Abstract
The Edinburgh Claudication Questionnaire (ECQ) was developed to help identify peripheral arterial disease (PAD) in the general population but has not been validated against diagnostic arterial imaging methods such as Duplex Vascular Ultrasound Scanning (DUS). In the present study, we assessed the accuracy of the ECQ for diagnosis using DUS. As part of a National Institute of Health Research funded project looking at novel diagnostic methods, 250 patients were studied from 15 general practices across North East England from May 2015 and November 2016. Practices identified those with a PAD diagnosis from their registers as well as age- and sex-matched controls. All the ECQs were recorded by a vascular specialist nurse. Duplex vascular ultrasound scanning was used as a reference standard for the diagnosis of occlusive PAD. The ECQ had a sensitivity of 52.5% (95% CI: 42.3%-62.5%), specificity of 87.1% (95% CI: 80.6%-92.0%), positive likelihood ratio of 4.06 (95% CI: 2.57-6.42), and negative likelihood ratio of 0.55 (95% CI: 0.44-0.68) compared with reference standard DUS. The ECQ has relatively poor overall diagnostic test accuracy in isolation. It may be helpful in ruling out PAD or as a supplementary test to improve diagnosis of symptomatic disease in General Practice.
Introduction
Peripheral arterial disease (PAD) due to atherosclerosis is a relatively common condition. In the Edinburgh Artery Study, 1 4.5% of men and women older than 55 years had intermittent claudication (IC), with a further 25% having evidence of asymptomatic disease. In one-third of the asymptomatic group, evidence of a major vessel occlusion was apparent. 1 Further evidence of this high prevalence comes from the PAD awareness, risk, and treatment: New resources for survival (PARTNERS program) study which screened 6979 patients older than 70 years or patients aged 50 to 69 years with a risk factor for vascular disease across 320 primary care practices in the United States. 2 The study found 5.5% had symptomatic PAD and in total 29% of patients had PAD. 2 Thus, a GP practice with 20 000 registered patients would expect to see 40 newly diagnosed patients with PAD each year. 2
Although the focus is often on potential loss of the limb, progression to amputation is in fact relatively uncommon in PAD. In the Edinburgh Study the annual risk of limb loss was <1% to 2%. 3 The risk of associated major cardiovascular events was much greater, with 5% to 10% of patients experiencing 1 such event/year, commonly a myocardial infarction or stroke. 3 For most patients the benefit of an early diagnosis of PAD is that early risk factor management can prevent heart attack and stroke, although monitoring lower limb disease symptoms is a significant concern for the vascular physician. The Edinburgh Claudication Questionnaire (ECQ) was developed from the Edinburgh study to aid in monitoring lower limb symptoms and identifying PAD at an early stage. 3 The aim of the present case control study was to evaluate the effectiveness of the ECQ as a means of assessing PAD in primary care.
Methods
Study Design
This case–control study was part of a broader National Institute of Health Research funded (ISRCTN13301188) project looking at novel diagnostic methods for patients registered as having PAD from general practices in the North East of England and the accuracy of the diagnosis. A total of 250 participants, with 125 having a registered diagnosis of PAD and 125 age- and sex-matched controls were recruited from 15 different general practices between May 2015 and November 2016. Two patient responses were excluded from analysis due to unclear responses on the ECQ.
Duplex vascular ultrasound scanning (DUS) undertaken by vascular scientists from a tertiary care center provided the reference assessment for a true diagnosis of PAD. Duplex vascular ultrasound scanning was considered a good reference standard for PAD diagnosis given its comparable diagnostic rates to angiography 4 and has the added advantage of being a noninvasive investigation that allowed for all measurements to be taken within the primary care setting. Although Ankle Brachial Pressure Index (ABPI) is arguably a more widely available tool in primary care, previous work has highlighted its limitations 5 and it has also been found to be nonsuperior to risk factor questionnaires when used in screening primary care populations. 6 The ECQ was undertaken by a specialist nurse during initial patient assessment. The nurse specialist was not blinded to the GP registry status of the patient.
