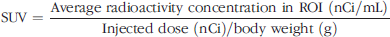Abstract
Dubin-Johnson syndrome (DJS) is caused by a deficiency of the human canalicular multispecific organic anion transporter (cMOAT). A new lipophilic copper-64 complex of 1,4,7-tris(carboxymethyl)-10-(tetradecyl)-1,4,7,10-tetraazadodecane (5) was prepared and evaluated for potential as a diagnostic tool for DJS. The prepared ligand was labeled with 64Cu citrate in high radiochemical purity. In vivo uptake and clearance of the complex was determined through biodistribution studies using normal Sprague-Dawley rats and mutant cMOAT-deficient (TR−) rats. In normal rats, the radioactive copper complex was cleared quickly from the body exclusively through the hepatic pathway. The 64Cu complex was taken up rapidly by the liver and quickly excreted into the small intestine and then the upper large intestine, whereas < 1% ID/organ was found in the kidney at all time points post injection. Whereas activity was accumulated continuously in the liver of TR− rats, it was not excreted into the small intestine. MicroPET studies of normal and TR rats were consistent with biodistribution data and showed dramatically different images. This study strongly suggests that cMOAT is involved in excretion of 64Cu-5. The significant difference between the biodistribution data and microPET images of the normal and TR− rats demonstrates that this new 64Cu complex may allow noninvasive diagnosis of DJS in humans.
Keywords
Introduction
Dubin-Johnson syndrome (DJS) is a rare autosomal recessive disorder characterized by a defect in the transfer of endogenous and exogenous anionic conjugates from hepatocytes into the bile, which causes conjugated hyperbilirubinemia and deposition of black pigments in the liver [1]. A mutation in the human canalicular multispecific organic anion transporter gene (cMOAT) was found to cause DJS [2–5]. cMOAT, also known as multidrug resistance protein 2 (MRP2) or ATP-binding cassette (ABC) transporter ABCC2, is expressed predominantly in the liver and only weakly in the kidneys, and mediates the transport of various amphipathic anions [6]. Previously, DJS was diagnosed from increased levels of coproporphyrine isomer in the urine, a delayed rise in the sulfobromophthalein sodium test, and liver biopsy to see pigments [7]. Because of the complexity and invasiveness of these diagnostic methods, hepatobiliary scintigraphy [8,9] and CT imaging [7] were recently tried as a new diagnostic approach to DJS. In hepatobiliary scintigraphy, 99mTc-labeled iminodiacetic acid derivatives such as 99mTc-HIDA [10], 99mTc-disofenin [9], and 99mTc-mebrofenin [8] showed swift blood clearance and liver uptake followed by retardation of excretion into the bile and gallbladder.
An animal model of human DJS is the transporter-deficient (TR−) Wistar rat [11]. The TR− rat has a defective cMOAT and shows the typical phenotypic and diagnostic features of DJS such as hyperbilirubinemia and delayed hepatic clearance of intravenously injected sulfobromophthalein [12,13].
The hepatobiliary excretion of a compound depends on several physicochemical characteristics [14,15]: an ideal molecular weight of 300-1000; the presence of a strong anionic polar group ionized at plasma pH; the presence of a nonpolar group to decrease renal excretion [16]; lipophilic character; and binding to plasma proteins, which prevents filtering by the glomeruli of the kidney and promotes transfer into the hepatocyte.
We prepared a Cu(II) complex of a DO3A derivative that fulfills the above physicochemical properties and can be labeled with copper-64 for imaging by positron emission tomography (PET). Copper-64 (t1/2 = 12.7 hr; β+, 19%; β−, 39%) has favorable properties as a radionuclide for use in both PET imaging and targeted radiotherapy [17–19]. DO3A derivatives are known to form stable metal complexes with many metal ions and are widely used as bifunctional chelators [20] and chelating agents for gadolinium magnetic resonance imaging contrast agents [21,22].
Herein, we report the synthesis of a DO3A derivative that has a tetradecyl group; its radiolabeling with 64Cu and log p determination, and the in vivo behavior of this 64Cu complex as determined by biodistribution and microPET imaging.
