Abstract
In vivo monitoring of cells labeled with paramagnetic iron oxide particles by magnetic resonance imaging (MRI) is complicated by intrinsic contrast of blood vessels. Distinction between T2* effects caused by blood vessels from those caused by labeled cells was so far only possible after carefully following the location of hypointense regions through subsequent slices of T2*-weighted 3-D MRI datasets, a procedure that is time consuming and not always reliable in the case of smaller blood vessels. Here, we demonstrate that the modification of the inhalation gas mixture from the routinely used composition 35% O2 and 65% N2O to a mixture containing 95% O2 and 5% CO2 results in a contrast suppression of most small blood vessels and reduces the intrinsic T2* effect of large vessels dramatically in an animal model. This change in protocol of physiological conditions was well tolerated by all studied animals, even over prolonged experimental times. The changed inhalation gas mixture thus provides a more reliable identification method for small clusters of iron oxide labeled cells in vivo.
Introduction
Three-dimensional (3-D) magnetic resonance imaging (MRI) can achieve microscopic resolution to monitor dynamic cellular processes. In order to discriminate implanted cells from host tissue, they are labeled in vitro, before transplantation. Cells can be effectively loaded with iron oxide nanoparticles, thus generating a pronounced contrast in T2*-weighted MR images. This approach is by now well established and has already led to a variety of applications in diverse medical but also in developmental biological areas. In regenerative medicine, stem cells have been labeled with iron oxide particles (USPIO), implanted in various animal models, and their migration to the side of injury has been monitored in vivo by MRI [1–6]. It was shown that labeled stem cells implanted into the contralateral hemisphere migrate to the side of an induced stroke in animal models [3,4]. This observation was based on decreased T2 and T2* relaxation times. However, other effects can also contribute to decreased T2 and T2* relaxation times and may complicate the image interpretation for cell migration studies. The major intrinsic source of T2 and T2* effects in the brain is caused by the paramagnetic state of deoxyhemoglobin in red blood cells, leading to depiction of vessels in T2*-sensitive images. Minor blood vessels are difficult to track even on high-resolution 3-D MR images and can thus be mistaken for clusters of labeled stem cells. One way to minimize potential confusion is the acquisition of 3-D MRI datasets before the implantation of labeled stem cells, in combination with careful co-registration with subsequent MRI datasets. However, this procedure is time consuming and may fail when dynamic processes are involved that change MRI tissue properties during disease progression.
Changes in the deoxyhemoglobin level are the basis of MRI studies of the effect of blood oxygenation level-dependent (BOLD) contrast and have resulted in such applications as functional MRI [7,8]. Another broad field for the application of BOLD MRI is the monitoring and characterization of tumor growth and treatment, performed in combination with inhalation of carbogen (95% O2, 5% CO2) [9–12]. Although the modulating effect of the inhalation gas on the BOLD effect is known [7], little attention has been paid to its application and importance for cell tracking by MRI.
We have studied the influence of the anesthesia inhalation gas mixture on the contrast of implanted, USPIO-labeled stem cells and on the intrinsic T2* effect of blood vessels in rats.
Materials and Methods
Animal Model
All experiments were performed in accordance with the National Institutes of Health animal protection guidelines and were approved by the local governmental authorities. All surgical procedures were performed on spontaneously breathing male Wistar rats (body weight 250–550 g), anesthetized with 1% halothane in 35% O2:65% N2O. The present study comprised the following groups of animals: (a) normal healthy control animals (n = 8), (b) animals implanted with iron oxide labeled embryonic stem (ES) cells (n = 4), and (c) animals with focal cerebral ischemia and implanted with iron oxide labeled ES cells (n = 2).
Temporary occlusion of the left middle cerebral artery (MCA) for 60 min was performed on three animals by the intraluminal thread occlusion technique as described earlier [13]. Retraction of the endovascular suture allowed successful reperfusion of the territory supplied by the MCA. After induction of stroke, animals were allowed to recover from anesthesia and, under close control, were placed in their cages in the animal facility. Two weeks after transient cerebral ischemia, animals underwent implantation surgery as described earlier [3]. In brief, animals, spontaneously breathing under halothane anesthesia, were fixed in a stereotactic headholder, and a depot of 60,000 ES cells was grafted into the cortex, just above the corpus callosum (0.5 mm anterior from the bregma, 3.0 mm lateral from the bregma, 2.5 mm ventral from the dural surface) of the right hemisphere, contralateral to the ischemic territory.
