Abstract
The Naples score is a new prognostic score developed according to inflammatory and nutritional status and frequently evaluated in cancer patients. The present study aimed to evaluate using the Naples prognostic score (NPS) to predict the development of decreased left ventricular ejection fraction (LVEF) after acute ST-segment elevation myocardial infarction (STEMI). The study has a multicenter and retrospective design and included 2280 patients with STEMI who underwent primary percutaneous coronary intervention (pPCI) between 2017 and 2022. All participants were divided into 2 groups according to their NPS. The relationship between these 2 groups and LVEF was evaluated. The low-Naples risk group (Group-1) included 799 patients, and the high-Naples risk group (Group-2) had 1481 patients. Hospital mortality, shock, and no-reflow rates were found to be higher in Group 2 compared with Group 1 (P < .001, P = .032, P = .004). The NPS was significantly inversely associated with discharge LVEF (B coefficient: −1.51, 95% CI-2.26; −.76, P = .001). NPS, a simple and easily calculated risk score, may help identify high-risk STEMI patients. To the best of our knowledge, the present study is the first to demonstrate the relationship between low LVEF and NPS in patients with STEMI.
Introduction
Reduced left ventricular ejection fraction (LVEF) following acute myocardial infarction (AMI) is an important indicator of cardiac prognosis. 1 ST-segment elevation myocardial infarction (STEMI) is associated with a high morbidity and mortality. 2 Reduced LVEF is seen in approximately 30–40% of patients with STEMI. 3
Many clinical, demographic, and laboratory parameters and risk scores predict hospitalization, adverse effects, and mortality for acute coronary syndrome (ACS).4,5 Galizia et al. 6 evaluated the Naples prognostic score (NPS) in a cohort of 562 patients undergoing first-time colorectal cancer surgery. The NPS is a new prognostic score developed according to the inflammatory and nutritional status and frequently evaluated in cancer patients. Considering the activation of inflammatory processes in the pathogenesis of AMI, indicators related to inflammation such as neutrophil-lymphocyte ratio (NLR), lymphocyte-monocyte ratio (LMR), and platelet-lymphocyte ratio (PLR) can be used as predictors of prognosis in AMI.7–9
There is an unmet need for patients with AMI to elaborate on the prognostic impact of the NPS on ejection fraction. Therefore, this study aims to evaluate NPS as a predictor of reduced LVEF development after AMI.
Methods
Study Population
