Abstract

Keynote Presentation
Abstract ID: 001
Current problems of genetic heterogeneity and the poor penetration of visible light through small animal tissues have compromised commercial techniques for imaging small animal cancer volumes in growth and regression, a most important metric for the quantitative evaluation for growth factors and anticancer drugs. In order to provide quantitative data on the distribution of markers of genetic expression in small animal cancers in growth and recession, we have applied serial section imaging to cryopreserved small animal cancers, with current single photon resolution of 40microns and an expected 2 photon resolution of 2microns, in 2D. The 3rd dimensional resolution is achieved by serial images obtained by low temperature milling currently at 100microns and prospectively at 10microns. The animal cancer (studied in cohorts) is snap frozen in 160Kdegree isopentane, the cancer mounted and ground smooth with the mill wheel at liquid nitrogen temperatures (77Kdegree) and scanned with fiber coupling (40microns resolution) with roughly 40000 voxels per square centimeter affording not only a color display of the distribution of the fluorescent signals but also a histogram display of the heterogeneity. The 2D images of 10 or more sections form a 3d image from which the volume of the cancer can precisely be calculated and the heterogeneity and changes thereof displayed as histograms. Three categories of signals are obtainable: #1Intrinsic signals of the mitochondrial redox state (of interest in apoptosis), matrix space NADH and citric cycle flavoprotein. #2oxyhemoglobin, deoxyhemoglobin and total hemoglobin. #3Blood-pooling agents such as ICG, molecular beacons, particularly Somatastatin, LDL, Integrin as affinity labeling and stealth beacons activated by Cathepsin B. #4Fluorochromes indicating genetic expression (GFP YFP RFP) can be accurately imaged. #5Luciferase, while giving no signal at liquid nitrogen temperature, can be rapidly brought up to zero degrees where luminescence can be observed in small voxels with laser temperature jump technology.
B. Chance, None.
Plenary Session I: Advancing Molecular Imaging Approaches in Biology and Medicine
Abstract ID: 002
Genetically encoded tags and indicators are molecular spies that reveal specific gene products and biochemical processes in living cells and organisms. The best known examples are fluorescent proteins originally from jellyfish and corals, which have been bred to cover the entire visible spectrum and to eliminate multimerization, facilitating their widespread use to track cells and fusion proteins. A novel and powerful method for evolving new fluorescent proteins is to hijack somatic hypermutation (SHM), the machinery normally used in B lymphocytes to optimize antibodies. For example, SHM of monomeric red fluorescent protein has produced a variant with increased photostability and a 649 nm emission maximum, currently the longest wavelength for an autofluorescent protein. With further engineering, fluorescent proteins can report local dynamic signals such as redox potential, kinase/phosphatase activities, protein-protein interactions, and ion and neurotransmitter (e.g. glutamate) concentrations. For clinical applications one would prefer not to have to introduce genes or be limited to optical detection. Arginine-rich sequences are known to mediate uptake of a wide variety of contrast agents into cells and tissues in vivo. We find that such uptake can be prevented by appending certain polyanionic sequences and selectively re-activated by cleavage of the linker. This new mechanism offers the exciting possibility that radioactive, magnetic, and infrared contrast agents and therapeutic drugs may be concentrated in diseased tissues expressing particular extracellular proteases. Complementary advantages and biological applications of genetically encoded and totally synthetic probes will be highlighted.
R.Y. Tsien, None.
Abstract ID: 003
We demonstrate that in aqueous solution, hydrophobic conjugated-multi(porphyrin)-based near-infrared fluorophores (NIRFs) cooperatively self assemble with amphiphilic diblock copolymers to form polymersomes (100 nm–20 μm diameter polymer vesicles). The thick membranes of these synthetic vesicles uniquely segregate and uniformly disperse large numbers of high emission dipole strength NIRFs. Long-wavelength optical excitation of such assemblies generates intense, highly localized emissive signals capable of penetrating through the dense tumor tissue of a live animal. Robust, NIR-emissive polymersomes thus define a soft matter platform with exceptional potential to facilitate deep-tissue fluorescence-based imaging for in vivo diagnostic and drug-delivery applications.
M.J. Therien, None.
Abstract ID: 004
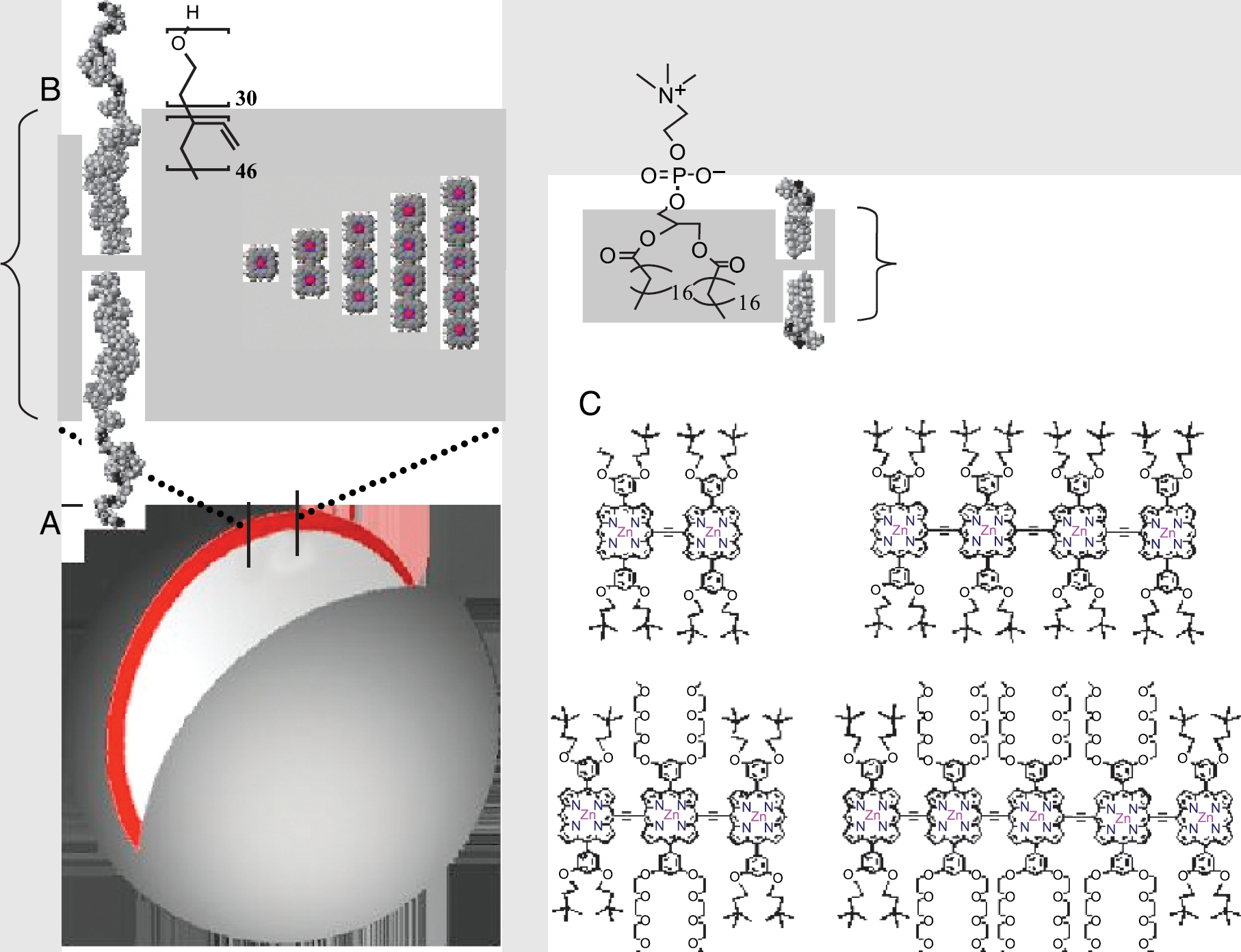
Schematic representation of NIR emissive polymersomes highlighting: (A) an excised cross-sectional slice of the polymer vesicles, (B) comparative metrical parameters with respect to a typical liposome membrane comprised of phospholipids, and (C) chemical structures of high emission dipole strength NIR fluorophores PZn2-PZn5.
Estrogens modulate target cell activities through a multiplicity of mechanisms involving genomic and non genomic actions of their conjugated receptors (ERα and ERβ). The two ERs are hormone-modulated transcription factors, which activity can be stimulated in the absence of ligand by molecules involved in the intracellular signaling of growth factor receptors. To understand the physiological meaning of these diverse mechanisms operating on ERs, we recently developed a transgenic mouse model (named ERE-Luc) that reports the transcriptional activity of ER in vivo. The transgene use to engineer the mouse consists of a multimerized estrogen responsive element (ERE) linked to a TK minimal promoter driving the transcription of a luciferase reporter gene. Several studies demonstrated a strict association between accumulation of luciferase and augmented transcriptional activity of ERs in the ERE-Luc tissues. In addition, our data reported a strong ER activation in the absence of gonadal production of estradiol, (prepuberal and ovariectomized female mice). Blocking the synthesis of estradiol with aromatase inhibitors we demonstrated that this latter ER activation is not sustained by local production of estrogens. Thus molecules other than estrogens may be responsible for the activation of both ERs. In vivo imaging of ER activity carried out in single mice during the estrous cycle supported this hypothesis; these experiments indicated that in reproductive organs luciferase accumulates at proestrus, when the level of circulating es-tradiol is highest; conversely in non-reproductive organs the maximal accumulation of luciferase occurs at diestrus. Treatment with the IGF-1 receptor antagonist JB-3 abolishes the pick of ER transcriptional activity observed in diestrus in non-reproductive organs, but not the pick at proestrus in reproductive tissues. Altogether the data accumulated suggest that estrogen may principally exerts its action in reproductive tissues, while IGF-1 play a major role in the activation of ER in non reproductive organs.
A. Maggi, None.
Plenary Session II: Biophysical Limits and Challenges in Molecular Imaging
Abstract ID: 005
Nuclear techniques are among the most sensitive available for molecular imaging in mouse models, and include methods known as planar imaging, single photon emission computed tomography (SPECT) and positron emission tomography (PET). Challenges in utilizing these techniques in the mouse include balancing the need for both high spatial resolution and sensitivity, physical limitations related to the process of radioactive decay, and obtaining data that are quantitative either in a relative or absolute sense. There also is a need to better understand the detection limits of these technologies, and to develop improved instruments that are accessible and easy-to-use, and that at the same time preserve the quantitative potential of the data. The challenges and opportunities of using nuclear imaging technologies for sensitive and quantitative molecular imaging in the mouse will be presented and discussed. This will cover a range of topics, including imaging instrumentation, image reconstruction, image processing and analysis, physiologic gating, radiation dosimetry, radiotracer mass and specific activity, and multimodality imaging as they relate to the current and future practice of in vivo nuclear molecular imaging.
S.R. Cherry, Concorde Microsystems 2; Radiation Monitoring Devices 1.
Abstract ID: 006
Imaging with red or Near-Infrared (NIR) light can be used for quantifying hemoglobin, water, lipids, scatterer concentration or endogenous and exogenous fluorophores. However decisions in the system design will inherently limit the ability to extract information from the measurements, and unlike standard clinical imaging systems, very few systems are of a similar design. The fundamental performance of this type of system will be presented with the perspective of contrast-resolution analysis, and with respect to what the systems are being used for. While high resolution is required in all detection and sceening applications, it is the contrast-resolution that is required for tissue characterization or spectroscopy applications. Resolution may be maximized by projection imaging or surface imaging approaches, yet contrast-resolution is maximized through tomographic approaches. Examples of these principles will be presented from near-infrared breast cancer imaging. In this setting, broadband spectral and temporally-resolved or phase-resolved systems are required for accurate quantitative imaging of hemoglobin in a scattering media. Also, coupling NIR systems to MRI or CT are now providing perhaps the most accurate measurements of these parameters in vivo. In fluorescence imaging, quantification methods have been developed over many years using microsampling probes and diffuse fluorescence spectroscopy algorithms, and now the microsampling approach can be used for quantitative surface imaging of fluorescence. Advanced tomographic fluorescence imaging can provide a complement to endogenous chromophore imaging by allowing imaging of metabolic parameters, yet even in this setting, the system design will inherently limit the information that can be derived from the measurements. A contrast-resolution analysis of fluorescence imaging systems will help determine the optimal configuration for detection/screening or tissue characterization applications.
B.W. Pogue, None.
Abstract ID: 007
There are a variety of processes that the brain performs at a very local level requiring high resolution imaging approaches. Inspired by the success of functional MRI techniques to monitor changes in hemodynamics due to changes in neural activity, there is growing interest in increasing the information available from MRI of the brain. We have demonstrated the usefulness of two molecular and cellular imaging agents for MRI of brain; manganese ion and micron sized iron oxide particles (MPIOs).
Manganese enhanced MRI (MEMRI) can give at least three useful types of contrast. Due to its ability to enter cells on voltage gated calcium channels and report on calcium influx, MEMRI enables imaging of active regions of the brain. Once inside cells in the brain, manganese will move along appropriate pathways enabling MEMRI based neuronal track tracing. Finally, MEMRI gives contrast that enables non-destructive imaging of cytoarchitecture. Recent experiments demonstrate that MEMRI is specific at the level of individual glomeruli in the olfactory bulb and neuronal layers throughout the brain.
Nanometer sized, iron oxide particles (USPIOs) are finding use for MRI studies of cell tracking. It would be very useful to label endogenous stem cells in the rodent brain that are known to migrate and populate the olfactory bulb. A drawback of USPIOs is that millions need to get incorporated into a cell. Recently, we have demonstrated that polymer coated MPIOs are readily taken up by a wide variety of cells and that single MPIOs can be detected in cells in culture. MPIOs should enable in vivo labeling strategies that do not require efficient uptake. Injection of MPIOs into the ventricle of rats leads to labeling of cells that migrate from the ventricle to the olfactory bulb. This should enable clarification of the role of these cells in olfactory bulb function.
A.P. Koretsky, None.
Symposium I: Linking Imaging, Proteomics and High Throughput Screens
Abstract ID: 011
Each member of a phage-display library is a natural or random peptide fused genetically to a coat protein on the surface of a bacterial virus. The displayed peptide is specified by the coding sequence of the recombinant coat protein gene inside the virus particle. When the virus is propagated by infecting fresh bacterial host cells, therefore, the displayed peptide replicates concomitantly. An individual virus particle or viral clone displays one or more copies of a single peptide, but a typical large phage display library would comprise about 10 billion clones altogether, and therefore represent 10 billion different peptides. Because the peptides are available on the surface of the virus particles, peptides that bind some target receptor can be specifically isolated by “affinity selection,” a simple application of standard affinity purification techniques. Thus the target is immobilized on a suitable medium (e.g., the plastic surface of a petri dish), the surface is reacted with the entire library, unbound viruses are washed away, and finally target-bound phage are eluted under conditions that loosen the bonds between the immobilized target and the virus-borne peptide without interfering with viral infectivity. Because a peptide that has been affinity-selected in this manner remains attached to the phage particle that contains its coding sequence, it can in effect be cloned and replicated indefinitely simply by infecting fresh bacterial host cells. By this means, a huge initial population of peptides can be effectively surveyed for exceedingly rare peptides with high affinity for any given target_including cancer-specific biomolecules or the surfaces of whole cancer cells. Application of phage display to cancer imaging will be illustrated by affinity-selection and optimization of peptide ligands for the Thomsen-Friedenreich antigen, a carbohydrate epitope that is highly expressed in many types of cancer cell.
G.P. Smith, None.
Abstract ID: 012
Glioblastoma multiforme (GBM) is a primary brain tumor with poor prognosis and low survival rate. Morphologically GBM is heterogeneous, and tissue sampling for micoarray analysis from these tumors are difficult due to this heterogeneity. Contrast-enhanced MRI using Gd(DTPA) is a powerful technique to identify regions with increased vascular permeability, and vessel density. Patients diagnosed with GBM, without any prior surgical, chemotherapy or radiotherapy procedures were scanned on a GE 1.5T MRI scanner using standard T1-and T2-weighted pulse sequences and Magnevist (Gd(DTPA), Berlex Inc., NJ) as contrast agent. Samples from regions with contrast agent accumulation (contrast-enhancing, CE) and regions that do not take up contrast agent (non-enhancing, NE) were collected for gene expression profiling using oligonucleotide microarray analysis. Tissue samples from the CE and NE areas of 13 patients reveal significantly distinct gene expression patterns. These results show laminin receptor, insulin-like growth factor binding protein −2 (IGFBP-2), IGFBP-3, IGFBP-5, heat shock protein 90 and autotaxin were all up-regulated in the CE regions as compared to the NE region. Immunohistochemical staining confirmed correlation of protein expression patterns with the observed genomic profile. Since in the CE region of the tumor the BBB is compromised, we evaluated the presence of proteins with low molecular weight (MW<30 kD) in the serum. We hypothesized that these proteins could enter systemic circulation and be detected in the patient serum due to the high vascular permeability. Preliminary results from ELISA performed on 7 patients indicate that IGFBP-2 has a higher mean value (88.1 ng/ml ±25.2) in GBM serum as compared to healthy individuals (55.0 ±11). However, other potential markers such as IGFBP-3, and aFGF do not exhibit any difference between GBM patients and controls. We conclude CE-MRI guided sampling and microarray analysis can be used to evaluate targets in permeable regions of the tumor.
S. Guccione, None.
Abstract ID: 013
In order to identify novel molecular targets for ovarian adenocarcinoma we used the “one-bead one-compound” combinatorial library method to create random peptide libraries on 90μm Tentagel beads. Using a “whole cell binding assay” these libraries were screened to identify novel ligands that were specific for these tumor cells. Specificity studies as well as structure activity relationship and computer modeling studies were performed on the identified peptides to confirm true receptor-ligand interactions. The amino acid sequence of the peptide beads were determined using Edman degradation and peptide “OA02“-Ebes-Ebes-Lys(DOTA) was synthesized on Rink Amide MBHA resin using standard Fmoc/tBu methodology. Ethylene glycol based hydrophilic linkers were used to separate the peptide from Lys(DOTA) moiety. The allyloxycarbonyl protecting group was removed selectively using 0.5eq Pd[PPh3]4 and 15eq phenylsilane in DCM for 2×30min. Protected DOTA (tBu)3-OSu 1.5eq was coupled to the ε-NH2 of lysine residue in DMF, pH was adjusted to 9 using DIEA and the mixture was incubated at room temperature for 18hrs. Product was characterized by RP-HPLC and MALDI-TOF mass spec. The DOTA-peptide was radiolabeled using Cu-64 obtained from Washington University School of Medicine (NCI R24 CA 86307). Various buffers, pH and temperature conditions were investigated. Purification was performed using C18-SepPak and analysis performed using radio-RP-TLC. Preliminary PET studies were performed using MicroPET in nude mice bearing subcutaneous ovarian tumor xenografts. Radioactivity was observed in the tumor with some liver and kidney uptake however no obvious radioactivity was observed in the negative control tumor. Further in vivo imaging studies are underway to investigate tumor uptake and specificity.
O. Aina, None.
Abstract ID: 014
A cysteine constrained phage library was used to search for peptides that bind to the TAG72 antigen as possible new targeting agents for gastric, colon, ovarian and breast cancers. An affinity column of TAG72 positive B72.3 antibody was used to purify commercial TAG72. The purified antigen was absorbed to plates along with the column eluant free of TAG-72 and BSA to select against non-specific phage, and used in succession prior to addition of the library to the TAG72 plates. Twenty-three clones from the third round of selection with the f88-4/cys6 phage library (G. Smith, U. of Missouri) were sequenced. Consensus phage were identified and were radiolabeled with 99mTc via MAG3 along with a random phage as control. The binding properties of the radiolabeled phage were evaluated with TAG-72 positive LS174T cells in vitro and as tumors in vivo in mice. Three consensus peptides were identified: NPGTCKDKWIECLLNG (I); NLIWCRKEFARCTSDM (II); and LKNYCRKCSNRCTPTG (III). One clone representing each sequence was evaluated. The phage were radiolabeled at 20%-60% efficiency and were 90% radiochemical pure after PEG/NaCl purification. Binding in LS174T cells (6×105) of each phage (at 1.5 ×1011 virions/well) showed a 4.5-fold and 1.5-fold greater accumulation for phage I and II over the control, while phage III accumulated similarly to the control. Administration of 99mTc-phage I, II and control phage to mice with tumors showed significantly greater accumulation in the tumored thigh of 0.51% ID/organ (sd 0.11, n = 3) for phage I compared to phage II and to the control (0.28%, sd 0.06 and 0.29%, sd 0.06, n = 3) respectively (p = 0.049). In all cases the highest activity at 4 hr was in the stomach, small and large intestine and liver. These results suggest that one peptide may have been identified by phage selection with potential as a ligand to the TAG72 antigen for scintigraphic tumor detection.
M. Rusckowski, None.
Abstract ID: 015
Peptide nucleic acids (PNAs) have shown great promise as antisense agents to block gene expression at the mRNA level, but little and controversial information is available on the bioavailability of PNAs administered systematically. Data indicate that these compounds are stable in plasma, excreted in the urine, and that their uptake in other organs is very low. Functionalization of the PNA backbone may change the physico-chemical properties of the PNA, and it is plausible that it could also influence PNA biodistribution and pharmacokinetics behaviour. Furthermore, such changes may be introduced without major influence on the DNA/RNA hybridisation potency of the PNA. Therefore, it could be of significant medicinal chemical interest to develop methods that would allow (predictable) modulation of the pharmacokinetics profile of a given PNA.
In this perspective, PNA fmoc-protected thymine monomers containing glycosyl substituents (galactosyl, mannosyl, fucosyl, N-acetyl-galatosaminyl and N-acetyl-glucosaminyl) or peptidic substituents (lysine, leucine, arginine and glutamic acid), in the á-amino acid position were synthesized and then incorporated into a PNA decamer. A cysteine was attached to the amino terminal of the PNA decamers for easy conjugation to a [18F] radiolabeled N-(4-fluorobenzyl)-2-bromoacetamide. The pharmacodistributions of the labeled glycosylated PNA were studied in four rats simultaneously with the Siemens EXACT HR+ PET camera during two hours after intravenous administration.
The pharmacodistribution in rats of PNA oligomers was profoundly changed by glycosylation, and these changes were accurately and reliably documented by PET imaging. Liver subcellular localization of the PNA sequence containing three N-acetyl-galatosaminyl substitutions was also observed by direct in vivo microscopy, showing membrane adsorption followed by intracellular uptake of this particular derivative. These results could be of great significance for PNA drug development as they should allow modulation and fine tuning of the pharmacokinetics profile of antisense PNA drug leads, and help to turn PNAs into pharmacologically active compounds.
B. Tavitian, None.
Symposium II: Instrumentation and Computational Challenges in Molecular Imaging
Abstract ID: 016
Position sensitive gamma-radiation detectors equipped with collimators (Single Photon Emission Computed Tomography, SPECT) or without collimators (Positron Emission Tomography, PET), have been used for quantitative in vivo imaging of the distribution of radionuclides with low concentrations (pico- and nano molars) in animals and humans for a few decades. Until recently, the best resolution achieved in practice in in vivo rodent imaging is on the order of 1.5 to 2 mm. With SPECT utilizing pinhole gamma-cameras, it is possible to achieve a better resolution if a sufficient fraction of emitted photons can be detected in the mean while. After a short introduction and overview of pinhole SPECT imaging systems it will demonstrated how resolution improvements can be obtained by focusing a high number of dense, shaped, micro-pinhole collimator apertures exclusively on the tissues of interest. Images of test distributions of labeled molecules, as well as different molecule distributions in the mouse representing bone metabolism in the spine and myocardial perfusion have been obtained at a resolution below 0.5 mm. The system can differentiate between organ components with 0.1 micro-litre volume, and shows tracer uptake in the papilary mucles in the heart and in parts of the spinal processes in the spine. Possible future applications and further increase of image resolution are discussed.