Four healthy rats received only implantation of ES cells. Two depots of ES cells of 10,000, 30,000, or 60,000 cells were grafted into the cortex of the left and right hemispheres at the same coordinates as for the animals with cerebral ischemia.
Animals were sacrificed directly following the last MRI measurement. Brains were removed and fixed in formalin for Prussian blue staining (2% potassium hexacyanoferrate and 2% HCl). Sections (coronal, 40 μm) were analyzed for ferric iron by microscopy.
Cell Culture
Murine ES cells of the D3 cell line, stably transfected with the pCX-(β-actin)-enhanced green fluorescence protein expression vector [14], were cultivated in Dulbecco's modified eagle medium (DMEM, Biochrom, Berlin, Germany) containing 15% fetal calf serum (Biochrom), nonessential amino acids (stock solution 1:100), penicillin-streptomycin (stock solution 1:100), 50 μM β-mercaptoethanol (Sigma-Aldrich, Taufkirchen, Germany), and leukemia inhibitory factor (100 nM). Labeling of the cells was achieved by lipofection of the magnetic resonance contrast agent SINEREM (generous gift of Dr C. Corot, Guerbet, Roissy, France) consisting of USPIO particles. The lipofection reagent Metafectene (Biontex, Munich, Germany, 1 μL mL−1) was co-incubated with SINEREM (10 mg mL−1) in serum-free DMEM for 60 min, which led to the encapsulation of the contrast agent by Metafectene, consequently facilitating the contrast agent incorporation into the cells [3]. ES cells were cultured with SINEREM/Metafectene for 24 hr. After harvesting, cells were washed three times, diluted, and injected immediately.
Magnetic Resonance Imaging
MR images from animals with stem cell implantation (controls and MCA occlusion) were acquired within 12 hr after implantation of the USPIO-labeled cells. MRI measurements were repeated up to four times for up to two weeks after the initial MRI experiment for control animals. All MRI experiments were performed on experimental animal scanners at 4.7 or 7.0 T (Bruker BioSpin, Ettlingen, Germany) equipped with actively shielded gradient sets 100 mT m−1 (4.7 T) or 200 mT m−1 (7.0 T). For rf irradiation and signal detection, custom-built coils were used. A 12-cm diameter Helmholtz coil arrangement served for rf excitation, while signal detection was achieved with a 2.3-cm (4.7 T) or 3.0-cm (7.0 T) diameter surface coil. The coils were decoupled from each other.
After acquisition of 2-D multislice localizer images, 3-D FLASH images were obtained. A matrix of 256 × 256 × 64 was recorded for a field of view of 20 × 20 × 10 mm3, resulting in a spatial resolution of 78 × 78 × 156 μm3. The experimental scan time of 1:42 h per 3-D dataset was achieved with four averages, TR = 100 msec, TE = 20 msec, and a flip angle of 25°. 3-D images were processed with the NIH software “Image J.” Division of images was performed in “Image J” for better visualization of changes between images. The change of blood vessel volume was estimated by pixel counting of hypointense areas in the original images and compared with areas of hyperintensity in the divided images using “Image J.”
MRI experiments were performed with three different inhalation gas mixtures as carriers for the anesthesia gas. The spontaneously breathing animals were anesthetized with 1% halothane in either:
Changes between inhalation gas mixtures were executed with a 15–20 min adaptation period while the animal remained in the magnet to exclude repositioning or misalignment problems for the data processing. Gas mixtures were changed from mixture 1 to mixture 2 (n = 20) followed by a change from mixture 2 to mixture 3 (n = 3) and from mixture 1 to mixture 3 with a subsequent change from mixture 3 to mixture 2 (n = 2). During the whole magnetic resonance experiment, body temperature was measured with a rectal thermocouple and kept constant at 37°C with a feedback-controlled warm-water blanket.