In this retrospective study, we identified the clinical data of 2590 patients who attended Ataturk University Medical Faculty Research Hospital and Erzurum City Hospital between 2017 and 2022 with acute STEMI who underwent primary percutaneous coronary intervention (pPCI). Information was collected for analysis from the hospital medical record system. Patients with end-stage renal disease estimated Glomerular Filtration Rate (eGFR) under 15 mL/min/1.73 m2 or on chronic hemodialysis at the time of the procedure, missing creatinine values before or within 48 h following the procedure, missing preprocedural hemoglobin, albumin, monocyte, lymphocyte, and cholesterol values, also the patients who died before LVEF measurement were excluded from the study. In addition, patients with a previous history of coronary artery bypass graft (CABG) surgery and PCI were not included in the study. The remaining 2280 patients formed the study population (Figure 1(A)). (A): Consort flow patient inclusion/exclusion diagram. Abbreviations: STEMI, ST-segment elevation myocardial infarction; PCI, Percutaneous coronary intervention; CABG, Coronary artery bypass graft; NPS, Naples prognostic score. (B): Parameters of Naples prognostic score.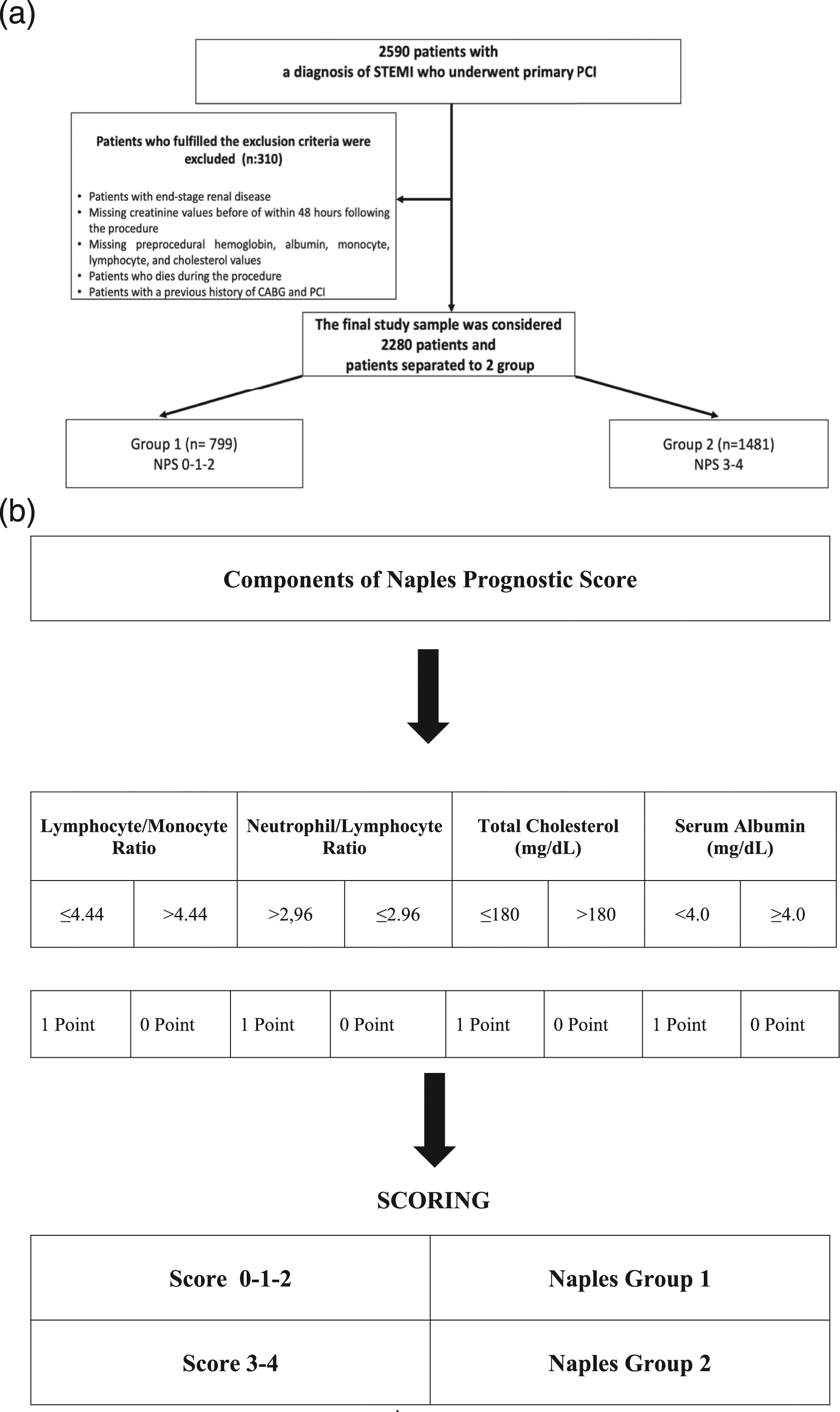
Patients who underwent pPCI with stent implantation within 12 h of symptom onset were included. After hospital admission, the patients were given a single dose of clopidogrel (600 mg) or prasugrel (60 mg) or ticagrelor (180 mg) and aspirin (300 mg) orally. Unfractionated heparin (first dose: 5000–10000 I.U., additional doses: achieves to activate clotting time >250 sec) was adjusted according to operator preference.
Echocardiography Analysis
All patients underwent transthoracic echocardiography (TTE) (Vivid 7; GE Medical System, Horten, Norway), and LVEF was determined using Simpson’s method. The data of patients who died within 1 year were based on the last TTE recorded in the system.
Study Protocol and Definitions
Age, sex, presence of hypertension (HT), diabetes mellitus (DM) and previous coronary artery disease (CAD), tobacco use, and complete physical examination were recorded from all study patients. Baseline laboratory values including missing preprocedural hemoglobin, albumin, monocyte, lymphocyte, and cholesterol values were recorded.
The NPS consists of 4 parameters, namely, serum albumin and total cholesterol concentrations, NLR, and LMR. 6 All patients in this study were divided into 2 groups according to their NPS scores. Patients with a score of 0, 1, 2, or 3, 4 were assigned to groups 1 and 2, respectively (Figure 1(B)).
