Abstract
Trefoil factor 3 (TFF3) has been studied in processes leading to atherosclerosis. Data are scarce in manifest disease and missing in peripheral artery disease (PAD). This study aims to elucidate TFF3 with disease stages, degrees of atherosclerosis, and outcomes. TFF3 was measured in serum in 364 PAD patients without critical limb ischemia and mild to moderate chronic kidney disease (CKD). Mortality data were retrieved from the Austrian central death registry (median observation 9.6 years). Survival analyses were performed using Cox regression and the Kaplan–Meier method. A negative association between ankle-brachial index and TFF3 (P < .001) was observed, while levels were similar in asymptomatic and symptomatic PAD. TFF3 increased with history of cardiovascular and cerebrovascular disease (P < .001). TTF3 was associated with the estimated glomerular filtration rate (R = −0.617, P < .001) and urinary albumin-creatinine ratio (R = 0.229, P < .001). One SD increase in TFF3 showed a worsening in all-cause mortality (hazard ratio 1.68, CI 1.37–2.05) which persisted after multiple adjustment for cardiovascular risk, inflammatory, and angiogenetic markers (hazard ratio 1.35, CI 1.01–1.81). This study is the first to link TFF3 with both disease markers and outcomes in PAD. TFF3 demonstrated associations with renal function, PAD severity measured by ankle-brachial index, and additional atherosclerotic burden in PAD.
Introduction
Trefoil factor 3 (TFF3) is a protein that is not typically associated with atherosclerosis. The small 6kD glycoprotein with a characteristic structure formed by disulfide bonds, the eponymous trefoil domain,1–3 is part of a family consisting of three subtypes (TFF1, TFF2, and TFF3). Trefoil factor 3 was first been described over 30 years ago is typically expressed by mucous epithelia with several functions, 4 including implications in rheologic properties of mucus. 5
While TFF3 is mainly known for being involved in the regulation of mucus and the modification of its properties, it has not only been found to be secreted in goblet cells but in a variety of organ systems, including the hypothalamus and the pituitary gland, lymphatic tissue, liver, heart, muscle, ulcerative colitis,3,6–8 neural tissue,9,10 and skeletal repair. 11
Trefoil factor 3 has also been described to be associated with previous malignant diseases such as prostate and ovarian cancer as well as the promotion of colorectal cancer,12–14 and mammary carcinoma, where it has been linked to angiogenesis and is involved processes like wound healing and cellular migration.8,15,16
Trefoil factor 3 has also been linked to renal disease in clinical studies, 17 which in turn has evolved as early risk factor for the ontogenesis and progression of atherosclerosis, 18 and explicitly PAD in the past years. 19 In addition, observations related to vascular disease have described several possible mechanisms which support a possible direct link between vascular disease and TFF3.20–22
Proangiogenic properties are similar to that of the vascular endothelial growth factor (VEGF), which is dependent upon cyclooxygenase-2 (COX2) and epithelial growth factor receptor (EGF-R) signaling.3,20 In addition to an upregulation of COX2 and an increase in prostaglandin synthesis, TTF3 can increase perfusion through an increase of nitric oxide (NO) by activation of inducible nitric oxide synthase (iNOS).20–22
Those findings suggest a possible stimulus for the expression of TFF3 in hypoxic tissue, which is common in patients with symptomatic peripheral artery disease (PAD). However, despite strong implications, to date, no study investigating the impact of TFF3 on clinical and outcome parameters in patients with PAD has been published.
Methods
Study Cohort
The study was approved by the institutional ethics committee and complies with the Declaration of Helsinki, including current revisions and Good Clinical Practice guidelines.23,24 The procedures followed were in accordance with institutional guidelines, and all patients gave written informed consent before inclusion into the study.