Materials and Methods
General
1,4,7,10-Tetraazacyclododecane (cyclen) was purchased from Strem Chemicals (Newburyport, MA) and all other solvents and reagents were obtained from Aldrich (St. Louis, MO) and used as received without further purification. Water was distilled and then deionized (18 MΩ/cm2) by passing through a Milli-Q water filtration system (Millipore Corp., Bedford, MA). 1H and 13C NMR spectra were measured using a Varian Gemini 300 instrument, and chemical shifts are reported in parts per million on the δ scale relative to TMS or solvent peak. Proton chemical shifts are annotated as follows: ppm [multiplicity, coupling constant (Hz), integral]. Elemental microanalyses were performed by Galbraith Laboratories (Knoxville, TN). Mass spectra were obtained from Washington University Mass Spectrometry Resource.
Copper-64 was prepared on the Washington University School of Medicine CS-15 cyclotron by a 64Ni(p,n)64Cu nuclear reaction at a specific activity range of 50–200 mCi/μg as previously described [18]. EM Science Silica Gel 60 F254 thin-layer chromatography (TLC) plates were purchased from Fisher Scientific (Pittsburgh, PA). Radio-TLC was accomplished using a Bioscan 200 imaging scanner (Bioscan, Inc., Washington, DC). Radioactivity was counted with a Beckman Gamma 8000 counter containing a NaI crystal (Beckman Instruments, Inc., Irvine, CA).
Female Sprague-Dawley rats were purchased from Charles River Laboratories (Wilmington, MA) and TR− Wistar rats were obtained from a colony maintained at Washington University. Blood was obtained by cardiac puncture from healthy Sprague-Dawley rats. The rat serum was obtained by spinning down blood using 13 mL Vacutainer® (SST gel and clot activation, Becton Dickinson, Franklin Lakes, NJ). Serum was stored on ice before use. All animal experiments were performed in compliance with the Washington University Animal Care Committee guidelines.
Ligand Synthesis
1-(Benzyloxycarbonyl)-1,4,7,10-tetraazacyclododecane·3HCl (
Synthesis of 1,4,7-tris(tert-Butoxycarbonylmethyl)-10-(Benzyloxycarbonyl)-1,4,7,10-Tetraazacyclododecane (2)
To a solution of 1-(benzyloxycarbonyl)-1,4,7,10-tetraazacyclododecane·3HCl (865 mg, 2.09 mmol) in 35 mL of acetonitrile were added N,N-diisopropylethylamine (2670 μL, 15.33 mmol) and tert-butyl bromoacetate (935 μL, 6.33 mmol). The reaction mixture was slowly heated to 80°C and allowed to stir for 2 days. After evaporation of solvents in vacuo, the residue was dissolved in 20 mL of methylene chloride and washed with 30 mL of H2O. The aqueous layer was further extracted by 2 × 20 mL of methylene chloride. The combined organic layer was dried over MgSO4 and rotary evaporated. The residue was purified via flash column chromatography on alumina, eluting with ethyl acetate/hexane (1:2) to afford a clear oil (999 mg, 74%): 1H NMR (CDCl3) δ 1.37 (s, 9H), 1.38 (s, 9H), 1.39 (s, 9H), 2.68 (br s, 4H), 2.72 (br s, 4H), 2.89-2.93 (m, 4H), 3.18 (s, 2H), 3.26 (s, 4H), 3.45-3.47 (m, 4H), 5.04 (s, 2H), 7.20-7.30 (m, 5H); 13C NMR (CDCl3) δ 28.19 & 28.24 (CCH3), 47.0, 47.5, 51.8, 52.0, 52.5, 52.7, 53.6 & 53.7 (NCH2CO2), 54.9 (NCH2CO2), 57.2, 57.7, 66.7 (OCH2Ph), 80.75 & 80.81 (OCCH3), 127.6, 127.8, 128.4, 137.1 (Ph ring C), 156.3 (NCO2), 170.8 & 170.9 (NCH2CO2). Anal. Calcd for C26H50N4O6·2H2O: C, 59.63; H, 8.83; N, 8.18. Found: C, 59.45; H, 8.47; N, 8.27. HRMS (ESI): m/z 649.4147 ([M + H]+, C34H57N4O8, calcd 649.4176).