Edinburgh Claudication Questionnaire
The ECQ was administered face-to-face to determine the presence of IC symptoms. It comprises of 6 questions: Q1 assessing whether patients get any pain or discomfort on walking; Q2 evaluating whether patients get this pain when they are standing or sitting still; Q3 estimating whether patients get pain when they walk uphill or walk in a hurry; Q4 determining whether patients get this pain walking at an ordinary pace on level ground; Q5 assessing whether if patients stand still the pain continues for more than 10 minutes or disappears within 10 minutes; and Q6 requiring patients to mark on a diagram, where they actually get pain or discomfort. 1
The definition of positive claudication requires all of the following responses: yes to Q1; no to Q2; yes to Q3; no (grade 1) to 4; yes (grade 2) to 4; and yes to 5 if pain usually disappears in 10 minutes. If these criteria are fulfilled, based on question 6, a patient with definite claudication is one who indicates pain in the calf, regardless of whether the pain was also marked at other sites. A patient with atypical claudication was one who indicated pain in the thigh or buttock in the absence of any calf pain. Patients were not considered to have claudication if pain was indicated in the hamstrings, feet, shins, or joints or appears to radiate, in the absence of any pain in the calf.
The ECQ symptom data were summarized with a positive grade 1 or positive grade 2 giving PAD+ diagnosis and a negative giving PAD−. Cases where the ECQ could not be reliably determined (eg, ambiguous answers or missing data) then no diagnostic result could be provided for the patient and the result assigned not assessable as a test failure.
Duplex Ultrasound Scan
All patients underwent bilateral lower limb DUS in the primary care setting. Each scan was performed by a trained vascular scientist blinded to the GP registry data and the ECQ result. Each leg was examined in this way from the groin to the ankle. Doppler ultrasound waveforms were obtained and velocities were measured by the operator to gauge the degree of narrowing of the artery and where disease was present. A summary judgment of the patient’s flow status was then made from the flow waveforms. Degree of vessel disease was scored and assessed by the duplex operator as mild, moderate, or severe. The scan protocol is given in Appendix A.
Statistical Analysis
The R programming language 7 was used for data cleaning and analysis. Pearson χ2 test with Yates’ continuity correction was applied to dichotomous data to test the null hypothesis of no association. For continuous data (age, body mass index [BMI]), the Mann-Whitney-Wilcoxon test was used to test the null hypothesis of independence between the 2 groups. Data were represented using parametric statistics of mean and standard deviation. Measures of diagnostic test accuracy (sensitivity, specificity, etc) were calculated using R package meta-analysis of diagnostic accuracy software with 95% CIs estimated by the method of Clopper and Pearson. A P < .05 was considered significant.
Results
The results of those with ECQ reported positive and negative PAD status with main patient demographics are shown in Table 1. Risk factors for cardiovascular disease including diabetes, hypertension, smoking status, and BMI were assessed. Common comorbidities of cardiovascular disease including atrial fibrillation (AF), stroke/transient ischemic attack (TIA), and ischemic heart disease (IHD) were also included (Table 2).