Trefoil factor 3 serum concentration was measured once in patients from the Vascular Medicine Center (VMC) Vienna cohort. The VMC Vienna cohort comprises PAD patients (asymptomatic or claudication symptoms) without critical limb ischemia or vascularization procedure at the time of inclusion. Patients with ulcerations, known cancer, or end-stage kidney disease were excluded. Detailed inclusion and exclusion criteria as well as data and analyses of comorbidities and medication have been published. 25
Laboratory Parameters
Fasting blood samples were collected at the baseline study visit. Samples for other measurements were sent to the institutional central laboratory of the General Hospital of Vienna for immediate analysis. The following parameters were measured: Fasting plasma glucose, HbA1c, triglycerides, total cholesterol, high-density lipoprotein cholesterol (HDL-C), low-density lipoprotein cholesterol (LDL-C), C-reactive protein (CRP), and serum creatinine. The estimated glomerular filtration rate (eGFR) was calculated using the Chronic Kidney Disease Epidemiology Collaboration (CKD-EPI)—equation. 26
Patient Assessment and Outcome Monitoring
At baseline, patients were evaluated by standardized questionnaires to obtain smoking status, body composition, and previous cardiovascular or cerebrovascular events. In addition, coronary heart disease and carotid artery disease were assessed by review of the patient’s history in a retrospective manner. PAD was defined as an ankle-brachial index (ABI) <0.9 or known previous peripheral revascularization. The Fontaine classification was used for the clinical assessment of self-reported pain-free walking distance. 27
ABI was measured by trained technicians using non-invasive Doppler sonographic measurements (ELCAT VL5000, Wolfratshausen, Germany). In the case of ABI >1.4 or noncompressible arteries media sclerosis was diagnosed. Blood samples for TFF3 and VEGF measurements were collected after an overnight fast, immediately centrifuged, and stored at −80°C prior to analysis.
Patients were followed in a prospective manner for up to 10 years to assess mortality. Outcome data were obtained by the Austrian central death registry and adjudication to assess cardiovascular causes was carried out by review of the patient’s history.
Measurement of TFF3
Serum concentrations of TFF3 were measured by bead-array technology using a commercially available kit (R&D Systems, Inc., Minneapolis, MN, USA). Samples from 364 patients were available for analysis.
The intra-assay and inter-assay coefficient of variation was 6.93% and 6.66%, respectively.
Statistical Analysis
