Abstract
We hypothesized that the glucose metabolism of prostate cancer is modulated by androgen. We performed in vivo biodistribution and imaging studies of [F-18] fluorodeoxyglucose (FDG) accumulation in androgen-sensitive (CWR-22) and androgen-independent (PC-3) human prostate cancer xenografts implanted in castrated and noncastrated male athymic mice. The growth pattern of the CWR-22 tumor was best approximated by an exponential function (tumor size in mm3 = 14.913 e0.108 × days, R2 = .96, n = 5). The growth pattern of the PC-3 tumor was best approximated by a quadratic function (tumor size in mm3 = 0.3511 × days2 + 49.418 × day −753.33, R2 = .96, n = 3). The FDG accumulation in the CWR-22 tumor implanted in the castrated mice was significantly lower, by an average of 55%, in comparison to that implanted in the noncastrated host (1.27 vs. 2.83, respectively, p < .05). The 3-week maximal standardized uptake value (SUVmax) was 0.99 ± 0.43 (mean ± SD) for CWR-22 and 1.21 ± 0.32 for PC-3, respectively. The 5-week SUVmax was 1.22 ± 0.08 for CWR-22 and 1.35 ± 0.17 for PC-3, respectively. The background muscle SUVmax was 0.53 ± 0.11. Glucose metabolism was higher in the PC-3 tumor than in the CWR-22 tumor at both the 3-week (by 18%) and the 5-week (by 9.6%) micro-PET imaging sessions. Our results support the notions that FDG PET may be useful in the imaging evaluation of response to androgen ablation therapy and in the early prediction of hormone refractoriness in men with metastatic prostate cancer.
Introduction
Prostate cancer is the most common cancer affecting men in the United States [1,2]. As life expectancy increases, so will the incidence of this disease, creating what will become an epidemic male health problem. Imaging evaluation of prostate cancer remains challenging [3,4]. As oncology departs from nonspecific diagnosis and treatment toward patient-specific approach to therapy, accurate knowledge of the presence and extent of metastatic disease becomes even more crucial in order to tailor the treatment plan appropriately [5]. This information, in combination with the physiologic, histologic, antigenic, molecular, and genetic markers of the disease, will provide unprecedented opportunities for the new era of imaging-based cancer diagnosis and therapy [5]. Current imaging tests, including ultrasound, computed tomography (CT), magnetic resonance imaging (MRI), bone scintigraphy, and In-111 capromab pendetide (Prostascint; Cytogen, Princeton, NJ) are not sufficiently accurate to detect local recurrence or metastatic disease in prostate cancer [6–9].
Positron emission tomography (PET) with [F-18] fluorodeoxyglucose (FDG) has become an important diagnostic imaging tool for identification of a diverse group of common and rare malignancies [10,11]. Early animal and preliminary clinical studies of FDG PET in prostate cancer have shown that FDG PET may be useful in the evaluation of advanced androgen-independent disease and in patients with high Gleason scores and serum prostate-specific antigen (PSA) levels, in the detection of active osseous and soft tissue metastases, and in the assessment of response after androgen ablation and treatment with novel chemotherapies [12–36]. We have therefore performed biodistribution, autoradiography, and micro-PET studies of the glucose metabolism in implanted androgen-sensitive and androgen-independent human prostate cancer mouse xenografts.
Materials and Methods
Biodistribution Studies
In vivo biodistribution studies were performed after subcutaneous injection of PC-3 (androgen-independent) and CWR-22 (androgen-dependent) human prostate cancer cells (American Type Culture Collection, Manassas, VA) at a concentration of 10 × 106 cells per 0.2 mL in the shoulders and thighs, respectively, of 4- to 6-week-old, 20–30 g, castrated and noncastrated male athymic mice (BALB/c nu/nu) and allowing enough time for tumors to grow to at least 1000 mm3. Castrated and noncastrated mice served as models for the absence and presence, respectively, of androgens. No testosterone pellets were employed. Tumor volume was calculated using the formula S2 × L/2 where S and L represent the small and large diameters of the lesion. The lesion dimensions were measured using calipers at every 2- to 3-day interval. The CWR-22 human prostate cancer cell line expresses androgen receptors and PSA and is stimulated by dihydroxytestosterone. The androgen-independent PC-3 human prostate cancer cell line was initiated from a bone metastasis of a Grade IV prostatic adenocarcinoma and displays low testosterone-5α reductase activity.