F.J. Beekman, Philips 1; MILABS 4.
Abstract ID: 017
Terahertz (THz) radiation is electromagnetic wave situated between infrared light and microwave radiation (approximately in the frequency range of 0.1 to 10 THz). Recently, THz technology has attracted a lot of attention for biomedical imaging due to its noninvasiveness and low Rayleigh scattering compared with optical waves. In particularly, many large proteins and DNA molecules have collective vibrational and rotational modes in the THz range, which is suitable for THz molecular imaging application. In order to achieve high spectral resolution molecular imaging, high-power high-efficiency widely-tunable-wavelength narrowband THz sources are needed for the improvement of image contrast and system sensitivity. Recently, we had demonstrated a novel edged-coupled membrane photonic transmitter that is based on a high bandwidth-efficiency product MSM traveling-wave photodetector and a co-planar-waveguide fed slot antenna, which has advantages including record-high conversion efficiency (1.1×10−3 at 645GHz), high THz output power (demonstrated average power is 3.9μW at 645GHz), frequency tunability, and compactness. Based on this device we constructed a novel quasi-CW THz image system, which is promising on high signal-to-noise ratio (SNR>100), low data-acquisition time, and wide wavelength-selectivity for molecular imaging application. Utilizing this new system, we have successfully acquired 2D transmission images of both dried and fresh (with water) biological samples at different radiation frequencies. During experiments all samples were contained in thick optically opaque plastic boxes and are thus invisible, but could be seen through by THz waves due to molecular absorption at specific THz frequencies. In the conference, wavelength-dependent bio-imaging will be discussed optically and biologically.
J. Lu, None.
Abstract ID: 018
Magnetic resonance imaging offers exceptional potential for clinical molecular imaging due in part to its high resolution, superior soft tissue contrast, and lack of ionizing radiation. However, contrast agents based on T1 and/or T2 contrast effects are plagued by competing background proton signal and the need for high payloads for visualization. The use of fluoridated contrast agents in conjunction with 19F MRI may enable unique and quantifiable visualization of targeted epitopes because of the lack of endogenous fluorine in any measurable quantity in the body. Our lab has developed a liquid perfluorocarbon nanoparticle contrast agent (20% v/v perfluoro-15-crown-5) for molecular imaging that incorporates a targeting ligand into its outer lipid layer. To define the lower limits for detection of this agent with 19F MRI, we used a clinical 1.5 T Philips MR scanner outfitted with a special channel tuned for fluorine nuclei and a 4-inch square surface coil constructed in our lab. To find the minimal amount of agent needed for detection, we acquired spectra (8kHz bandwidth, 144 averages, 4092 samples, 4 s pre-delay) from different volumes of emulsion. For imaging, the emulsion was serially diluted and imaged using a 2D gradient echo sequence (TR = 37ms, TE = 1.95ms, resolution = 1×1times10mm). Spectroscopy revealed that the lowest detectable amount of perfluorocarbon nanoparticles is 5.5 μmol of 19F or about 1×1010 nanoparticles. In order for the particles to generate enough signal-to-noise to be visibly apparent (SNR = 5), they should be present in a concentration of 650 pM or greater (see Figure). These results suggest that 19F MR molecular imaging of cellular epitopes present in concentrations in the high picomolar range could be feasible with 19F MRI and targeted fluorocarbon nanoparticles at 1.5 T.

(Left) 19F MR image of perflourocarbon nanoparticles in a concentration of 550 pM. (Right) Signal to noise ratio (SNR) as compared with nanoparticle concentration. The lowest concentration that will produce adequate contrast (SNR=5) is estimated to be 650 pM.
A.M. Morawski, None.
Abstract ID: 019
Different groups recently showed the feasibility of molecular imaging by using specific ultrasound contrast media (USCM). USCM are an ideal sensor for molecular imaging primarily because of the outstanding sensitivity of their detection. Based on Stimulated Acoustic Emission (SAE), a unique “signature” of microparticles (MPs), even single MPs can be detected in tissue. However, limited by the spatial resolution of ultrasound, these signals are displayed in mm-size. Consequently, two or more MPs within 1 mm cannot be discriminated. Concentrations above 1000 MP/mL lead to signal-saturation in the image and a quantification above that concentration is not possible.
We developed Single Particle Acoustic Quantification (SPAQ), a new quantitation-mode, which enables the quantification of MPs up to 100000 MPs/mL, thereby decreasing the slice-thickness of the ultrasound image (color Doppler) down to 10 μm.
We first verified the method in an agar phantom containing 30000 MPs/mL and then corroborated our findings in the livers of rats, which had received tenfold different MP-doses. We could find a good correlation between the MP-concentrations in the livers and the given doses using SPAQ. In conventional color Doppler mode however, no difference was detectable due to a complete signal saturation in all livers.
Molecular imaging requires the identification and spatial localization of a specific marker. However, many questions concerning target density or up- and down-regulation of marker-molecules under treatment also require a quantitative analysis. While common ultrasound techniques fail to quantify high MP concentrations, SPAQ enables the quantification of MPs up to 100,000/mL. Moreover, the imaging slice thickness is reduced to the lower μm-range and large volumes can be mapped with high accuracy and reproducibility.
All these advantages make SPAQ an ideal tool for quantitative molecular imaging.
Abstract ID: 020
In this talk I will review the fundamentals of signal, noise, contrast and imaging time relevant to in vivo MRI of targeted cells. The fundamental variables are field strength, tissue relaxation, animal size, tissue conductivity, RF coil dimensions and conductivity. I will also explain the important concept of tissue noise dominance, which is both necessary and sufficient for optimal SNR.
Contrast in targeted cell tracking depends on both the tagging methods (e.g., magnetic nanoparticles, or paramagnetic contrast agents) as well as the imaging sequences employed. Both positive and negative contrast imaging methods exist and fundamental tradeoffs will be discussed. In particular, partial voluming effects in a negative contrast method can obscure the dropout of a small volume of targeted cells. With positive contrast methods, partial voluming issues are minimized. Hence, this could enable more reliable cell quantitation, and could allow for faster projection imaging. I will discuss two new pulse sequences that create positive contrast for cells labeled with magnetic nanoparticles. One is a gradient recalled echo sequence [1,2] where the slice selection is not refocused, so that only spins near a magnetic dipole field are refocused. The other is a frequency selective off resonance spin echo sequence that uses the dipole field much like a slice selection gradient [3].
We will also include an analysis of the minimal detectable number of tagged cells. Because the magnetic dipole field from the tagged group of cells extends several radii away from the center, considerable volume amplification of the MRI signal is possible. Future improvements in targeted cell MRI will be discussed.
S.M. Conolly, None.
Symposium III: Advances in PET and SPECT Radiochemistry
Abstract ID: 021
The importance of the specific radioactivity (SA) has been well recognized in the field of the PET neuroreceptor imaging with 11C-labeled compounds, such as [11C]FLB457 (for dopamine D2 receptor) and [11C]PE2I (for dopamine transporter). However, it has also been recognized that it is not easy to synthesize 11C-labeled compounds with a high SA. [11C]CH3I is the most widely used precursor in the radiosynthesis of 11C-labeled compounds, and is synthesized via [11C]CO2 or [11C]CH4 generated directly in the target chamber or by the reduction of [11C]CO2 with a catalyst under a H2/He gas flow. The method via [11C]CO2 was developed originally and used widely for the synthesis of [11C]CH3I, having the advantage of a high radiochemical yield and disadvantages of a low SA and tedious preparation procedures. The deterioration of the SA could easily be caused by contamination by CO2 from the atmosphere, the target chamber, the LiAlH4/THF solution, and so on. On the other hand, the method via [11C]CH4 was developed later and has the advantages of a high SA and repeated synthesis of [11C]CH3I without tedious work, and the disadvantage of a low radiochemical yield. It is more difficult for the deterioration of the SA is to occur in this method, since a major carrier source of carbon, the LiAlH4/THF solution, can be avoided and the abundance of CH4 (1.6 ppm) in the air is lower than that of CO2 (330 ppm).
By improving the procedures of the irradiation, the reagent preparation and so on, we have successfully synthesized [11C]Ro15-4513 (for benzodiazepine receptor) and [11C]FLB457 with SA higher than 400 GBq/mmol by the former method, and with SA around 4000 GBq/mmol by the latter method.
Many of the attempts at our institute to achieve a high SA will be presented in this talk.
K. Suzuki, National Institute of Radiological Sciences 1, 5.
Abstract ID: 022
M. Yu, None.
Abstract ID: 023
Malignant melanoma is the sixth most commonly diagnosed cancer with an increasing incidence in the United States. Moreover, melanoma metastases are resistant to conventional chemotherapy and external beam radiation therapy. Therefore, it is crucial to develop novel agents for early detection of primary lesions and their metastases.
Y. Miao, None.
Abstract ID: 024
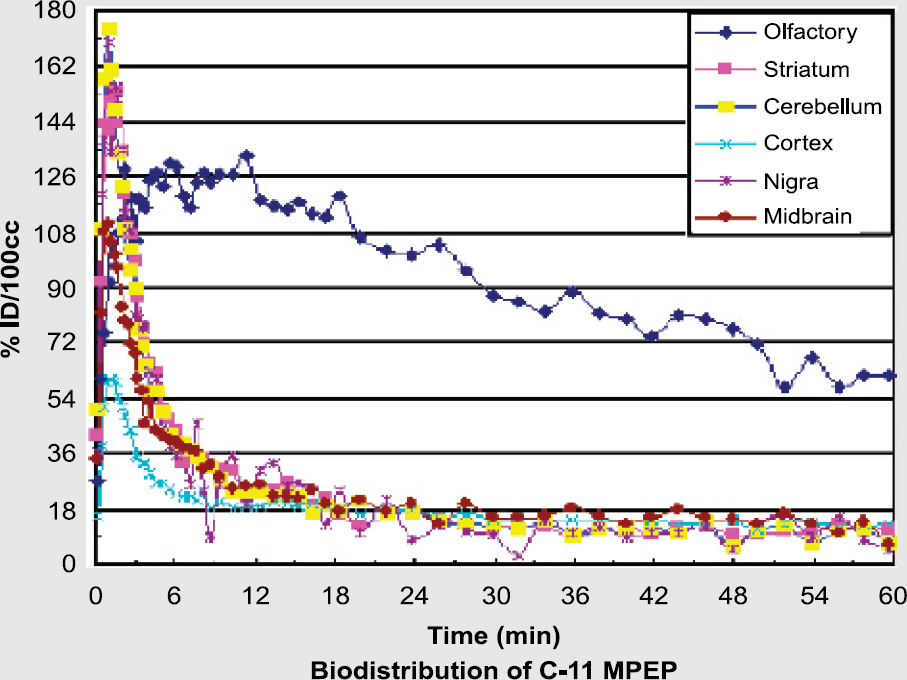
Metabotropic glutamate subtype-5 receptors (mGluR5) are implicated in many cerebral disorders, such as depression, schizophrenia and Parkinson's disease. mGluR5 are localized post-synaptically mostly in thalamic nuclei, cortex and hippocampus. We aim to develop a radioligand that might be used with PET to explore the role of mGluR5 in neuropsychiatric disorders. 5-Methyl-(3[(2-methyl-1,3-thiazol-4-yl) ethynyl]pyridine) (5-Me-MTEP) has moderate lipophilicity (clogP=2.42) and also higher affinity (IC50= 3 nM) than some related and weakly sensitive PET radioligands. These favorable properties suggest that [11C]5-Me-MTEP might be a good candidate radioligand for imaging mGluR5. Here we report the radiosynthesis of [11C]5-Me-MTEP and initial findings on its behavior as a PET radioligand.
Treatment of 5-trimethylstannyl-MTEP (1.5 mg) in tetrahydrofuran with [11C]iodomethane in the presence of tris-(dibenzylideneacetone) dipalladium(0) and copper(I) iodide for 5 min at 80°C gave [11C]5-Me-MTEP (15–20 mCi; >1.5 Ci/μmol) ready for i.v. administration into rhesus monkey and monitoring of its regional brain kinetics with PET.
After radioligand (5.73 mCi) injection alone, the standard uptake value (%ID/cm3 × g body mass) reached 530% in cerebellum at 2 min, decreasing to 50% at 48 min. The highest ratios of radioactivity to that in cerebellum were seen in striatum and inferior frontal cortex (~1.6 at 12.5 min). Pretreatment of the same monkey with the mGluR5 ligand, 2-methyl-6-(3-methoxy-phenylethynyl)pyridine (2 mg/kg i.v.) at 10 min before radioligand (5.64 mCi) injection reduced all ratios to near unity.
[11C]5-Me-MTEP is an mGluR5-selective PET radioligand that merits further investigation of itself and also serves as a lead for further radioligand development

