Abstract
The best characterized mechanism of multidrug resistance (MDR) in cancer involves the MDR1 efflux transporter P-glycoprotein (Pgp). The positron-emitting radiotracer hexakis (2-methoxyisobutylisonitrile)-94mTc (94mTc-MIBI) was synthesized and validated in cell transport studies as a substrate for MDR1 Pgp. In vivo small-scale PET imaging and biodistribution studies of mdr1a/1b (−/−) gene deleted and wild-type mice demonstrated the use of 94mTc-MIBI to detect Pgp function. The reversal effect of a Pgp modulator was shown in tissue distribution studies of KB 3–1 (Pgp-) and KB 8–5 (Pgp+) tumor-bearing nude mice. The current 94mTc-MIBI experiments parallel previous studies employing 99mTc-MIBI, showing essentially identical performance of the two technetium radiotracers and providing biological validation of 94mTc-MIBI for PET imaging of multidrug resistance.
Introduction
Multidrug resistance (MDR) impedes successful chemotherapeutic treatment of cancer in humans. Therefore, the ability to determine the MDR status of a patient's tumor burden could guide therapeutic choices and allow treatment to be tailored to the patient's individual needs. Several genes are associated with the MDR phenotype [1,2]. The best characterized of these mechanisms involves overexpression of the MDR1 gene product, P-glycoprotein (Pgp), a 170-kDa transmembrane glycoprotein. Pgp is a member of the ATP-binding cassette (ABC) superfamily of membrane transport proteins and behaves as an efflux pump, opposing the passive influx of many chemotherapeutic drugs and altering intracellular drug accumulation [1–4]. Pgp recognizes and acts on several clinically important chemotherapeutic compounds, including taxanes, etoposides, and Vinca alkaloids [1]. Recently, newer cancer-targeted agents such as imatinib mesylate (Gleevec) also have been found to be transported by Pgp [1,5]. The transport activity of Pgp can be blocked by the administration of nontoxic compounds known as MDR modulators and results in the enhanced accumulation of a coadministered chemotherapeutic agent [1,6–8]. Although the exact mechanisms of Pgp function and modulation are not known, studies have shown a strong link between the level of Pgp expression and treatment outcome; patients with higher levels of Pgp expression have poorer responses and are more likely to become refractory [1,9].
The compound hexakis(2-methoxyisobutylisonitrile)-99mTc (99mTc-MIBI) is a single photon emission computed tomography (SPECT) radiopharmaceutical (Figure 1). Originally developed as a myocardial perfusion imaging agent [10], it was later shown to be a Pgp transport substrate [11]. This nonmetabolized, monocationic, and lipophilic tracer passively enters malignant and normal cells and accumulates in the mitochondria in response to electronegative plasma and mitochondrial membrane potentials [12]. For cells that have Pgp-mediated resistance, 99mTc-MIBI is recognized and expelled from the cell, resulting in net accumulation inversely proportional to the level of Pgp expression [11,13]. The accumulation and expulsion of 99mTc-MIBI in Pgp-nonexpressing and -expressing cells (and therefore tumors) provide the rationale for imaging a cancer patient with 99mTc-MIBI to assess for the MDR phenotype before the patient's treatment course is planned.
The use of 99mTc-MIBI as a tracer to detect Pgp-mediated MDR and to predict patient response has been validated in numerous clinical trials [14–25]. However, single photon agents such as 99mTc-MIBI have certain restrictions as a direct result of the limitations associated with the SPECT instruments used to collect the images [26]. Although able to yield high-quality images for analysis of superficial tissues (e.g., breast tumors), SPECT imaging suffers from attenuation artifacts and distortions when deeper lesions are studied (e.g., lung tumors). Compared to SPECT, clinical positron emission tomography (PET) instruments provide tomographic images of higher spatial resolution with greater sensitivity and temporal resolution. PET instruments also have the ability to quantitatively correct for attenuation, which decreases image distortion and allows quantification of the regional distribution of the radiotracer [26]. Although the image resolution of small-animal SPECT and PET cameras (microSPECT and microPET, respectively) is more comparable than their large-scale counterparts, microPET is more quantitative and has greater sensitivity than microSPECT. Technetium-94m (94mTc) has the same chemical properties and behavior as 99mTc, but has emission characteristics suitable for imaging using PET (t1/2 = 52.0 min, β+ = 70%, Eβ + max = 2.44 MeV). The focus of the work here involves replacing 99mTc with 94mTc to yield the PET analog 94mTc-MIBI and demonstrating the transport of 94mTc-MIBI by MDR1 Pgp. Therefore, while exploiting the quantitative, high-resolution advantages of PET, the extensive biochemical and clinical experience from 99mTc-MIBI imaging of MDR can be retained.