Statistical Analysis
Numerical variables were represented as mean ± standard deviation while categorical variables were represented as absolute numbers and percentages. The primary outcome of the study was LVEF. The main predictor was NPS. The one-way ANOVA or Wilcoxon rank and the χ2 statistics were applied to evaluate the baseline and clinical characteristics among patients based on the NPS 0, 1, 2 vs 3, 4 group.
Simplified Grouping
We labeled patients as fit (NPS <3), and NPS 3, 4. Because there are only 5 NPS levels, we could not group the participants equally with a median NPS value.
Statistical modeling was carried out by using multivariable ordinary least square regression. Statistical models were built based on clinical rationale. Firstly, we built a full model using DM, HT, gender, age, shock, C Reactive Protein (CRP), Syntax score admission, total ischemic time, previous statin usage, admission cardiopulmonary resuscitation (CPR), anterior MI, no-reflow, and the main predictor, NPS. Finally, we developed a reduced model to make a simpler model with a stepdown backward method and alfa set as .05. The association between LVEF and NPS was quantified by the Beta coefficient and 95% Confidence Interval (CI, 95%). Continuous variables are included in the models using restricted cubic spline transformation with 3 knots to capture the non-linear relation between predictors and outcomes. A P < .05 was considered statistically significant. All statistical analyses were performed using the “RMS” package with R studio version 4.1 (R Project, Vienna, Austria).
Results
Baseline Demographic Clinic Characteristics Comparison According to Group-1 and Group-2.
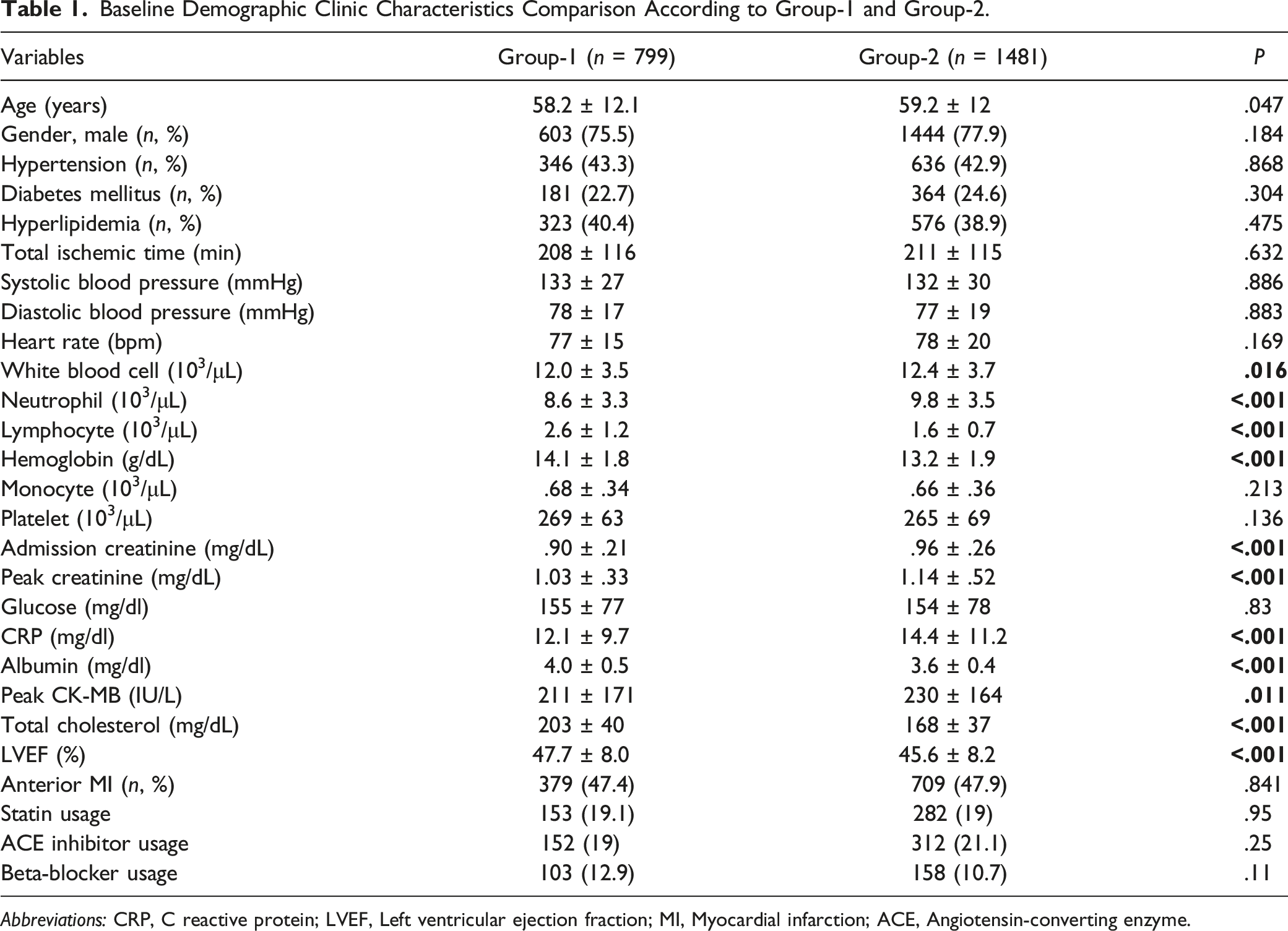
Abbreviations: CRP, C reactive protein; LVEF, Left ventricular ejection fraction; MI, Myocardial infarction; ACE, Angiotensin-converting enzyme.