Synthesis of 1,4,7-tris(tert-Butoxycarbonylmethyl)-1,4,7,10-Tetraazacyclododecane (3) To a solution of 1,4,7-tris(tert-butoxycarbonylmethyl)-10-(benzyloxycarbonyl)-1,4,7,10-tetraazacyclododecane (999 mg, 1.54 mmol) in 35 mL of methanol was added 10% Pd on activated carbon (193 mg). The reaction was stirred under hydrogen gas at room temperature for 18 hr. The Pd catalyst was filtered off using a Celite pad and the filtrate was concentrated under reduced pressure. The residue was redissolved in 30 mL of methylene chloride and then reduced to complete dryness under vacuum (739 mg, 93%): 1H NMR (CDCl3) δ 1.398 (s, 9H), 1.404 (s, 18H), 2.57 (br s, 4H), 2.72 (s, 8H), 2.79 (br m, 4H), 3.27 (s, 2H), 3.28 (s, 4H); 13C NMR (CDCl3) δ 28.3 and 28.4 (OCCH3), 47.7 (CH2CH2NH), 51.0, 52.0, 52.4, 57.4 (NCH2CO2), 81.0 (OCCH3), 171.1 and 171.4 (CO2). HRMS (ESI): m/z 515.3798 ([M + H]+, C26H51N4O6, calcd 515.3809).
Synthesis of 1,4,7-tris(tert-Butoxycarbonylmethyl)-10-(Tetradecyl)-1,4,7,10-Tetraazacyclododecane (4)
To a solution of 1,4,7-tris(tert-butoxycarbonylmethyl)-1,4,7,10-tetraazacyclododecane (152 mg, 0.30 mmol) in 35 mL of acetonitrile were added N,N-diisopropylethylamine (127 μL, 0.59 mmol) and 1-bromotetradecane (79 μL, 0.32 mmol). The reaction mixture was slowly heated to 70°C and allowed to stir for 64 hr. After evaporation of solvents in vacuo, the residue was dissolved in 30 mL of methylene chloride and the layer was separated between methylene chloride and 30 mL of water. The aqueous layer was further extracted with 2 × 20 mL of methylene chloride. The combined organic layer was dried over MgSO4 and concentrated to dryness. The crude product was chromatographed using alumina to wash out impurities first by methylene chloride, then to elute the product by MeOH:methylene chloride (1:20) (101 mg, 48%): 1H NMR (CDCl3) δ 0.88 (t, J = 6.6 Hz, 3H), 1.25 (s, 24H), 1.45 (s, 27H), 2.36 (m, 2H), 2.59 (t, J = 5.6 Hz, 4H), 2.82 (s, 12H), 3.28 (s, 4H), 3.31 (s, 2H); 13C NMR (CDCl3) δ 14.3 (CH2CH2CH3), 22.9 (CH2CH2CH3), 27.4, 27.9, 28.2, 28.5 (OCCH3), 29.6, 29.9, 32.1, 52.08 and 52.28 and 52.34 and 52.58 (cyclen CH2), 56.50, 56.57, 56.9 (NCH2CO2), 80.8 (OCCH3), 171.4 (CO2). HRMS (ESI): m/z 711.5960 ([M + H]+, C40H79N4O6, calcd 711.6000).
Synthesis of 1,4,7-tris(Carboxymethyl)-10-(Tetradecyl)-1,4,7,10-Tetraazacyclododecane Hydrochloride Salt (5·4HCl)
A 3 M HCl solution (20 mL) was added to 22 mg (0.031 mmol) of 1,4,7-tris(tert-butoxycarbonylmethyl)-10-(tetradecyl)-1,4,7,10-tetraazacyclododecane. The flask was slowly heated to 110°C and allowed to stir for 14 hr. The clear solution was concentrated to dryness overnight using Savant SpeedVac to give white solid
Synthesis of Cu(II) Complex of 1,4,7-tris(Carboxymethyl)-10-(Tetradecyl)-1,4,7,10-Tetraazacyclododecane (Cu-5)
To an aqueous solution (15 mL) of 1,4,7-tris(carboxymethyl)-10-(tetradecyl)-1,4,7,10-tetraazacyclododecane hydrochloride salt (
Radiochemistry
Copper-64 chloride was converted to 64Cu citrate by stirring with 100 μL of 0.1 M ammonium citrate, pH 6.5. Copper-64 citrate (1.2-12.5 mCi) was added to a ligand solution containing less than 0.3 mg of ligand in 100 μL of the same buffer. The reaction mixture was incubated at 40°C for 1 hr. The radiochemical purity was determined by radio-TLC using silica gel plate (Rf = 0.70, H2O:MeOH = 1:2).