Statistical analysis was performed using IBM SPSS 28.0 (IBM Corp., Armonk, NY, USA). Data are presented as mean (±standard deviation) or median (25th percentile; 75th percentile) as appropriate. A P < .05 (two-tailed) was considered statistically significant. Standard distribution was assessed by the Kolmogorov–Smirnov test. If necessary, parameters were log-transformed to obtain a normal distribution. ANOVA with post-hoc analysis, Student’s t-test, Kruskal–Wallis test, or Whitney–Mann U-test were performed to investigate differences between groups, as appropriate. Bivariate correlation analysis was performed using Pearson coefficients. To assess the association of TFF3 and ABI, univariate linear regression and a multivariable linear regression model for the prediction of systemic atherosclerosis, including TFF3, vascular and sociodemographic risk factors, CRP, and eGFR, was conducted.
Outcome analysis was carried out by using the Kaplan–Meier method with log-rank test and Cox regression analysis to assess a possible association with survival.
Results
TFF3 measurements were obtained from 364 Patients (33.8% female, age 69 ± 10.3 years) with PAD. There was a high prevalence of comorbidities with 143 (39%) patients having a history of cerebrovascular disease, 116 (31%) having a history of cardiovascular disease, and 56 (15%) patients who had both.
Baseline characteristics for TFF3 tertiles.
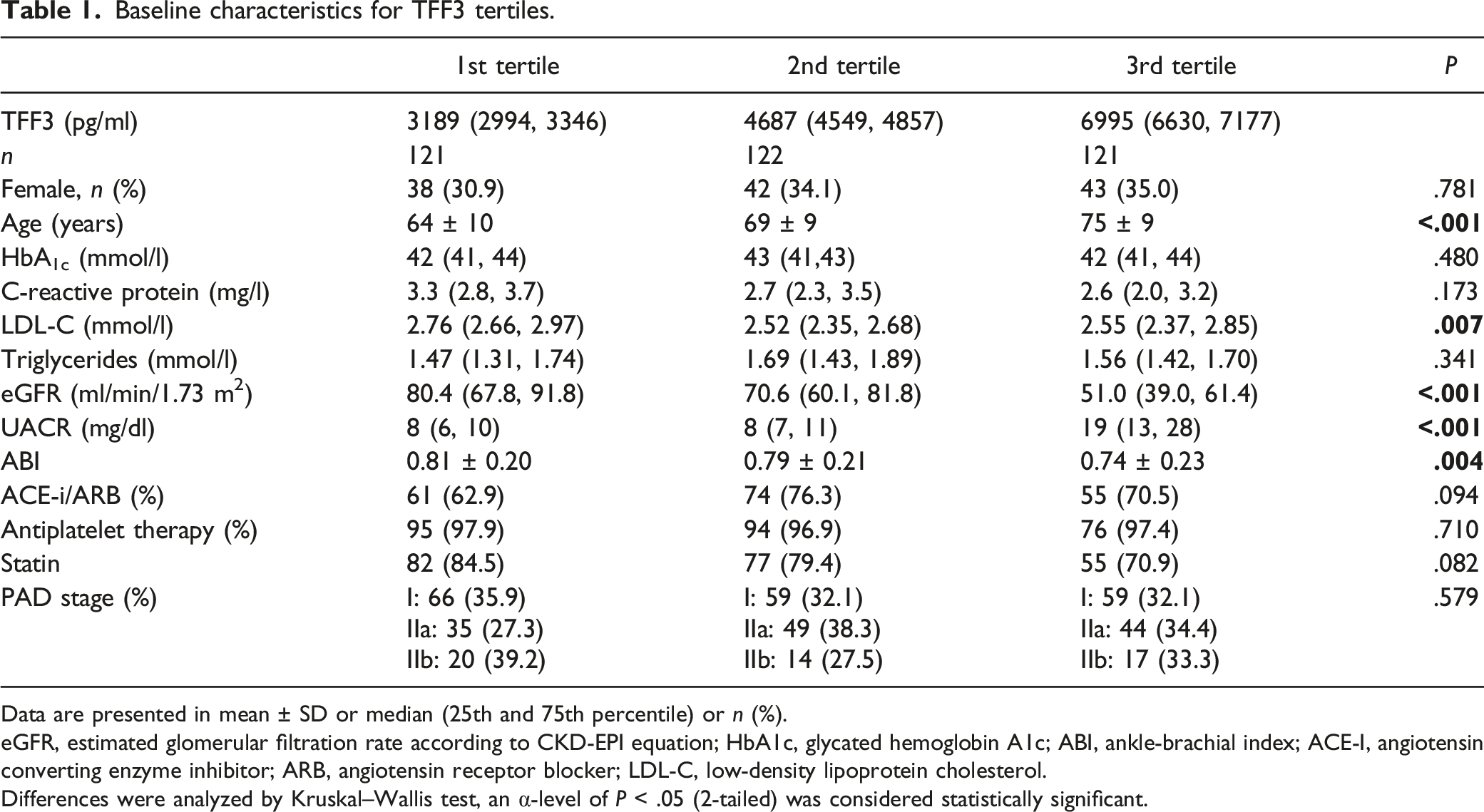
Data are presented in mean ± SD or median (25th and 75th percentile) or n (%).
eGFR, estimated glomerular filtration rate according to CKD-EPI equation; HbA1c, glycated hemoglobin A1c; ABI, ankle-brachial index; ACE-I, angiotensin converting enzyme inhibitor; ARB, angiotensin receptor blocker; LDL-C, low-density lipoprotein cholesterol.
Differences were analyzed by Kruskal–Wallis test, an α-level of P < .05 (2-tailed) was considered statistically significant.
TFF3 and Renal Function
129 patients had an impaired renal function with an eGFR <60 mL/min/1.73 m2 while no patients had an eGFR <15 mL/min/1.73 m2.
When analyzing according to CKD stages, a stepwise increase according to CKD stages 1 to 4 with the highest TFF3 values in CKD 4 could be observed (P < .001).
To assess the association between TFF3 and renal damage, which has been linked to PAD as well as cardiovascular events, we correlated TFF3 with both main clinical attributes of renal function, albuminuria, and renal function defined by eGFR.
We could observe a significant negative correlation between eGFR and TFF3 (P < .001, R = −0.617). The urinary albumin-creatinine ratio could be measured in 347 patients, of those 87 patients had overt albuminuria defined by a urinary albumin/creatinine ratio ≥30 mg/g. 28 There was a significant difference in TFF3 levels between patients with overt albuminuria and patients without (P < .001). Additionally, when including all patients, a significant correlation between albuminuria and TFF3 levels could be observed (P < .001, R = 0.241).