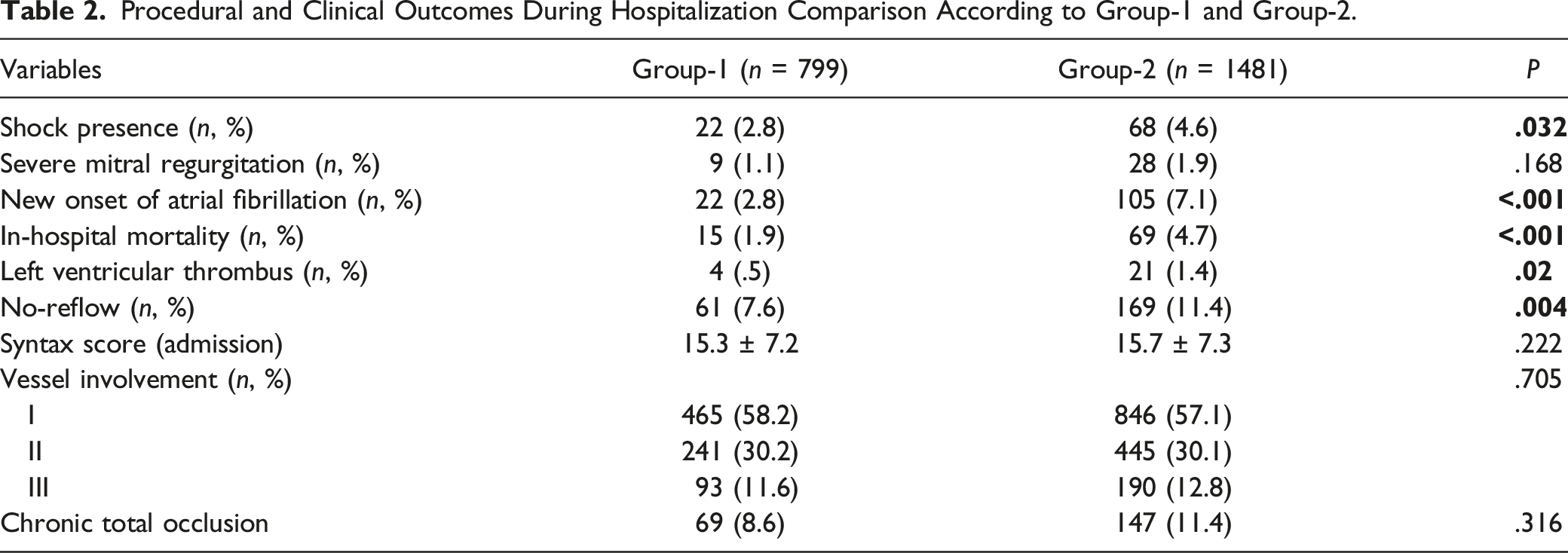
Procedural and Clinical Outcomes During Hospitalization Comparison According to Group-1 and Group-2.
