Abstract
Noninvasive near-infrared fluorescence reflectance imaging (FRI) is an in vivo technique to assess physiological and molecular processes in the intact organism. Here we describe a method to assess gastric emptying in mice. TentaGel™ beads with covalently bound cyanine dye (Cy5.5) conjugates as fluorescent probe were administered by oral gavage. The amount of intragastric beads/label was derived from the fluorescence signal intensity measured in a region of interest corresponding to the mouse stomach. The FRI signal intensity decreased as a function of time reflecting gastric emptying. In control mice, the gastric half-emptying time was in agreement with literature data. Pharmacological modulation of gastric motility allowed the evaluation of the sensitivity of the FRI-based method. Gastric emptying was either stimulated or inhibited by treatment with the 5-HT4 receptor agonists tegaserod (Zelnorm®) and cisapride or the α2-receptor agonist clonidine, respectively. Tegaserod and cisapride dose-dependently accelerated gastric emptying. In contrast, clonidine dose-dependently delayed gastric emptying. In conclusion, FRI using fluorescently labeled beads allows the reliable determination of gastric emptying as well as the assessment of pharmacological interventions. The technique thus offers the potential to characterize molecular targets and pathways involved in physiological regulation and pharmacological modulation of gastric emptying.
Keywords
Introduction
Noninvasive in vivo imaging with light photons represents an intriguing avenue for studying molecular and physiological processes in the intact organism. As hemoglobin and water, the major absorbers of visible and infrared light, have their lowest absorption coefficient in the near-infrared (NIR) region of the electromagnetic spectrum around 650–900 nm, the NIR window is ideally suited for in vivo applications [1]. Depth of penetration for NIR light in tissue is several centimeters, allowing for whole body imaging of small animals as well as investigation of superficial tissue in larger species including humans. Examples are the detection of tumors [2,3], their characterization by visualization of molecular events such as enzymatic activities or drug receptor interactions [4–6], or the study of bone formation in rodents [7]. In these studies, the high sensitivity of optical techniques has been exploited to monitor fluorescent reporters at low tissue concentrations. Alternatively, NIR imaging could be used to assess the physiological functions of organs using nontargeted labels.
Classically, gastric emptying is studied in laboratory animals using invasive methods, frequently requiring postmortem analyses: The rate of emptying is determined by measuring the gastric content or a marker thereof [8–10]. Few alternative methods based on noninvasive readouts have been proposed, such as X-ray measurements monitoring the passage of radio-opaque tracers [11] or a modified breath test [12–14]. In the latter case, the amount of an isotope-labeled metabolite (CO2) of an orally administered substrate is determined in the exhaled air. The measure, however, is affected by a cascade of physiological and metabolic processes, gastric passage being only one of them and, hence, constitutes an indirect measure of gastric emptying. Not surprisingly, the use of these two techniques in small animal studies has been very limited. The availability of novel imaging modalities such as optical imaging developed during the last years might offer attractive noninvasive alternatives to the established methods used for studying gastric emptying in animals.
In this article, we describe a noninvasive method to assess gastric emptying of a nonnutrient semisolid meal in mice using fluorescence reflectance imaging (FRI). TentaGel beads conjugated with cyanine dye Cy5.5 have been used as a fluorescent sensor. This technique has been applied to characterize gastric emptying in mice under normal conditions and following pharmacological stimulation or inhibition with the serotonin type-4 (5-HT4) receptor agonists tegaserod (Zelnorm®) and cisapride, or the α2-adrenergic receptor agonist clonidine, respectively. These pharmacological agents have been shown to alter gastric emptying in both animals and humans [15–20].
Materials and Methods
Animals
Adult male Balb/C mice (20–25 g; Charles River, France) were used. Animals were housed in groups of up to six animals per cage under standard conditions and with food and water available ad libitum. All procedures were in accordance with the Swiss animal welfare laws and approved under the animal research license BS 1827.