Log p Measurement
The partition coefficients were determined by adding 15 μL of 64Cu-labeled complex (21 μCi) to a solution containing 1.5 mL of octanol and 1.5 mL of buffer solution (0.01 M ammonium acetate, pH 7.4), which were obtained from saturated octanol buffer solutions. The resulting solutions were then vortexed for 1 min and centrifuged for 5 min at 5000 rpm. A 1-mL aliquot of the octanol layer was removed, back extracted with 1 mL of buffer, vortexed, and centrifuged for 2 min at 5000 rpm. Aliquots (150 μL) of octanol and buffer were removed, weighed, and counted (first partition coefficient). A 700-μL aliquot of the previous octanol layer was removed and back extracted again with 700 μL of buffer, vortexed, and centrifuged as before. Aliquots (150 μL) of octanol and buffer were removed, weighed, and counted (second partition coefficient). A 400-μL aliquot of octanol layer of second back extraction was removed and back extracted one more time with 400 μL of buffer, vortexed, and centrifuged as before. Aliquots (150 μL) of octanol and buffer were removed, weighed, and counted (third partition coefficient). The partition coefficient was calculated as a ratio of counts in the octanol fraction of unit mass to counts in the buffer fraction of unit mass per back extraction. The average log p value of the three back extractions is reported.
Protein Binding Study
The fraction of the labeled compound to proteins was determined by ultrafiltration (Amicon Centrifugal Filter Devices, Centricon YM-10, 10,000 MWCO, Beverley, MA). To 7 mL of the rat serum, 5 μL of the labeled compound 64Cu-
Biodistribution Study
The radioactive complex was diluted with saline for injection. Mature female Sprague-Dawley rats (n = 4 per time point) weighing 180–200 g were anesthetized with 1-2% isofluorane and injected with 15 μCi of activity in a volume of 140 μL via the tail vein. At selected time points post injection (5, 15, 30 min, 1, 2, and 4 hr), rats were sacrificed. For comparison, the labeled compound was injected into mature TR− Wistar rats (n = 4 per time point) weighing 300–400 g after dilution with saline (15 μCi in 150 μL each). At 30 min, 2 hr, and 4 hr post injection, rats were sacrificed. Organs of interest were removed, weighed, and counted. The percent injected dose per gram (% ID/g) and percent injected dose per organ (% ID/organ) were calculated by comparison to weighed, counted standard solutions.
MicroPET Imaging Study
The microPET imaging studies were carried out using the microPET-R4 rodent scanner (Concorde Microsystems, Knoxville, TN) [25]. Images were reconstructed using Fourier rebinning followed by 2-D filtered back projection [26,27]. A normal mature Sprague-Dawley rat weighing 139 g and a mature TR− Wistar rat weighing 199 g were anesthetized with 1-2% vaporized isofluorane and injected with 1.4 mCi of activity in 140–145 μL saline via the tail vein. At specific time points, the rats were reanesthetized and then immobilized in a supine position on custom-built support beds with attached anesthetic gas nose cones for data collection. The regions of interest (ROIs) were quantified by viewing these areas in the selected tissues and averaging the activity concentration over the contained voxels. ROI analysis software consisted of two programs: Analyze AVW 3.0 (Biomedical Imaging Resource, Mayo Foundation, Rochester, MN) and a viewing application program developed in-house by R. Laforest (Washington University School of Medicine, St. Louis, MO) in International Data Language (Research Scientific, Boulder, CO).
Dynamic Imaging. The rats were first anesthetized with 1-2% isofluorane and placed near the center of the field of view (CFOV) of the microPET scanner where the highest image resolution and sensitivity are obtained. Dynamic imaging was performed for 30 min beginning at the injection of 1.4 mCi of activity in 140–145 μL saline into a normal mature Sprague-Dawley rat weighing 139 g and a mature TR− Wistar rat weighing 199 g via a microcatheter (Harvard Apparatus, Holliston, MA) placed in the external jugular vein. The image frame duration was 300 sec per frame × 6 frames.