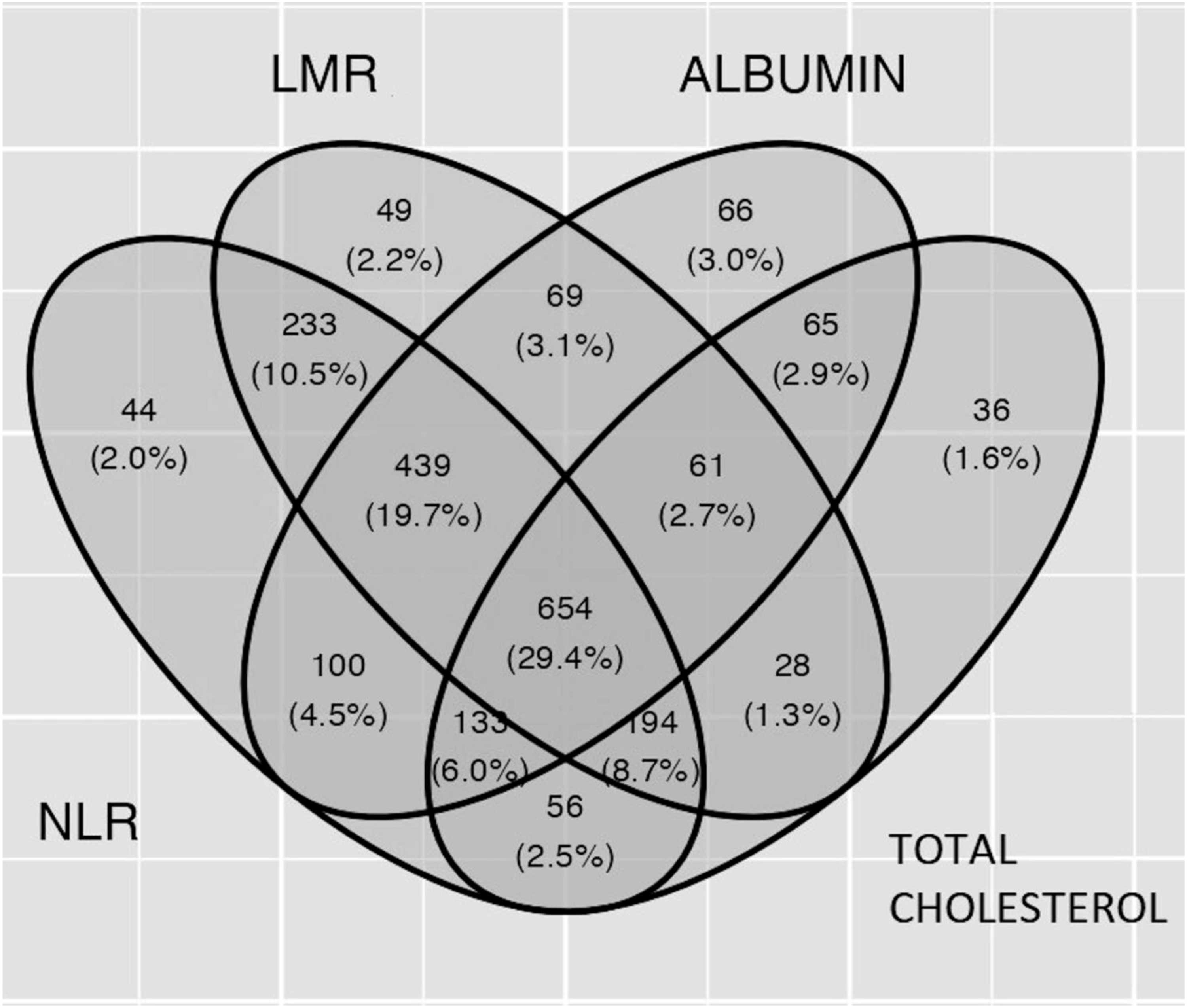
Venn diagram shows relationships between 4 parts of Naples prognostic score. Abbreviations: LMR, Lymphocyte-monocyte ratio; NLR, Neutrophil-lymphocyte ratio.
In order to determine the roles of NPS in the prediction of discharge LVEF, initially, a full model was produced including DM, HT, gender, age, shock, CRP, Syntax score admission, total ischemic time, previous statin usage, admission CPR, anterior MI, no-reflow, and the main predictor, NPS, were used. The predictors related to discharge LVEF were evaluated by multivariable linear regression.
Multivariable Linear Regression for Predicting Discharge LVEF in STEMI Patients.