Synthesis and Stability of Fluorescence Conjugates
Particles with reactive amino alkyl surface groups were used for the synthesis of the fluorescent conjugates. Controlled pore glass, TentaGel beads, or polystyrene beads were reacted with the amino reactive Cy5.5 succinimidyl ester in 0.1–100% molar ratios (calculated on particles surface free amino groups) for 2 hr in N, N-dimethylformamide. Labeled beads were obtained as blue (controlled pore glass) or blue–green to black solids (TentaGel, polystyrene beads) after several washings. Relative fluorescence spectra of labeled beads were recorded on a SPEX Fluorolog spectrometer equipped with a cooled R928 detector (1 mm slit) (Jobin Yvon, Edison, NJ, USA).
The blue–green color of the TentaGel beads (1% loading with Cy5.5) changed to yellow–green after several days, indicating a partial decomposition of the chromophor on the beads. For that reason, a protected form of the chromophor (NVP-AGY749) was prepared. TentaGel beads were reacted with the amino reactive Cy5.5 succinimidyl ester in a 1% molar ratio, as described above. At the end of the reaction, the remaining free amino groups were capped by addition of five equivalents of acetic acid anhydride. The absence of free amino groups after this treatment has been proved using the trinitrobenzenesulfonic acid test [22]. Following these procedures, NVP-AGY749 (protected TentaGel beads loaded with 1% Cy5.5) was obtained as stable blue solid. Stability of NVP-AGY749 at low pH was assessed by determining time-related changes in the fluorescent spectra of a sample maintained at pH = 2.0 for up to 23 hr. NVP-AGY749 was suspended in 0.01 N HCl (pH ~2.0) and the relative fluorescence spectra at 15 min and 3, 6, and 23 hr were recorded on a SPEX Fluorolog spectrometer, as described above.
Drugs and Chemicals
Controlled pore glass (Aminopropyl-CPG-1400 Å, particle size 120–200 mesh, capacity 0.04 mmol/g; Fluka Chemie, Buchs, Switzerland), TentaGel beads (TentaGel MB-NH2, particle size 200–250 μm, capacity 0.2–0.3 mmol/g; Rapp Polymere, Tübingen, Germany), polystyrene beads (Polystyrene AM-NH2, particle size 160–200 μm, capacity 0.8–1.2 mmol/g, Rapp Polymere), and the fluorescent dye Cy5.5 succinimidyl ester were used for the synthesis of fluorescent conjugates. Tegaserod [5-methoxy-indol-3-carboxaldehyd-amino-(pentyl-amino) methylenhydrazon-hydrogenmaleinat; Novartis Pharma, Basel, Switzerland] was dissolved in 10% (vol/vol) 1-methyl-2-pyrrolidon in normal saline. Cisapride [cis-4-amino-5-chloro-N-(1-(3-(p-fluorophenoxy) propyl)-3-methoxy-4-piperidyl)-o-anisamide; Novartis Pharma] was dissolved in 1% (vol/vol) lactic acid in distilled water. Clonidine hydrochloride [2-(2,6-dichloroanilino)-2-imidazoline hydrochloride; Novartis Pharma] was dissolved in physiological saline solution. All solutions were freshly prepared before each experiment.
Experimental Protocols for in Vivo Determination of Gastric Emptying
Prior to the experiments, animals were fasted for 4 to 5 hr, with water available ad libitum resulting in an almost complete emptying of the stomach in mice [10]. To avoid circadian variations, fasting was always started between 8:00 and 9:00 a.m. and all measurements were carried out between 12:30 and 4:30 p.m.
The fluorescent probe was administered by oral gavages (400 mg kg−1 of NVP-AGY749 suspended in 0.5% methylcellulose in water, 0.1 mL per animal). FRI measurements were performed immediately after the administration of the probe (time 0) and at different time-points thereafter. For FRI, the animals were temporarily anaesthetized with isoflurane (1.5% vapor concentration in nitrous oxide/oxygen 2:1; Forene, Abbott, Baar, Switzerland). Anesthesia was induced in a chamber and maintained during the imaging time with a face-mask. For gastric emptying measurements, animals were placed in right lateral position exposing the left side (where the stomach is localized) to the NIR excitation/detection system. The spatially integrated fluorescent activity corresponding to the anatomical location of the stomach (region of interest [ROI]) was determined at 0, 30, 60, 120, and 180 min after the administration of the fluorescent probe; in some cases, an additional imaging dataset was collected at 90 min. At each time point, the imaging procedure lasted about 5 min; in-between measurements, animals were returned to their home cage and were left undisturbed until the next measurement time point. In order to ensure the same positioning at each time point of gastric emptying measurements, that is, to minimize co-registration errors, the anaesthetized mice were placed in a positioning device. The positioning device consisted of wax and its shape was adapted to the individual contour of each animal prior to an experiment. In addition, the ROI was marked with black dots on the skin of the animals for the reproducible illumination with the laser beam (Figure 2). The ROI was defined from the fluorescent area recorded immediately after the oral gavage of the labeled beads (time 0).