F.G. Simeon, None.
Symposium IV: Tracking Infectious Agents and Imaging Host Response
Abstract ID: 026
Herpes simplex virus 1 (HSV-1) is a common and significant neurotropic human pathogen. HSV-1 replicates peripherally in epithelia, enters axonal terminae, and is transported retrogradely to sensory nerve ganglia where the virus may establish latency or progress to life-threatening infection of the central nervous system. HSV1 also can produce fatal systemic disease, typically in neonates or immunocompromised patients. Studies of viral and host factors that influence pathogenesis largely have used experimental mouse models that rely upon sacrifice of infected mice to determine distribution and titer of virus. While this experimental paradigm has provided important data, it precludes real-time investigations of the same animal over the entire course of disease progression. We have developed recombinant HSV1 reporter viruses that express firefly luciferase or GFP to enable imaging of viral infection in living mice. The recombinant viruses reproduce the growth kinetics of wild-type HSV1 in cell culture and in vivo, and viral titers at a defined anatomic site correlate directly with bioluminescence quantified with non-invasive imaging. This system has been used to monitor efficacy of anti-viral therapy in mouse models. Using mice with genetic deletion of interferon receptors, we have used these viruses to identify essential functions of innate immunity in preventing systemic dissemination of HSV1 infection. These studies demonstrate how noninvasive imaging can be applied to studies of viral-host pathogenesis and show that this experimental approach can identify new mechanisms of viral disease.
G.D. Luker, None.
Abstract ID: 027
Modern medical and surgical practice has come to rely increasingly on various types of implanted medical devices and prosthetics. A major problem with many of these devices is their colonization by microorganisms that form biofilms. Not only are these biofilm infections highly resistant to antimicrobial treatment, but reestabishment of the infection may occur soon after treatment has ended. Little is known about such biofilm infections, especially in vivo, mainly due to a lack of reliable nonintrusive longitudinal monitoring techniques. We have developed a direct real-time biophotonic monitoring method to visualize implanted catheter-based infections on intravascular or urinary catheters in mice using bioluminescent bacteria and a low-light imaging system (Xenogen Corp, Alameda, CA). Jugular vein catheterized mice showed increased risk for catheter related colonization and metastatic disease following bloodstream infection with Staphylococcus aureus. Similarly, placement of catheters precolonized with uropathogens such as Pseudomonas aeruginosa and Proteus mirabilis in the bladder caused spread of infection from the implant to the bladder causing cystitis and infection of the kidneys. In the latter model, catheter associated infections not only persisted significantly longer than in mice challenged by infecting the bladder with bacterial suspensions, but also catheter related infections relapsed a few days after termination of antibiotic treatement. In contrast, bacteria injected in suspension were easier to irradicate. Whole body bioluminescent imaging provides a noninvasive method for studying device-related infections from outside the infected animal without exogenous sampling. It allows investigators to not only track the course of infection, but also to rapidly monitor the efficacy of antimicrobial therapies both without disruption of the biofilm and in individual animals over time.
J.L. Kadurugamuwa, Xenogen Corporation 5.
Abstract ID: 028
Pneumonia is an infection of the lungs, which can be caused by a variety of microorganisms, including viruses and bacteria. During the host response in pneumonia, the bacteria are cleared from the lungs. There are two important parts of host defense against bacteria: recruitment of white blood cells (particularly neutrophils) and the killing of the infecting bacteria. Presently, a common method of measuring bacterial clearance utilizes dilutional plating and colony counts. An alternative method of measure, bioluminescence imaging, offers a quicker and less variable measure of bacterial clearance and the ability to make measurements in the same animal over time.
E. coli bacteria (Xen17, Xenogen Corp.) expressing the luc gene were instilled into the left lungs of C57Bl/6 mice. Images were taken at various time points post-bacteria instillation. Figure 1 shows the accumulation of bacteria during each time point. In the left lung, the number of bacteria initially increased (6 h) and then decreased by 24 h, and clearance was complete by 48 h. At 24 h, bacteria were present and the nasopharynx and the abdomen. No bacteria were observed at 72 h. To verify which organ contained the bacteria at 24 h, the abdominal cavity was opened and imaging showed that the bacteria were located in the colon. Further imaging studies verified that the bacteria was located in the inner lumen of the colon and verification was made using cell cultures. The ability to characterize bacterial clearance during pneumonia using bioluminescence imaging will allow a more effective alternative to studying the molecular mechanisms behind host response. Further studies will include the role of ICAM-I and the effect of IFN- γ.
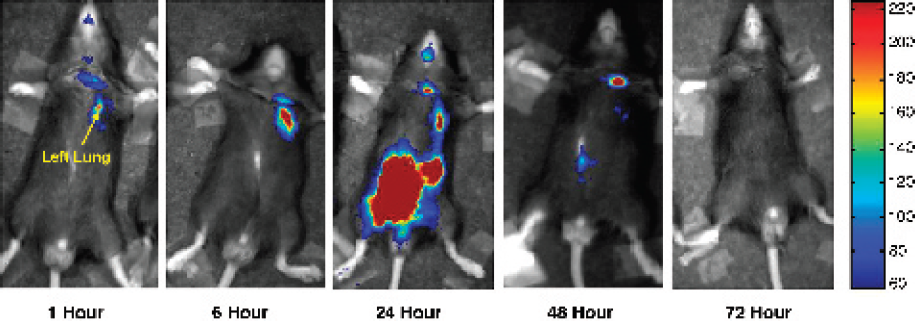
An initial accumulation of bacteria was detected in the nasopharynx and the left lung at the 1 and 6 h time points. Bacteria was present at the site of installation (neck). At 24 h, the bacteria could still be detected in the lungs but the majority was present in the abdominal area. After 72 h, there was no bacteria detected.
P. Cheung, None.
Abstract ID: 029
Since cells infected by viruses expose viral antigens on their membranes, ligands specifically recognizing these antigens can be used as vectors of contrast agents to design innovative contrast media. We cloned by phage display technology from a human library, obtained from bone marrow donor patient, a human recombinant antibody's fragment (HrFab) specific for an Herpes Simplex Virus (HSV) (both Type 1 and 2) infection-associated antigen.
To assess its possible use for imaging, a set of in vitro experiments was performed using VERO cells infected with HSV-1 VR733 ATCC-strain and HSV-2 VR734 ATCC-strain. We conjugated anti-HSV HrFab with Gd3+ hexahydrate. To assess conjugation efficacy, dialized immunoconjugates were detected by MRI and compared with controls (Table 1).
Since conjugation with a contrast agent may result in impaired affinity to target, immunoreactivity of the immunoconjugate was confirmed in vitro by ELISA in order to test post-labeling affinity. MRI images were obtained in 1.5T MRI unit. The in vitro experiments showed excellent detection of infected cells with a minimal HrFab/Gd complex dose.
In order to evaluate the in vivo use of this HrFab preliminary experiments were performed on an animal model. Rabbit herpetic keratitis is a suitable HSV infection model because it is easy to perform, and allows to monitor infection avoiding the risk of death of animals by encephalitis.

L. Selan, None
Abstract ID: 030
RNA interference (RNAi) inhibitors are an exciting new class of therapeutics that have the potential to treat many diseases. RNAi is an antiviral mechanism that responds to double-stranded RNAs by silencing homologous genes. This endogenous pathway can be used to silence any target RNA by introduction of short interfering RNAs (siRNAs) or short hairpin RNAs (shRNAs) expressed from DNA templates.
We have used bioluminescence imaging (BLI) as a tool to follow gene silencing in living mice in real time. Initial proof of principle experiments used siRNAs targeting luciferase. This model has been very useful for optimizing parameters involved in efficient gene silencing. We used BLI to assess the efficacy of naked RNAi delivery as well as expression of shRNAs from plasmids and adenoviruses. Chemically stabilized RNAi triggers were also tested.
After optimization with BLI, we used RNAi to target hepatitis B virus (HBV) in mice. Four hundred million people infected with HBV are at significant risk for chronic liver disease and hepatocellular carcinoma. We show that RNAi inhibits HBV replication in cultured cells and in immunocompetent and immunodeficient mice. Transfection with an HBV plasmid initiated viral replication. Co-transfection with plasmids expressing shRNAs homologous to HBV mRNAs induced RNAi. Northern and Southern analyses of mouse liver RNA and DNA showed dramatically reduced levels of HBV RNAs and replicated HBV genomes upon RNAi treatment. Secreted HBV surface antigen was reduced by 94.2% in culture and 84.5% in mouse serum. HBV core antigen immunohistochemistry revealed a >99% reduction in stained hepatocytes upon RNAi treatment. Thus, RNAi effectively inhibited HBV replication in cultured cells and mammalian liver. This approach can be adapted to treatment of diverse diseases but efficient delivery of RNAi remains a major obstacle. BLI remains an attractive tool to assess delivery methodologies.
A.P. McCaffrey, Benitec 6; Alnylam 6.
Plenary Session III: Imaging Signal Transduction, Transcription and Cell Cycle in Vivo
Abstract ID: 031
We have developed a transgenic mouse that expresses firefly luciferase from the E2F1 promoter. This construct allows bioluminescence imaging of cycling cells, primarily those in S and G2 phases of the cell cycle. We have crossed this line to our tv-a transgenic lines allowing somatic cell gene transfer into glial progenitors with RCAS vectors. In this double transgenic line we are able to generate oligodendrogliomas at high penetrance by infecting with a PDGF-encoding RCAS retroviral vector. These glioma-bearing mice can be imaged using bioluminescence for the presence of gliomas. These tumors can be followed over time for tumor development and for response to therapy in preclinical trials. Using this system we are using small molecule inhibitors of signaling components to dissect the requirements of signaling pathway activity in the proliferation of glioma cells in vivo.
E.C. Holland, None.
Abstract ID: 032
Cell cycle checkpoints responding to damaged or unreplicated DNA delay the activation of cyclin dependent protein kinases (CDKs) to temporarily arrest the cell division cycle. Cell cycle checkpoints are essential for maintenance of genomic integrity. We identified the CDC25 family of protein phosphatases as key targets of negative regulation by the DNA damage and replication checkpoints. The CDC25 phosphatases positively regulate cell division by dephosphorylating and activating cyclin dependent protein kinases (CDKs). In humans and rodents, there are three family members CDC25A, −B, and −C, encoded by three genes. CDC25 family members can be distinguished based on their intracellular localization, their abundance and/or activity throughout the cell cycle, the CDKs that they target for activation and whether they are overexpressed in human cancers. Furthermore, the CDC25A-and CDC25C-regulatory pathways are inhibited by UCN-01, a protein kinase inhibitor currently in clinical trials for cancer treatment. My laboratory is interested in defining the contributions made by individual members of the CDC25 family to cell cycle- and checkpoint control in mammals and in determining whether CDC25-regulatory pathways are valid targets for the development of anti-cancer agents. With this goal in mind, molecular imaging strategies are being applied to study CDC25-regulatory pathways in intact cells and in mice. Finally, Chk1, a Cdc25A regulatory kinase, is being evaluated as a therapeutic target for cancer treatment.
H.M. Piwnica-Worms, None.
Plenary Session IV: Imaging Cell Fates and Function: Stem Cell Biology and Immunology
Abstract ID: 034
Leukocytes circulating in the blood vessels are recruited to specific sites of tissue injury or inflammation by interacting with molecules expressed on the vascular endothelium during an immune response. Cancer cells that are shed into the circulation may similarly interact with the endothelial cells in the initial steps of metastasis, i.e., attachment to blood vessel walls and migrating into the extravascular space. In vivo flow cytometry is a new method for real-time detection and enumeration of circulating cells in live animals without the need to draw blood samples. With this method we have determined the circulating kinetics of leukocytes and leukemic cancer cells, and measured how the circulation kinetics is affected by interfering with their binding to the endothelial cells. The combination of in vivo flow cytometry and in vivo microscopy allows us to track both the circulating compartment and the tissue compartment with single cell resolution in live animals.
C.P. Lin, None.
Abstract ID: 035
Recently there has been a tremendous progress in labeling different kinds of stem cells and other mammalian cells with superparamagnetic iron oxides (SPIO) to monitor the temporal-spatial migration of cells by magnetic resonance imaging (MRI). Although SPIO nanopaticle is an excellent susceptibility MR contrast agent for monitoring cell migration the iron in the SPIO core has the potential to be toxic to the cells if it is dissolved and released in the cytoplasm. It is important to note the fate of the incorporated SPIO particles in the endosomes/lysosomes of the labeled cells, their effect on the cellular iron homeostasis and free radical formation, cellular viability, apoptosis and functional capacity. This presentation will discuss the issue of toxicity of the SPIO nanoparticle labeling of cells and how magnetically labeling techniques can be utilized to monitor the migration of labeled stem cells and other cells for repair, replacement and therapeutic strategies in a variety of experimental systems. A.S. Arbab, None.
Abstract ID: 036
The early events in hematopoietic reconstitution from stem cells occur infrequently and are randomly distributed throughout the hematopoietic compartments, thus sensitive whole body imaging approaches are necessary to study these processes and their regulation. We used in vivo bioluminescence imaging (BLI) to monitor engraftment and hematopoietic reconstitution from highly purified luciferase-labeled hematopoietic stem cells (HSC) after transfer into irradiated syngeneic recipient mice. We used small numbers of HSC, and even single cells, in an attempt to assess the dynamic patterns of engraftment and reconstitution. BLI enabled us to count in vivo colony forming units resulting from transplanted HSC; discrete foci were detected in the spleen and bone marrow (BM), at a frequency that correlated with BM compartment size. These data also indicated a 20% engraftment frequency. The foci that were initially detected were observed to expand locally, seed other sites in BM or spleen, and/or recede. These different outcomes proceeded with different kinetics indicating several different patterns of reconstitution could result from a given HSC. These studies revealed dynamic and variable patterns of engraftment from highly purified HSC and indicated that the final overall contribution of an individual HSC to hematopoiesis does not depend on the specific anatomic site of initial engraftment and expansion.
C.H. Contag, Xenogen Corp. 2.
Symposium V: Imaging Markers of Cardiovascular Disease
Abstract ID: 037
Programmed cell death, also termed apoptosis, is the physiological process used by multi-cellular organisms to selectively eliminate cells that are no longer needed, have been damaged or are dangerous. Apoptosis plays an important role in embryogenesis, homeostasis and many diseases. Insight in the role of programmed cell death in disease will allow us to understand the mechanism of pathological cell loss, such as is seen in myocardial infarction, or lack of programmed cell death, for instance in cancer. Consequently, novel modes of therapy can be devised to influence the amount of programmed cell death, by either inhibiting unwanted cell death or stimulating lack thereof.
A better understanding of the kinetics of apoptosis in diseases of cell loss is crucial to guide therapeutic intervention in diseases associated with cell loss, such as myocardial infarction and stroke. In addition, a way to monitor apoptosis in diseases characterized by cell accumulation, such as cancer, will enhance the development of novel cell death inducing compounds. Therefore, a key advantage in the assessment of novel cell death modulating therapies is the development of a technology that allows to monitor or to visualize programmed cell death in patients. The most widely used method for non-invasive imaging of programmed cell death is detection of labeled Annexin A5. In this presentation will discuss the use of labeled Annexin A5 for the in vivo imaging of programmed cell death in cardiovascular diseases, such as myocardial infarction, and the unstable atherosclerotic plaque.
L. Hofstra, PharmaTarget 4.
Abstract ID: 038
Our group is applying PET-CT, SPECT and MRI techniques to monitor stem cell engraftment in a canine model of myocardial infarction. We report: 1) a PET/CT method to track transplanted bone marrow cells after intramyocardial injection; 2) evaluation of stem cell injection on infarct size using PET and MRI; and 3) development of a reporter gene approach for the monitoring of stem cell function. For the first two objectives, bone marrow monocytes were isolated and labeled with 10.1 MBq [18F]FDG. 2×107 cells were either injected directly into the infarct or through an intracoronary catheter, followed by 4h of PET/CT imaging. PET detected 27% of the label within myocardium immediately after direct injection, with 2.5% for the intracoronary route. MRI indicated a 20% greater reduction in infarct volume for the direct injection technique. To develop a reporter gene strategy, mesenchymal cells were isolated and transfected with a cDNA construct containing the coding sequence of herpes simplex virus thymidine kinase fused in-frame to green fluorescent protein (HSV1tk-gfp). After incubation with 10 μCi [131I]-FIAU for 3h, transfected cells showed a 22-fold greater uptake of [131I]-FIAU than non-transfected cells. For in vivo studies, 5×106 cells were labelled with 0.3 MBq [111In]tropolone prior to injection in order to localize the cells using SPECT. Following injection of labeled cells, 43.6 MBq [131I]-FIAU was injected i.v. Following imaging by SPECT for 22h, the dog was sacrificed and the tissue containing the injected cells was collected. There was significantly (p<0.05) greater accumulation of 131I in the tissue containing stem cells than in control tissue, indicating that FIAU was specifically taken up at the site of injection. We conclude that transplanted stem cells can be tracked using a multi-modality approach, and that functional imaging using a reporter gene strategy shows promise in a large animal model of myocardial infarct.
S. Dhanvantari, None.
Abstract ID: 039
F.A. Jaffer, None.
Abstract ID: 040
J.V. Frangioni, None.
Abstract ID: 041
MRI is particularly attractive for molecular imaging applications due to high spatial resolution, lack of ionizing radiation and ability to gather both anatomic and physiological information simultaneously. The critical limitation of MRI, however, is low sensitivity to traditional contrast agent compounds, which require millimolar concentrations for adequate image enhancement. Clearly, such agents would be useless for molecular imaging of physiological targets at nanomolar concentrations.
To overcome this limitation, we have developed a nanoparticulate site-targeted contrast agent consisting of a lipid encapsulated liquid perfluorocarbon core. The nanoparticle “platform” approach provides a natural scaffold to support an enormous paramagnetic payload (>50,000 gadolinium ions per particle). The resulting paramagnetic impact (i.e. relaxivity per binding site) is approximately one million times higher than traditional contrast agents. The structure is also ideally suited for incorporation of a wide variety of targeting ligands (antibodies, peptides and peptidomimetics) and therapeutic agents (doxorubicin, paclitaxel and fumagillin). The nanoparticle size tends to be ~200 nm in diameter, in order to support the large numbers of paramagnetic chelates. This particle size is well suited for targeting vascular epitopes expressed on the luminal aspect of endothelial cells. This provides a wide range of possible targets (angiogenic adhesion molecules, fibrin, tissue factor and collagen) associated with various pathologies (vulnerable plaques, early atherosclerosis, thrombosis and vascular injury). Incorporating therapeutic drugs into the nanoparticle provides local drug delivery, which can be confirmed and quantified noninvasively at the target of interest. This approach may improve safety and effectiveness of the drug by lowering the dosage and concentrating the compounds directly at the site of pathology. The integration of molecular imaging and targeted drug delivery may allow patients to be screened and stratified for therapy, delivery of treatment, confirmation and quantification of drug delivery, and monitoring of therapeutic effectiveness.
P.M. Winter, None.
Abstract ID: 042
Optical contrast agents, such as indocyanine green and its functionalized derivatives, have been studied to enhance the sensitivity and specificity of diagnostic optical imaging with near-infrared (NIR) light. However, due to the overwhelming scattering of light in biological tissues, the spatial resolution of traditional optical imaging modalities degrades rapidly as the imaging depth increases. Here, for the first time, we present results on non-invasive photoacoustic angiography of animal brain with NIR light and an optical contrast agent. When ICG-PEG was injected into the circulatory system of a rat, it greatly enhanced the absorption contrast between the blood vessels and background tissues. Because NIR light can penetrate deep into the brain tissues through the skin and skull, we were able to successfully reconstruct the vascular distribution in the rat brain from the photoacoustic signals. Based on differential optical absorption with, and without, contrast enhancement, a high-resolution photoacoustic angiograph of a rat brain was achieved in vivo to reflect the concentration of the exogenous contrast agent. Photoacoustic tomography can in principle be used to image various molecular contrast agents with high spatial and temporal resolution. Its potential for dynamic in vivo imaging is enormous.
L.V. Wang, None.
Abstract ID: 043
Fluorescence imaging of tissue can be made quantitative by the use of microsampling methods to image fluorescence in vivo. Through the use of both specially designed fiber probes and raster scanning imaging techniques, we demonstrate that accurate quantification of the fluorescence from porphyrins is possible. When either large fibers or broad beam imaging approaches are used the ability to quantify fluorescence in vivo, without the aid of computer modeling, there is a non-linear response between the signal and the actual concentration present. We use microsampling methods to image endogenously produced protoporphyrin IX in vivo which is induced by administration of aminolevulinic acid. The production of this fluorophore results from over active metabolism in cancer tumors in the heme synthesis pathway, and is well known to be a high contrast marker of many tumor types. In this study, we demonstrate for the first time that this can be used as a measure of tumor burdon and can be used to monitor response to therapy. Using the Dunning prostate tumor model, we track response to radiation therapy and show that non-invasive monitoring of tumor viability can be achieved. The use of noninvasive measurement of tumor metabolism through this novel marker of heme pathway synthesis can provide a useful tool to optimize therapy in experimental tumors, and is already FDA approved for use in human cancer therapy.
B.W. Pogue, None.
Abstract ID: 044
Optical molecular imaging can provide early diagnostic signs of disease, frequently before morphological changes occur. Molecular bond-specific imaging has been demonstrated in living cells using Coherent Anti-Stokes Raman Scattering (CARS) microscopy, which detects the intensity of the CARS signal from thin sections. We have developed a novel technique and molecular imaging system that we call Nonlinear Interferometric Vibrational Imaging (NIVI) which uniquely takes advantage of the coherent nature of the CARS signal. NIVI uses principles from optical coherence tomography (OCT), including sensitive interferometric heterodyne detection and phase resolution to perform optical ranging and spatial localization of the coherent CARS signal. Using various interferometric methods, two CARS signals are separately generated and interfered. One is generated from a known reference molecular species and a second is generated from a material or biological sample with unknown molecular composition. The intensity of the interference signal is a measure of the concentration of the selected bonds present in the sample focal volume, and corresponding with the bonds in the reference molecular species. The interference fringes themselves provide additional phase information that allow for the reconstruction of the vibrational characteristics of the molecules in the sample focal volume. In addition, we have shown that detection of the interferometric signal enables the differentiation of the resonant CARS signal from the problematic non-resonant background signal due to four-wave-mixing processes. We present the theoretical basis and experimental implementation of this new technique, along with interferometric and molecularly-specific image data from molecules with well-characterized optical signatures. Cross-sectional and en face images are used to demonstrate multi-dimensional depth-resolved molecular imaging. NIVI offers the potential for non-invasive imaging of selected endogenous molecular species in living tissue.
S.A. Boppart, None.
Abstract ID: 045
Diffuse Optical Tomography (DOT) shows promise as a quantitative imaging method for mapping molecularly targeted fluorescent probes in vivo. A current challenge in developing small animal DOT systems is to simultaneously provide optimal resolution, full body field of view and reasonable scan times (<1 minute). Recent DOT systems, based on charge coupled device (CCD) detection, have demonstrated the advantages of large CCD data sets, particularly in the planar transillumination geometry. Simultaneous dense spatial sampling and large imaging domains on the detection plane can be achieved. However previous systems have employed slow fiber switches which introduce an undesired asymmetry between illumination and detection. In the planar geometry, the spatially sampling on the source plane is relatively sparse which limits either the sampling density, or the field of view, or both. We present a fast scanning fluorescence DOT system with a 10× larger imaging domain (5cm × 5cmx1.5cm) compared to an equivalent fiber-switched system while maintaining the same resolution (point spread function FWHM ≤ 2.2 mm), sensitivity (<1 pmole) and scan time (<1min). As opposed to previous systems that used slow (Tswitch~250ms) fiber-optic switches to multiplex the illumination over a few fixed positions (36-45 positions), the new system scans the source laser using a galvanometer mirror pair (Tswitch~1ms) over flexible source patterns. The fast source switching is complemented by a high frame rate low noise, 5 MHz electron multiplying CCD camera that increases data acquisition rates by >10× over previous 1 MHz CCD systems. Using a limited view planar geometry, anesthetized animals are suspended in a matching fluid between the source and detectors windows during <1 minute measurements. Preliminary phantom and in vivo results, demonstrate feasibility of providing quantitative whole body bio-distribution assays of molecularly targeted fluorescent probes.
J.P. Culver, None.
Abstract ID: 046
We present recent advances in an emerging technology, Fluorescence Molecular Tomography, developed to enable quantitative three-dimensional visualization of fluorescence in whole animals in vivo. We address several important new developments employing non-contact instrumentation and 3D surface extraction that allows for high performance implementation. Different approaches that are suitable for obtaining the surface geometry of the specimen with high accuracy in the same experimental setup will be exposed showing how this approach considerably simplifies the experimental conditions and increases the signal-to-noise ratio.
The two main domains where relevant in-vivo results have been obtained, namely in the Near Infrared (NIR) and in the visible will be showcased. First, results on Fluorescent Molecular Tomography of NIR activatable probes will be presented. Secondly, the implementation of Fluorescent Protein Tomography in the visible will be explained and in-vivo results of 3D fluorescent protein imaging will be shown.
Finally, different applications in biology and medicine as outlined by cohort efforts recently materialized on optical imaging and tomography in Europe and in the United States will be discussed.