For each group of studies, 25 μCi of FDG was injected into a tail vein in fasted animals. The animals were scarified at 30, 60, and 90 min after FDG administration (n = 3 per time frame per host). Prostate tumors, blood, and organs were then harvested, weighed, and counted for radioactivity. Tissue tracer uptake was calculated as %ID/g normalized to a 20-g mouse and expressed as ratio to the muscular uptake [37]. All animal studies were approved by our Institutional Animal Care and Use Committee, the Biological Safety Committee, and the Radiation Safety Committee. Anesthesia was induced using one intraperitoneal injection of each of ketamine 30mg/kg and xylazine 10 mg/kg with additional one-third of doses as needed. Euthanasia was by intraperitoneal injection of 120 mg/kg pentobarbital followed by either dissection or cervical dislocation.
Autoradiography Studies
Castrated and noncastrated animal tumor model preparations were similar to those described above for the biodistribution studies in two separate athymic male mice. Autoradiography (Bright Instrument, Huntington, UK) was performed in the usual manner 60 min after intravenous administration of 200 μCi FDG into a tail vein [38].
Micro-PET Studies
Castrated and noncastrated animal tumor model preparations were similar to those described above for the autoradiography studies in two separate anesthetized athymic male mice. Dynamic micro-PET imaging (Concorde Microsystems, Knoxville, TN) was performed for 90 min after intravenous administration of 200 μCi FDG into a tail vein. Images were reconstructed using 2D-OSEM algorithm. Regions of interest over the tumors were defined using isoactivity contours set at 80% of the maximal lesion activities and maximal standardized uptake values (SUVmax) were calculated according to the following relationship:
Statistical Analysis
The least-squares error best-fit (i.e., maximum goodness-of-fit R2 where 0 ≤ R2 ≤ 1) was determined using the built-in mathematical functions of Microsoft Excel software (Microsoft, Redmond, WA). Two-tailed Student's t test was used for comparison of means between two groups. A difference with probability of less than .05 was considered statistically significant.
Results
Biodistribution Studies
The growth pattern of androgen-sensitive CWR-22 tumor was best approximated by an exponential function (tumor size in mm3 = 14.913 e0.1086 × days, R2 = .96, n = 5). The growth pattern of androgen-independent PC-3 tumor was best approximated by a quadratic function (tumor size in mm3 = 0.3511 × days2 + 49.418 × day −753.33, R2 = .96, n = 3) (Figure 1). The time to a tumor size of 1000 mm3 was on average 39 days for CWR-22 tumor and 30 days for PC-3 tumor. The androgen-independent tumor had a more rapid growth and a shorter time (by 9 days) to a tumor size of 1000 mm3. The CWR-22 tumor-to-muscle FDG uptake ratios (TMRs) were 1.58 ± 0.38 in the noncastrated and 0.72 ± 0.54 in the castrated hosts, respectively (mean ± SD). The TMRs for PC-3 were 0.93 ± 0.68 (noncastrated) and 1.58 ± 0.52 (castrated). The mean CWR-22 to PC-3 tracer uptake ratio was 1.3 in the noncastrated and 0.5 in the castrated mice. The FDG accumulation in the CWR-22 tumor implanted in the castrated mice was significantly lower, by an average of 55%, in comparison to that implanted in the noncastrated host (1.27 vs. 2.83, respectively, p < .05). No statistically significant interhost change was seen in the level of tracer accumulation in the PC-3 tumors (2.5 vs. 2.17, respectively, p > .5). Figure 2 shows the results of the biodistribution studies.

Growth patterns of (A) androgen-sensitive and (B) androgen-independent human prostate cancer mouse xenografts implanted in noncastrated athymic male mice.

Biodistribution of FDG in the implanted xenografts of androgen-sensitive (CWR-22) and androgen-independent (PC-3) human prostate cancer in noncastrated and castrated nude mice at 30, 60, and 90 min (n =3 for each time frame per host) after tail vein administration of the tracer. The results were similar in the castrated mice except for an approximately 55% decline in the mean level of FDG uptake in the CWR-22 xenograft and no significant effect on the FDG uptake in the PC-3 xenograft. The vertical lines represent one standard deviation from the mean value.