The structure of 99mTc-MIBI is shown. Ligands in an octahedral geometry surround the central 99mTc(I) core, encasing it and generating a lipophilic compound with a delocalized monocationic charge.
Materials and Methods
General
Acetone was purchased from Fisher Scientific (Pittsburgh, PA), normal saline (0.9% NaCl, 10-mL sterile vials) from American Pharmaceutical Partners, Inc. (Schaumburg, IL), and absolute ethanol from AAPER Alcohol and Chemical Company (Shelbyville, KY). Sep-Pak Light C-18 and Sep-Pak Light alumina N cartridges were purchased from Waters Corporation (Milford, MA) and Cardiolite sestamibi kits (for 99mTc) from Bristol-Myers Squibb Medical Imaging, Inc. (North Billerica, MA). Aqueous solutions were prepared using distilled, deionized water (Milli-Q; > 18 MΩ resistivity). All other chemicals and materials, unless otherwise specified, were purchased from Aldrich Chemical Company, Inc. (Milwaukee, WI) or Sigma Chemical Company (St. Louis, MO).
A CS-15 cyclotron (Cyclotron Corporation, Berkeley, CA) with proton beam energy of 14.5 MeV was used for the in-house production of 94mTc. Radioactive samples were counted in a radioisotope calibrator (Capintec, Inc., Ramsey, NJ) for determination of activity in millicuries and in a Beckman 8000 (Beckman, Fullerton, CA) or a Cobra II (Packard, Meridian, CT) gamma counter for activity in counts per minute. Radiochemical purity was determined by analytical radio-thin layer chromatography (radio-TLC) analyzed on a Bioscan System 200 imaging scanner running WinScan 3 software (Bioscan, Inc., Washington, DC). Radionuclide purity was determined on a Canberra multichannel analyzing gamma spectrometer (Canberra Industries, Inc., Meriden, CT). Microwave synthesis procedures were based on methods reported by Gagnon et al. [27] and were carried out in a 1040 W conventional microwave oven (model AR610, Emerson Radio Corporation, Parsippany, NJ). MicroPET imaging was performed on a microPET-R4 instrument (Concorde MicroSystems Inc., Knoxville, TN).
Preparation of 94mTc-MIBI
Technetium-94m is a positron emitter, which allows for imaging using small-animal PET as well as standard tissue dissection biodistribution analysis. Methods for the production and purification of 94mTcO4− have been previously described ([28–30]; Bigott HM, Laforest R, Ruangma A, Welch MJ, submitted for publication). Briefly, 94mTc was produced by proton irradiation (approximately 13 MeV) of an enriched molybdenum target, according to the 94Mo(p,n)94mTc nuclear reaction. After irradiation, 94mTc was separated from the molybdenum starting material by sublimation and 94mTcO4− was isolated with a Sep-Pak Light alumina N cartridge.
Synthesis of the radiolabeled compound 94mTc-MIBI was achieved with a one-step Cardiolite kit formulation, containing solid stannous chloride (0.075 mg) as a reducing agent and MIBI as the Cu(MIBI)4BF4 salt (1.0 mg, where MIBI is 2-methoxy isobutyl isonitrile).
A normal saline solution containing purified 94mTcO4− (~ 20 mCi) was aseptically added to the vial. The volume added was either 0.4 or 3 mL, which was determined by the exclusion or inclusion, respectively, of a secondary purification step (for removal of inactive kit components, see below). In both cases, the resulting solution was heated exactly 12 sec in a microwave and cooled briefly. The injectable solution of 94mTc-MIBI was drawn from the kit and divided into syringes in the required dosage. Radiochemical purity was found to be > 97%, as assessed by radio-TLC on alumina oxide plates using absolute EtOH as the mobile phase. (Cautionary note: The following steps were needed to avoid sparking and/or explosion during the microwave heating of the metal-capped kit. The internal pressure of the kit was lowered by drawing two 20-mL syringe volumes of air from the kit headspace. The metal cap was then fitted with a piece of styrofoam and the kit was enclosed in a plastic shield. Venting the kit through a 0.22-mm sterile filter after heating is also advised for easy and safe removal of the pressurized kit contents).
For the in vivo studies, a secondary purification to remove the inactive components of the kit was used. The 94mTc-MIBI solution was loaded onto a Sep-Pak Light C-18 cartridge that had been preconditioned with 3 mL of a 90:10 solution of absolute EtOH:normal saline followed by 5 mL of normal saline. The loaded cartridge was washed with 10 mL of normal saline, and the 94mTc-MIBI was eluted in 2 mL of the EtOH:saline solution used above. The bulk of the EtOH was quickly removed under a flow of N2, and water/normal saline was used to obtain the desired dosing concentration. This process yielded 94mTc-MIBI as an isotonic solution with a maximum EtOH concentration of 10% and was well tolerated (with slow injection) by the mice.