For each animal, a maximum five experiments were conducted with the treatment regimen varied in a randomized manner. Between two consecutive experiments, animals were allowed to recover for at least 3 days.
Validation and Pharmacological Studies
In a first set of experiments, the reproducibility of the technique and the normal gastric emptying pattern was determined by carrying out three identical experiments in the same nontreated animals (N = 8–10).
In a second set of experiments, the pharmacological modulation of gastric emptying was studied following oral administration of the 5-HT4 receptor agonists tegaserod (0.03 or 0.1 mg kg−1, N = 7–8) and cisapride (0.3 or 1.0 mg kg−1, TV = 8) or following intraperitoneal administration of the α2-receptor agonist clonidine (0.03 or 0.1 mg kg−1, N = 4-5). Test compounds or the appropriate vehicles (N = 6–13) were administered 15 min before the administration of the fluorescent probe (0.1 mL).
Determination of Gastric Emptying and Statistical Analysis
For each individual animal, the gastric emptying in percent, GE%(t), was determined as a function of time t from the changes in total fluorescent activity in the respective ROI, according to
where FRI(t) and FRI(0) represent the corrected fluorescence intensities (Equation 2) measured at times t and zero, respectively. In Equation 1, we assumed that the FRI activity is a direct measure for the amount of beads in the ROI.
For each individual animal, the gastric half-emptying time, T1/2, defined by GE%(T1/2) = 0.5GE%(0), was determined by nonlinear regression analysis (sigmoidal Boltzmann fit) followed by interpolation/extrapolation. In 8 out of 105 experiments, an extrapolation of T1/2 values could not be performed due to the low incremental increase in gastric emptying with time. These experiments (5 with vehicle, 2 with cisapride, and 1 with clonidine) were excluded from data analysis.
All results are expressed as mean ± SEM (n = number of animals). Differences between treatment groups were assessed using analysis of variance (ANOVA) followed, if necessary, by a multiple comparison test (Dunnett's or Student's–Newman–Keuls method, as appropriate). p values #x003C; .05 were considered statistically significant.
FRI System
For FRI, a custom-tailored spectroscopic imaging system has been used (multi-wavelength NIR imaging system) [21]. The laser beam generated by a laser diode emitting at 660 nm with a power of 35 mW (Sharp, Osaka, Japan) was dispersed by a lens system to a circular diameter of 4 cm at an object distance of 40 cm. The fluorescent light emitted from the sample (mouse) was detected by a charge-coupled device (CCD) camera (Hamamatsu Photonics K. K., Hamamatsu City, Japan) equipped with a focusing lens system (macro lens 60 mm, 1:2.8; Nikon, Tokyo, Japan). The CCD features low noise and low dark signal enabling both the detection of low-intensity light as well as the use of long integration times. The matrix size of the images is 532 × 256 pixels. A liquid crystal tunable filter (LCTF, Cambridge Research and Instrumentation, Woburn, MA, USA) was used for selection of the detection wavelength (720 nm). Data acquisition (i.e., integration) times ranged from 3 to 16 sec depending on the intensity of the fluorescence signal. The data acquisition process was controlled by a PC using a LabView-based imaging software (B. Schattka, National Research Council, Winnipeg, Canada). An anatomical reference image was obtained at the excitation wavelength (660 nm) with diffuse illumination of the whole animal.