Autoradiography Studies
Autoradiography showed high tracer accumulation in both CWR-22 and PC-3 implanted tumors (Figure 3). No qualitative discernable difference could be observed in the level of tracer localization in the tumors in each of the two animal hosts. The hypermetabolism of the tumor xenografts provided the proof of principle for additional dynamic micro-PET imaging described next.
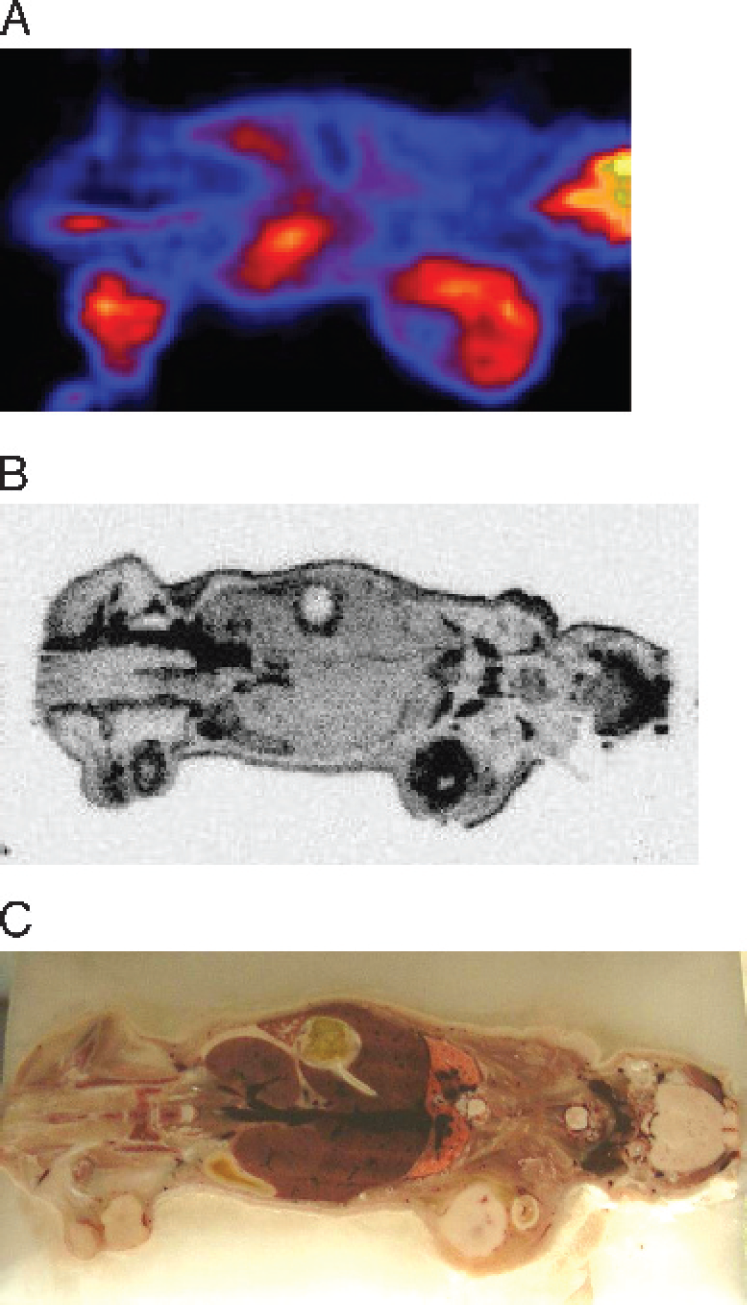
FDG uptake in the human prostate cancer xenografts 3 weeks after implantation in the same noncastrated athymic male mouse. Autoradiograph (A) and micro-PET (B) images show hypermetabolism of the implanted tumors in the right shoulder (androgen-independent PC-3) and in the right hind leg (androgen-sensitive CWR-22) as demonstrated on the corresponding optical image (C). The internal areas of photopenia within the tumors on both the micro-PET and the autoradiography images correspond to the areas of necrosis. The PC-3 tumor is larger than the CWR-22 tumor. In the microPET image, physiologic high FDG localization is noted in the brain and in the kidneys. In all the mouse images, the tail is on the left and the head is on the right.