Patient Demographics and Cardiovascular Risk Factors.a
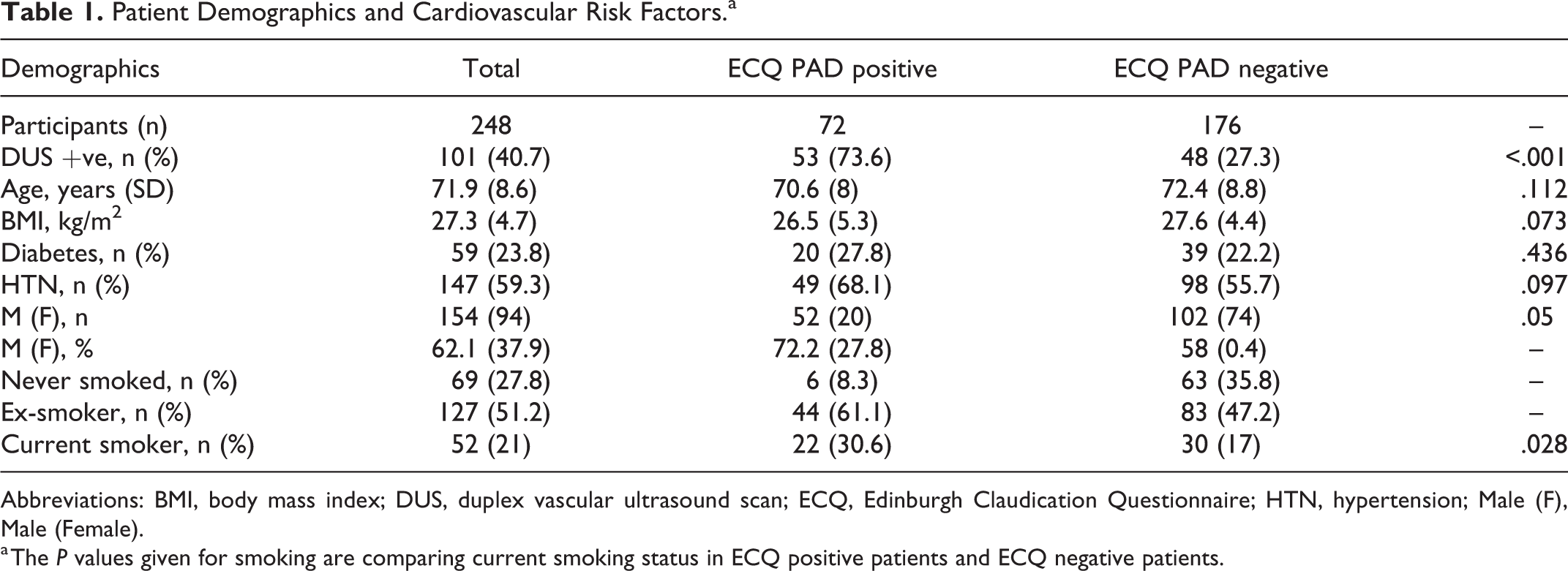
Abbreviations: BMI, body mass index; DUS, duplex vascular ultrasound scan; ECQ, Edinburgh Claudication Questionnaire; HTN, hypertension; Male (F), Male (Female).
a The P values given for smoking are comparing current smoking status in ECQ positive patients and ECQ negative patients.
Participant Comorbidities and ECQ Result.
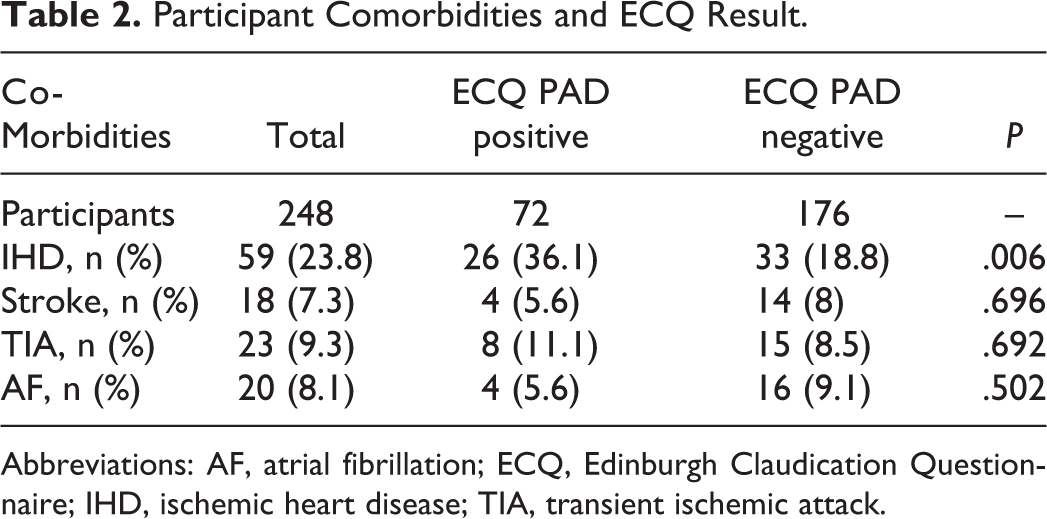
Abbreviations: AF, atrial fibrillation; ECQ, Edinburgh Claudication Questionnaire; IHD, ischemic heart disease; TIA, transient ischemic attack.