Cell Culture and Cell Transport Studies
Stock solutions of LY335979, a difluorocyclopropyl dibenzosuberane [31] (gift of Eli Lilly and Co., Indianapolis, IN), and valinomycin were prepared in dimethyl sulfoxide (DMSO). The final concentration of DMSO in experimental buffers was ≪0.5%; this value was reported to have no effect on net uptake of 99mTc complexes in cultured cells [12]. The control solution for transport experiments was a modified Earle's balanced salt solution (MEBSS) containing (in mM): 145 Na+, 5.4 K+, 1.2 Ca2+, 0.8 Mg2+, 152 Cl−, 0.8 H2PO4−, 0.8 SO42−, 5.6 dextrose, 4.0 HEPES, and 1% bovine calf serum (vol/vol), pH 7.40 ± 0.05. A 130 mM K+/20 mM Cl− solution was made by equimolar substitution of potassium methanesulfonate for NaCl, as previously described [12,32].
Monolayers of human epidermoid carcinoma drug-sensitive KB 3–1 (Pgp-) cells and the colchicine-selected KB 8–5 (Pgp+) derivative cell line were grown as previously reported [11,33,34]. Briefly, cells were plated in 100-mm Petri dishes containing seven 25-mm glass coverslips on the bottom and were grown to subconfluence in DMEM (GIBCO, Grand Island, NY) supplemented with
Coverslips with near-confluent cells were used for studies of cell transport and kinetics as previously described [13]. Cells on coverslips were removed from culture medium and washed for 15–30 sec in MEBSS, then immersed in 60-mm glass Pyrex dishes containing 4 mL of loading solution consisting of MEBSS, drug, or vehicle, and 94mTc-MIBI (~ 5 μCi/mL). Coverslips with cells were removed after 30 min, rinsed three times in 25 mL ice-cold, radionuclide-free MEBSS for 8 sec each to clear extracellular spaces, and placed in 35-mm plastic Petri dishes. Cells were then extracted in 1% sodium dodecylsulfate with 10 mM sodium borate before protein assay by the method of BCA analysis (Pierce Chemical Co., Rockford, IL), using bovine serum albumin as the protein standard. Aliquots of the loading buffer and stock solutions also were obtained for standardizing cellular data with extracellular concentration of Tc complex. All cell extracts, stock solutions, and extracellular buffer samples were assayed for gamma activity in a Cobra II gamma counter. The absolute concentration of total Tc complex in solution was approximated from the activity of stock solutions, decay time, and decay scheme [35]. Data are reported as fmol Tc complex (mg protein)−1 (nMo)−1, with (nMo)−1 representing total concentration of Tc complex (dominated by 94mTc and 94gTc) in the extracellular buffer, a numerical presentation independent of the specific activity of any particular production run.
MicroPET Imaging Studies
PET imaging was performed on a microPET-R4 system, the first generation of high-resolution, commercially available Concorde Microsystems small-animal PET scanners that was based on the original design of Cherry et al. [36]. The microPET-R4 has a field of view of 8 cm axially by 11 cm transaxially and is capable of a spatial resolution of 2 mm and an absolute sensitivity of 920 cps/μCi in the middle of the field of view. Data were acquired in list mode and histogrammed into frames. Images were then reconstructed by Fourier rebinning (FORE) followed by 2-D-filtered back projection (2D-FBP) with ramp filter cutoff at the Nyquist frequency, using data from all tilt angles [37].
All animal experiments were conducted in compliance with guidelines established by the Washington University Animal Studies Committee. MicroPET imaging studies were carried out in two mouse models. The first model consisted of parental strain FVB mice (FVB/NTac) and FVB mdr1a/1b(−/−) gene deleted mice (males, 12–20 weeks old, Taconic, Germantown, NY). The mdr1a/1b(−/−) gene deleted mice and wild-type (WT) controls were matched and imaged in pairs. The second model, also imaged in pairs, were NCRNU-M mice bearing KB 3–1 and KB 8–5 tumors (males, 6–8 weeks old, Taconic). Subcutaneous implants of drug-sensitive KB 3–1 and drug-resistant KB 8–5 cells [(1-1.5) × 107 cells] on each shoulder resulted in growth of palpable tumors. The animals were imaged in studies 1–2 weeks after implant, when the tumors were approximately 0.5-1.0 cm (200-400 mg). The shoulders were selected for tumor implantation to minimize high background activity resulting from the high liver and kidney activity associated with 94mTc-MIBI uptake.