Static Imaging. For the baseline PET scans, rats were first anesthetized with 1-2% isofluorane and placed in the supine position in a custom-designed holder. The entire holder was placed near the CFOV of the microPET scanner. Ten-minute static scans were obtained 1, 2, 3, and 4 hr post injection. The methods of data collection and the reconstruction for microPET imaging have been described previously [28,29]. Briefly, all raw data were first sorted into 3-D sinograms, followed by Fourier rebinning and 2-D filtered backprojection reconstruction using ramp filter cutoff at the Nyquist frequency. An ROI was placed on organs in the transaxial microPET images that include the entire organ volume. The average radioactivity concentration within an organ was obtained from the average pixel values within the multiple ROI volume. From the dynamic study, time-activity curves were also calculated from the standard uptake value (SUV) of an organ, which was defined as the average radioactivity concentration divided by the total injected activity that is divided by body weight with the following formula:
In these microPET scans, the attenuation corrections were not applied because the accuracy of the measured attenuation correction was poor with this scanner and the amount of attenuation from a rat body was relatively small; instead, the attenuation correction factors were incorporated into the system calibration. With the image reconstruction algorithm and filter described above, the resolution of this microPET system was between 2.0 and 3.0 mm full width at half maximum in each of the three dimensions within a 5-cm imaging field of view around the central axis of the tomograph.
Results
Synthesis, Radiolabeling, and Lipophilicity
Mono-protected cyclen compound, mono-Cbz-cyclen (
The prepared ligand
The log p (octanol/water partition coefficient) of the radiolabeled compound was determined. Average log p values of three measurements of three consecutive back extractions was 1.18 ± 0.16.
Protein Binding Study
The bound and unbound fractions of the labeled compound were separated from serum samples by ultrafiltration. All values have been corrected for nonspecific binding of the lipophilic tracer to the filter material using binding data independently measured for 64Cu-
Biodistribution Studies
In vivo uptake and clearance of the complex was determined through biodistribution studies using normal Sprague-Dawley rats and cMOAT-deficient (TR−) rats. The TR− rat model is a rodent model of DJS, which is defective in cMOAT. Biodistribution results are summarized in Tables 1 and 2. Due to limited availability of TR− rats in our facility, there were fewer biodistribution time points for TR− rats than for normal rats. More direct comparisons in clearance organs of 64Cu-

Synthesis of ligand
Biodistribution of 64Cu-5 in Mature Female Sprague-Dawley Rats

Data are expressed as the %ID/g ± SD with four animals per data point.
The 64Cu-complex was taken up rapidly by the liver (49% ID/organ at 5 min), and quickly excreted through the small intestine (34% ID/organ at 30 min) and then the upper large intestine (91% ID/organ at 2 hr), whereas < 1% ID/organ of activity was found in the kidney at all time points post injection. The ratio of the percentage of injected dose per gram (% ID/g) of liver versus kidney is 5.9, 7.1, and 6.3 at 5, 15, and 30 min, respectively. Blood retention was gradually decreased from 1.6% ID/g at 5 min to 0.09% ID/g by 2 hr. Because most of the activity was already excreted into the upper large intestine by 2 hr, there was very little change in the % ID/g in organs except liver between the 2- and 4-hr time points. The liver activity at 2 hr (29% ID/g) was further excreted by 4 hr (1.0% ID/g). The uptake (% ID/g) in the kidney was actually lower than that of blood by 15 min, and from 30 min to 4 hr post injection, very constant uptakes were measured (0.74-0.87% ID/g).