Of the total 248 patients, 147 were found not to have significant PAD by DUS (59.3%; Table 2). The remaining 101 with a positive diagnosis of PAD had bilateral disease in 65 cases and unilateral disease in 36 cases. No significant differences were observed between those with a positive ECQ finding versus those with PAD negative scores in terms of BMI (P = .073), age (P = .112), diabetes (P = .436), or hypertension (P = .097). Ischemic heart disease was significantly more prevalent in the ECQ PAD positive cohort than the ECQ PAD negative cohort (36.1% vs 18.8%, P = .006). Stroke (P = .696), TIA (P = .692), and AF (P = .502) were not found to differ significantly between the 2 cohorts.
The ECQ had a sensitivity of 52.5% (95% CI: 42.3%-62.5%) and specificity of 87.1% (95% CI: 80.6%-92.0%). The positive predictive value (PPV) of the ECQ was 73.6% (95% CI: 61.7%-83.0%) and the negative predictive value (NPV) was 72.7% (95% CI: 65.4%-79.0%) compared with reference standard DUS for a sample disease prevalence of 40.7%. The positive likelihood ratio was 4.06 (95% CI: 2.57-6.42), the negative likelihood ratio was 0.55 (95% CI: 0.44-0.68), and the diagnostic odds ratio was 7.44 (95% CI: 4-13.83). There was a total of 48 false negatives from the ECQ of a total 101 patients with confirmed PAD on DUS, with a comparably lower rate of 19 false-positive results from a total of 147 DUS negative patients. Greater proportions of patients reported experiencing claudication on ECQ where more severe disease was present on DUS (Table 3). A total of 71.4% of patients with severe disease on DUS scored grade 1 or grade 2 on ECQ compared with only 25.0% who had mild disease (Table 3). Where patients with diabetes were analyzed separately, the ECQ was 64.3% sensitive (95% CI: 44.1%-81.4%), 93.5% specific (95% CI: 78.6%-99.2%) with a PPV = 90% (95% CI: 66.9%-98.2%) and NPV = 74.4% (95% CI: 57.6%-86.4%) compared with reference standard DUS for a sample disease prevalence of 47.5%. The positive likelihood ratio was 9.96 (95% CI: 2.54-39.16), the negative likelihood ratio was 0.38 (95% CI: 0.23-0.63), and the diagnostic odds ratio was 26.1 (95% CI: 5.12-132.96; Table 4).
Edinburgh Claudication Questionnaire (ECQ) Grade as Compared to Peripheral Arterial Disease (PAD) Severity by Duplex Vascular Ultrasound Scanning (DUS).a,b
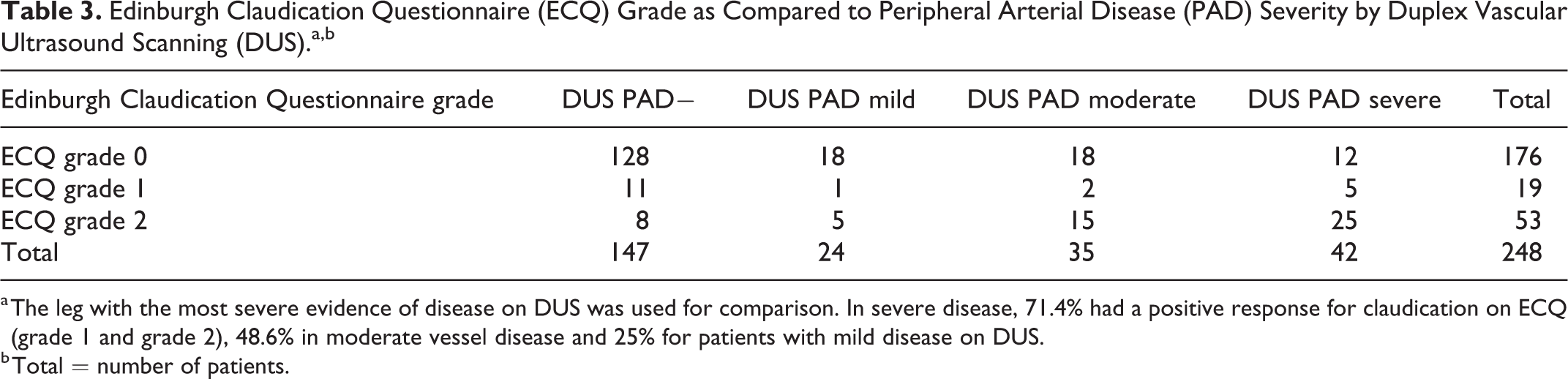
a The leg with the most severe evidence of disease on DUS was used for comparison. In severe disease, 71.4% had a positive response for claudication on ECQ (grade 1 and grade 2), 48.6% in moderate vessel disease and 25% for patients with mild disease on DUS.