Results
MRI of Normal Control Animals
Control animals were anesthetized with all three gas mixtures (n = 3) and with gas mixtures 1 and 2 (n = 8), without repositioning and with an adaptation time for equilibrium of the new gas mixture of 15–20 min before the next 3-D MRI experiment. All experimental parameters were kept identical for the comparative 3-D gradient-echo MRI experiments. All animals survived the experiments (up to 6 hr) and fully recovered within less than 60 min. Three animals were studied several times (up to four times within 10 days) without any adverse effect. The order of the applied gas mixture was varied between experiments and showed similar results.
The first rows in Figures 1 and 2 show representative 3-D gradient-echo MR images of rat brains acquired with the conventionally used inhalation gas mixture (35% O2/65% N2O). Arrows indicate hypointense areas that represent blood vessels. Small blood vessels usually appear as small lines, whereas major blood vessels may also appear as spherical hypointense areas (when cutting through the image plane). Such blood vessels can only be distinguished from depots of USPIO-labeled cells if they are of sufficient size to be tracked over several consecutive slices in the image stack.
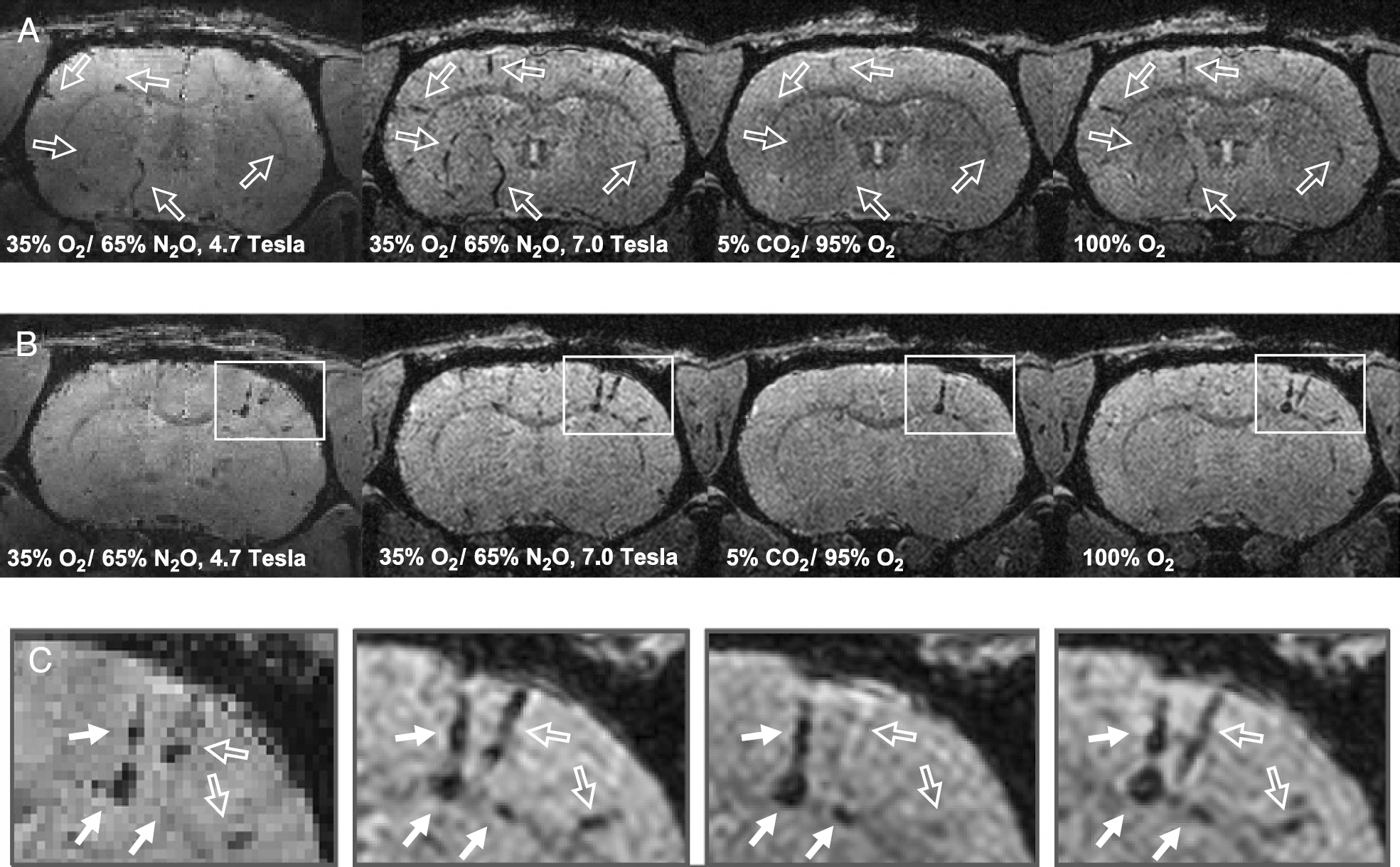
3-D FLASH MR images of a normal rat brain (A) and a rat brain 4 days after implantation of 30,000 USPIO-labeled ES cells suspended in 2 μL buffer into the right cortex (B and C). Images in C are enlargements of the marked areas in B. The resolution was 78 × 78 × 156 μm3 in all images. All images in each row are from the same animal. The inhalation gas was 35% O2/ 65% N2O [acquired at 4.7 T (left) and 7.0 T (second from left)], 5% CO2/ 95% O2 [acquired at 7.0 T (second from right)] and 100% O2 [acquired at 7.0 T (right)]. Hollowed arrows indicate blood vessels and solid arrows indicate depots of USPIO-labeled cells (including the implantation needle track). The MR images indicate that (i) blood vessels can be mistaken for USPIO-labeled ES cells when 35% O2/ 65% N2O was used as the inhalation gas (images B and C); (ii) the susceptibility effect of blood vessels is larger at higher magnetic field strength; (iii) blood vessels can be suppressed by reduction of the amount of deoxyhemoglobin in the image voxels, which is most efficient when 5% CO2/ 95% O2 was used as the inhalation gas; and (iv) magnetic susceptibility effects of USPIO-labeled cells remain unchanged, independent of the applied inhalation gases.