Biodistribution of 64Cu-5 in Mature TR− Wistar Rats
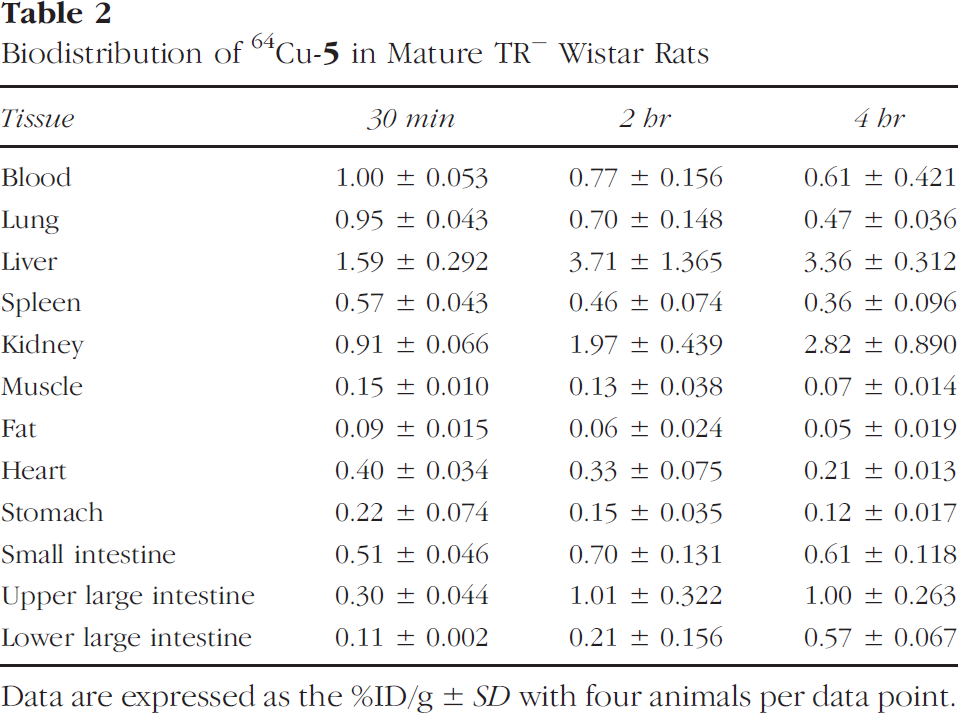
Data are expressed as the %ID/g ± SD with four animals per data point.
The TR− rats showed a completely different biodistribution pattern. The activity was accumulated continuously in the liver (22% ID/organ at 30 min and 42% ID/organ at 2 hr), but was not excreted into the small intestine (1.8% ID/organ at 30 min and 2.4% ID/organ at 2 hr). Although renal excretion is known to serve as the alternative route of clearance for all 99mTc-IDA complexes in cases of reduced hepatic function [30], the uptake of activity in the kidney was very slowly increased and was only marginally higher than that of normal rats (1.3% ID/organ at 30 min and 3.8% ID/organ at 4 hr). Liver uptake nearly doubled between 30 min and 2 hr (21.6% ID/organ at 30 min and 41.7% ID/organ at 2 hr); however, only 8% of the activity of 2 hr was further accumulated in liver by 4 hr post injection, whereas only 3.8% more of the activity was excreted to the gastrointestinal track. Compared to 91% ID/organ of 64Cu-
MicroPET Imaging Studies
MicroPET was also used to follow in vivo uptake and clearance of activity. Dynamic imaging was performed for the first 30 min after 64Cu-
MicroPET images of normal and TR− rats, shown in Figure 4, showed great differences in clearance between the two rats at all time points post injection. 3-D rotating images were captured as a projection image at the angles that best represented the activity uptake in organs. Sectional images could not show all liver, kidney, and intestine in the same plane.