b Total = number of patients.
The Edinburgh Claudication Questionnaire (ECQ) Compared With Duplex Vascular Ultrasound Scanning (DUS) for Diabetic Patients With a Positive Result for Peripheral Arterial Disease (PAD).a
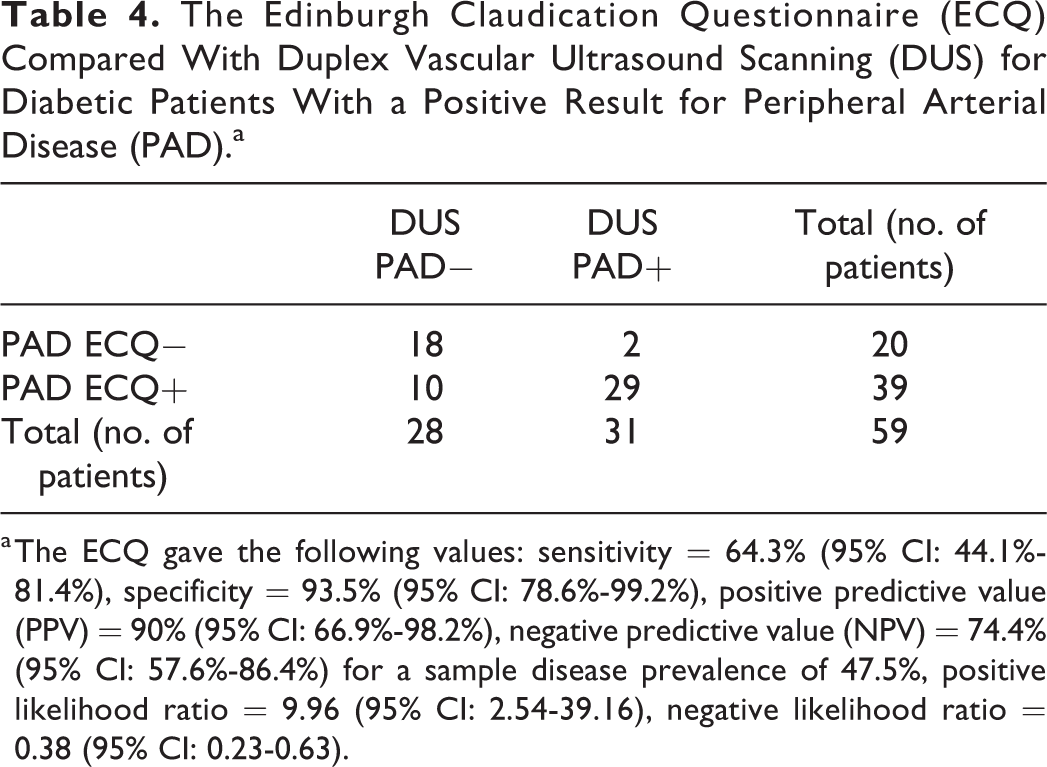
a The ECQ gave the following values: sensitivity = 64.3% (95% CI: 44.1%-81.4%), specificity = 93.5% (95% CI: 78.6%-99.2%), positive predictive value (PPV) = 90% (95% CI: 66.9%-98.2%), negative predictive value (NPV) = 74.4% (95% CI: 57.6%-86.4%) for a sample disease prevalence of 47.5%, positive likelihood ratio = 9.96 (95% CI: 2.54-39.16), negative likelihood ratio = 0.38 (95% CI: 0.23-0.63).