3D imaging of 700,000 CFSE loaded Cells implanted in a nude mouse resolved using Flourescence Molecular Tomography in non-contact geometry employing simultaneous surface extraction.
J. Ripoll, None.
Symposium VII: Probe Design: Novel Activation Strategies
Abstract ID: 047
Widespread interest in imaging molecular events in tissues by MRI has lead to renewed interest in development of novel imaging agents that can detect specific molecular binding events or report on biochemical variables such as gene expression, enzyme activity, pH, cellular redox state, oxygenation state, and hypoxia. Several Approaches can be taken in the design of biologically responsive imaging agents. One can alter the T1 of bulk water using paramagnetic complexes of Gd(III), T2 using various formulations of iron oxide or other nanoparticles, or the total bulk water signal using a paramagnetic
A. Sherry, Macrocyclics 4.
Abstract ID: 048
Current strategies for activatable “smart” optical probes incorporate self-quenching fluorophores conjugated to large nanoparticles or dendritic-type scaffolds, but these often suffer from poor tissue penetration. Here we present a novel small, cell-permeable, quenched imaging probe capable of detecting apoptosis via caspases 3 and 7 proteolysis.
Cell permeability of the quenched imaging probe, TcapQ647, was conferred by coupling a D-amino acid Tat-peptide derivative to the N-terminus of an L-amino acid caspase 3 and 7 recognition sequence, DEVDAPC. By flanking the cleavage sequence with the fluorophore Alexa Fluor 647 via thiol conjugation and the quencher QSY 21 via an internal ε-amine amide bond, the intrinsic fluorescence of AF647 was quenched with greater than 95% efficiency. The quenched imaging probe was efficiently cleaved and activated by both recombinant human caspases 3 and 7. Preincubation with the reversible inhibitor, DEVD-CHO, eliminated TcapQ647 cleavage.
Further, an all D-amino acid control peptide, identical in quenching properties and amino acid sequence to TcapQ647, was not cleaved by recombinant caspase. Confocal microscopy confirmed cell permeation and activation of our apoptosis imaging probe in doxorubicin-treated KB 3-1 cells. In a Jo2 anti-Fas-mediated liver apoptosis mouse model, pilot in vivo experiments with mice injected i.v. with our novel imaging probe resulted in a 3-4 fold signal increase over mice injected i.v. with the control, all D-amino acid peptide.
In summary, a novel quenched imaging probe, TcapQ647, is activated by recombinant and cellular caspases 3 and 7 as well as apoptotic tissue in vivo. The small size and efficient fluorescence quenching properties may render TcapQ647 useful for studying apoptotic initiators and chemotherapeutic efficiencies in vivo.
K. Bullok, None.
Abstract ID: 049
The invasive detection of nitric oxide (NO) will be useful for the diagnoses of many disorders related to NO. Several methods for NO detection have been developed. Among them, fluorescence methods are suitable for the real-time analysis of cellular NO functions in terms of sensitivity, selectivity, and experimental feasibility. We have successfully developed DAFs1), DARs2) and DAMBOs3) as fluorescent probes for NO. They react with NO to produce the corresponding fluorescent triazole ring compounds under physiological conditions. These probes have been widely used, and many biological applications of them have been reported. However, it is difficult to apply them to in vivo imaging, because they are needed to be excited by visible light, which is highly absorbed by biological molecules, like hemoglobin. On the other hand, the light in the near-infrared (NIR) region around 650–900 nm is known to be less absorbed by such molecules and can travel to deeper tissue. Moreover, it has another advantage that there is not autofluorescence in the region. Therefore, we aimed to develop NIR fluorescent probes for NO.
The NIR fluorescent NO probes were rationally designed. They are composed of two moieties, tricarbocyanine as the NIR fluorochrome and o-phenylenediamine as the NO-sensitive fluorescence modulator. Initially, this modulator decreases the fluorescence intensity of tricarbocyanine. Upon the triazole ring formation by the reaction with NO, the fluorescence quenching is removed, and then tricarbocyanine recovers the NIR fluorescence. We synthesized novel NO probes, diaminocyanines (DACs) and observed their fluorescence increase at 790 nm in an NO concentration-dependent manner under physiological conditions, as we expected.
H. Kojima, None.
Abstract ID: 050
C. Brekken, None.
Symposium VIII: Imaging Advanced Animal Models of Disease
Abstract ID: 052
In the past few years non-invasive visible light imaging has rapidly evolved from a purely exploratory experimental approach, to the integral part of Oncology drug discovery. As with all new technologies expectations are high when it comes to its impact on costs, timelines, and quality of future drug candidates. In order to fully benefit from it, we have to clearly understand its advantages, as well as its limitations. Tagging tumor cells with a light emitting entity (e.g. luciferase) allows us to non-invasively detect and quantify these cells in live mice. This can be particularly useful in orthotopic models, as well as in models of experimental metastasis, where it makes possible to follow effectiveness of new therapies in a single cohort of mice. We have however to keep in mind that the relationship between the light emission and the tumor burden may not be linear. In addition, the light intensity registered by the camera will depend on the absorption/dispersion properties of the tissue “hosting” the tumor, and on the distance from the tumor to the surface of the mouse. On top of that the concentration of luciferin will also be a limiting factor for light emission since this compound becomes toxic to animals before it can saturate the luciferase. Rapid elimination of luciferin calls for a precise and uniform timing of all imaging after the injection in order to obtain reproducible results. With all the above caveats the lucifearse-based visible light imaging technology can be extremely valuable to Oncology drug discovery. Examples of such applications will be described and discussed, including orthotopic models, models of experimental metastasis, and mechanistic models in hollow fibers.
P. Lassota, None.
Abstract ID: 053
In 1999, the National Cancer Institute (NCI/NIH) confronted the critical need for improved model systems to inform basic, clinical, epidemiologic, and translational research. The ability to manipulate the germline of mice, and the unprecedented store of data about genetic alterations implicated in human cancer prompted the NCI to implement a collaborative project of mouse cancer modeling. The resulting program, the Mouse Models of Human Cancers Consortium (MMHCC), has expertise in many aspects of basic, translational, clinical, and epidemiological research, and mouse genetics. The initial program of 19 groups was recently enlarged to 25 to accommodate an expanded set of goals, designed to leverage advances in many technologies, particularly in vivo imaging, computational modeling, and simulation.
The 300-member MMHCC cooperates with the NCI Center for Bioinformatics to evolve an integrative systems approach to human cancer research, providing the platforms to blend descriptive cancer model information with comparable human disease data. The Center maintains the Cancer Models and Cancer Images Databases, to which any researcher may submit data. This ensures that the databases reflect the experience of all cancer researchers who explore how well the models inform human cancer therapy, prevention, early detection, imaging, and population science. The eMICE website ( http://emice.nci.nih.gov ) is the interface to the NCI's preclinical models programs, resources, databases, and the NCI Mouse Repository.
The MMHCC members collaborate with the NCI to convene numerous roundtables and other open forums to promote state-of-the-art mouse cancer science. By year's end, the NCI-MMHCC will launch the Imaging Sciences Roundtable to promote collaborations among academic and private sector researchers who employ various in vivo imaging techniques to examine changes in tissues as cancers emerge, progress to invasive tumors and metastases, and respond to interventions or recur. More importantly, the Roundtable will encourage the application of real-time cell-based imaging strategies to intact living systems.
C.L. Marks, None.
Abstract ID: 054
J.P. Weichert, None.
Abstract ID: 055
Stem cell therapy can attenuate the clinical disease score in EAE. Central to the future success of stem cell transplantation in MS is the ability of transplanted cells to migrate from the site of transplantation to relevant foci of disease, while exhibiting remyelinating and trophic properties. We applied MR cell tracking to assess the distance and speed of cell migration after transplantation. Mouse neural BRDU+ and human ES-derived GFP+ neurospheres were labeled with Feridex and PLL. Following labeling cells differentiated normally into the three neural lineages. Intraventricular stem cell transplantation was performed on day 6 in a chronic EAE mouse model (n=16). Serial in vivo MRI (4.7 T) showed that at days 1–2 after transplantation, cells were mainly located within the ventricles. At 6 days (disease onset), cells started to migrate along white matter tracts, with further coninuation at days 13–14 (peak first relapse) and 30 (chronic phase). Most migration occurred early during the acute phase of disease. For the syngeneic system (mouse cells), the clinical disease score demonstrated a good correlation between disease severity and distance of migration. The histological distribution of labeled cells were found to match the MRI findings, in particular with the high resolution ex vivo imaging (9.4 T), with differentiation of cells occurring in vivo. The observation that the greatest degree of migration occurred very early in the course of disease highlights the narrow time window in which transplantation of remyelinating cells may be effective for obtaining clinical results. These results show that inflammatory signals associated with the clinical score modulate migration in a positive manner, and that serial MR cell tracking can provide a deeper biological insight of cell dynamics in vivo. Supported by RO1 NS045062 and NMSS PP0922.
J.W. Bulte, None.
Abstract ID: 056
Many molecular pathways relevant to cancer impinge upon the activities of ubiquitin ligases and their substrates. In many cases ubiquitin ligases bind to colinear targeting motifs within their substrates. Importantly, these motifs are often modular and can be used to target foreign proteins, in cis, for polyubiquitylation and hence proteasomal degradation. For example, p27 is a critical cell-cycle regulator and is polyubiquitylated by a complex containing the F-box protein Skp2. The binding of Skp2 to p27 is influenced by phosphorylation of p27 Thr187 by cyclin/cdk2 complexes. We fused p27 to firefly luciferase (p27-Luc) and showed that the resulting chimera was unstable and could be induced by various cdk2 inhibitors (including peptides, siRNA, and drugs) in cell culture. p27-Luc, but not wild-type luciferase, was also induced by cdk2 inhibitory drugs in vivo in nude mice xenograft assays. As a second example, we fused the region of HIF recognized by the VHL tumor suppressor protein (pVHL) to luciferase. The interaction of pVHL with HIF is governed by oxygen- and iron-dependent hydroxylation of HIF by members of the EGLN family. As expected, the HIF-luciferase reporter (′ODD-Luc) was induced by hypoxia or EGLN inhibitors in VHL+/+ cells grown in culture. In contrast, ODD-Luc was constitutively stable in VHL-/- cells. Next the ODD-Luc reporter or wild-type luciferase was introduced into the ubiquitously expressed ROSA26 locus in mice. As expected, the bioluminescent signal in the ODD-Luc mice was greatly diminished compared to the wild-type luciferase mice. Moreover, ODD-Luc was induced in mice exposed to hypoxia or treated with an EGLN inhibitor. Preliminary experiments also show accumulation of this reporter in VHL-/- tumors arising in VHL+/- mice. These studies establish the utility and feasibility of using luciferase fusion proteins for functional imaging in mice. In theory this approach can be extended to many other ubiquitin ligase/substrate pairs.
W.G. Kaelin, Imigen Systems 4.
Plenary Session V: Imaging/Therapy Combinations and Guiding Therapeutic Choices in Cancer
Abstract ID: 058
After intravenous injection the USPIO particles are transported into the interstitial space and subsequently via lymph vessels into the lymph nodes. Once within normally functioning nodes the iron particles are taken up by the macrophages, reducing the signal intensity of normal lymph node tissue in which they accumulate, because of the T2*- and susceptibility effects of iron oxide. In areas of lymph nodes replaced with malignant cells, there is absence of macrophage activity and hence lack of USPIO uptake. Thus the post USPIO MRI is able to identify metastatic areas in the lymph nodes independent of the lymph node size.
M.G. Harisinghani, GERRAF 1.
Abstract ID: 059
Bone marrow is a preferential site of metastasis in a variety of cancers. However, only a restricted number of solid cancers, especially breast and prostate cancer, are responsible for the majority of skeletal metastases. An emerging concept identifies the growth support provided by a specific organ microenvironment as the principal target for adjuvant therapies aiming to prevent progression of micrometastases to overt metastases. Bone is a highly dynamic tissue that is continuously remodelled by bone resorption and subsequent bone formation. Growth factors supporting bone metastatic growth are released especially during bone resorption. Differently from most of the other tissues, drugs that can limit local turnover, namely bisphosphonates (BP), are available for bone. Thus, we investigated whether pharmacological interference with bone turnover could repress development and growth progression of experimental bone metastasis. Whole body bioluminescent imaging (BLI) was adopted as a validated and reliable method for sensitive detection, monitoring and quantification in vivo of the growth progression of bone metastases induced by intra-cardiac or intra-osseous injection of luciferase-transfected breast cancer cells (MDA-231-B/luc+). Preventive BP-induced reduction of bone turnover, prior to bone colonization by cancer cells, inhibited by a great extent the number of developing bone metastasis. Nevertheless, tumor growth in the few, but still developing bone metastases, was affected only transiently. Reduction of bone turnover was also unable to control growth progression of bone metastases, either induced by haematogenous spreading or by intra-osseous inoculation, which were already established when BP-treatment was initiated. This, despite a substantial anti-osteolytic effect induced by BP. Therefore, bone metastatic cancer cells, after an initial growth phase dependent on the interaction with the local stroma, become independent on micro-environmental growth factor support and progress autonomously. Inhibition of bone turnover may represent a useful adjuvant therapy for cancer patients at risk to develop bone metastasis.
Plenary Session VI: Imaging in Aging and Neurological Disease
Abstract ID: 060
Formation and accumulation of beta amyloid (Abeta) aggregates in the brain are hallmarks of Alzheimer's disease (AD). Current methods of diagnosis of AD are based on cognitive decline and behavioral changes related to old age and AD, which are often very difficult to differentiate. Therefore, there is an urgent need for in vivo imaging agents, which are valuable as a specific biomarker to demonstrate the location and density of beta-amyloid plaques in the living human brain. Recent advances in developing tracers for binding Abeta plaques suggest that there are specific and saturable binding sites on the aggregates of Abeta peptides that can be selectively labeled and imaged in vivo. Several Abeta specific binding agents which are derivatives of naphthalene, benzothiazole, stilbene or other related heterocyclic derivatives containing an electron-donating group on one of the aromatic rings, have been reported. They are simple, relatively small, neutral and lipophilic molecules. On the basis of their exquisitely high binding affinity to Abeta aggregates (Ki at the range of 0.1-20 nM), they are suitable candidates as Abeta plaque-selective imaging agents. More importantly, they also showed ability to penetrate the intact blood-brain barrier, an essential pre-requisite for a useful plaque-imaging agent. In vitro and in vivo testing of these agents in transgenic mice specifically engineered to overproduce Abeta peptides in the brain showed promising characteristics in binding to Abeta plaques. In conjunction with positron emission tomography (PET) and single photon emission computer tomography (SPECT) the Abetaplaque-specific imaging agents will be useful for early detection and monitoring the progression and effectiveness of treatment of AD.
H.F. Kung, None.
Abstract ID: 061
Assessment of the efficacy of radiation and chemotherapy for brain cancer patients is traditionally accomplished by measuring changes in tumor size several weeks after therapy has been administered. The ability to use noninvasive imaging during early stages of fractionated therapy to determine if a particular treatment will be effective would provide an opportunity to optimize individual patient management and avoid unnecessary systemic toxicity and expense. We investigated whether changes in the Brownian motion of water within tumor tissue as quantified using diffusion magnetic resonance imaging (MRI) could be used as an early, surrogate marker for early prediction of treatment response in brain cancer patients.
Twenty brain tumor patients were examined by standard and diffusion MRI before initiation of treatment. Additional images were acquired at 3 weeks post-initiation of chemo- and/or radio-therapy. Images were co-registered to pretreatment scans, and tumor water diffusion values were calculated and correlated with subsequent response defined by change in tumor size on MRI by standard radiographic criteria.
Of the 20 patients imaged during the course of therapy, 6 were classified as having a partial response (PR), 6 as stable disease (SD) and 8 as progressive disease (PD). Changes in tumor diffusion values were found to predict patient response at 3 weeks from the start of treatment revealing that early changes in diffusion values could be used as a prognostic indicator of subsequent volumetric tumor response (specificity and sensitivity of 100%).
Diffusion MRI provides an early surrogate marker for predicting treatment response in patients with brain tumors.
B.A. Moffat, None.
Symposium IX: Oncogenesis, Angiogenesis and Disease Progression
Abstract ID: 063
The penetration of large therapeutic agents (e.g. monoclonal antibodies, viral vectors) in solid tumors is limited by transport across the tumor vasculature and through the tumor interstitial space. Antiangiogenic agents can improve the accumulation of therapeutic agents and oxygen levels in tumors. Based on these findings we hypothesized that anti-angiogenesis “normalizes” the vasculature and transport parameters in tumors. Inhibition of vascular endothelial growth factor (VEGF) receptor 2 with the monoclonal antibody DC101 prunes immature blood vessels and improves the integrity and functioning of the remaining vasculature. DC101 can also decrease the tumor interstitial fluid pressure (IFP) and induces an hydrostatic pressure gradient across the vascular wall, which leads to a deeper penetration of large molecules into tumors. A recent phase I clinical trial provided support for the “normalization” hypothesis, in patients with rectal cancer inhibition of the VEGF ligand with the monoclonal antibody bevacizumab “normalized” the vasculature and reduced the IFP in rectal tumors. As part of our studies on interstitial transport we adapted second harmonic generation to dynamically image and quantify fibrillar collagen in vivo and developed multi-photon fluorescence correlation spectroscopy to measure diffusion within tumors. Using these technologies we have identified fibrillar collagen as a significant transport barrier and identified a novel approach to overcome the interstitial barrier using relaxin, a collagen-modifying hormone. Strategies that enhance transvascular and interstitial transport in solid tumors may improve the penetration and efficacy of therapeutic agents.
Y. Boucher, None.
Abstract ID: 064
Activation of the transcription factor NF-κB is tightly regulated by IκBα which forms a cytoplasmatic heterodimeric complex with NF-κB, thereby preventing its nuclear localization and DNA-binding. Following stimulation (e.g., by TNFα or LPS), IκBα is phosphorylated by an upstream kinase (IKK) and in turn, is ubiquitinated and degraded by the proteasome. These events are rapid (minutes), resulting in the release and nuclear translocation of active NF-κB.
We hypothesized that monitoring the relative levels of IκBα in intact cells and living animals may offer a novel approach to monitor regulation and activation of NF-κB. We therefore engineered and stably expressed an IκBα-firefly-luciferase (IκBα-FLuc) fusion reporter in Hela cells and analyzed its bioluminescent signal before, during and after stimulation with TNFα. We observed a rapid decrease, followed by a gradual increase in bioluminescence, as compared to untreated cells or to unfused FLuc-expressing cells, due to degradation and re-synthesis/stabilization of IκBα-FLuc. The rapid decrease in bioluminescence could be inhibited in a dose-responsive manner by pre-incubating the cells with proteasome inhibitors (MG-132 or Bortezomib) or IKK-inhibitors (IKK-NBD peptide or Bay 11-7085). Furthermore, upon addition of TNFα, phosphorylation and degradation of endogenous IκBα and the fusion-reporter exhibited similar kinetics by western-blot analysis, comparable to the changes in bioluminescence.
In living mice co-expressing the IκBα-FLuc reporter and a Renillaluciferase (RLuc) control construct in hepatocytes, we observed 1 hour post-administration of LPS a significant decrease in liver bioluminescence (8-fold, normalized to RLuc). Pre-treatment with Bortezomib not only abrogated the LPS-mediated decrease in bioluminescence, but induced a 4.5-fold increase in bioluminescence, suggesting that both basal turnover and ligand-induced degradation of IκBα could be inhibited in-vivo. No significant change in bioluminescence was observed in control mice (LPS-untreated or mice expressing unfused FLuc). The IκBα-FLuc reporter may facilitate development and validation of NF-κB modulators acting on or upstream to IκBα-degradation.
S. Gross, None.
Abstract ID: 065
The significance of the interstitial-lymphatic continuum in the metastatic cascade until recently has been eclipsed by the focus on angiogenesis. Here, we present a noninvasive, in vivo MRI technique for identifying: (i) extravascular regions drained by lymphatic-convective channels and (ii) regions in which “pooling” of fluid exudate occurs. We conducted MRI on anesthetized MCF-7 tumor-bearing (>200mm3) mice (n=3), and intravenously administered (60mg/ml) Albumin-GdDTPA. Since intestitial drain in tumors is slow (≈hours), tumors were imaged in two phases: 30 min MRI, 60 min interval, followed by 30min MRI. Animals were then sacrificed, tumors excised and fixed. Tumor sections were double-stained for lymphatic vessels (LYVE-1), blood vessels (isolectin BS-I-B4). The two phases of the MR concentration time data were analyzed using a novel multiple regression approach (