Biodistribution of 64Cu-
Discussion
We synthesized a DO3A-based cyclen compound that has a long alkyl chain. The ligand 1,4,7-tris(carboxymethyl)-10-(tetradecyl)-1,4,7,10-tetraazacyclododecane (
The log p measurement using octanol/water partition coefficient showed that 64Cu-labeled complex was lipophilic. Previous studies [33] in our group have shown that the lipophilicity of Cu-cyclen complexes are influenced by the overall charge of the copper complex, the symmetry within the complex, and the lipophilicity of substituents on N atoms of cyclen ring. The radiolabeled compound, 64Cu-

Biodistribution of 64Cu-
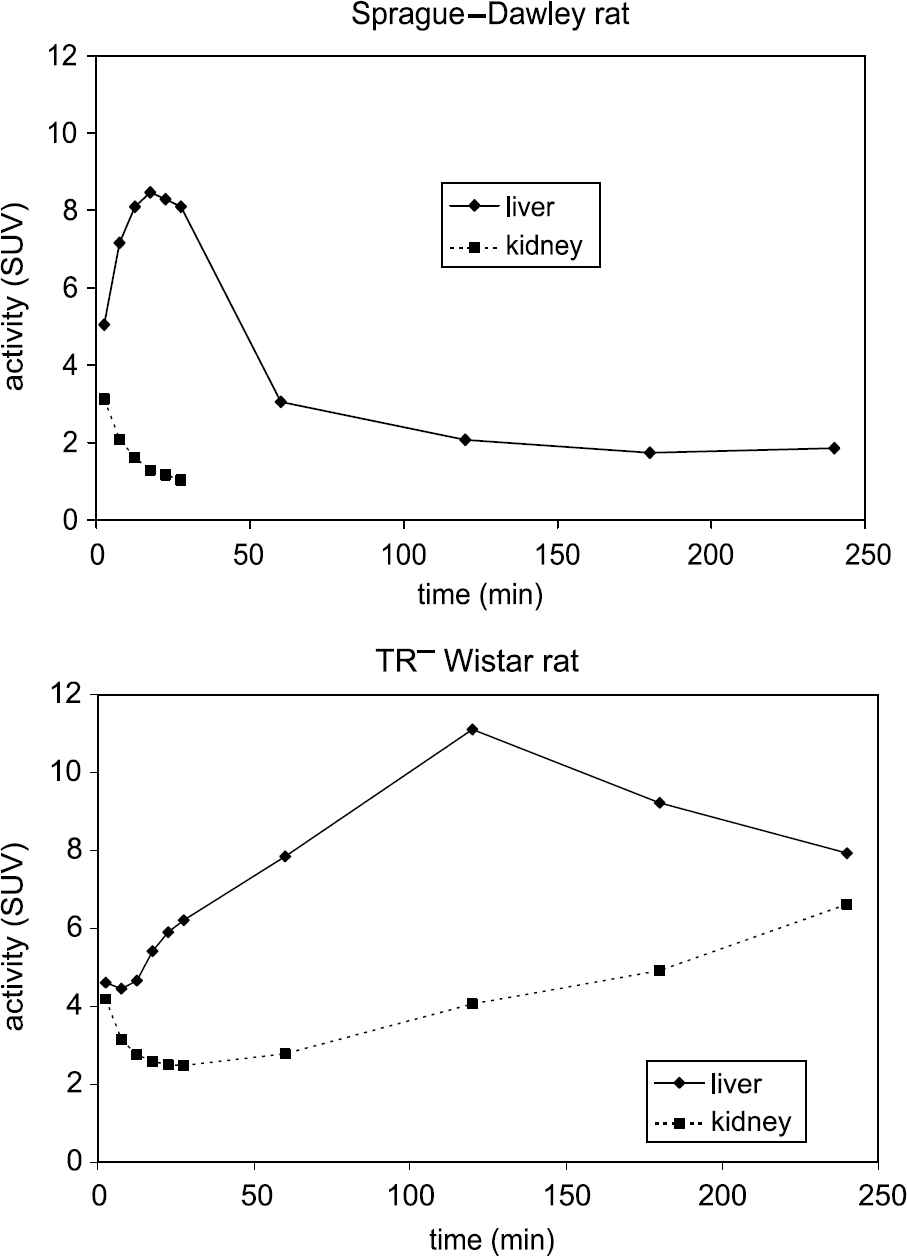
Time - activity curves for liver and kidney of one Sprague-Dawley rat and one TR− Wistar rat (n = 1 each).
Hepatocellular specific compounds have several common physicochemical characteristics such as range of molecular weight, the presence of a strong anionic group ionized at plasma pH, the presence of a nonpolar group, lipophilic character, and binding to plasma proteins [14,15]. The Cu(II) complex of
Biodistribution studies showed that the radioactive copper complex was cleared quickly from body exclusively through the hepatic pathway in normal Sprague-Dawley rats (Figure 1). Kidney uptake of this Cu(II) complex was lower than that of clinically used hepatobiliary specificity reagents such as disofenin (DISIDA) [35], even though blood pool and liver clearance of Cu-

Comparison of microPET images of a Sprague-Dawley rat (top) and a TR− Wistar rat at 5 min, 25 min, 1 hr, and 3 hr post injection of 64Cu-
No significant uptake was found outside of the clearance organs. Even though 64Cu-
The biodistribution data of 64Cu-
MicroPET imaging analysis data of normal and TR− rats were consistent with biodistribution data and showed a more detailed clearance scheme for liver and kidney (Figure 3). Especially at early time points, TAC of dynamic scan images provided very precise uptake and clearance information of liver and kidney because the bed position of the rat remained at the exact same position, and therefore the exact same ROI was applied.