Discussion
The ECQ was a tool designed to aid identification of IC. 8 In the initial study by Leng and Fowkes, the ECQ had a 95% sensitivity and 99.3% specificity compared with diagnosis by general physician. 8 The results of this study suggest that if a primary care physician or practitioner records a positive result from the ECQ, there is a reasonable chance that individual has DUS evidence of PAD. This is reflected by a specificity of 87.1% (95% CI: 80.6%-92.0%), a positive likelihood ratio of 4.06 (95% CI: 2.57-6.42) and PPV of 73.6% (95% CI: 61.7%-83.0%). It is perhaps less clear how they would interpret a negative ECQ result, given the poorer performing sensitivity of 52.5% (95% CI: 42.3%-62.5%).
Others have investigated and compared the ECQ in primary care to different reference investigation standards in different patient populations. 9,10 Bendermacher et al. assessed the ECQ in patients presenting with IC or one or more risk factor in general practice against ABPI. 9 The sensitivity of the ECQ in this setting performed similarly to the present study at 56.2%. 9 Başgöz et al. reported the ECQ was less sensitive, at 31.6%, when assessed against ABPI in a Turkish population. They did however report comparable findings to the present study, where the ECQ was 88.4% specific against ABPI. 10
There are a number of possible explanations for the limitations of the ECQ’s detection of PAD when compared with the ABPI and DUS. This may in part be due to the use of IC symptoms as a determining factor. Many PAD cases may be asymptomatic or symptoms overlap with other causes of exertional leg pain. In a study performed by Poots et al., 41% of patients referred to secondary services with IC had normal ABPI findings. 11 Patients who have IC and patients who have lumbar spinal stenosis share similar demographic profiles as well as a largely similar description of their presenting complaint. Studies by Han et al. and Uesugi et al. demonstrated that PAD is copresent in 4.1% and 6.7% of lumbar spinal stenosis patients, respectively. 12,13 Thus, there is potential diagnostic overshadowing and similar presentations being incorrectly labeled as IC. 12
Another limitation with using IC symptoms as a means of identifying PAD is that PAD is often asymptomatic, even in patients with moderate to severe disease. 14 This is highlighted by epidemiological data that suggest only about 10% to 30% of patients with ABPI values of <0.9 present with the classic features of IC. 15 Within this population, the presence of more severe disease improved the sensitivity of the ECQ to 71.4% in severe disease by DUS from 48.6% in moderate and only 25% in mild disease (Table 3). This may be because some patients do not ambulate sufficiently to provoke symptoms or that lesser degrees of PAD may not be sufficiently hemodynamically significant.