In all cases, the mice were anesthetized with 1-2% isoflurane/oxygen and placed prone or supine in an immobilizing scanner support. The administration of 94mTc-MIBI consisted of a bolus injection via the tail vein. The tumor-bearing mice received pretreatment of either 20 mg/kg of the MDR modulator LY335979 (drug), dissolved in 5% mannitol, or vehicle alone. When such pretreatments were performed, the vehicle or drug was a bolus injection via the tail vein, and was given 30 min before 94mTc-MIBI. Single position imaging was performed at five time points (5, 30, 60, 120, and 180 min post injection), each consisting of 10-min static data collections.
Biodistribution Studies
Tissue distribution studies were performed on mice at the conclusion of the microPET imaging sessions as well as on groups of mice that were not imaged. As in the imaging experiments, two types of mice were studied: mdr1a/1b(−/−) gene deleted and WT mice and KB 3–1 and KB 8–5 tumor-bearing nude mice. The mice were anesthetized with 1-2% isoflurane/oxygen, and a bolus injection of 94mTc-MIBI was administered via the tail vein. The tumor-bearing mice were pretreated as above with either vehicle or drug at 30 min before 94mTc-MIBI. The animals were sacrificed by cervical dislocation at the desired time post injection (PI) of 94mTc-MIBI. The tissues and organs were promptly removed, weighed, and the radioactivity of each sample was determined on a Beckman 8000 gamma counter.
Pyruvaldehyde bis(thiosemicarbazonato) copper(II) (Cu-PTSM) was labeled with 64Cu and was used in a tissue distribution study to assess blood flow to the organs and tissues of the KB 3–1 and KB 8–5 tumor-bearing mice. Methods for the synthesis of 64Cu-PTSM have been previously reported [38]. Similar to the 94mTc-MIBI biodistributions studies, the mice were treated with vehicle 30 min before the administration of a bolus tail vein injection of 64Cu-PTSM. The animals were sacrificed by cervical dislocation at 15 min PI, and the tissues and organs were treated as above.
Results
In Vitro Validation of 94mTc-MIBI as a Transport Substrate for MDR1 Pgp
To validate the MDR1 Pgp-mediated transport of 94mTc-MIBI, cell accumulation of the radiotracer was determined in drug-sensitive KB 3–1 and drug-resistant KB 8–5 cell lines. Previous Western blot analyses have shown that KB 3–1 cell lines express no immunodetectable MDR1 Pgp, whereas KB 8–5 cell lines express moderate levels of Pgp [39,40]. Measurements at 30 min after initial exposure to tracer amounts of 94mTc-MIBI in the uptake buffer showed a large difference in the accumulation of 94mTc-MIBI between KB 3–1 and KB 8–5 cells (Figure 2). Cell-associated counts were normalized to total 94mTc-MIBI concentrations in the loading buffer and to protein content. Uptake (30 min) of 94mTc-MIBI was 86.5 ± 4.0 fmol (mg protein)−1 (nMo)−1 in KB3-1 cells and 2.4 ± 0.5 fmol (mgprotein)−1 (nMo)−1 in KB 8–5 cells, comparable to the values of 104.6 ± 4.1 and 2.8 ± 0.1 fmol (mg protein)−1 (nMo)−1, respectively, previously obtained for 99mTc-MIBI under identical conditions [39]. The ratio of steady-state accumulation (30 min) of the radiopharmaceutical in KB 3–1 and KB 8–5 cells was used as a measure of functional MDR1 Pgp, thereby correcting for differences in absolute cell content of each Tc complex. The ratio of cell accumulation (KB 3-1/KB 8–5 cells) for 94mTc-MIBI was 36, highly comparable to the ratio for 99mTc-MIBI of 37, as expected. These data confirm that the sensitivity for detection of functional MDR1 Pgp in KB cells using 94mTc-MIBI was identical to 99mTc-MIBI.

The effect of LY335979 on cellular accumulation of 94mTc-MIBI in KB 3–1 and KB 8–5 cells (30 min incubation) and the effect of high K+ and valinomycin on cellular accumulation of 94mTc-MIBI in KB 3–1 cells (30 min incubation). Error bars represent SEM.