Dynamic micro-PET time-activity curves of FDG accumulation in the androgen-independent PC-3, (triangles connected by solid line) and the androgen-sensitive, CWR-22, (circles connected by dashed lines) implanted human prostate cancer xenografts in (A) noncastrated and in (B) castrated athymic male mice for 60 min following the tail-vein administration of the tracer. Note that in the noncastrated mouse the FDG uptake in CWR-22 tumor is rising monotonically, whereas in the castrated mouse, the FDG uptake in CWR-22 tumor is lower and plateaus below the level of FDG uptake in the PC-3 tumor.
Micro-PET Studies
The 3-week SUVmax was 0.99 ± 0.43 (mean ± SD) for CWR-22 and 1.21 ± 0.32 for PC-3, respectively. The 5-week SUVmax was 1.22 ± 0.08 for CWR-22 and 1.35 ± 0.17 for PC-3, respectively. The background muscle SUVmax was 0.53 ± 0.11. Glucose metabolism was higher in the androgen-independent PC-3 tumor than in the androgen-sensitive CWR-22 tumor at both the 3-week (by 18%) and the 5-week (by 9.6%) micro-PET imaging sessions (Figures 3 and 4). The earliest photopenic necrotic areas were evident at 3 weeks only in the PC-3 tumors.
Discussion
Imaging evaluation of prostate cancer remains challenging [4]. PET with FDG has become an important diagnostic imaging tool for identification of a diverse group of common and rare malignancies [10,11]. There has been great interest in exploring the specific clinical situations where FDG PET would be useful in such prevalent and important cancers as in prostate cancer. As experience accumulates with FDG PET in prostate cancer, it has become evident that FDG accumulation in high Gleason primary tumors and in metastatic lesions is elevated [12–36]. There may also be additional utility in the evaluation for treatment response and in men with rising serum PSA level despite therapy [12,21,24,29,35]. Detection of metastatic disease at PSA relapse is clinically important because it prompts palliative androgen ablation therapy. Other investigators have noted other essential issues such as the critical need for early detection of androgen-independent clinical state in view of the anticipated future new therapies for delaying or preventing androgen-independent disease [39–43].
In the context of above encouraging preliminary clinical results, we performed a series of animal tumor model experiments to evaluate the glucose metabolism of implanted androgen-sensitive and androgen-independent human prostate cancer xenografts in castrated and noncastrated mice. Both our biodistribution and imaging studies with autoradiography and micro-PET demonstrated a high level of glucose metabolism in the implanted tumors. The more biologically aggressive androgen-independent xenografts showed higher metabolism than that of androgen-sensitive tumors. Additionally, the glucose metabolism of the androgen-independent tumor was relatively insensitive to androgenic presence and stimulation. Conversely, the metabolic activity of the androgen-sensitive tumor declined significantly when the tumors were implanted in the castrated host in comparison to that implanted in the noncastrated host. Therefore, the rise in tumor glucose metabolism after suppression with androgen ablation may be considered as an early indication for the development of androgen refractoriness.
Our study is limited in that animal tumor model studies may not necessarily be translatable to human clinical studies. However, our studies are consistent with the findings of the other animal model and preliminary clinical studies, demonstrating the potential utility of FDG for monitoring metabolic changes of prostate tumor following treatment with androgen deprivation and for assessment of the development of hormone refractory state [44–54]. Given that the tumors were implanted subcutaneously, we neither controlled for nor quantified the vascular supply and blood perfusion (adequacy of tracer delivery) of the implanted tumors. However, because FDG uptake is generally not flow-dependent and in order to reduce the effect of this potential source of variability, all tumors were implanted using the same technique at similar anatomic sites in the animals and all tumors were assessed at a similar size. Although castrated and noncastrated mice served as models for absence and presence, respectively, of androgens, we did not measure or quantify the serum androgen levels. Future studies may be planned to correlate the serum androgen levels to the amount of FDG uptake in the tumor.
In summary, the findings of our animal model study support the dependence of androgen-sensitive tumor glucose metabolism on the androgen presence and stimulation. Our results are in line with the findings of the other investigators in that FDG PET may be clinically useful in the imaging evaluation of response to androgen ablation therapy and in the early prediction of hormone refractory state in men with metastatic prostate cancer.
Footnotes
Acknowledgments
This work was supported in part by a grant from The Wright Foundation.