A. P. Pathak, None.
Abstract ID: 066
MRI has been used to detect tumorigenic molecular events of lymphoma cells in a previoulsy described Notch3-transgenic mouse model (EMBO J., 19(13): 3337-48, 2000; PNAS, 99(6): 3788-93, 2002). In this model of transgenic mice the physiologic developmental program of T lymphocytes is deregulated by overexpression of the oncogenic receptor Notch3 in T cell precursors; this overexpression hampers the attenuation of the expression of the pTa chain of pre-TCR, which is not observed in mature lymphocytes migrating to peripheral lymphoid tissues. Signals generated from pTa are responsible for lymphomagenesis; transformed cells, bearing high expression of pTa, migrate to secondary organs (spleen and lymph nodes). pTa plays a mechanistic role in tumorigenesis, since Notch3 transgenic mice in which pTa gene was deleted, were unable to undergo lymphomagenesis.
To detect tumoral lesions in this model we used a contrast medium obtained by direct labeling of a highly specific monoclonal antibody anti-pTa with Gd3+ hexahydrate. In vitro detection of cells was performed on a 1.5T MRI unit, while in vivo detection of tumor lesions in animals was performed on a 4.7T MRI unit equipped with an animal dedicated magnet.
In vitro experiments showed that Gd-Ab complex stained efficiently only neoplastic cells isolated from the spleen of transgenic mice while did not stain splenocytes obtained from wild-type animals.
In vivo experiments were performed in Notch3 transgenic mice and in wild-type animals used as controls. When injected into Notch3 transgenic mice, Ab-Gd complex targeted neoplastic cells and allowed MRI detection of lesions in the spleen and lymph-nodes. The same amount of free Gd was unable to detect tumor cells and lesions in Notch3 transgenic mice. Spleen and lymph-nodes were not detectable in wild-type animals injected with both Ab-Gd complex and free Gd.
M. Artini, None.
Symposium X: Methods of Multimodality Imaging: Fusion Instruments & Software Solutions
Abstract ID: 068
The presentation will survey registration applications over a wide variety of situations (including multimodality, 2D/3D, in vivo/vitro, human/animal) and demonstrate existing solutions. Solutions are constructed using mutual information as an objective function, Nelder-Mead simplex as the minimizing algorithm, and thin plate splines as a warping interpolant.
C. Meyer, None.
Abstract ID: 069
As a member of the Network for Translational Research in Optical Imaging (NTROI), Siemens Corporate Research (SCR) has started a research and development effort, whose broad goal is to improve breast cancer detection, and clinical management of breast cancer, through the integration of multi-dimensional diffuse optical imaging with other leading imaging modalities (MRI and X-ray mammography).
We have developed a software platform for multi-modal integration and visualization of diffuse optical tomography (DOT) and magnetic resonance imaging (MRI) of breast cancer. The image visualization platform allows multimodality 3D image visualization and manipulation of datasets, such as a variety of 3D rendering techniques (direct volume rendering, multi-planar reformatting, or maximum intensity projection), and the ability to simultaneously control multiple fields of view. This platform enables quantitative and qualitative analysis of two types of diagnostic content:
−Structural data: derived from high-resolution MR images and possibly from light scattering changes in DOT images due to cell density increase associated with tumor growth.
−Functional data: derived from angiogenesis (Gadolinium in MR, blood volume in DOT images), hypermetabolism (blood saturation in DOT images), and vasculature permeability (indocyanine green kinetics).
The functional parameters, together with morphological parameters from MR can be suitably combined and correlated to the absolute diagnosis from histopathology. Fusion of the multimodal datasets may eventually lead to a significant improvement in the sensitivity and specificity of breast cancer detection. Fusion may also allow a priori structural information derived from MRI to enhance the reconstruction of diffuse optical tomography images.
We will present the early results of using the image visualization platform on multimodal breast cancer data (DOT and MRI) from the University of Pennsylvania Depts of Radiology and Diffuse Optical Imaging Group.
F.S. Azar, None.
Abstract ID: 070
Abdominal imaging in mice is an emerging field of interest with significant implications for research studies using mouse models of cancer. Correlations in imaging findings between mice and humans with similar tumors serve to test the validity of these mice as appropriate models of human disease. We have used the lfabp-cyclin D1 transgenic mice (Deane, N.G. et al., 2001) harboring multi-focal spontaneous primary hepatocellular adenomas (HCA) and carcinomas (HCC) in a comparative multi-modality imaging study of findings for HCC examining limits of tumor detection, accuracy of tumor detection, and estimation of overall tumor burden. Animal imaging was conducted using state-of-the-art small-scale, small animal scanners including a Visualsonics Vevo660 ultrasound (US) system, an Imtek microCATII (microCT), a Concorde Microsystems Focus microPET, and a 7T Varian MRI/MRS spectrometer. Animals were autopsied within 2 weeks of imaging and findings were correlated with autopsy results. We found that MRI provided the most accurate measures of tumor detection for HCA/HCC using inherent tissue contrast (sensitivity = 65%, specificity = 100%, PPV = 100%, NPV = 22%). MicroCT alone provided no inherent contrast between tumors and normal liver but predictive values improved where images were either (a) paired with microPET imaging and/or (b) aided with hepatocyte specific contrast agent (87.5% sensitivity, 100% specificity). MicroPET imaging using 18 F-deoxyglucose alone had relatively poor sensitivity for murine HCC but high specificity (26% sensitivity, 100% specificity). US screening provided the least accurate pictures of HCA/HCC tumor burden with 35% sensitivity and 70% specificity (PPV = 79%, NPV = 26%). The radiological findings in this mouse model were consistent with human studies for HCA/HCC and serve to support the validity of this genetically engineered mouse model for preclinical and molecular studies in liver cancer progression and therapeutics. N.G. Deane, Alerion Biomedical 6.
Abstract ID: 071
Increasingly, preclinical therapeutics research requires quantitative assessment of in vivo tumour growth, functional status of the vasculature, and molecular profile. Crude measurements of tumour burden are usually made using mechanical calipers in association with volume calculations based on ellipsoidal formulae. In this study we introduce ultrasound biomicroscopy and micro computed tomography (micro-CT) into longitudinal tumour growth studies, focussing on three-dimensional (3D) tumour volume evaluation and microvascular morphological analysis. Studies were performed on intradermally injected human malignant melanoma cancer cells (MeWo) or orthotopic injection of breast cancer cells (MDA-MB-231) in immunocomorthotopic promised mice. A high resolution (30 - 100 micron), 40 MHz ultrasound biomicroscope (UBM) (VisualSonics) revealed early MeWo xenografts growth, and was able to detect tumours less than 0.5 mm3 in volume. In addition, multiple ultrasound image planes with inter-plane spacing of 33 microns were acquired over the complete volume of MeWo tumour xenografts and reconstructed into 3D images. Segmentation of 3D images yield volume values which are strongly correlated to a conventional tumour volume model (v/6*w*d*l) where w, d and l were obtained from orthogonal ultrasound views (r=0.98). A lower degree of correlation was obtained in tumours less than 1 mm3 (r=0.80), suggesting that 3D segmentation volume measurement gives a more accurate estimate when small, initiating MeWo xenografts have yet to assume an ellipsoidal shape. Contrast-enhanced micro-CT revealed significant and progressive distortion in the vascular architecture. These changes complemented alterations in blood flow patterns as visualized by UBM speckle variance analysis. A three-week therapy of tumour-bearing mice with an anti-angiogenic drug DC101 (Imclone), significantly reduced the rate of tumour growth, possibly by inhibiting the expansion of vasculature required for tumour growth. 3D ultrasound-assisted tumour volume study, in conjunction with vascular morphology and functional analysis using micro CT and speckle variance, are valuable tools in pre-clinical assessment of anti-cancer therapeutics.
A. Cheung, None.
Abstract ID: 072
Despite tremendous strides made in molecular biology techniques, imaging, and the design of unique imaging probes, the cure for cancer remains beyond our grasp. Cancer is a complex disease and the apparent impenetrability of the disease is largely due to multiple, often redundant pathways, present in cancer cells. Finding effective treatments against cancer depends upon identifying and attacking targets critically important for the cancer cell to survive, or failing this, to invade and metastasize. Multi-modality or multi-parametric functional imaging provides unprecedented opportunities for identifying and imaging key common pathways specific to cancer cells, and imaging the effectiveness and outcome of therapies targeting these pathways. Altered choline metabolism is a prime example of a common feature of cancer revealed by MR spectroscopy, which can be exploited for treatment. Similarly the existence of tumor hypoxia is another characteristic common to most tumors which can be used for therapeutic as well as imaging strategies. In fact the lethal phenotypic traits of cancer, such as invasion and metastasis, may arise from the unique physiological environment of tumors, such as hypoxia and extracellular acidosis. Using combined MR and optical imaging of a prostate cancer tumor model engineered to express GFP under hypoxia, we have gained useful insights into the dynamics between the tumor environment, vascularization and metabolism. MRI and MRSI were used to obtain co-localized maps of vascular volume, permeability, total choline and lactate/lipid, and optical imaging to obtain co-localized maps of hypoxia. We found that hypoxic regions were characterized by increased total choline and permeability. These data suggest that hypoxia may cause, in part, the high total choline levels observed in tumors.
Z.M. Bhujwalla, None.
Symposium XI: Imaging in Drug Discovery and Drug Development
Abstract ID: 073
Although NET are relatively uncommon, the impact of their symptoms (uncontrollable diarrhoea and flushes) can be tremendous; such patients may be unable to lead normal lives. At the time of the initial diagnosis, up to 75% of the patients have already spread of disease, very often to the liver.
Since about 10 years somatostatin receptor scintigraphy (SRS) is used by many nuclear medicine centres worldwide. It appeared superior for staging of NET in the whole body compared to (a combination of) CT, MRI, Ultrasound and angiography, the standard imaging tools for these tumours so far. SRS is now the initial imaging choice in NET staging and determines the choice of therapy.
As a sequel to SRS, the same principle has been applied for radiotherapy of NET using therapeutic radionuclides.
We have carried out radiotherapy since 1992 in more than 400 patients with NET expressing somatostatin receptors. In general, patients undergo such treatments 3 to 4 times with intervals up to 9 weeks. Especially, with the second and third generation of somatostatin analogs labelled with beta-particle emitters, impressive tumour shrinkage (in up to 50 % of patients for third generation analog) and tumour growth stabilization have been observed in patients with growing tumours at the start of treatment. On average, this effect lasts for at least 36 months. Overall Quality of Life improved with less fatigue, insomnia and pain. Finally, more than 50 % of the patients treated with the second generation analog are still alive after 5 years. These survival results contrast very positively with our findings in a similar group of patients after treatment with the first generation analogs. Also, these data suggest much better responses with regards to duration of effect and survival than after treatment with chemotherapy.
E.P. Krenning, Novartis 1, 2; Mallinckrodt 1, 2.
Abstract ID: 074
A critical characteristic of antibody therapeutics is the specificity to target tissues. Here we demonstrate the potential of integrated SPECT/CT to provide quantitative characterizations of the biodistribution patterns of antibody derivatives. This was done by comparing the full-length antibody, Herceptin, with its Fab and AB.Fab derivatives. AB.Fab is a Fab fragment engineered to bind endogenous albumin through an albumin binding peptide [J. Biol. Chem., 2002; 38:35035-35043].
Herceptin and derivative antibody fragments were radiolabeled with In-111 as DOTA conjugates. Antibodies were injected by tail vein into nu/nu mice carrying huHER-2 expressing tumors. SPECT and contrast-enhanced CT images were acquired consecutively in the same instrument (X-SPECT, Gamma Medica, Inc.) multiple times over four days post injection. After the final image, tissues were harvested, weighed, and the radioactivity was measured in a gamma well counter for later comparison with image-based measurements. Regions of interest (ROI) were defined using the CT volumes and used to compute mean ROI intensities for the corresponding SPECT images, which were scaled according to a standard of known activity placed in every image. A comparison of ex vivo signals with in vivo image intensities was used to determine empirical recovery coefficients for each tissue and collimator.
As predicted, the Fab fragment showed rapid accumulation in the kidney while Herceptin showed slow and progressive accumulation into tumor tissue. The AB.Fab, which has an extended half-life compared to the Fab, resulted in tumor tissue uptake comparable to Herceptin. Comparison of the CT-guided SPECT results with ex vivo gamma well counter data confirmed that the higher resolution pinhole collimator allows a more accurate characterization of the biodistribution.
Ventral view of 3D fused SPECT/CT images showing distribution of labeled antibodies (Left to right. Full length Herceptin Fab, and Herceptin at 18 hours post injection.