To further define the interactions of 94mTc-MIBI with cells, monolayers of confluent KB 3–1 cells were incubated with tracer amounts of the Tc complex in buffer alone or buffer containing 130 mM K+/20 mM Cl− and 1 μg/mL of the potassium ionophore valinomycin. Under high K+ plus valinomycin conditions, electrical potentials of the mitochondrial membrane (ΔΨ) and plasma membrane (Em) are depolarized toward zero, eliminating the inward driving force for uptake of hydrophobic cations such as Tc-MIBI [12,41]. The residual net accumulation of a radiopharmaceutical under isoelectric membrane potential is one measure of nonspecific adsorption of hydrophobic cationic complexes to lipid compartments within cells [12,42]. Previous studies have established that Tc-MIBI is a Nernstian probe of membrane potential with minimal adsorptive binding to lipid bilayers [12,43,44] and, thus, in the presence of high K+ and valinomycin, residual uptake of Tc-MIBI maps intracellular water space [12,43]. For 94mTc-MIBI, net uptake in KB 3–1 cells (30 min incubation) under isoelectric membrane potential conditions was 3.7 ± 0.4 fmol (mg protein)−1 (nMo)−1 (n = 4; Figure 2), similar to 99mTc-MIBI under the same conditions [4.7 ± 0.3 fmol (mg protein)−1 (nMo)−1 (n = 4)] [39]. Dividing normalized uptake values by cell water space (typically about 5.4 mL/mg protein [45]) results in the in-to-out ratio of radiotracer. Both values for 94mTc-MIBI and 99mTc-MIBI (0.7 and 0.9, respectively) were close to 1, further confirming the lack of significant nonspecific binding of Tc-MIBI to cell membranes and lipid compartments.
To determine if cell content of 94mTc-MIBI could be increased by inhibition of Pgp, a potent modulator of MDR1 Pgp function, LY335979, was tested in KB cells. Inhibition of a putative efflux transport of 94mTc-MIBI would be indicated by an increase in net accumulation of the tracer. Cells were incubated for 30 min in uptake buffers in which 1 μM LY335979 had been added. Accumulation of 94mTc-MIBI was significantly enhanced in KB 8–5 cells with the modulator, whereas little effect on net tracer accumulation in KB 3–1 cells was observed under identical conditions (Figure 2). The uptake level in KB 8–5 cells was 55.5 ± 1.6 fmol (mg protein)−1 (nMo)−1 for LY335979 treated cells, corresponding to a 24-fold increase in 94mTc-MIBI cell content. This is comparable to the reported enhancements of 99mTc-MIBI in KB 8–5 cells with other Pgp modulators (46- and 37-fold, for cyclosporine A and GF120918, respectively) [7,39].
Imaging and Biodistribution Studies in mdr1a/1b(−/−) and Wild-Type Mice
To illustrate the in vivo utility of 94mTc-MIBI as a transport marker of Pgp function, microPET imaging sessions and biodistribution studies were performed on mdr1a/1b(−/−) gene deleted and WT control mice after injection of 94mTc-MIBI. Because Pgp is normally expressed in the brain, liver, and kidneys, differences in the accumulation and washout of 94mTc-MIBI were expected in these organs. Figure 3 shows representative coronal microPET image slices of a mdr1a/1b(−/−) gene deleted and WT mouse pair at 30 and 180 min PI of 94mTc-MIBI. Initial uptakes in the livers of mdr1a/1b(−/−) and WT mice were similar at 30 min. In contrast, at 180 min PI, enhanced retention in the liver is clearly observed in the mdr1a/1b(−/−) mice compared with the WT controls, consistent with the lack of Pgp-mediated efflux of 94mTc-MIBI from this organ.