TFF3 and Atherosclerosis
A significant negative correlation between ABI and TFF3 could be observed when excluding patients with media sclerosis (n = 258, P < .001, R = −0.277) (Figure 1). Consecutively, there was a significant difference in ABI (P < .001) with a stepwise increase from the highest (0.67 [0.24–1.05]) and the middle (0.77 [0.31–1.17]) to the lowest TFF3 tertile (0.79 [0.34–1.25]). TFF3 in serum and ankle-brachial index scatter-plot of the patient’s individual logarithm TFF3 serum levels and ankle-brachial index measurements. R represents the Pearson correlation coefficient. A P < .05 (two-tailed) was considered statistically significant.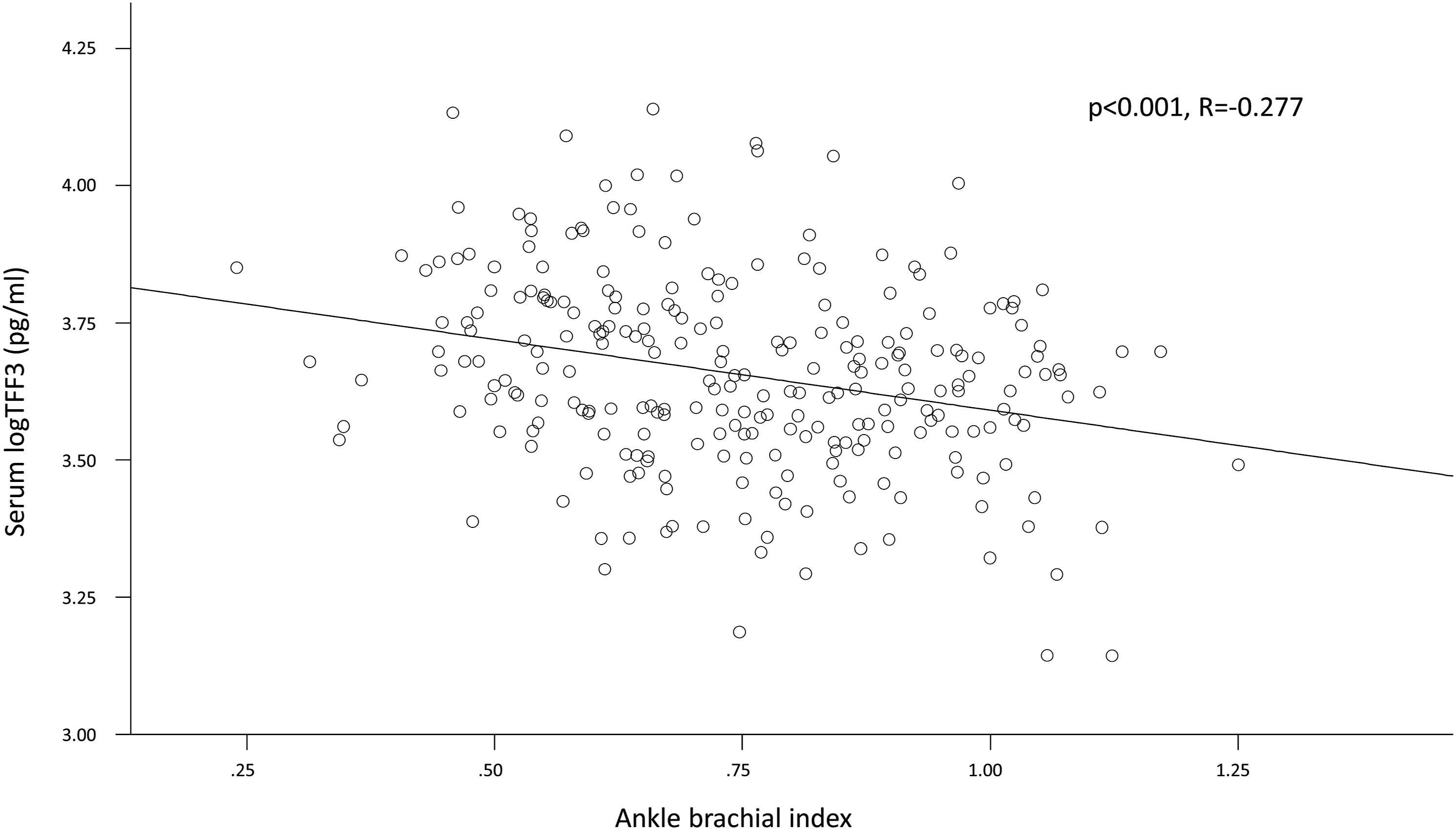
To evaluate TFF3 as an independent predictor, we performed a stepwise regression analysis in three models. For further analyses with regard to atherosclerosis, the variables age, gender, HbA1c, LDL-C, and eGFR (model 1), as well as CRP to represent inflammation (model 2), and VEGF to represent angiogenesis (model 3) were used; TFF3 remained a significant predictor of the ABI in all three models.
Regarding PAD parameters, there was no difference in clinical PAD stages (P = .461); thus, we did not observe a difference between asymptomatic and symptomatic patients.