Influence of the Inhalation Gas Mixture on Magnetic Susceptibility Effects Caused by Blood Vessels
Representative 3-D MR images of normal control animals after application of the three gas mixtures are shown in Figures 1 and 2. For all animals, the gas mixture 2 (95% O2/ 5% CO2) resulted in the largest reduction of the endogenous magnetic susceptibility effects and thus in the apparent size of blood vessels. Most small cortical and striatal blood vessels are completely suppressed under 95% O2/ 5% CO2 inhalation. The effect of the changed inhalation gas mixture is also demonstrated for blood vessels mimicking reservoirs of USPIO-labeled cells, as shown in Figure 2A. For better demonstration of the effect of the inhalation gas mixture, images of gas mixtures 1 and 2 were divided, as shown in the right column of Figure 2. The hyperintense areas demonstrate the regions suppressed by the use of gas mixture 2. The reduction of the apparent blood vessel size after application of pure oxygen (gas mixture 3) was only marginal (Figure 2, right column) compared with the conventionally used gas mixture 1 (35% O2/ 65% N2O).

3-D FLASH MR images of a normal rat brain (A), and a rat with a stroke in the left hemisphere and a depot of USPIO-labeled ES cells (60,000) in the right hemisphere (B). All images were acquired at 4.7 T with a resolution of 78 × 78 × 156 μm3. The images in the left column were acquired with gas mixture 1 (35% O2/ 65% N2O), and the images in the center column with gas mixture 2 (5% CO2/ 95% O2). The right column shows the division of images from gas mixture 2 by images from gas mixture 1. Hyperintensity refers to suppressed areas of vascular origin when the gas mixture was changed from inhalation mixture 1 to mixture 2. Arrows in (A) indicate blood vessels that mimic depots of USPIO-labeled cells. Such blood vessels can be suppressed by decreasing the amount of deoxyhemoglobin in the blood. Visualization is improved by division of the images. This is particularly important in the case of small blood vessels that change during a disease process as shown in the case of stroke in (B) (arrows in the left hemisphere). Depots of USPIO-labeled ES cells are not affected by the inhalation gas mixture and cannot be visualized by division of the images (see arrows in the right hemisphere of B).