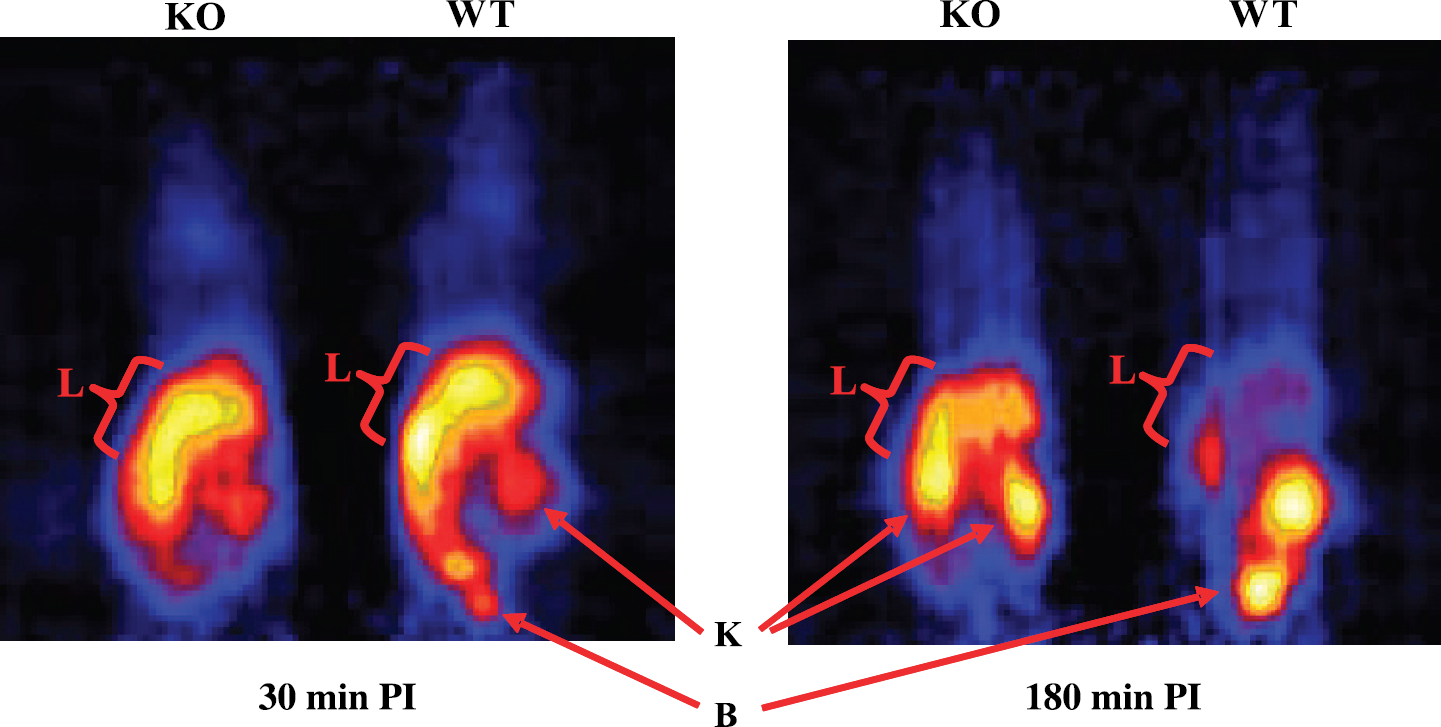
Representative coronal microPET image slices of mdr1a/1b(−/−) (KO) and wild type (WT) control mice at 30 (left) and 180 (right) min post tail vein injection of 94mTc-MIBI (400 μCi). Liver (L), kidney (K), and bladder (B) are highlighted with arrows.
Tissue distribution studies of the mdr1a/1b(−/−) gene deleted and WT mice were performed immediately after the final microPET imaging sessions (3.5 hr PI). These data, presented in Table 1, show the delayed liver washout in the mdr1a/1b(−/−) mice that was observed in the microPET images. There is a trend toward retention of 94mTc-MIBI in the kidneys, but this failed to reach statistical significance. Brain retention, although not seen in the images, was 4-fold greater in the mdr1a/1b(−/−) mice versus the WT controls (0.20 ± 0.08 and 0.05 ± 0.02, respectively, p < .05). This is consistent with reported literature findings with 99mTc-MIBI [7] in which a 3.5-fold enhancement in brain uptake was initially observed (5 min).
Imaging and Biodistribution Studies in KB 3-1 and KB 8-5 Tumor-bearing Mice
To further characterize the Pgp-mediated transport of 94mTc-MIBI in vivo, KB 3-1 (Pgp-) and KB 8-5 (Pgp+) tumor-bearing mice were investigated in microPET imaging and tissue distribution studies experiments. Pairs of tumor-bearing mice were imaged with 94mTc-MIBI on a microPET scanner on consecutive days after pretreatment with vehicle (Day 1) or LY335979 (Day 2). On Day 1, the KB 3-1 tumors, which lack Pgp, were expected to have higher tracer concentrations and appear as brighter spots on the image compared to the KB 8-5 tumors. On Day 2, pretreatment with LY335979 was expected to result in the modulation of Pgp action and to increase the KB 8-5 tumor activity to a level comparable with the KB 3-1 tumors. Unfortunately, neither tumor type was visible on either day in the microPET images. However, a similar uptake pattern, as observed previously in non-tumor-bearing animals, was detected in the liver, kidneys, intestines, and bladder. Coregistration of microPET images with microCT confirmed the tumor locations and the absence of detectable activity in these sites (data not shown).
Biodistribution of 94mTc-MIBI at 35 hr PI in FVB WT and mdr1a/1b(−/−) Micea

a% Injected dose per gram. Values are reported as the mean ± SD; n = 4 for each time point.
*p < .05, indicates a statistical difference in WT versus mdr1a/1b(−/−).
Biodistribution of 94mTc-MIBI at 2 and 3.5 hr PI in KB 3-1 and KB 8-5 tumor-bearing NCRNU micea

a% Injected dose per gram. Values are reported as the mean ± range (n = 2) or SD (n > 2). NCRNU mice were pretreated with vehicle or LY335979 before administration of 94mTc-MIBI; n > 4 for each time point.
bn = 2.