To further differentiate between sites of atherosclerotic lesions, we formed the study population into groups of PAD alone and PAD in addition to other vascular regions (carotid artery disease and cardiovascular disease). We observed an increase with the highest TFF3 levels in patients with all atherosclerotic manifestations (P < .001) while there was no significant difference between one or two additional atherosclerotic manifestations (P = .481).
Outcome Analysis
143 patients died (59% cardiovascular) within a median observation period of 9.6 (6.9, 10.0) years. Both all-cause and cardiovascular mortality were significantly higher in patients in the highest TFF3 tertile compared with middle and lowest tertile (P < .001) and TFF3 was significantly associated mortality in univariate Cox regression analyses (HR 1.68, CI 1.37–2.05; per SD of TFF3) (Figure 2). It remained significant as an independent marker when correcting for risk factors in model 1 (HR 1.38, CI 1.03–1.83), model 2 (HR 1.36, CI 1.02–1.82), and model 3 (HR 1.35, CI 1.01–1.81) (Table 2). TFF3 and long-term mortality Kaplan–Meier plot for the prediction of all-cause (A) and cardiovascular (B) survival according to serum TFF3 tertiles over 10 years. Tertiles were compared using the log-rank test (P-value). All-cause mortality by standard deviation of TFF3 serum levels. Cox regression analyses for all-cause mortality by TFF3. HR, hazard ratio; CI, confidence interval; HbA1c, glycated hemoglobin A1c; LDL-C, low-density lipoprotein cholesterol; eGFR, estimated glomerular filtration rate; VEGF, vascular endothelial growth factor.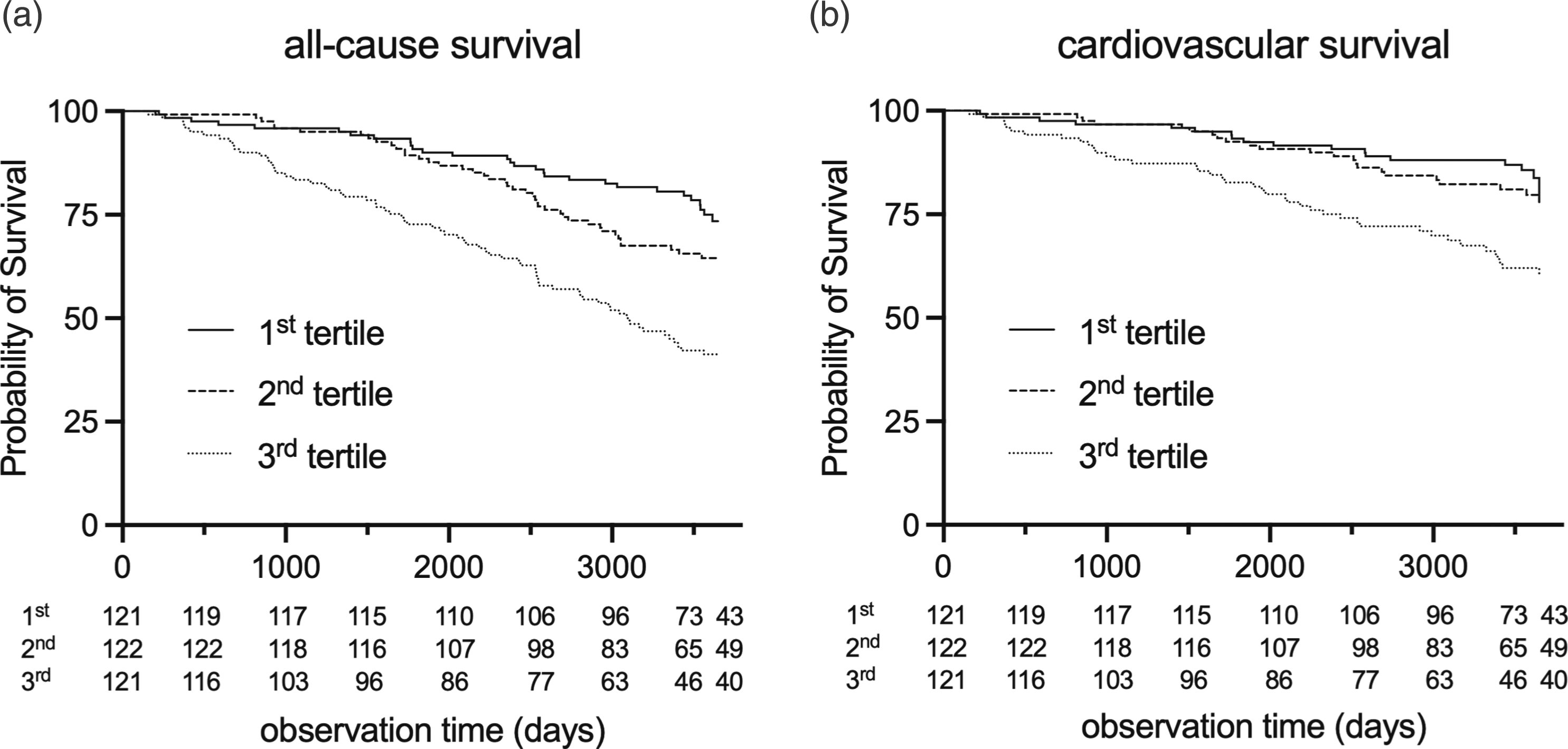

Discussion
In current literature, TFF3 was mainly investigated with a scope outside the cardiovascular field. This study is the first to demonstrate an association with TFF3 with the ABI, a very relevant prognostic factor for all-cause and leg survival in patients with PAD and one of the few independent predictors for survival in cardiovascular disease. In addition, our data suggest an association of TFF3 with the development of other macrovascular disease, namely, cardiovascular and cerebrovascular disease. Furthermore, those associations do not only show in measurements of surrogates but also in all-cause and cardiovascular survival.