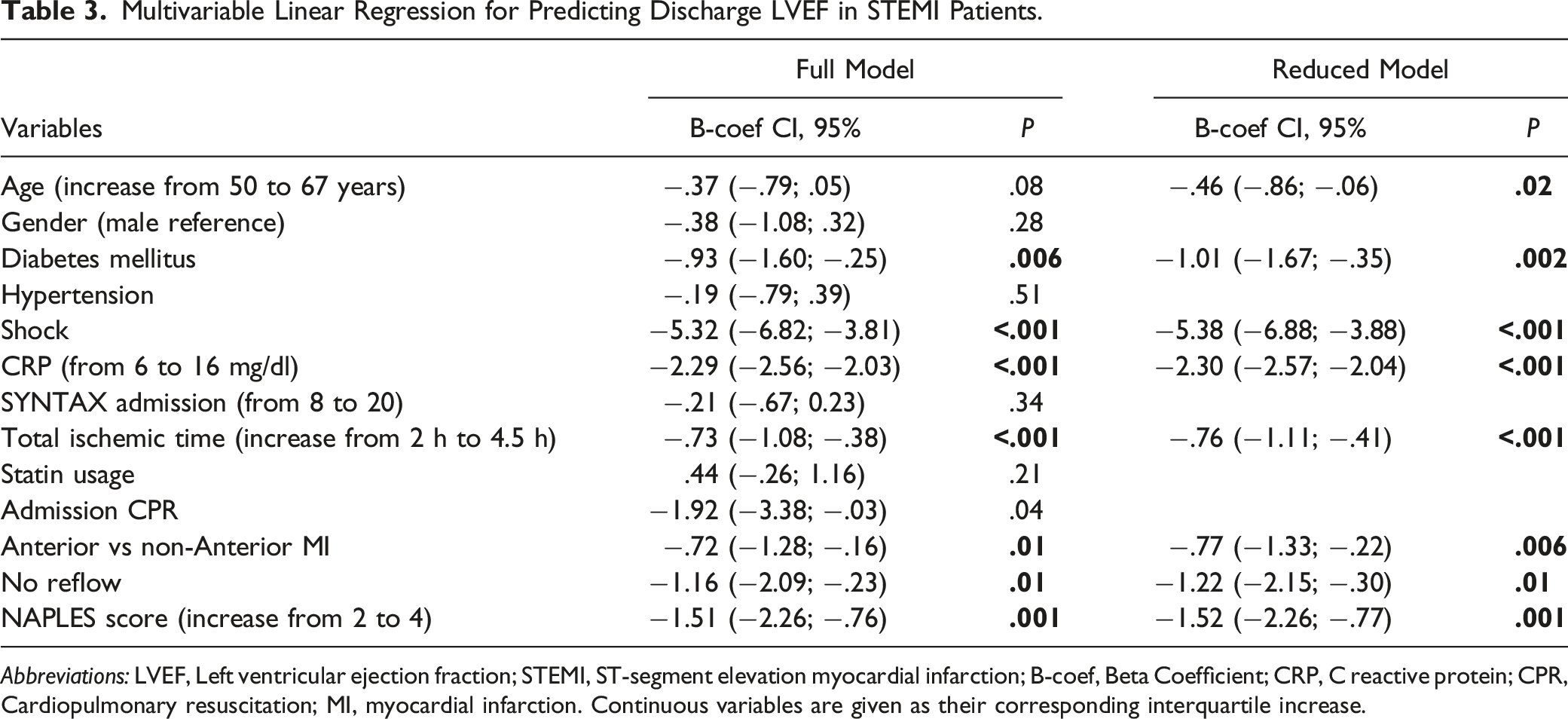
Abbreviations: LVEF, Left ventricular ejection fraction; STEMI, ST-segment elevation myocardial infarction; B-coef, Beta Coefficient; CRP, C reactive protein; CPR, Cardiopulmonary resuscitation; MI, myocardial infarction. Continuous variables are given as their corresponding interquartile increase.