Concurrent tissue distribution data for tumor-bearing mouse experiments are given in Table 2. The mice were divided into vehicle- and LY335979 (drug)-treated groups, injected with 94mTc-MIBI, and sacrificed at 2 hr PI. Mice from the imaging experiments were also studied; these animals were sacrificed immediately following the final imaging session (3.5 hr PI). As noted in the microPET images, the liver, kidneys, and intestines of both vehicle and drug treated groups had appreciable activity concentrations at both 2 and 3.5 hr PI. No difference in the concentration of 94mTc-MIBI in the brain tissue of the vehicle- and drug-treated groups was observed, and the absolute activity concentration at 2 and 3.5 hr PI was low (Table 2). These findings are comparable to studies by Slapak et al. that demonstrated no significant difference in the brain pharmacokinetics of 99mTc-MIBI in FVB WT mice after pretreatment with either vehicle or LY335979 [7].

Tumor-specific tracer concentration at 2 and 3.5 hr PI of 94mTc-MIBI in KB 3-1 and KB 8-5 tumors of NCRNU mice pretreated with either vehicle or LY335979 (* indicates p < .05).
By biodistribution, the KB 3-1 tumors exhibited significant 2- and 3-fold enhancements in tumor activity versus KB 8-5 tumors at 2 and 3.5 hr PI, respectively (Figure 4 and Table 2), demonstrating Pgp-mediated transport of the radiotracer from the KB 8-5 tumors. In addition, as shown by the equalization of activity in the tumors of the drug treated group at both time points, LY335979 modulated the Pgp transport activity in the KB 8-5 tumors. The tracer concentration in both KB 3-1 and KB 8-5 tumors, however, was quite low (< 1% of the injected dose per gram of tumor tissue). Tumor to blood ratios ranged from 2:1 to 5:1.
Cu-PTSM has been validated as a tracer of blood flow for multiple organs, including tumors [46]. Copper-64-labeled PTSM was used in a tissue distribution study to directly assess blood flow to the organs and tissues of the KB 3-1 and KB 8-5 tumor-bearing mice. As in the 94mTc-MIBI biodistributions studies, the mice were treated with vehicle 30 min before the administration of the 64Cu-PTSM. The data, given in Table 3, show relatively low blood flow to the tumors compared to other organs; tumor flow was only ~30% that of the brain and ~15% that of the heart.
Biodistribution of 64Cu-PTSM at 15 min PI in KB 3-1 and KB 8-5 tumor-bearing NCRNU micea

a% Injected dose per gram. Values are reported as the mean ± SD; n = 4 for each time point. NCRNU mice were pretreated with vehicle before administration of 64Cu-PTSM.
Discussion
To validate the use of 94mTc-MIBI for PET imaging of Pgp transport activity, transport assays were performed in drug-sensitive (Pgp -) KB 3-1 epidermal carcinoma cells and drug-selected MDR (Pgp+) KB 8-5 cells. Net tracer accumulations were 86.5 ± 4.0 and 2.4 ± 0.5 fmol (mg protein)−1 (nMo)−1, respectively, indicating that 94mTc-MIBI is, as expected, efflux transported by Pgp. Furthermore, a 24-fold enhancement of net accumulation of 94mTc-MIBI in KB 8-5 cells treated with the Pgp modulator LY335979 demonstrated the in vitro blocking of Pgp-mediated transport of 94mTc-MIBI.
In addition, microPET imaging demonstrated the biodistribution of 94mTc-MIBI in mdr1a/1b(−/−) gene deleted mice and WT control mice. Compared to WT controls, 94mTc-MIBI showed delayed clearance from the liver and kidneys of mdr1a/1b(−/−) gene deleted mice, consistent with disruption of Pgp at the biliary canalicular surfaces of hepatocytes and luminal surfaces of renal tubules. Biodistribution data, obtained after the microPET images, verified greater retention of 94mTc-MIBI in the liver and kidneys of mdr1a/1b(−/−) gene deleted versus WT control mice. Additionally, a 4-fold difference in brain retention of 94mTc-MIBI was observed; however, because the absolute value of 94mTc-MIBI in the brain tissue of the mice was low, the brain was not observed in the microPET images.
Biodistribution studies of 94mTc-MIBI were also carried out in vehicle- and LY335979-pretreated KB 3-1 and KB 8-5 tumor-bearing NCRNU mice. Mice treated with vehicle had 2- to 3-fold higher uptake in the drug-sensitive KB 3-1 tumors versus the drug-resistant KB 8-5 tumors, demonstrating Pgp-mediated transport of the radiotracer from the KB 8-5 tumors. A second group of tumor-bearing mice was treated with the Pgp modulator LY335979 to block the Pgp function of the KB 8-5 tumors; as a result, a subsequent leveling of activities in the KB 3-1 and KB 8-5 tumors was observed because of enhanced tracer accumulation in the KB 8-5 tumors.