Image Analysis
Images were analyzed using the BioMap 2.4 software (M. Rausch, Novartis Institutes for BioMedical Research, Basel). The camera-offset signal was removed by automatic baseline correction prior to quantitative analysis. For this purpose, the value of the lowest peak in the intensity histogram of the image was subtracted from all image pixels. Thereafter, images were corrected for inhomogeneities of the laser diode excitation profile by dividing the fluorescence signal by a reference signal recorded at the excitation wavelength for each pixel, according to
where
Results
Preparation and Characterization of the Fluorescent Sensor
TentaGel beads, polystyrene beads, and controlled pore glass beads conjugated with the cyanine dye Cy5.5 as fluorochrome were stable and showed maximal fluorescence emission at 710–720 nm, except for controlled pore glass beads that showed more variable emission spectra (Figure 1A-C). A fluorochrome loading of 1% was optimal for the fluorescence properties of the different conjugates; fluorescence intensities decreased with higher Cy5.5 loadings (Figure 1D). Because of their physico-chemical properties and easy handling, TentaGel beads with a 1.0% Cy5.5 loading were selected for further characterization. The blue–green color of the TentaGel beads (1% loading) changed to yellow–green after several days, indicating a partial decomposition of the fluorochrome. For that reason, protected TentaGel beads loaded with 1.0% Cy5.5 (NVP-AGY749) were prepared. NVP-AGY749 was obtained as blue solid, which shows no decomposition (as monitored by color change) for months when stored in the dark. The emission spectrum of NVP-AGY749 was virtually identical to that of the beads–dye conjugate before the capping of the free amino groups, with a maximal emission at 720 nm (Figure 1E). Moreover, NVP-AGY749 was stable at low pH (~2.0) for up to 23 hr, as demonstrated by the lack of changes in the emission spectrum (data not shown).

Fluorescence intensity and emission spectra of Cy5.5 labeled carriers. Fluorescent emission spectra of TentaGel beads (A), polystyrene beads (B), and controlled pore glass (C) alone or labeled with different concentrations of Cy5.5 dye. Comparison of the integrated fluorescent intensity for the different carriers tested in relation with the concentration of Cy5.5 dye (D). Comparison of the fluorescence emission spectra obtained for either nonprotected or protected (NVP-AGM749) TentaGel beads loaded with a 1.0% Cy5.5 dye (E).
Visualization of Gastric Emptying in Vivo by Noninvasive NIR FRI