Comparison of the signal behavior of the same blood vessels at 4.7 and 7.0 T (after careful image co-registration) showed that the reduction of the apparent boundaries of blood vessels was even more prominent at the higher field strength due to the larger susceptibility effect of paramagnetic deoxyhemoglobin (blooming effect that extends further beyond vessel walls). This was confirmed for all intraindividual comparisons of those animals scanned both at 4.7 and 7 T (n = 4).
Quantitative estimations of the inhalation gas effect indicated strong dependency on vessel size. For small blood vessels, the area of hyperintensity in the divided images (gas mixture 2/gas mixture 1) was identical to the initial area of hypointensity in images obtained under gas mixture 1, confirming a complete suppression of those vessels by using gas mixture 2. The suppression of the largest blood vessels was at least 70% or 60% at 7.0 and 4.7 T, respectively.
Influence on Ischemic Tissue and Stem Cell Visualization
Variation of intrinsic contrast is a particular issue if dynamic processes like in focal ischemia are involved that lead to variations in blood circulation in some vessels. The area of focal ischemia is shown in Figure 2B. Blood vessels that are otherwise difficult to detect were visualized by division of the images acquired with different gas mixtures (Figure 2B, right).
Depots of USPIO-labeled, implanted stem cells were not affected by the change of the inhalation gas mixture. As shown in Figure 1C, the size of the USPIO-labeled cell depot was unchanged at both field strengths. This was confirmed by unchanged signal intensity after division of the images obtained with gas mixture 1 by images obtained with gas mixture 2 (data not shown).
Discussion
Blood vessels cutting through the image plane often cause spherical areas of hypointensity, which can mimic depots of USPIO-labeled stem cells. Only few of such blood vessels can be tracked over several slices and thereby be distinguished from depots of USPIO-labeled cells. Small blood vessels may appear similar to small clusters of migrating, USPIO-labeled cells.
Earlier studies have already established a strong relation between the BOLD effect and the blood flow and blood oxygenation in the venous blood [7]. Our present studies demonstrate that paramagnetic effects of deoxyhemoglobin in blood vessels can effectively be suppressed or reduced by careful adjustment of the inhaled gas mixture. We found that the combined effect of 95% O2, shifting the equilibrium from deoxy- to oxyhemoglobin, and 5% CO2, increasing the blood flow and thereby further diluting the deoxyhemoglobin, was necessary for an effective suppression of intrinsic hypointensity of blood vessels. This was confirmed by the only marginal effect that pure oxygen has on the signal behavior of the blood vessels. Using a gas mixture of 95% O2 and 5% CO2 increases the arterial partial oxygen pressure from 93% to 100%, and thereby the oxyhemoglobin concentration [9]. This effect has been applied successfully for the visualization of tumor vascularization in the past [9,10].
In contrast to the T2* effect in blood vessels, the hypointensity caused by USPIO-labeled, implanted stem cells is not affected by the change of the inhaled gas mixture. The utilization of 95% O2 and 5% CO2 instead of the commonly used 35% O2 and 65% N2O will therefore greatly enhance the detection and assignment reliability of labeled, migrating cells in the brain. Distinction of T2* effects caused by blood vessels from those caused by USPIO-labeled cells was so far only possible after carefully following the location of hypointense regions through subsequent slices of T2*- weighted MR images, a procedure that is time consuming and not always reliable, particularly in the case of smaller blood vessels. Although USPIO-labeled cells can be distinguished from blood vessels, other areas of hypointensity like hemorrhages can still be confused with labeled cells. However, such areas are usually distinguished from migrating stem cells by the large area they occupy.
Although long-time inhalation of pure oxygen is considered toxic, no adverse effects were noticed in the animals studied several times for 3 to 6 hr within a relatively short time frame of session repetition. This indicates the suitability of the changed inhalation gas mixture for longitudinal studies.
Footnotes
Acknowledgments
The authors thank Dr. C. Corot (Guerbet, Roissy, France) for the generous gift of SINEREM. Financial support by the Hertie Foundation and the European Commission (European program EMIL, LSHC-2004-503569) are gratefully acknowledged.