L. McFarland, Genentech 5.
Abstract ID: 075
P53, a key protein in the response to DNA damage, has been extensively studied in cultured cells but only in a more limited fashion in whole organisms. A transgenic mouse model wherein firefly luciferase is expressed from the p53 responsive MDM2 promoter was developed to dynamically follow p53 expression. Following total body radiation (TBI) bioluminescent imaging (BLI) revealed a dose, time, and spatial dependence of p53 activation. Western blots of radiated tissues demonstrated an increase in both p53 and luciferase proteins confirming BLI. The greatest BLI increases occurred in tissues that are known to be radiosensitive, the abdomen and oral cavity (10.0 +/− 1.6 and 3.4 +/-0.6 fold induction, respectively). Post mortem BLI indicated that small intestine accounted for the predominant signal within the abdomen (11.3 +/− 0.4 fold induction). To extend these studies animals were treated with common radiosensitizers (5-Fluo-rouracil or Gemcitabine) and even at the maximum tolerated dose there was no increase in BLI. Interestingly, the combination of radia-tion and either of these agents did not result in an increase in the peak bioluminescent signal compared to XRT alone. However, in both instances with concurrent chemoradiotherapy there was a prolonga-tion of the BLI signature consistent with prolonged activation of p53. In contrast, pretreatment of mice with a known radioprotector (Amifostine) resulted in no alteration in baseline BLI, but a > 50% decrease in peak BLI following TBI. Further studies to correlate gene and protein expression as well as biologic end-points are on-going to further validate this model system. Thus, the MDM2-luciferase mouse is a valuable tool to assess gene activation dynamically and non-invasively in response to ionizing radiation with or without concurrent radiation modifiers and may be utilized to screen for novel compounds to alter p53 pathways in vivo.
D.A. Hamstra, None.
Abstract ID: 076
FTY720 is a sphingosine-1-phosphate receptor agonist that has been shown to redirect homing of lymphocytes from the peripheral blood to secondary lymphoid organs. By that it can reduce general inflammatory activity in the brain and might serve as a new agent for treatment of multiple sclerosis (MS). In the present study FTY720 was used for treatment of rats in a model of chronic relapsing EAE at an oral dose of 0.3 mg/kg/day. Magnetic resonance imaging (MRI) based on in vivo tracking of macrophages labeled with super-paramagnetic nano-particles (USPIO), immunohistological staining (IHC) and neurological readouts were used to study the burden of disease in treated and untreated animals. While untreated animals showed severe paralysis of the hind paws, intense accumulation of macrophages in brain tissue and areas of blood brain barrier (BBB) disruption, FTY720 treated animals displayed no signs of inflammatory activity or neurological impairment. These observations were made for both acute phase and first relapse. In conclusion, tracking of macrophages by MRI provides direct evidence of the immuno-modulatory efficacy of FTY720 in the EAE model and correlates well with neurological symptoms and histology.
M. Rausch, None.
Abstract ID: 077
Technology-oriented alliances with academic institutions play an increasingly important role in the business strategies of pharmaceutical and biotechnology companies. These interactions offer an industrial partner competitive advantages via access to new, as well as, basic technologies available within the university environment. Academic institutions also benefit financially from industrial collaborations which help support research, drug-development programs, postdoctoral student fellowships, scientific meetings, and graduate education programs. In addition, the academic partner is often able to access novel therapeutic agents that allow for novel experimentation. This interplay leads to both an improvement in scientific integrity of industrial R&D efforts and to a better focus on practical applications of academic research interests. In this two-part talk, an historical perspective will be presented that outlines the long-term scientific relationship between Washington University (W.U., St. Louis) and Pfizer, Inc. (and its legacy companies.). This academic-industrial relationship spans various areas of biotechnical and preclinical pharmaceutical research employing magnetic resonance imaging (MRI) technologies. In pharmaceutical sciences, these MRI methods (1) allow in vivo serial monitoring of disease progression and regression; (2) provide early markers of pathology and toxicology; (3) permit studies of mechanisms of drug action, metabolism, and efficacy; and, (4) yield important pre-clinical data used to support and guide costly clinical trials. The second part of this presentation will highlight how Pfizer's involvement with W.U. (and other academic institutions) has helped guide and impact the industrial/pharmaceutical decision-making process. Often such decisions are made at an early stage in product development where uncertainty is high and the possibility of obtaining relevant information needed to make a decision is limited. Thus, this presentation demonstrates how the W.U.-Pfizer MRI collaboration has enabled more informed Go/NoGo decisions to be made prior to the launch of a new product and/or the transition of a preclinical drug candidate into clinical trials.
J.J. Ackerman, Varian NMR Systems, Palo Alto, CA 1; Sigma-Aldrich, St. Louis, MO 6; Doty Scinetific, Columbia, SC 1; Luna Innovations, Blacksburg, VA 1.
Symposium XII: Late Breaking Session
Abstract ID: 078
Tumor necrosis factor-alpha (TNF-α) is a potent pro-inflammatory cytokine that is involved in the pathogenesis of inflammatory bowel disease. TNF-α stimulates IκB-kinase, which then phosphorylates IκB and releases NF-κB to freely translocate to the nucleus and bind its cognate recognition sites in the interleukin-8 (IL-8) promoter to induce IL-8 gene transcription. Curcumin, the major component of the spice turmeric, blocks TNF-α-mediated NF-κB activation and IL-8 gene expression by inhibiting IκB-kinase activity. To develop a high throughput system to identify dietary anti-inflammatory compounds that would inhibit TNF-α function, we generated HCT-116 colon adenocarcinoma and HeLa cervical adenocarcinoma cells that constitutively expressed a chimeric IκBα-luciferase (IκBα-Luc) fusion protein. To determine the effect of curcumin in these cells, curcumin (0-30 μM) was added to the cells 30 min before treatment with TNF-α (10 ng/ml). Immediately following addition of TNF-α, D-luciferin (150 μg/ml) was added to the cells and luminescent signals were measured using the IVIS Model 100 for over a period of 90 min. As compared to untreated controls, curcumin demonstrated a dose-dependent inhibition of TNF-α-mediated degradation of IκBα-Luc. Furthermore, low levels of inhibition of IκB-Luc degradation were easily measurable. No significant change in bioluminescence was observed in cells expressing un-fused luciferase. To confirm this inhibition, transfection studies using a plasmid encoding the luciferase gene under the control of a minimal TK promoter containing NF-κB binding sites were performed. Curcumin also demonstrated a dose-dependent inhibition of NF-κB transcriptional activity. Furthermore, curcumin also inhibited IL-8 promoter activity in these cells. These data demonstrate that non-invasive imaging of IκBα degradation is feasible for the identification of novel anti-inflammatory dietary factors that regulate cellular signaling and gene expression.
N. Murmu, None.
Abstract ID: 079
In this study we investigated the application of the human sodium iodide symporter (hNIS) as a reporter gene to non-invasively image in vivo gene transfer and expression in lung tissue in real time.
G. Niu, None.
Abstract ID: 080a
Recent work has shown genetic disruption of the RNA binding protein Apobec-1 reduces intestinal tumorigenesis in an in-vivo model of adenomatous colorectal cancer APCmin/+, in part through alteration of vascular endothelial growth factor (VEGF) mRNA stability1. This study was undertaken to evaluate APCmin/+ vs APCmin/+Apobec-1−/−mice using a non-invasive imaging protocol, as well as biodistribution studies, with [F-18]-FLT, followed by evaluation of polyp morphology and gene expression profiling.
For imaging and biodistribution studies, mice were anesthetized, and IV injected with 18F-FLT (285–399μCi, or 20μCi, respectively). Imaging studies were obtained at 1hr post injection (10min static), with microPET FOCUS-F220 Scanner® (Concorde Microsystems, Knoxville, TN). Biodistribution studies were carried out at 1hr post injection, the organs were dissected and counted in a gamma counter. MicroPET data was analyzed (SUV) by drawing ROI's over the intestinal polyps. Data showed an increase of 18F-FLT uptake throughout the intestinal polyps in the APCmin/+ mice when compared to APCmin/+Apobec-1−/−mice (1.24±0.05 vs 0.655±0.035). Biodistibution data was analyzed as %ID/gram, and validated an increase of radiotracer uptake throughout the intestinal polyps in the APCmin/+ mice when compared to APCmin/+Apobec-1−/− mice (2.48±0.95 vs 1.25±0.24). VWF immunostaining revealed decreased angiogenesis in the polyps, specifically in the flat polyp in APCmin/+Apobec-1−/− compared to APCmin/+. Real time PCR analysis showed a 5-fold decrease in VEGF mRNA in the APCmin/+Apobec-1−/− compared to APCmin/+. BrdU labeling (%) showed a decrease in staining of APCmin/+Apobec-1−/− when compared to APCmin/+ (0.18 vs 0.42; p=0.0001), consistent with altered proliferation.
These data suggest promising results for non-invasive imaging, as well as correlative radiopharmaceutical biodistribution of 18F-FLT, polyp morphology, and gene expression profiling in the APCmin/+ mouse when compared to APCmin/+Apobec-1−/− mouse.
D. Mukhopadhyay, None.
Abstract ID: 080b
A. Winkeler, None.
Abstract ID: 081a
We have developed a noninvasive imaging methodology that enables the investigation of biology inside the femurs of living mice. We describe combined in vivo magnetic resonance imaging (MRI) and bioluminescence imaging (BLI) of reporter gene (TGL) marked mouse bone precursor cells passively labeled with particles (SPIOs) containing superparamagnetic iron oxide and a red fluorophore (FR). Fluorescence activated cell sorting yielded double red (SPIO labeled) and green (BLI reporter gene marked) fluorescent cells which were transplanted into mouse femurs. High resolution images were acquired using a home-built solenoidal coil in combination with microimaging gradients at 7.05T. Sagittal 2D images of the femurs were obtained at 43 × 25 μm in-plane resolution. Following intramedullary transplantation of 2.5 × 105 4 TGL SPIO-FR cells, loss of contrast was observed throughout the femur (Fig. 1a, b). Axial 3D images at 50 × 50 × 375 μm resolution were acquired for quantitative analysis of image contrast within the mouse femur prior to and following injection of SPIO labeled cells. Region-of-interest analysis in pre- and post-injection images yielded a significant signal intensity difference of 2.02 × 106 ± 2.56 × 105 per ml (p<0.0001). Following MRI, BLI registered reporter gene expression only from the 4 TGL SPIO-FR harboring left femur (Fig. 2a, b; 15-fold higher signal intensity as compared to the femur of the control animal). The FR and EGFP labels were used to verify the presence of viable cells bearing both the SPIO label and the BLI reporter gene respectively by <i>ex vivo analysis. Thus, our combined MRI-BLI approach provides a versatile tool specifically designed to resolve the biological environment within the mouse bone marrow cavity.


T.P. Gade, None.
Abstract ID: 081b
Figure legend. A: in vivo and B: ex vivo MRI. Arrows point at regions with negative signal enhancement caused by CLIO-Cy5.5 within plaques at the lesser curvature and the aortic root. C: fluorescent Cy5.5 signal and D: Texas red immunoflurescence staining of macrophage antigen MAC 3 merged with dapi stain for nuclei at 400× magnification. C and D were obtained from the same slide. Arrows point at 2 macrophages that took up CLIO-Cy5.5
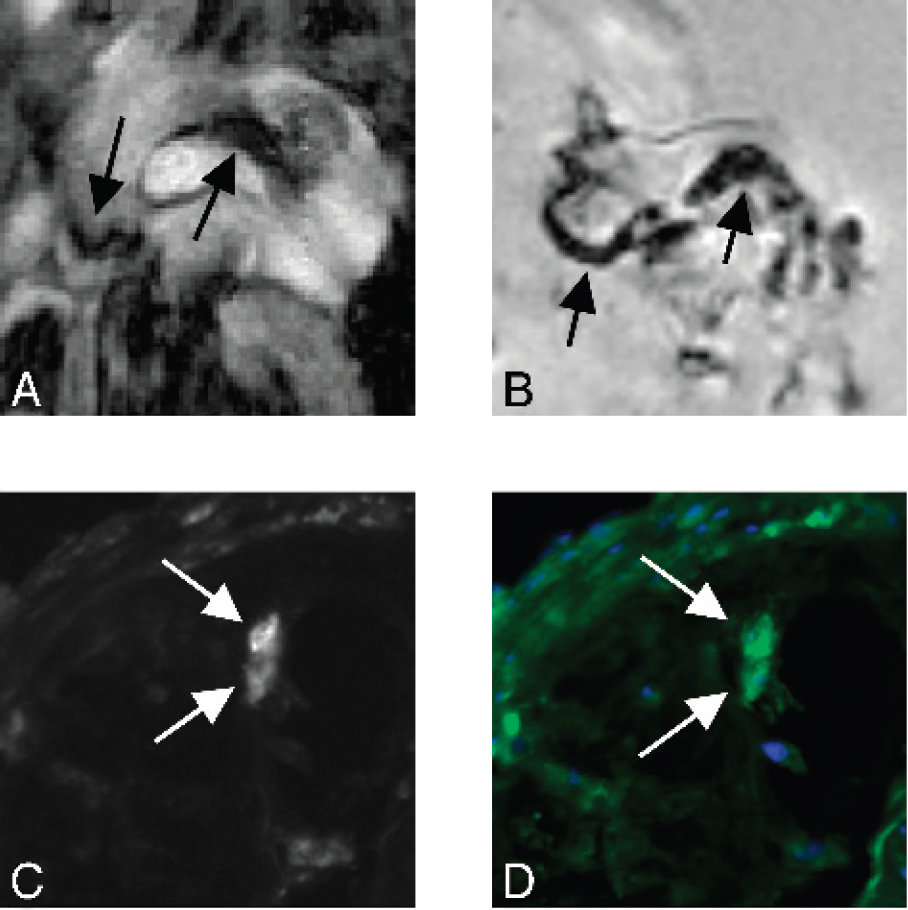
M. Nahrendorf, None.
Abstract ID: 082
H. Alencar, None.
Abstract ID: 083
Dynamic fluorescence images were obtained from xenografts bearing a subcutaneous human Karposi's sarcoma (KS1767) and human melanoma tumor (M21) model immediately following the i.v injection of an integrin-receptor (αvβ3) targeting Cy5.5-c(RGDf) at an equivalent dose of 3 nanomoles of c(RGDf). Both M21 and KS1767 models were integrin expressing. The fluorescence images were acquired using an intensified charge-coupled device (ICCD) system and were analyzed with a pharmacokinetic (PK) model to determine short term PK parameters of each tumor model over a period of 20 minutes. Imaging of both KS1767 and M21 models was also conducted in the presence of a competitive binder, free c(RGDf) administered i.v at a dose of 600 nanomoles. Our results show that the KS1767 exhibited early and rapid uptake of Cy5.5-c(RGDf) that could be mediated by the prior administration of c(RGDf) as a competitive binder to (αvβ3). However, the M21 tumor model showed different PK values from the KS1716 model. Indeed, there was little difference between the PK uptake rate constants between normal and M21 tissue regions at early times. Moreover, short term PK uptake rates were unchanged by the introduction of c(RGDf) prior to Cy5.5-c(RGDf) administration in M21. Finally, though KS1767 was visible from early dynamic fluorescence images, M21 was not readily contrasted at early times following administration even though it was contrasted by fluorescent uptake after 24 hours following Cy5.5-c(RGDf) administration. In vivo PK parameters may be reflective of the difference in tumor biology and molecular target expressions between KS1767 and M21.
S. Kwon, None.
Symposium XIII: Advances in Clinical Molecular Imaging
Abstract ID: 084
There is a misconception by many radiologists that “Molecular Imaging” is not going to be a major part of clinical radiology in the near to intermediate future because it takes many years and multi-million dollars to take a new imaging probe to market. To dispel this misconception we need a more clinical definition of “Molecular Imaging” which I would define as “the practice of medical imaging in the era of molecular medicine”. Obviously, this definition is designed to be vague and all inclusive but this is precisely what we need to do so that we don't get tunnel vision and the field of “Molecular Imaging” can enjoy the rapid growth similar to molecular diagnostic testing. How then can a clinical radiologist become a “Molecular Imager”? I believe it can be divided into 4 different stages with increasing levels of sophistication. Stage 1. “See morphology think molecular biology”. Stage 2. “Combine imaging information with molecular diagnostic information”. Stage 3. “Obtain molecular information using imaging”. Stage 4. “Personalizing treatment using combined molecular imaging and therapy”. To achieve the first stage of development requires only acquisition of knowledge and applying the knowledge in the daily practice of clinical radiology and even everyday life. With this type of approach, any interested clinical radiologist can become a “Molecular Imager” without waiting for new imaging probes or imaging equipment. However, new teaching materials need to be developed to enable the interested radiologists to become active participants which many organizations are beginning to address. With the increase in momentum in the whole field we are optimistic that “Molecular Imaging” will be part of everyday clinical radiology practice in the near future.
K.C. Li, None.
Abstract ID: 085
Regions of interest analysis was used to measure FIAU concentrations in tumour and background brain, before and after virus administration, to calculate retention indices and allow comparison of FIAU uptake.
M.F. Dempsey, None.
Abstract ID: 086
Pgp is part of the multidrug resistance system possibly implicated in the poor response to anti-epileptic therapy. The ABCB1 gene encoding for Pgp is functionally expressed also in the brain as well as in circulating leukocytes. Pgp activity is maximal and minimal in the “dominant” and the “recessive” homozygote genotype, while variable levels are observed in the heterozygous genotype. Pgp activity can be assessed in vivo by 99mTc-MIBI scintigraphy, a substrate of the Pgp-mediated transport. Aim of this study was to evaluate the ABCB1 genotype of epileptic patients resistant to medical treatment and to relate this to the pattern of 99mTc-MIBI uptake in the brain. Pgp genotype was evaluated in 16 drug-resistant epileptic patients. Patients were divided into two groups, respctively with high (n=6) and low (n=10) seizure frequency (cut-off >4/month), a negative prognostic factor. SPECT data were acquired 20-min after injection of 740 MBq of 99mTc-MIBI. ROI were drawn on intracerebral pixels displaying 99mTc-MIBI activity, corresponding to the choroid plexuses. To reduce sensitivity to partial volume effect, only the 3 slices with highest activity were selected. The dominant and recessive genotypes were both found in 2 patients. Activity counts in the group with high frequency seizures were significantly (p<0.05) lower than in the low frequency group (4.0±1.1 vs. 5.9±2.1). When only heterozygous patients were considered, statistical significance of this difference between the two groups increased (p<0.01). Lower early uptake of 99mTc-MIBI was detected in epileptic patients with clinically unfavourable prognostic factors. This finding may be related to reduction of drug delivery or increasing drug clearance. Because of the lower incidence of homozygous genotypes the relationship between ABCB1-genotype and 99mTc-MIBI scintigraphic findings could not be adequately evaluated. The ongoing analysis of uptake at later acquisition time-points, after inclusion of an adequate number of controls, will allow to properly address this issue.
P.A. Erba, None.
Abstract ID: 087
Superparamagnetic Iron Oxide (SPIO) nanoparticles, either modified or in combination with other macromolecules, are being used for intracellular magnetic labeling of stem cells and other cells to monitor cell trafficking, by Magnetic Resonance Imaging (MRI), as part of repair, replacement or treatment strategies. By combining SPIO with polycationic transfection agents (TA), endosomal incorporation of the nanoparticles occurs with high labeling efficiency, however commercially available TA are not made according to clinical good manufacturer practices (CGMP) nor are they FDA approved. Protamine Sulfate (USP) is an FDA-approved drug used to reverse heparin anticoagulation and can be complexed via electrostatic interaction to Ferumoxides (Feridex® Berlex, Wayne NJ) and used to magnetically label cells. Adherent cells (e.g., Mesenchymal stem cells (MSC), monocytes, macrophages), and cells grown in suspension (e.g., CD 34+ hematopoietic stem cells (HSC), T-cells) are efficiently labeled with ferumoxides-protamine sulfate complexes (FE-Pro). FE-Pro labeled cells demonstrated no short- or long-term toxicity or changes in function, differentiation capacity, or phenotype when compared to unlabeled cells. FE-Pro labeled HSC and MSC are able to home to tumors and areas of inflammation, and these migrations can be visualized with MRI. Since FE-Pro consists of two CGMP products, scale up procedures have been developed to allow for magnetic cell labeling in cell processing section of a blood bank, thereby facilitating its ultimate use in clinical trials where it is important to determine the temporal and spatial migration of stem cells to target tissues.
J.A. Frank, None.
Abstract ID: 088
Imaging has played an essential role in the diagnosis and staging of cancer since the introduction of radiography. Over the last twenty years, it was debated if chemotherapy was useful in improving symptoms or prolonging life, surgical treatment constituted the effective therapy for some cancers and chemo- and radiotherapy presented limited options and were generally used for short-term palliative effect. Today, even if the first line of chemotherapy fails, second and third line regimens have been developed and approved. As more choices become available, the need for rapid assessment of response emerged as an integral responsibility for management of treatment. Improved imaging choices have accompanied the changes in treatment available today and an early determination of the efficacy of a particular regimen is needed because agents are expensive and toxic. CT and MRI provide the tomographic imaging of anatomic structures and can measure the size of a tumor, are clearly useful and an accepted standard of care, but provide little information about its physiologic status. Positron emission tomography (PET) provides a way of measuring tumor physiology. While PET using fluorodeoxyglucose (FDG) is useful in a number of situations, it does not directly measure tumor proliferation. Agents to image this aspect of tumor metabolism have been widely sought, in order to detect cancers and measure their response to therapy. To this end a number of potential tracers have been explored over the years these have included [C-11]thymidine, [F-18]FLT (3′-deoxy-3′-fluorothymidine), and FMAU (2′-fluoro-5-methyl-1-beta-D-arabinofuranosyluracil). Of these, FLT is now the most widely available and is undergoing a thorough evaluation. Further work is ongoing to determine the relative merits of proliferative tracers and FDG. Depending on the type of tumor under study, its location, and the type of treatment being employed one may find certain advantages with one tracer or another.
A.F. Shields, PETNet 2; GE 2.
Symposium XIV: Enhancing Delivery and Traversing Barriers
Abstract ID: 089
The vascular endothelium forms a significant barrier preventing tissue-specific penetration of a wide variety of imaging and pharmacological agents injected into the circulation. Our proteomic mapping of the endothelial cell surface and its caveolae identifies key tissue- and tumor-induced proteins that can be immunotargeting to acheive single organ or solid tumor delivery in vivo. Live dynamic imaging via gamma-scintigraphy and intravital microscopy show that apon intravenous injection, antibodies targeting caveolae can be transported across the endothelial cell barrier in <1 min to penetrate effectively into the tissue parenchyma of one tissue of the body. We have targeted endothelial caveolae of solid tumors that improves pentration throughout the tumor and thus facilitates radioimmunotherapy to acheive complete remissions. This novel strategy of targeting caveolae to improve tissue penetration and targeting in vivo creates new opportunities to access the inside of specific tissues for drug and gene delivery as well as many functional imaging agents.
J. Schnitzer, None.
Abstract ID: 090
D.S. Wang, None.
Abstract ID: 091
Nanoplatforms are nanoscale structures that are being designed as general platforms to create a diverse set of multifunctional diagnostic and therapeutic devices. Synthetic imaging platform may be problematic due to biocompatibility and toxicity limitations. Low-density lipoproteins (LDL) are an endogenous transport vehicle in mammalian systems. These naturally existing nanoparticles (22nm) consist of a phospholipid monolayer encapsulating a hydrophobic core. Spanning the lipid monolayer is the apolipoprotein B-100 which is a recognition protein for targeting LDL to cells expressing the LDL receptor. LDL can be modified to incorporate contrast agents for magnetic resonance imaging (MRI). Here we describe preliminary animal experiments utilizing a novel LDL-receptor targeted MRI contrast agent.
The lipophilic chelate DTPA-Bis (stearylamide) (DTPA-SA) was synthesized and incorporated into LDL at a molar ratio of 200:1 (probe:apoB-100). Gd-DTPA-SA-LDL (0.04 mmol/kg) was administered i.v. into nude mice bearing subcutaneous HepG2 tumors in the hind limb. Animals not receiving any contrast agent served as controls. In controls, there is little intrinsic signal contrast between the liver/ dorsal thoracic muscle (A) and tumor/leg muscle (B). At 5 hrs post injection there is marked signal enhancement within the liver (70%) compared to baseline (C). However, at this time point the tumor enhances little (9%) relative to control (D). By 24 hrs the signal enhancement within the liver drops to 28% (E). In contrast the signal enhancement within the tumor shows a striking significant increase (33% over controls) (F).
The results obtained from these experments are important as it lays the frame work for high resolution imaging of such targeting nanoplatform schemes. The accumulation/amplification mechanisms of LDLR pathway allow us to surpass the threshold (~1mM) for intracellular MRI detection.