Gastric emptying was monitored as reduction of the integrated corrected fluorescence intensity FRI(t) in the ROI corresponding to the anatomical location of the stomach over time. Figure 2A shows a series of representative images obtained from an animal administered with TentaGel beads-dye conjugate in 0.5% methylcellulose (400 mg kg−1, 0.1 mL, p.o.) at time 0. FRI(t) values decreased as a function of time reflecting the progressive reduction of the number of labeled beads in the stomach and thus gastric emptying (Figure 2B-C).
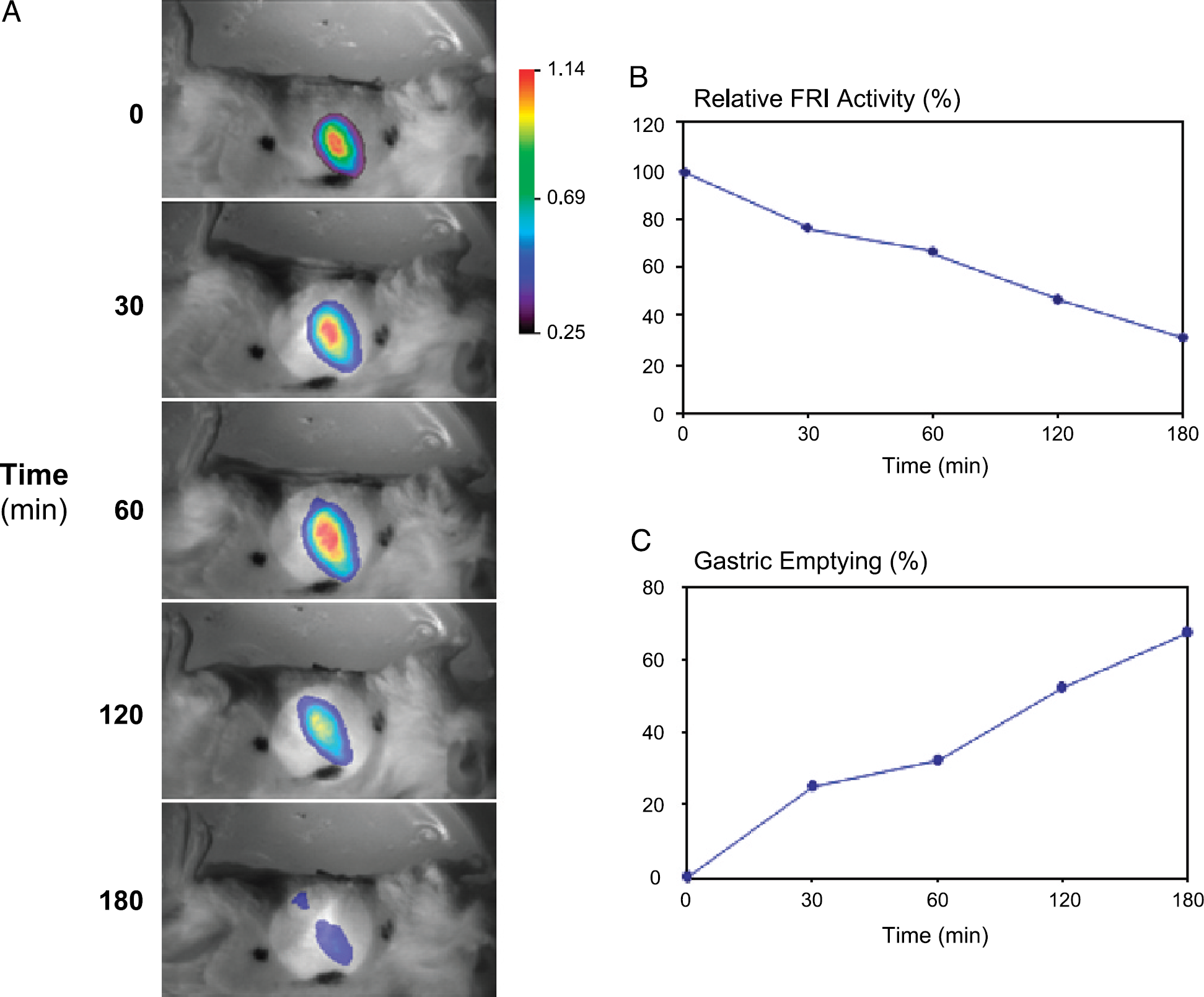
Representative images showing the FRI fluorescence intensity in the region of interest (covering the anatomical location of the stomach) of TentaGel beads loaded with Cy5.5 (1.0%) in a mouse. TentaGel beads loaded with Cy5.5 (1.0%), suspended in methylcellulose 0.5% aqueous solution (400 mg kg−1, 0.1 mL), were administered orally at time t = 0 and FRI images were recorded at time points t = 0, 30, 60, 120, and 180 min after administration of the probe (A). The FRI images represented in a color-coded scale are superimposed to the white light reference image. At t = 180 min, two spatial distinct areas of FRI signals could be detected: stomach (lower area) and small intestine (upper area). The black dots on the skin of the animal represent the area marked for the reproducible illumination with the laser beam (A). The fluorescence intensity in the ROI (in percent of the initial signal) declined with time (A, B) reflecting increasing gastric emptying GE%(t) (C).
Repetitive imaging in the same animal revealed good quantitative reproducibility of gastric emptying measurements. In three consecutive experiments, the mean T1/2 values amounted to 115 ± 18 min, 107 ± 15 min, and 124 ± 13 min (mean ± SEM, n = 8–10 animals), respectively. At the end of the 3-hr experimental period, FRI(180 min) amounted to 42.2 ± 5.6%, 37.0 ± 7.2%, and 42.0 ± 8.0% of the initial value corresponding to the respective gastric emptying rates of GE%(180 min) = 57.8%, 63.0%, and 58.0% (Figure 3). The coefficients of variation for intraindividual differences, that is, the day-to-day variability, amounted to 0.32 ± .0.05 and 0.30 ± 0.05 for T1/2 and GE%(180 min), respectively (mean ± SEM, n = 3 experiments in n = 8–10 animals). The coefficients of variation for interindividual differences, that is, the variability between the mice within one experiment, amounted to 0.39 ± 0.10 and 0.34 ± 0.04 for T1/2 and GE%(180 min), respectively (mean ± SEM, n = 8–10 animals in n = 3 experiments).