MicroPET imaging studies of Pgp transport and its modulation in mice bearing KB 3-1 and KB 8-5 tumors were unsuccessful. As seen in the 64mCu-PTSM biodistribution studies, the blood flow to the subcutaneous tumors relative to other organs was low. The modest 94mTc-MIBI activity accumulation in the tumors resulted in low tumor-to-background ratios in the images. This is consistent with previous reports of low 99mTc-MIBI uptake values in tumor-bearing mice [47,48]. The low tumor uptake was further complicated by the nuclear decay characteristics of 94mTc, which have been shown to degrade the quality of images on the microPET (Bigott HM, Laforest R, Ruangma A, Welch MJ, submitted for publication). Briefly, 94mTc has a relatively long positron range (mean range, ~2.7 mm; maximum range, ~12 mm) [49], the distance traveled by the emitted positron before annihilation and subsequent detection of the annihilation photons. For high-resolution small-animal PET scanners with reconstructed resolutions of 2-3 mm, the long positron range becomes the limiting factor for resolution, and a strong contrast reduction is observed in the images. The decay of 94mTc also gives rise to a high flux of 871 keV photons; 94% of 94mTc decays lead to the first excited state of 94Mo (80% direct and 14% indirect) and are followed by emission of a gamma photon upon de-excitation to the 94Mo ground state [50]. The 871 keV energy of these photons falls within the energy detection window of the microPET scanner, interfering with the accurate detection of the positron decay events and further degrading the quality of the image. Taken together, the low activity in the tumors and limitations of 94mTc in microPET imaging prevented the visualization of the tumors in this model.
Although the high resolution and quantitative advantages of PET cannot currently be fully exploited in small-animal 94mTc PET imaging, 94mTc still has a role to play in nuclear medicine. As shown above, microPET imaging of 94mTc was able to demonstrate, with organ resolution superior to single photon planar imaging [51], Pgp-mediated transport differences of 94mTc-MIBI in mdr1a/1b(−/−) gene deleted versus WT mice. Also, a novel reconstruction algorithm, Maximum a posteriori (MAP) [52], is being developed for nonstandard isotopes of PET (including 94mTc); preliminary results have shown promise for improving the quality and quantification of 94mTc microPET images ([53] and Bigott HM, Laforest R, Ruangma A, Welch MJ, submitted for publication). Furthermore, as well as serving a role in animal research, 94mTc radiotracers, including 94mTc-MIBI, are being applied in human PET studies [54–56]. Because the reconstructed resolution of these large scanners is lower (5-10 mm) [26], images from clinical PET scanners are less affected by the positron range of 94mTc than microPET cameras. Additionally, the tissues targeted in humans are typically of greater size than in animals, also making them easier to visualize in the images.
Conclusions
In an effort to take advantage of quantitative PET imaging, the 94mTc-MIBI radiotracer was synthesized and validated in KB 3-1 (Pgp -) and KB 8-5 (Pgp +) cell transport studies as a substrate for MDR1 Pgp. In vivo microPET imaging and tissue distribution studies of mdr1a/1b(−/−) gene deleted (Pgp -) and WT (Pgp +) control mice further demonstrated the use of 94mTc-MIBI to detect Pgp function. Mice bearing KB 3-1 and KB 8-5 tumors were also studied, and the Pgp modulation of LY335979 was shown. Tissue distribution studies showed higher 94mTc-MIBI activity in the KB 3-1 tumor of vehicle-treated mice and the equalization of tumor activities in the LY335979-treated mice, as expected. However, due to low tracer uptake levels of the tumors as well as the nuclear decay characteristics of 94mTc, the tumors were not visualized in the microPET imaging experiments. As advances in the reconstruction software for microPET imaging of nonstandard PET radionuclides are made, the current role of 94mTc in small-animal imaging will improve.
Footnotes
Acknowledgments
This work was supported by United States Department of Energy grants DE-FG02-87ER60212 (MJW) and DE-FG02-94ER61885 (DPW) and the United States National Institutes of Health (NIH) grant P50 CA94056 (DPW). Technetium-94m produced at Washington University Medical School was partially funded through National Cancer Institute (NCI) grant R24 CA86307 (MJW). MicroPET imaging was supported by NIH/NCI SAIRP grant 1 R24 CA83060. We thank the Small Animal Imaging Core of the Alvin J. Siteman Cancer Center at Washington University and Barnes-Jewish Hospital in St. Louis, Missouri, for additional support of the microPET imaging. The Core is supported by NCI Cancer Center Support grant 1 P30 CA91842.