Long arrow indicates tumor; Short arrow head indicates liver parenchyma)
I.R. Corbin, None.
Abstract ID: 092
W. Hundt, None.
Abstract ID: 093
Barriers exist throughout the body that inhibit delivery of therapeutic agents. Such barriers may occur within the vasculature, where thrombosis may pose a barrier for entry and activity of thrombolytic agents, or at the cell surface where the cell membrane prevents entry or within a subcellular compartment. Acoustically active agents, in concert with ultrasound, afford the potential to overcome these barriers to achieve enhanced pharmacologic effect. Microbubbles can be cavitated by variations in ultrasound frequency and power transmission. Cavitation effect can be used to treat vascular thrombosis and to overcome barriers to drug and gene delivery. We have conducted both pre-clinical and clinical trials, studying the efficacy of microbubbles and ultrasound to lyse clots in occluded grafts and vessels.
Furthermore, a second generation of “targeted” microbubbles is being developed in our laboratory in conjunction with ultrasound for treating other intravascular occlusive diseases that may reside on the endothelial cell surface as in myocardial infarction and vulnerable plaque. Targeted microbubbles should improve upon therapy by concentrating microbubbles primarily in the area of disease thereby localizing ultrasound-mediated cavitation.
Finally, we are using ultrasound, in conjunction with targeted-delivery systems to overcome physiological barriers to efficiently deliver drug payloads via two strategies: 1) targeted microbubbles with drug payloads can be given intravascularly followed by ultrasound-mediated localization to endothelial surfaces and subsequent binding to receptors, or 2) targeted nanodroplets (<350 nm) that can diffuse into the interstitial space, bind to extracellular receptors, followed by ultrasound-mediated intracellular delivery. We are currently combining targeted microbubbles or Acoustically Active Lipospheres (AALs) with ultrasound-mediated delivery to explore localization and delivery of drug in the intravascular space. Alternatively, we are utilizing targeted nanodroplet technology and ultrasound to explore drug delivery systems that target extravascular or interstitial cell-surface receptors (i.e. regional disease) followed by ultrasound energy-mediated intracellular delivery of therapeutics.
E.C. Unger, None.
Symposium XV: Chemistry of Multifunctional Probes
Abstract ID: 094
New imaging techniques and agents are revolutionizing the study of biology and medical science1,2. For example it is now possible to do single molecule detection within fixed biological matrices and potentially during live cell imaging. Molecular imaging methodologies now allow very early-stage tumor detection in-vivo, evaluation of metabolic activity of suspect lesions and therapeutic efficacy.
Used clinically for many years, Lanthanide complexes are unique molecular imaging agents which provide a diversity of signatures. Unique to the Lanthanide complexes is long-lived luminescence that is spectroscopically shifted from the excitation (Stokes' shift ~300nm), inviting the use of simple gated imaging in the zero noise regime.
Multi-modal molecular imaging has the ability to significantly aid visualization and characterization of disease. Metal ion exchangeability makes Lanthanide chelates inherently compatible with multi-dimensional imaging methods. Recently we exploited Lanthanide complex multi-dimensionality by performing cellular scale bi-modal imaging using a Peripheral Benzodiazepine Receptor PBR targeted molecular imaging agent3. We have recently expanded upon these observations demonstrating subcellular detection of Ln-PK11195 by electron microscopy. In this presentation we report on new efforts that employ small molecule ligands and multiple signature Ln-chelates. These targeted agents have long circulating properties and include dendritic species. The synthesis of new and improved antennae for Lanthanide sensitization will be presented showing red-shifted and NIR Ln-chelates are possible. It will be shown that the multi-dimensionality of our Ln-chelate imaging “tool box” can aid the study of biology and medicine.
D. J. Bornhop, None.
Abstract ID: 095
J.V. Frangioni, None.
Abstract ID: 096
Combining both optical and nuclear imaging can provide coherent and more complete evaluation of the molecular target of interest than each method used lone. In this study, we report the first nuclear and NIR fluorescence-based optical imaging study using a single probe targeted to integrin αvβ3 in human melanoma xenografts. Cyclic pentapeptide containing Arg-Gly-asp (RGD) motif, c(KRGDf), was conjugated to both an NIR dye (ex/em, 765/792 nm) and a radiometal chelator DTPA. The conjugate was labeled with indium-111 with high radiochemical purity and radiochemical yield. Nude mice bearing s.c. human M21 (αvβ3+) and M21-L (αvβ3-) melanoma were divided into two groups: each mouse in group 1 received an intravenous injection of the imaging probe alone at a dose of 90 μCi equivalent to 5 nmol of fluorescent tracer (18 Ci/mmol), while in group 2, each mouse received the parent RGD peptide (600 nmol) followed by the injection of the imaging probe 1 h later. Both gamma scintigraphy and NIR optical imaging demonstrated selective targeting of the imaging probe to integrin αvβ3 in M21 melanoma. Furthermore, optical imaging generated greater signal-to-noise ratio and better resolution than nuclear imaging. Our results warrant future clinical evaluation of dual-labeled imaging probe in melanoma patients (Supported by Prostate Cancer SPORE Career Development Award CA90271, NIH grants R01 EB00174 and John S. Dunn Foundation).
S. Ke, None.
Abstract ID: 097
The possibility of a multi-functional probe that combines proteasetriggered photo activation, singlet oxygen quenching, photon activation, singlet oxygen liberation, together with a mitochondrial flavoprotein damage assessment provides a unique approach to cancer detection and therapy on a rational basis. Very recently, a protease–triggered photosensitizing (PS) beacon and a short peptide sequence, specific to an over expressed protease quenches singlet oxygen generation due to the FRET1. Upon cleavage of the Caspase-3 substrate, singlet oxygen is copiously generated and damages the tumor upon NIR activation. An in vivo PDT assay is afforded by the extreme oxidation of the “recently rediscovered” flavoprotein fluorescence of mitochondrial ketoglutarate dehydrogenase as has been observed in BChl PDT (unpublished data) and provides an unique signal of mitochondrial damage, presumably by apoptosis. The principle of the beacon is indicated in Fig. 1

The Mitochondrial flavoprotein, redox state is shown to be rapidly oxidized as a sensitive in single oxygen induced apoptosis, and thus provides an on-line, in vivo assay of tumor.
Abstract ID: 098
Radioligands that are specific for the serotonin 5-HT1A receptor will be useful in characterizing the physiological action of this receptor subtype. With radioligands of varying pharmacokinetic properties, investigators can measure not only receptor density, but also the effect of endogenous serotonin. To this end, four fluorinated analogs of WAY-100635 were prepared and evaluated as 5-HT1A receptor ligands of varying pharmacokinetic properties. The four compounds are cis-4-fluoro-, trans-4-fluoro-, cis-3-fluoro-, and trans-3-fluoro-N-{2-[4-(2-methoxyphenyl)piperazinyl]ethyl}-N-(pyridin-2-yl)cyclohexane-carboxamides (FCWAYs). All four compounds were radiolabeled with fluorine-18. We then determined in vitro inhibition constants at the 5-HT1A receptor; in vitro metabolic profile using rat hepatocytes and LC/MS; in vitro subtype specificity for 5-HT1A; the rate of defluorination, and hippocampus to cerebellum ratio ex vivo. This led to the conclusion that high affinity 4-trans F-18 FCWAY had the best properties for measuring receptor density given its high H/Cb ratio and 3-cis F-18 FCWAY had the best properties for measuring dynamic change in receptors, with lower affinity and faster pharmacokinetics. The closest competitor is the previously reported compound with 1,3 pyrimidine substituted for pyridine in FBWAY (FPWAY). It has slightly poorer properties when compared to cis 3-FCWAY with a H/Cb ratio of 6.5 compared to a H/Cb ratio of 10 for cis 3-FCWAY. Neither of these compounds has yet to be shown to be sensitive to changes in serotonin concentration in vivo and, until that has been shown, trans 4-FCWAY is the compound of choice. However, two compounds, cis 3-FCWAY and FPWAY, are now available to test whether serotonin ligands can be used to measure endogenous serotonin. An optical derivative of this class of compound using coumarin in place of cyclohexane carboxylic acid did not have high affinity for the 5-HT1A receptor. Small changes in the structure of the acid moiety has profound effects on the biochemical properties.
W.C. Eckelman, None.
Symposium XVI: Imaging Gene Therapy and Advanced Vector Design
Abstract ID: 099
A significant challenge in optical imaging of bioluminescence reporter genes in vivo is to obtain high-level expression in order to visualize signals within tissues of live animals. Our laboratories developed an imaging paradigm based on the concept of two-step transcriptional amplification (TSTA). The TSTA system comprises a cell-specific promoter driving an optimized version of the potent artificial transactivator GAL4-VP16, which in turn activates firefly luciferase greater than 100-fold above that of one-step systems. We tested the ability of this system to detect promoter activity deep within tumors implanted into SCID mice and to respond dynamically to systemic signals that controlled promoter activity. The prostate specific antigen (PSA) enhancer is driven by the androgen receptor (AR), a DNA binding transactivator that requires the ligand dihydrotestosterone. A PSA TSTA adenovirus injected into prostate cancer xenografts generated robust levels of firefly luciferase as detected by the Xenogen In Vivo Imaging System. The TSTA approach permitted visualization of tumor progression and response to drug therapies much more sensitively than standard benchmarks like serum PSA levels. The TSTA approach can be modified to measure two or more signals simultaneously. The PSA-TSTA-MAPK system, replaces GAL4-VP16 with GAL4-ELK1, and activates luciferase expression only in the presence of AR and MAPK. The TSTA-MAPK system responds dynamically to signals that activate AR and MAPK and is inhibited by small molecule pharmaceuticals that intervene in the AR and MAPK pathways. We describe a series of vectors, where the cell specific promoter, GAL4-derivative or reporter gene can be easily swapped out for other applications in cancer and disease research.
M.F. Carey, None.
Abstract ID: 100
Supported in part by MSWF-516-400 002 99, ZMMK-TV46 and DFG-Ja-981/1-2.
A.H. Jacobs, None.
Abstract ID: 101
We recently developed a new chimeric suicide gene by fusing HSV-tk to the extracellular and transmembrane domains of human CD34 (CD34-tk). In this study, we used in vivo bioluminescence imaging to determine whether delayed donor lymphocyte infusion (DLI) of CD34-tk-expressing T cells and treatment with ganciclovir (GCV) could provide a graft-versus-leukemia (GVL) effect in the absence of graft-versus-host disease (GVHD) after allogeneic bone marrow transplantation (BMT). To induce leukemia, BALB/c mice were lethally irradiated and reconstituted with T cell depleted (TCD) BM only or with TCD BM and A20-luc/egfp leukemia cells (B cell lymphoma of BALB/c origin expressing a luciferase-EGFP fusion gene). Ten days post-BMT, we administered a DLI of CD34-tk-expressing T cells. Eight of ten recipients who received TCD BM and A20-luc/egfp cells exhibited tumor engraftment and growth. In contrast, we found no significant tumor signal in untreated leukemic mice that received a DLI. However, these untreated animals developed lethal GVHD. To evaluate the ability of GCV to prevent this GVHD and maintain a GVL effect, we administered a 7-day course of GCV beginning 1, 4, or 10 days post-DLI. Both day 1–7 and day 4–10 treated leukemic animals were protected from GVHD. However, 60% of the day 1–7 treated mice and 58% of the day 4–10 treated animals developed leukemia, with an overall survival that was not significantly different than the leukemia controls. In contrast, only 20% of the mice developed leukemia if GCV treatment was delayed until day 10 post-DLI. Importantly, this GVL effect was obtained in the absence of GVHD, as evidenced by day 10–16 GCV treated mice having an overall survival, weight gain, and lymphoid reconstitution that were not significantly different than TCD BM only controls. These results further demonstrate the potential of the HSV-tk/GCV system to reduce acute GVHD without impairing GVL activity.
M. Rettig, None.
Abstract ID: 102
Treatment and prevention of Hepatitis C virus (HCV) infections remains a major challenge for controlling this worldwide health problem; existing therapies are only partially effective and no vaccine is currently available. RNA interference offers the potential of a novel therapeutic approach for treating HCV infections. Toward this end, we evaluated small hairpin interfering RNAs (shRNAs), targeting the conserved internal ribosome entry site (IRES) element of the HCV genome, for their ability to control gene expression in cells and animals. We used a dual reporter luciferase plasmid (generously provided by Peter Sarnow, Stanford University) where firefly luciferase (fLuc) expression was dependent on the HCV IRES and Renilla luciferase (rLuc) was constitutively expressed. Direct transfection of HCV IRES shRNAs, or alternatively shRNAs expressed from polIII-promoter vectors, efficiently blocked HCV IRES-mediated fLuc expression in 293 cells. Control shRNAs containing a double mutation had little or no effect on fLuc expression, and shRNAs containing only a single mutation showed partial inhibition. These shRNAs were also evaluated in a mouse model where DNA constructs were delivered to cells in the liver by hydrodynamic transfection via the tail vein. The dual luciferase expression plasmid, the shRNAs, and another control plasmid encoding secreted alkaline phosphatase (pSEAP2) were used to transfect cells in the liver, and the animals were imaged at time points over 12 to 96 hours using the IVIS in vivo imaging system (Xenogen Corp.). Imaging revealed that HCV IRES shRNA directly, or alternatively expressed from a polIII-plasmid vector, inhibited HCV IRES-dependent reporter gene expression; mutant or irrelevant shRNAs had little or no effect. These results suggest that shRNAs, delivered as RNA or expressed from viral or nonviral vectors, may be effective antivirals for the control of HCV and related viruses.
Q. wang, None.
Abstract ID: 103
“Molecular imaging”, as distinct from structural (anatomic) or functional imaging, aims to non-invasively characterize and quantify cellular and sub-cellular events in intact organisms. Cystic fibrosis (CF) is the most common inherited disease of Caucasians, and can serve as a paradigm for how molecular imaging may be applied in the future to the management of lung disease. Much of the morbidity and mortality in this disease is the result of airway infection and inflammation. Neutrophils are the prominent inflammatory cell in the lungs of patients with CF. Accordingly, neutrophilic airway inflammation has become a therapeutic target in CF. A number of molecular imaging methods are being developed to monitor and quantify inflammation, including methods to directly label inflammatory cells or mediators, or to measure the rate of uptake of [18F]FDG by activated neutrophils. Recently, we found a positive correlation with the rate of [18F]FDG uptake and the rate of FEV-1 decline in patients with CF.
With the prospect of preventing, not just treating, pulmonary disease, gene replacement therapy of the underlying defect in the cystic fibrosis transmembrane regulator (CFTR) gene has been considered the most exciting potential therapeutic strategy for CF. However, a significant limitation to evaluating gene therapeutics in CF patients is a lack of clinical endpoints. Non-invasive, molecular imaging for reporter genes, delivered concurrently with CFTR, could provide information regarding the onset and duration of transgene expression. The ability to perform serial measurements should help with evaluations of gene delivery systems and to provide information relevant to vector dosing. Recent studies by our group demonstrate the feasibility of employing molecular imaging techniques to monitor the distribution of transgene delivery, the efficiency of different delivery systems, and the duration of transgene expression in the lungs.
D.P. Schuster, None.
ABSTRACTS FOR POSTERS PRESENTATIONS
Linking Imaging, Proteomics & High Throughput Screens
Abstract ID: 104
We have examined MR contrast heterogeneity in tumor tissue at the genetic and cellular levels using a murine squamous cell carcinoma model. Using MR-guided tumor tissue sampling and microarray analysis, the receptor of the mitogenic gene product, platelet-derived growth factor (PDGF-R), was found to be upregulated in the contrast-enhanced areas of the tumor relative to the non-enhanced areas. This finding was confirmed by immunohistochemistry, and autoradiography. In addition. I125-labeled antibody to PDGF-R was used to image the distribution of this targt in vivo using whole body nuclear scintigraphy. These data support a role for PDGF-R in the molecular mechanism that leads to contrast heterogeneity of a tumor, implicating a key mitogenic signaling pathway that can be imaged in vivo.
S. Guccione, None.
Abstract ID: 105
Efficacious, individualized carcinoma diagnosis/therapy may involve targeting of patient-specific biomarkers. In vivo combinatorial phage display utilizes affinity selection to identify peptides that bind to relevant biomarkers in the context of the whole body. Previously, we have optimized in vivo selections to identify phage that extravasate to target carcinoma, rather than vascular biomarkers. We hypothesize that in vivo selections will identify peptides with superior in vivo stability and tumor-targeting ability. A fUSE5 15-amino-acid peptide library, depleted of peptides binding normal targets, was injected into severe combined immune deficiency mice with heterotransplanted human PC-3 prostate tumors. Extravasated, tumor-bound phage were eluted in 0.5% CHAPs detergent buffer. All eluted phage were amplified for the next selection round. Prostate-avid phage yield was enriched 100-fold between rounds 2 and 4. A “tumor-to-cell-micropanning” assay identified phage with high affinity for prostate tumor tissue/heterotransplanted cell lines relative to normal (irrelevant) targets. Synthesized peptides (G1, D4) were analyzed using live cell surface binding (A) and laser scanning confocal microscopy of fixed cells (B). (A) The peptides had significantly higher affinity (Kd ≈ 800 pM) for PC-3 cells compared to PC-3M (Kd ≈ 80 nM) or MDA-MB-435 cells (no binding observed). (B) The peptides labeled PC-3 with higher intensity than PC-3M or MDA-MB-435 cells. Fluorescent-labeled D4, but not G1, was internalized by PC-3 cells, which suggested that different biomarkers were targeted. Thus, the in vivo-selected peptides have potential applications as agents to target diagnostic and therapeutic chemicals to carcinoma cells.

