for reconstruction or by the subsequent scarring, and pushing the reconstructive tissue against the base of skull. The fact that it does not dissolve until 30 to 45 days after placement allows the SES to provide lasting structural support in the post-operative period after removal of non-resorbable packing with minimal to no patient discomfort. 11 Thus, the use of this device could also provide significant longer-term benefits to our patient cohort.
Use of SES in FESS is not, however, without adverse effects. A recent retrospective study examined the literature for reports of patients who received an SES and subsequently had complications. 15 A total of 28 cases were identified. The most common adverse event reported was post-operative infection (11 patients), 5 of whom had fungal sinusitis. Given the fact that FESS has a baseline risk of post-operative infection, 16 it is not clear whether the SES increased the incidence of infection or was merely associated with it. The presence of steroids in the sinus could contribute to a diminished local immune response, increasing susceptibility to infection. A pre-clinical rabbit model, however, did not show an increased incidence of fungal infection in animals receiving SES versus non-drug-eluting stents. 17 Additionally, the ADVANCE trials in humans did not report a significant increase in post-operative infection with the use of SES,13,14 and neither systemic absorption of steroids nor changes in plasma cortisol levels have been observed with SES use. 18 Therefore, while low, the risk of post-operative infection could be increased by the use of SES. While we did not observe any cases of post-operative infection in our cohort, caution should be taken to prevent complications of CNS infection in post-operative patients, especially given that post-operative meningitis can occur following standard CSF leak repair without the use of SES. 19
Other relevant adverse events reported in the literature include stent migration in 6 patients, which occurred most commonly in the oropharynx but did not require surgical intervention, and CSF leaks in 2 patients, which were likely iatrogenic. 15 Only 1 case of increased intra-ocular pressure following SES use has been reported, 15 and none of the 155 patients in the ADVANCE trials showed increased intra-ocular pressure in the post-operative period.13,14
The limitations of our study should be acknowledged. This was a retrospective study with a small sample size. Without a control group, we were not able to assess the efficacy and benefits of SES use compared to the current standard of care. Also, resorbable and non-resorbable packing were used in addition to the SES for bolstering. This prevents us from truly understanding the bolstering function of SES and whether they can be used alone for anterior skull base reconstruction support. With our small sample size, it could be difficult to detect rare, yet serious complications, especially in the setting of CSF leaks where the CNS is vulnerable. A larger, prospective controlled study will be necessary to study the efficacy of this method compared to other bolster/buttress techniques.
Conclusion
Steroid-eluting stents are safe and helpful as an adjunct to bolstering anterior skull base reconstruction grafts and flaps during endoscopic endonasal CSF leak repair, providing longer term structural support with minimal to no patient discomfort and preserving sinus drainage patency.
Footnotes
Declaration of Conflicting Interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: David Jang and Ralph Abi Hachem receive research funding from Amgen and GlaxoSmithKline. David Jang is a consultant for Medtronic.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