In normal rats, liver uptake of 64Cu-
MicroPET imaging of a single TR− rat and subsequent data analysis provided detailed information on in vivo uptake and clearance (Figure 3), whereas a biodistribution study would have required a much larger number of these transgenic rats. The microPET study showed that 64Cu-
In the micro PET images of the normal Sprague-Dawley rat (Figure 4), most of the activity was accumulated in the liver at 7.5 min then excreted rapidly into the intestines. Kidney was not easily identified in microPET images at all time points due to low uptake. However, the bladder was clearly visualized at 7.5 min, which suggests that even though the kidney uptake was low, some activity was still cleared through the renal tubules, which is presumably due to the presence of a small amount of cMOAT in kidney [6].
In the TR− rat, however, liver and kidney were both visualized at all time points (Figure 4). However, the images of liver and kidney at 7.5 and 27.5 min were not clear due to the high retention of activity in blood. The intestines were barely visualized due to severe retardation of liver clearance into the small intestine. At 1 hr, most of the activity remained in the liver and kidney in the TR− rat, which was very different from the microPET image of the normal rat, in which only the intestines were visualized. At 3 hr post injection, the liver and kidney in the TR− rat was more easily visualized than in the earlier images, which was likely due to decreased background activity. Due to the limited availability of the TR− rats, we could not carry out multiple microPET studies.
It is known that the bile flow rate in TR− rat liver is 50% that of normal rats [43]. However, the secretion of a nonsubstrate of cMOAT such as bile salt was normal in TR− rat. Also, some organic anions showed relatively normal excretion patterns in TR− rats. For example, the hepatobiliary secretion of bilirubin ditaurate in TR− rats was entirely normal [43]. Although the bile flow of TR− rats was reduced to 50% of normal, the biliary concentration of bilirubin ditaurate in TR− rats was twice that in normal rats. Therefore, the substrate specificity of cMOAT can be deduced in TR− rats from compounds that are not secreted or poorly secreted into the bile [12,43]. The data from biodistribution and microPET studies presented here show the severe retardation of secretion of 64Cu-
Copper-64 is one of a limited number of positron-emitting radionuclides that can be produced on both a nuclear reactor and a medical cyclotron. A method was developed at Washington University School of Medicine to produce large quantities of 64Cu with low-energy biomedical cyclotrons [18]. Copper-64 offers the advantages of a positron-emitting tracer for imaging and has potential for use as an internally administered therapeutic radionuclide. The potential wide availability of 64Cu, both from reactors and biomedical cyclotrons, suggests that 64Cu is a radionuclide with great potential for the clinical use. The human studies of 64Cu-labeled anticolorectal carcinoma monoclonal antibody (MAb 1A3) [44,45] and 64Cu-TETA-OC (where TETA is 1,4,8,11-tetraazacyclotetradecane-N,N′,N¶ime;,N′¶ime;-tetraacetic acid and OC is
Owing to its availability from a generator, Cu-62 (β+ 97.6%) might be used for PET imaging in centers relatively remote from a cyclotron unit [47]. However, in order to use 62Cu instead of 64Cu in case of Cu-
Conclusion
In conclusion, we synthesized a novel DO3A derivative having a long alkyl chain and labeled it with 64Cu. The log p measurement using octanol/water showed that the labeled Cu(II) complex was lipophilic, whereas the complex was assumed to carry a −1 charge. In normal rats, 64Cu-
Footnotes
Acknowledgments
This work was supported by the United States Department of Energy (DEFG02-087ER-60512). We thank the Research Resource in Radionuclide Research (R24 CA86307) for the production of 64Cu. We also thank Lori A. Strong, Nicole M. Fettig, John A. Engelbach, Lynne A. Jones, and Terry L. Sharp for their technical support in the biodistribution study and microPET imaging.