The 5-HT4 agonists tegaserod (0.1 mg kg−1) and cisapride (0.3 or 1 mg kg−1) significantly accelerated gastric emptying as reflected by the shortened gastric half-emptying times (T1/2 = 44–69 min) and the increased gastric emptying rates (GE%(180 min) = 70–83%) (Figure 4A and B, Table 1). The α2-adrenergic agonist clonidine (0.1 mg kg−1) significantly prolonged (doubled) gastric half-emptying times and reduced (halved) gastric emptying rates (Figure 4C, Table 1).

Reproducibility of gastric emptying pattern measured by FRI in mice. To determine the reproducibility of the FRI measurements, the rate of gastric emptying was determined in three consecutive experiments in the same group of animals. For each time point, the integrated fluorescence intensity covering the anatomical location of the stomach was used to calculate gastric emptying over a 3-hr period. Each data point represents the mean ± SEM of 8–10 animals.
Discussion
We have shown that FRI in combination with fluorescently labeled beads can be used to visualize gastrointestinal functions such as gastric emptying in vivo. The fluorochrome used, Cy5.5, belongs to the cyanine-type dyes extensively used in biomedicine and has been previously used as an enzyme-responsive reporter to measure cathepsin B enzyme activity in vivo [6]. Conjugation with different carriers did not alter the fluorescent characteristics of the dye. TentaGel beads–, controlled pore glass–, or polystyrene beads–dye conjugates were stable and showed similar fluorescent activity when excited. Loading with different concentrations of Cy5.5 dye showed a maximal fluorescent activity with a 1.0% loading. The further increase in loading up to 10% resulted in a reduced activity, most probably due to internal quenching. The observed tendency of the chromophor to decompose with time was prevented by capping the free amino groups at the end of the dye loading reaction. The protected conjugate (NVP-AGY749) was very stable, even in acidic solutions, mimicking the intragastric environment. No changes in the emission spectrum were observed when NVP-AGY749 was maintained at a pH ~2.0 up to 23 hr. The time-dependent reduction of integrated fluorescence intensity in the mouse stomach can therefore be attributed to transport of beads out of the stomach rather than label decomposition and loss of fluorescence activity. Hence, temporal changes of the integrated fluorescence intensity are a measure of gastric emptying.
It is critical when using contrast-enhanced imaging approaches that the labels and procedures used do not influence the parameters to be measured. In this regard, the size (diameter: 200–250 μm) and composition of the TentaGel beads are considered not to affect the physiological pattern of gastric emptying. Firstly, the beads are stable in the gastric environment and do not stick to the gastric surface (data not shown). Secondly, their size allows normal pyloric evacuation during gastric emptying, as the internal diameter of the mouse pylorus has been shown to be greater than 1 mm [23]. Thirdly, the TentaGel beads–Cy5.5 conjugate was suspended in a 0.5% methylcellulose solution, forming a viscous suspension which, together with the beads, mimics the texture of a nonnutrient semisolid meal.
The suitability of the technique to monitor gastric emptying in vivo is demonstrated by three facts: (i) the signal derived from the fluorescent marker can by detected and analyzed easily; (ii) measurements are reproducible among different experiments carried out under similar conditions, giving a consistent pattern of gastric emptying; and (iii) the technique has high enough sensitivity as to detect both the stimulation and the inhibition of the normal emptying pattern.

Effects of cisapride, tegaserod, and clonidine on gastric emptying in mice. Accelerated gastric emptying was observed for the 5-HT4 agonists cisapride (A) and tegaserod (B), while the α2 inhibitor clonidine has clearly inhibitory effects (C). Each data point represents the mean ± SEM of 4–13 animals.
The sensitivity of the technique for studying pharmacological effects was evaluated using the gastroprokinetic 5-HT4 agonists cisapride and tegaserod as well as the inhibiting α2-adrenergic agonist clonidine [19,20,24,25]. The results demonstrated that the FRI technique is able to detect the dose-related stimulatory and inhibitory drug effects on gastric emptying and, hence, constitutes an attractive tool for drug discovery and development.
Effects of Cisapride, Tegaserod, and Clonidine on Gastric Emptying Rate and Gastric Half-Emptying Time (T1/2)

Data represent the mean ± SEM, n = number of animals.
p #x003C; .05 versus respective vehicle group.