The combined use of objective diagnostic tests and questionnaires in primary care to aid in early detection has been the intuition of many GPs looking to optimally manage at-risk patients. For individuals who present with IC, the ABPI has traditionally been recommended as the primary means of confirming/refuting a probable diagnosis of PAD and has a reported comparable efficacy to angiography. 16 However, despite commonplace use, a number of studies have highlighted both ABPI and CV risk factor scoring systems have low yields in identifying PAD when used in the community setting. 6,17
With regard to the ECQ, there are a number of other potential limitations to its use as a screening tool. The accuracy of the ECQ depends on whether it is self-reported or performed by a trained specialist. 14 Furthermore, there are also reported difficulties when the questionnaire is applied to first generation Black African-Caribbean and South Asian UK migrants. 18 Often the ECQ is used in patients with related cardiovascular diseases such as IHD and stroke to screen for PAD. 16 However, for patients with IHD undergoing cardiac surgery, the ECQ was found to be only 5.6% sensitive at confirming PAD against ABPI values <0.9, 19 although ABPI is perhaps not the best standard of comparison in these patients given that vessel calcification producing falsely high readings that do not reflect poor vessel flow in these patients. 20
Diabetic patients have worse outcomes with PAD. 21 Early detection to allow for effective risk factor management is therefore especially important. 22 There is some evidence that IC as a symptom is less commonly reported by diabetic patients which would arguably make the use of tools like the ECQ less useful. 23 When comparing ABI to ECQ in diabetic patients, Pita- Fernández et al. found that the ECQ was only 50.2% sensitive and 82.6% specific. 24 However, in this analysis, the ECQ was more sensitive in diabetics than for nondiabetic patients with a sensitivity of 64.3% (95% CI: 44.1%-81.4%), specificity 93.5% (95% CI: 78.6%-99.2%), PPV = 90% (95% CI: 66.9%-98.2%), NPV = 74.4% (95% CI: 57.6%-86.4%) for a sample disease prevalence of 47.5%, positive likelihood ratio = 9.96 (95% CI: 2.54-39.16), negative likelihood ratio = 0.38 (95% CI: 0.23-0.63). It is worth noting that these numbers are relatively small with large CIs. Where IC was present, the ECQ, when assessed by Mancero-Romero et al., had an overall clinical accuracy of 75% in diagnosing PAD in diabetic patients when assessed against Doppler imaging. 25
Limitations
Participating practices were taken from a list of those that were part of a research registry collaborative and eligible centers were therefore chosen based on willingness to partake in the study and not at random. All practices were within the North East of England and as such results should be generalized to other regional populations with caution. The vascular nurse specialist was not blinded to the PAD registry status of the patient although the DUS performed after the ECQ was recorded by a vascular scientist who was blinded to the ECQ result.
Conclusion
The ECQ is an easy-to-use, widely applicable and replicable resource which has good specificity (87.1%) but too poor a sensitivity (52.5%) to be relied on as a means of diagnosing PAD in primary care. The positive likelihood ratio of the ECQ in this study was 4.06 (95% CI: 2.57-6.42) and the negative likelihood ratio was 0.55 (95% CI: 0.44-0.68) compared with reference standard DUS. Its place is likely as part of a multifaceted approach to assessing PAD in the community. There remains an identifiable need for improved approaches to diagnosis of PAD in this setting in order to allow effective risk factor management.
Supplemental Material
Supplemental Material, sj-pdf-1-ang-10.1177_0003319720984882 - Reliability of the Edinburgh Claudication Questionnaire for Identifying Symptomatic PAD in General Practice
Supplemental Material, sj-pdf-1-ang-10.1177_0003319720984882 for Reliability of the Edinburgh Claudication Questionnaire for Identifying Symptomatic PAD in General Practice by Luke Boylan, Craig Nesbitt, Lesley Wilson, John Allen, Andrew Sims, Ina Guri, Philip Mawson, Crispian Oates, Gerard Stansby and On Behalf of the NOTEPAD Investigators in Angiology
Footnotes
Authors’ Note
All our authors contributed to (1) substantial contributions to conception and design, or acquisition of data, or analysis and interpretation of data, (2) drafting the article or revising it critically for important intellectual content, and (3) final approval of the version to be published. This report is independent research funded by the National Institute for Health Research (NIHR) (Invention for Innovation, “Innovative photoplethysmography technology for rapid non-invasive assessment of peripheral arterial disease in primary care”, II-C1-0412-20003). The views expressed in this publication are those of the author(s) and not necessarily those of the National Health Service (NHS), the National Institute for Health Research or the Department of Health and Social Care.
Declaration of Conflicting Interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: Between 2014 and 2018, Dr John Allen was the Chief Investigator on an NIHR i4i funded grant (II-C1-0412-20003) to develop a miniaturized version of the multisite photoplethysmography (PPG) vascular technology—specifically for peripheral arterial disease (PAD) detection in a primary care setting. He is a coauthor on published patents in relation for the multisite PPG device.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: NIHR funded (ISRCTN13301188)
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