For sensitivity analysis, we performed another analysis, when other variables retained reduced model inclusion of categoric NPS 3, 4 vs 0, 1, 2 (B coefficient: −.88 95% CI-1.47; −.29; P = .004). Added variable plot for reduced model demonstrated that when NPS increased, LVEF decreased (Figure 3). Variable plot for reduced model demonstrating that Naples prognostic score increased when left ventricular ejection fraction decreased.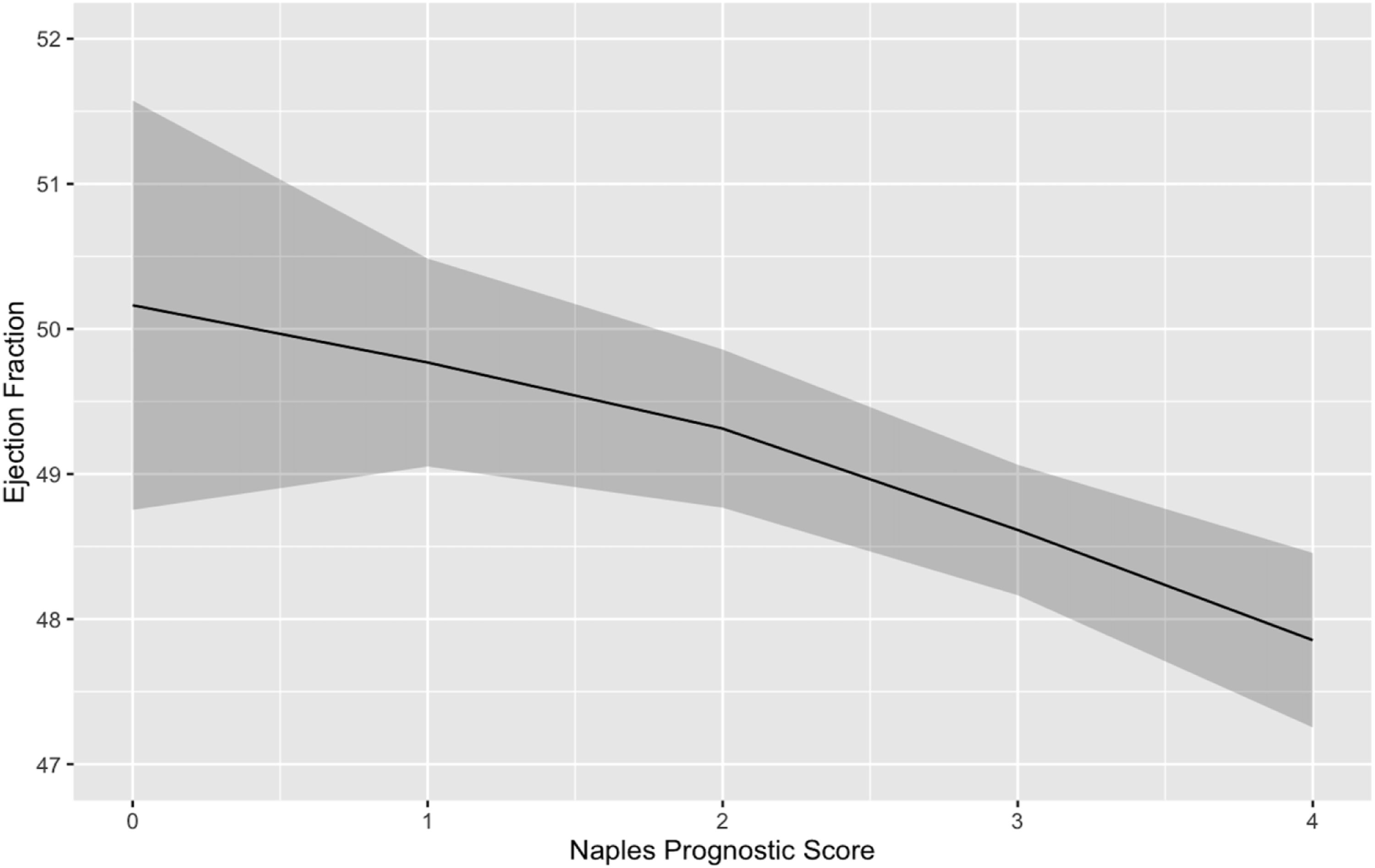
We performed a correlation analysis whether an association was present among variables used in the reduced model; the analysis depicted NPS as slightly and positively correlated with age (Pearson’s r = .255, P = .01).
Discussion
The main finding of our study is that the NPS is an independent factor for the prediction of decreasing LVEF for those admitted with STEMI. To the best of our knowledge, the present study is the first to identify the prognostic significance of NPS on LVEF in these patients.