Limitations of FRI are caused by the light absorption and scattering of tissue compromising accurate spatial localization of the fluorescent source and quantification of the fluorescence activity. The intensity values measured depend on both the intensity of the fluorescence source (the number of labeled beads) and its depth with regard to the surface of the animal. Increasing the distance that the light photons have to propagate through tissue will attenuate and disperse the signals detected. Nevertheless, semiquantitative image analysis is only feasible assuming reproducible tissue geometry, that is, the ROI has to be located at the same position with respect to the animal's surface throughout the imaging study. However, in lack of external references, the very exact repositioning of the animals is difficult and co-registration errors might affect the quantitative measures of gastric emptying by inclusion of anatomical regions adjacent to the stomach.
However, these limitations are suggested of minor importance as our present findings are in accordance with data derived from other approaches. In basal conditions, we observed gastric half-emptying times (T1/2) of a semisolid meal of 70–120 min, which are in line with the T1/2 values of 20 and 150 min of respective liquid and solid meals measured with an adapted breath test in mice [12,13]. The breath test is based in the production of labeled CO2 after the ingestion of a labeled substrate (usually with either 13C or a radioactive isotope). In addition, the gastric emptying rate of a solid nutrient meal in mice has been reported to be 30% to 60% at 2 hr following the meal [9,10] as compared to 40% to 70% determined at 3 hr following a nonnutrient semisolid meal in our study. It should, however, be mentioned that using classical approaches in nonanaesthetized mice, gastric half-emptying times and gastric emptying rates of nonnutrient semisolid meals have also been reported to be respective 30 min [26] and 40–60% within 15 to 30 min [27,28]. The discrepancy to the present data might be due to the repeated anesthesia used in our study. High doses of isoflurane have been shown to cause a prolongation of gastric half-emptying times in man and animals [29,30].
Classical approaches to determine gastric emptying in laboratory rodents are invasive techniques that might require chronic surgery and, in most of the cases, lead to the death of the animal at the end of the experiment. This has several major implications: An animal provides only one gastric emptying value for a fixed time point. Hence, time-course or comparative studies have to be performed in different animals, which increase the degree of variability associated to interindividual differences. Studies using classical techniques are limited to transversal comparisons of single measurements and, therefore, the number of animals needed is relatively high. In contrast, the noninvasive FRI technique allows repetitive measurements in the same animal. The pattern of gastric emptying can be monitored over time; the same animal can be used in several experiments and for comparative studies, in which gastric emptying can be measured before and after treatment. In addition, the animals are not subjected to any surgery, which might compromise the validity of the results.
The pharmacological validation of the technique provided evidence that the 5-HT4 receptor agonist, tegaserod (Zelnorm®), has gastroprokinetic properties. Doses of 0.03 and 0.1 mg kg−1 stimulated gastric emptying, consistent with preliminary evidences in dogs, rats, and mice [6,17,18]. Maximum efficacy was observed at a dose of 0.1 mg kg−1 and was comparable to that of cisapride at a dose of 1.0 mg kg−1, suggesting a higher potency of tegaserod as compared to cisapride. This observation suggests that tegaserod has therapeutic potential for the treatment of pathophysiological states with impaired gastric emptying, such as diabetic gastroparesis or postoperative gastric ileus, or states in which gastroprokinetic drugs might contribute to a general symptom improvement, such as functional dyspepsia. However, whether the mouse model of gastric emptying predicts the efficacy of drugs in human beings might depend on the drug target. Although we demonstrated inhibitory effects of clonidine in mice, this might be a rodent-specific effect, as several clinical studies suggest that clonidine does not inhibit human gastric emptying [31,32].
In summary, our results demonstrate that in vivo FRI, in combination with TentaGel bead–cyanine dye (Cy5.5) conjugates as fluorescent probes, is an appropriate technique for monitoring gastric emptying in small rodents. The technique displays good reproducibility and sensitivity allowing the detection of both accelerated and delayed gastric emptying pattern and thus offers the potential to characterize molecular targets and pathways involved in physiological regulation and pharmacological modulation of gastric emptying.
Footnotes
Acknowledgments
We thank Dr. Luigi LaVecchia for his contributions to the label synthesis and Matthias Irouschek for his technical assistance.