While the literature on TFF3 in vascular disease is scarce, there are some studies that provide a possible explanation for our observations. In the ARIC (Atherosclerotic Risk In Communities) studies, 17 TFF3 was investigated as a possible marker for CKD in a sub-cohort of 143 patients with an eGFR <60 mL/min/1.73 m2, which were compared to 143 healthy control subjects. Median urinary TFF3 measurements were significantly higher in patients than in the control group. Although the authors concluded that TFF3 was expressed as part of a repair mechanism in the renal tubules, TFF3 remained a significant marker of mortality independent of renal function. While in ARIC, TFF3 was measured in urine samples, we found similar associations in serum. Thus, our results suggest that TFF3 might reflect damage in other parts of the kidney, which can be associated with mortality and vascular damage. 29
In turn, loss of renal function is associated with the development of PAD and mortality.18,19 Our data fit those well-documented observations. Patients with nephropathy showed higher TFF3 levels than patients with normal renal function. When investigating TFF3 under the aspect of atherosclerosis and PAD, we could observe an increase of TFF3 with an increase in atherosclerotic burden. TFF3 was highest in patients who had systemic manifestations and suffered from peripheral, cardio-, and cerebrovascular disease. Within the entity of PAD, patients who had a more severe manifestation documented by ABI had higher TFF3 levels. Interestingly, the patient’s symptoms documented by the clinical PAD stage did not show a similar pattern. However, in our study, only patients with Fontaine stages I and II were included. Thus, we suspect that TFF3 mainly reflects chronic ischemia, while intermittent ischemia, which is often the underlying mechanism of Fontaine stages I and II, is not a significant contributor to TFF3. This hypothesis is supported by several observations made on TFF3 expression, which showed an upregulation of COX2 and an increase in prostaglandin synthesis as well as an activation of iNOS as effects of TFF3.21,22
While we could demonstrate the associations between atherosclerosis and outcome, the underlying pathophysiological mechanisms behind TFF3 remain unclear. While in our study, TFF3 did not seem to be associated with inflammation and did not correlate with CRP, it has been associated with angiogenesis in malignancies. 15 Again, this could be interpreted as an indicator of a potentially chronic ischemic setting in which TFF3 is mainly expressed in.
In this study, several limitations have to be considered. First, no patients with critical ischemia were included. While it is likely that patients with a higher degree of atherosclerotic burden and especially a higher degree of ischemia would show a further increase in TFF3 levels, our data cannot be applied to those patients. Second, TFF3 was only measured at baseline and longitudinal measurements are not available. Thus, we do not know if changes in TFF3 in a single patient have an effect on the outcome. Our results are hypothesis-generating and not definitive.
Our data suggest, for the first time, a significant role of TFF3 in PAD regarding both atherosclerotic burden and outcome.
Footnotes
Author contributions
GP performed the literature search and study design, conducted the statistical analyses, interpreted the results, and drafted the manuscript. BZ, CH, and RK participated in the interpretation of the results and revised the manuscript. GHS designed the VMC study cohort, supervised the statistical analyses, interpreted the results, and revised the manuscript. All authors read and approved the final manuscript.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: Funding was provided by Medical University Vienna, Vienna, Austria.