Inflammation plays a role in the pathogenesis of many diseases. 10 Inflammatory mediators play key roles in atherosclerosis which is the underlying pathology of cardiovascular disease. 11 Cardiovascular diseases are responsible for approximately one-third of deaths and the most common form is ischemic heart disease. 12 Despite significant improvements in treatment options in recent years, STEMI is an important cause of cardiovascular morbidity and mortality. 13 Ischemic myocardial injury during STEMI induces an inflammatory response, which is an important determinant of ventricular remodulation. 14 This response is associated with the extent of infarct size, decreased LVEF, malignant arrhythmia, and even death. In addition to being an important determinant of mortality, decreased LVEF is associated with reduced functional capacity and recurrent hospitalizations. 15 In a study of STEMI patients, lesion localization, presence of no-reflow, and prolonged ischemia time were important predictors of LVEF decline. 16 In STEMI patients, CRP levels are associated with infarct size and decreased LVEF in addition to mortality, indicating that ischemia size is indirectly associated with inflammation. 17
The present study showed that neutrophil, lymphocyte, and monocyte cells involved in inflammation have the potential in assessing the prognosis of patients with STEMI. Inflammation associated with atherosclerosis may cause myocardial infarction as a result of destabilization and rupture of the atherosclerotic plaque. 18 It has been shown that the evaluation of these cells, each of which plays a role in a different inflammatory pathway, can also be used in cardiac diseases.19,20
Neutrophils secrete inflammatory mediators that can cause vascular wall degeneration. In contrast, lymphocytes regulate the inflammatory response and thus have an anti-atherosclerotic role. Therefore, the NLR has been proposed as an inflammatory biomarker and potential predictor of risk and prognosis in coronary artery disease.21,22 NLR has been investigated in many cardiac diseases and a significant relationship has been found in many studies.7,19,23–27
Lower lymphocyte counts and higher monocyte counts were associated with adverse cardiovascular endpoints in patients with CAD.20,28 Therefore, LMR has been studied as a systemic inflammatory marker, considering that a composite inflammation ratio including lymphocyte and monocyte levels may provide additional information in cardiovascular risk assessment. Many studies have shown that LMR is also associated with cardiac disease severity and mortality.8,29–34 According to the results of our study, it has been shown that NLR, which is the parameter of the NPS, will be a good marker in evaluating the inflammatory response. Considering that parameters including more than 1 inflammatory pathway give better results, it will be seen that the NPS including LMR gives good results in our study.
It has been shown that increased inflammation and malnutrition can cause atherosclerosis, endothelial damage, and consequent plaque rupture. 35 In addition, hypocholesterolemia, a parameter of the NPS, is associated with an increased risk of death from all causes in patients with CAD. 36 In addition to being a marker of malnutrition, hypoalbuminemia is also a marker of systemic inflammation as a result of the proinflammatory effect of various cytokines that reduce albumin concentration. 37 It has been shown that low serum albumin level, which is also associated with inflammation and a parameter of the NPS, is associated with CAD and AMI. 35
Various modalities are required to predict the prognosis due to possible complications that may be experienced. In conclusion, although some biomarkers and clinical features are associated with poor prognosis in STEMI,38–40 a risk classification that can identify high-risk patients is an important need in clinical practice. To date, it has been demonstrated that various risk scores, including parameters related to inflammation and nutrition, are associated with outcomes in patients with STEMI. However, all these scores are insufficient and have not been widely accepted in clinical practice.41–44
This study demonstrated that the NPS has the potential in predicting patients with reduced LVEF after AMI. Early detection and treatment of malnutrition and inflammation can lead to better patient outcomes.45–47 The NPS, in which these parameters are evaluated, reflects the patient inflammation and nutritional status in detail. To summarize the results, the NPS, which represents systemic inflammation and nutritional status, is an easily accessible and prognostic evaluation method in AMI patients. The NPS evaluated at admission has prognostic value in identifying patients with decreased LVEF after AMI.
Our study has several limitations. First, we did not evaluate other inflammatory and nutritional indices such as the controlling nutritional status score, prognostic nutritional index, systemic immune-inflammation index, and CRP/albumin ratio. Second, factors such as localization of vascular lesions, time to hospital admission, and procedural success were not evaluated in this score. Finally, we did not compare the NPS with the scores used in AMI, such as the GRACE (The Global Registry of Acute Coronary Events) and TIMI (The Thrombolysis in Myocardial Infarction) risk scores.
In conclusion, we found an association between NPS and reduced LVEF in STEMI patients. To the best of our knowledge, the present study is the first to demonstrate this association. NPS, a simple and easily calculated risk score, may help identify high-risk STEMI patients.
Footnotes
Author Contributions
All authors contributed to: (1) substantial contributions to conception and design, or acquisition of data, or analysis and interpretation of data, (2) drafting the article or revising it critically for important intellectual content, and, (3) final approval of the version to be published.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