L.A. Landon, None.
Abstract ID: 106
Telomerase is a ribonucleoprotein with an included RNA-template included for reverse transcriptase activity. Increased telomerase activity is found in the majority of malignant tumors but is absent from or significantly lower in normal cells, it therefore serves as a marker for malignancy e.g. in cytology specimen. Measuring telomerase activity is usually done with an assay that includes PCR and is therefore prone to artifacts. We have developed an assay to sense telomerase activity with MRI based on the magnetic relaxation switch (MRS) technology. In the current study we exploit the regulation of telomerase by kinases and phosphataes. We also expand the MRS technique to detect the presence of the catalytical subunit hTERT of telomerase in crude cell extracts by MRI as well as an approach to evaluate the length of the telomeres, the capping structure at the end of each single chromosome. Our results show that these assays require considerably less steps than conventional assays and are highly suitable to allow for an integrated approach to detect enzyme function (activity), processivity (telomere length) and expression (hTERT) within the same system.
J. Grimm, None.
Abstract ID: 108
Tissue molecular complexity and in vivo inaccessibility of cells within a tissue hinder genomic and proteomic analysis in discovering useful targets for directing tissue-specific pharmacodelivery. We use high-through put proteomic analysis of tissue subcellular fractions to reduce the molecular complexity of tissues and thus uncover the few key proteins induced at the tissue-blood interface inherently accessible to antibodies injected intravenously. Expression profiling and γ-scintigraphic imaging validates targets as specifically exposed in vivo to permit immunotargeting of single organs and solid tumors in < 1 hour. Targeted radio-immunotherapy destroys tumors to increase animal survival. These endothelial targets are expressed in multiple human primary and metastatic tumors. Immunotargeting caveolae permits transendothelial transport within minutes to improve tissue penetration. By coupling our mapping of tissue- and disease-modulated expression of endothelial cell surface and caveolar proteins with in vivo imaging, we can discover and rapidly validate key promising iv-accessible targets useful in directing functional imaging agents as well as drug and gene therapies in vivo.
J. Schnitzer, None.
Abstract ID: 110
Near infrared fluorochromes (NIRF) (Cy5.5, Cy7) are being used with great efficacy in tumor detection. The NIRF must be specifically targeted to a carcinoma biomarker by a homing molecule. Our laboratory has isolated tumor-homing bacteriophage, which target the carcinoma-associated Thomsen-Friedenreich antigen (TFA). Three peptides (P30, P30-1, P30-2), when synthesized or displayed on bacteriophage, bind TFA with high affinity. In vivo biodistribution studies in mice show that bacteriophage virions are stable and well tolerated at high concentrations. Importantly, extended incubation in vivo allows bacteriophage to extravasate to target tumor cells. Here, we hypothesize that bacteriophage displaying peptides, which specifically target carcinoma cell antigens, can be used as specific, high affinity targeting vehicles for NIRFs in tumor detection. We have labeled bacteriophage displaying anti-TFA peptides with radiolabels (99mTc, N2P2/THP chelators) and optical labels (Oregon Green 488 (OG)/Cy5.5 analog Alexa Fluor 680 (NIRF), N-succinimidyl ester linkers) and have found that the labels do not interfere with binding of the displayed peptides to TFA. In confocal microscopy studies (A-E), optically-labeled peptide-displaying bacteriophage (A, D, E), but not peptide-free bacteriophage (B), bind to TFA on MDA-MB-435 human breast carcinoma cells. In biochemical assays, the unlabeled peptide-displaying bacteriophage bind to TFA with high affinity (P30-1, fUSE5, 3 peptide copies; P30-2, f88 bacteriophage 100 peptide copies; P30, f88, Kd ≈ 1012 virions/ml). Similar experiments indicated that the NIRF label does not reduce the affinity of bacteriophage for TFA. These results indicate that bacteriophage have prospective uses as targeting vectors for NIRF in fluorescence detection of carcinoma tumors.
J.R. Newton, None.
Abstract ID: 111
The domain of molecular imaging is the merger of current advances in the fields of molecular biology and imaging research. Yet, an information gap exists between the scientists who discover new gene products and the imaging scientists who can exploit this information. The research objective is to bridge this information gap through the development of a molecular imaging knowledge base consisting of molecular images, literature, and genomic information using biomedical informatics techniques.
To achieve this research goal, we developed a molecular imaging terminology to represent the concepts in the molecular imaging domain. The terminology will form the foundation for the knowledge base creation. In order to populate the knowledge base, we are using automated information extraction techniques to extract key entities from the molecular imaging literature. BioMedLEE is a natural language processor for the automated extraction of biological information consisting of biomolecular substances and phenotypic data. We are adapting BioMedLEE to recognize relevant molecular imaging entities, and to identify valuable relationships between molecular imaging probes and targets, and molecular targets and diseases.
We have processed molecular imaging abstracts from the Molecular Imaging Journal and other scientific journals. These documents will be integrated with relevant images, literature documents, and genomic information. In addition, we will link to molecular interactions automatically extracted by GeneWays. GeneWays is a system for mining text and integrating data on molecular pathways. In order to facilitate the visualization of images, literature and genomic information, various visualization tools have been developed.
Ultimately, the development of this knowledge base, coupled with the integration of disparate data sources and links to external knowledge bases, will lead to a valuable information resource for physicians and researchers.
P. Tulipano, None.
Abstract ID: 112
H. Kenis, None.
Abstract ID: 113
J.V. Frangioni, None.
Instrumentation & Computational Challenges in Molecular Imaging
Abstract ID: 114
The development of small animal tomography for quantitative determination of endogenous or exogenous fluorescent dye concentrations within tissue compartments could have significant implications for increasing throughput within the drug discovery pipeline. Yet there are significant challenges that must be met before small animal fluorescence tomography can be realized. With continuous wave, or time-invariant methods, the fluorescent contrast can be small and can be impacted and attenuated by optical property variation of internal organs. In addition, owing to the small animal volumes probed, the diffusion approximation to the radiative transport equation describing light propagation is not likely met, further complicating the development of small animal optical tomography.
Herein we present developments of time-dependent, frequency-domain photon migration (FDPM) measurements between points of illumination and collection on a cylindrical surface housing a nude mouse. We present the tomographic approach for reconstructing quantitative fluorophore concentration. FDPM measurements consist of launching intensity modulated (100 MHz) excitation light at visible wavelengths at points along the circumference of the cylinder and determining the phase delay and amplitude attenuation of the propagated excitation light as well as the generated fluorescent light. The tomographic reconstruction algorithm is based upon the radiative transport equation weighted backprojection (WB) method which provides greater performance than the diffusion weighted backprojection, owing to its increased accuracy for predicting light transport in small volumes.
In this presentation, we demonstrate that FDPM phase and amplitude measurements can be used to tomographically reconstruct the fluorophore concentration even in the presence of heterogeneous absorption. These results can be expected owing to the increase in contrast arising from fluorophore lifetime that can be probed effectively only with time-dependent methods. In addition, comparison of the forward imaging solution using the diffusion approximation and full radiative transport equation show that the weighted backprojection using diffusion analysis represents the worse case image.
J.C. Rasmussen, None.
Abstract ID: 115
Our institution has obtained a novel research prototype pre-clinical CT scanner that utilizes dual, digital flat panel detectors and a standard CT x-ray tube (developed by GE Global Research Center, Niskayuna, NY). Each volume data set, with z-axis coverage from 1.4 - 4.2 cm per gantry rotation, is obtained in 2 −8 seconds. The system is operated in either single or dual panel scan mode, with maximum fields of view in each mode of approximately 12cm and 30cm respectively. Multiple steps can be acquired in one scan, providing the capability to image entire small animals like mice and rats, and entire organs of larger animals such as dogs. The spatial resolution within reconstructed volumes was 1.9 - 3.1 lp/mm at 10% MTF, using various reconstruction filters. This provides resolution of 170 - 260 microns for typical applications. With the help of our veterinary team, we have developed a breath-hold technique that maintains a constant pressure of lung inflation during the scanning procedure. This technique has been successfully applied to laboratory imaging applications for animals as small as 15 g. Preliminary imaging has produced high quality images of mouse and rat lungs with minimal motion artifact (single panel mode coronal view after IV contrast injection, figures 1&2), as well as a magnified axial view of a lung tumor in a dog (dual panel mode, Figure 3). The scan acquisition parameters used for generating figures 1–3 are shown in the Table.


D.D. Cody, General Electric Global Research Center 1.
Abstract ID: 116
The apparent diffusion coefficient (ADC) of 23Na+ was determined in rat brain. Na+ resides primarily in the extracellular space in healthy tissue. For intact brain, the intracellular-to-extracellular content ratio for Na+ is ~8:2, whereas it is the reverse for H2O. Thus the ADC of Na+ reflects diffusion in the extracellular space, while that of water is weighted more to diffusion in the intracellular space. ADC values for healthy rat brain and following global ischemia are shown below. The values for water ADC are taken from the literature.

The reduction in ADC from solution to intact brain is 30% for Na+ and 70% for H2O. Assuming that the determinants of Na+ and H2O ADC are similar, this suggests that the ADC of water in the extracellular space is roughly two fold greater than that of the intracellular space. The brain Na+ ADC decreased ~70% after global ischemia, consistent with a marked increase in extracellular restriction/hindrance to diffusion due to cell swelling and with the ingress of Na+ into the more restricted/hindered intracellular space upon loss of cellular ion homeostasis.
J. Goodman, None.
Abstract ID: 117
J.V. Frangioni, None.
Abstract ID: 120

Z. Cao, None.
Abstract ID: 121
Molecular imaging requires the identification and spatial localization of a specific marker and its quantitative analysis. Based on the stimulated acoustic emission (SAE) of single air-filled microparticles (MPs) we established single particle acoustical quantification (SPAQ) which is an ultrasound-derived imaging method with spatial resolution in the lower micrometer range. SPAQ enables the quantification of acoustical signals of static MPs with high accuracy.
To test the potential biological and medical feasibility of SPAQ, we performed contrast-enhanced ultrasound imaging in rat experimental autoimmune encephalomyelitis (EAE), an established inflammatory disease model of human multiple sclerosis (MS), with MPs targeted to intercellular adhesion molecule 1 (ICAM-1) expressed on lesion-associated blood vessels.
Specific MPs were created by using streptavidine loaded MPs and a biotinylated anti-rat-ICAM-1-antibody. We checked for feasibility and specificity of our system first, in an ex vivo approach followed by in vivo experiments.
The brains from EAE rats showed significantly more MP-based acoustical signals compared with their respective controls. Furthermore, pre-treatment with anti-ICAM-1 antibodies clearly diminished the acoustical signals from the CNS tissue of EAE rats, demonstrating the specificity of the ICAM-1-specific MPs. Finally we could demonstrate a clear signal reduction in EAE rats that were treated with corticosteroids before the application of the ICAM-1-specific MPs (monitoring of treatment effects).
The results demonstrate that SPAQ of antibody-MP conjugates enables depiction of molecular events at the blood-brain barrier (BBB) and provides monitoring of treatment effects at the molecular level with high sensitivity and specificity in vivo.
P. Hauff, Schering AG 5.
Abstract ID: 122
One of the central challenges in molecular imaging is to achieve high-resolution 3-D mapping of molecular contrast agents in vivo. High-resolution imaging (<10 μm) using the traditional optical imaging methods, such as fluorescence microscopy or confocal microscopy, have limited depth-mapping ability (penetration depth limited to 200 μm in most tissues). Optical coherence tomography (OCT), however, is a cellular-resolution imaging modality with superior depth penetration (typically 2 mm). Through time-frequency analysis of the interference fringes of the back-scattered light, many absorbing molecular probes can be mapped using spectroscopic OCT (SOCT).
We have constructed and optimized the hardware and software components for performing SOCT. Using a broadband Ti:sapphire laser centered around 800 nm wavelength, the system is able to generate images with 5×5 μm lateral resolution and 3 μm depth resolution. By using appropriate signal detection and estimation method, near-infrared dyes at concentrations as low as 10 μM can be mapped in three dimensions.
This technique is applicable to many types of molecular probes, ranging from common fluorescence molecular probes to newly developed plasmon-resonant nanoparticles, as long as the particular probe has significant absorption within the available laser spectrum. For protein or nucleic acid imaging, hybrid probes can be constructed by conjugating near-infrared chromophores to specific antibodies or dyes.
We have demonstrated the mapping of a near-infrared fluorescent dye in vivo and compared SOCT images with those acquired with fluorescence microscopy. We conclude that in addition to high-resolution anatomical imaging, SOCT provides a high-resolution, deep-penetration molecular imaging modality that links the coherence gating properties of OCT with the detection of a wide range of molecular probes that have been traditionally designed for fluorescence studies.
C. Xu, None.
Abstract ID: 123
A semiconductor (SC) gamma camera combined with high resolution and high sensitivity could be a potent modality for providing information on small objects. In this study, we assessed the efficacy of the SC gamma camera for small animal imaging, using rats.
A gamma camera based on the Cadmium-Telluride SC detector and equipped with a parallel-hole collimator was compared with a conventional SPECT camera based on a sodium iodide scintillation detector, with a pinhole collimator. The right middle cerebral arteries (MCA) of rats were occluded, and 24 hr later, [99mTc]HMPAO (370 MBq) was injected, followed by 1-min static data acquisitions for the SC camera (planar images) and 20-min acquisitions for the SPECT camera (summed images). The brains were sectioned at 2-mm thickness to obtain coronal slices, which were imaged with the SC camera (5 min). In addition, using 50 MBq of [123I]MIBG, the rat heart imaging was verified with dynamic acquisition at 1-min intervals for the SC camera. The effect of desipramine on [123I]MIBG uptake to the cardiac muscle was also investigated.
Reduced CBF in the right MCA territory was clearly visualized with both cameras in the in vivo imaging experiments, however, the image quality was higher with the SC camera, despite a shorter acquisition time for this instrument. Ex vivo imaging of brain slices with the SC camera showed a significantly decreased accumulation of [99mTc]HMPAO in the MCA territory, which is consistent with the quantitative result obtained by the gamma counting. Furthermore, the in vivo heart accumulation curve of [123I]MIBG was obtained. Thirty-minute pretreatment with desipramine decreased the accumulation in its early stage.
Thus, the SC camera visualized the focal ischemia in MCA-occluded rats, and the cardiac sympathetic function. These preliminary results suggest that the gamma camera based on the SC detector provides a useful means for small animal imaging.
H. Kawashima, None.
Abstract ID: 124
Gd-based contrast agents are the primary choice of labeling in MRI. They have been successfully incorporated into molecular events related to cellular development stages. Contrast enhancement drops drastically at magnetic field strength higher than 3T due to the relaxivity decreasing as the field strength increases. To compensate for the loss of relaxivity, higher concentrations of contrast agents are needed. Previously, we had reported a unique method, paramagnetic relaxation enhancement in off-resonance rotating frame, to overcome the relaxivity loss without using more contrast agents (H. Zhang et al,
H. Zhang, None.
Abstract ID: 125
In this work, we determine the optimal pinhole configuration for a high-resolution seven-pinhole SPECT system used for imaging tumors and gene expressions. The pinhole configuration was determined based on geometric criteria viz: 1) place the pinholes evenly in the vertical and horizontal directions of the collimator so that a large number of transaxial planes in the animal are completely sampled with minimal overlap, 2) select the pinhole arrangement that gives the aximum sensitivity, 3) select a configuration that maximizes the distance between adjacent pinholes and 4) arrange the pinholes such that the projections from the VOI containing the tumor and surrounding tissues are not truncated. Based on these criteria, two seven-pinhole configurations were selected. The first seven-pinhole configuration gave the maximum sensitivity in the projection data while the second seven-pinhole configuration had the maximum distance between the pinholes (less overlap in projection data). The reconstructed images from the two seven-pinhole configurations were evaluated with those obtained from a single-pinhole SPECT system using a simulated mouse phantom. Two lesions with diameter of 6.23 mm and 3.58 mm were simulated on either side of the abdomen close to a hot bladder to study the worst-case scenario. Image degradation factors such as pinhole diameter, crystal PSF, decrease in sensitivity and Poisson noise were modeled in the projection. The two seven-pinhole configurations resulted in a faithful reconstruction of a larger volume, reducing the chance of systematic errors propagating into the quantitative values from the VOI. A 100% improvement in contrast-to-noise ratio and a 40% reduction in the normalized standard deviation was measured for the seven-pinhole system configuration compared to the single-pinhole case.
The MOBY mouse phantom with a lesion of 6.2 mm and 3.5 mm on either side of the abdomen. (A) Original phantom. Reconstructed image (5 iter, 15 subsets) obtained using (B) single pinhole and (C) seven-pinhole collimator. The image is scaled down to show the reduction in noise in the background tissue for the seven-pinhole case compared to single pinhole

G. Bal, None.
Abstract ID: 126
The purpose of the present study is to conduct local and global analyses of FRET microscopy in linearized regions of interest in cellular plasma membranes using the multi-resolution properties of the continuous wavelet transform. The genetically encoded fluorescent reporter of protein kinase C (PKC) phosphorylation activity was expressed in COS1 cells. FRET microscopy was employed to observe PKC activity stimulated by either phorbol-12,13-dibutyrate (PDBu) or acetylcholine (ACh). The plasma membrane regions within these images were re-defined into continuous regions of interest (ROI) from statistical comparisons of FRET matrices at various time points. FRET values within the ROI were linearized and subjected to texture and wavelet analysis (Matlab, Mathworks). The wavelet transform produces a matrix of coefficients localized in both the frequency and space domains. Treatment with PDBu and ACh resulted in a small transient increase in the mean FRET intensity, followed by a continuous stable decrease. Mean FRET values in the re-defined ROI were much greater than those in the manually derived ROI. Visual inspection of 2D-pseudo-colormaps of coefficient matrices revealed the heterogeneity of FRET signals at high and low scales. Standard deviations of wavelet coefficients calculated as a function of scale supplied an objective measure of coefficient dispersion, and prolonged exposure to PDBu and ACh yielded more homogeneous FRET distribution in the plasma membrane, which was in agreement with the results from the standard texture analysis. The developed analysis of FRET microscopy revealed heterogeneity in the functional signaling microdomains representing a dynamic balance of PKC and phosphatase activity. Wavelet-based image analysis of FRET microscopy provides a means of detecting and quantifying dynamic changes in functional signaling domains of PKC activity in plasma membranes.
E. Kobrinsky, None.
Abstract ID: 127
In vivo 1H NMR spectroscopy is increasingly used for biochemical and biomedical research in biological objects. However, in many cases the spectral information is limited because the spectra often suffer from poor resolution due to variations in the isotropic bulk magnetic susceptibility in biological objects, resulting in a broadening of the NMR lines. In principle this broadening can be averaged to zero by magic angle spinning (MAS). However, a problem is that in a standard MAS experiment spinning speeds of a kHz or more are required in order to avoid the occurrence of spinning sidebands (SSBs) in the spectra. At these spinning speeds the large centrifugal forces cause severe structural damage in larger biological objects, restricting this technique for studies of small cell agglomerates. It has been shown recently that with a method called phase-corrected magic angle turning (PHORMAT), where a special radio frequency pulse sequence is combined with MAS, even at a 1 Hz MAS speed a spectral resolution was obtained similar to that observed with fast MAS. In this presentation, the development of a first generation of volume-selective PHORMAT using pulsed field gradients will be reported along with possible ways for further improving the method. It is anticipated that in vivo PHORMAT will significantly enhance the utility of proton MR spectroscopy for biomedical research, molecular diagnostics, and therapy evaluations in live animals, and perhaps even in humans, in the latter case by rotating the external magnetic field rather than the patient.
J. Hu, None.
Abstract ID: 128
High resolution imaging devices to assess human disease models in small animals, including a microSPECT-CT system, have been realized recently. Many biological experiments would benefit from the ability to quantify radionuclide uptake on an absolute basis. In clinical SPECT, absolute quantification has been challenging due to a large fraction of scattering and attenuation in human bodies. To investigate absolute quantification in small animals, we have developed a mouse lesion phantom, which consists of a Co-57 (122 keV) containing warm cylindrical body having a diameter of 2.5 cm with 1 to 5 mm cubic lesions to simulate organs and tumors. The phantom was scanned with SPECT and CT using X-SPECT (a microSPECT-CT system, Gamma Medica, Inc., Northridge, California) fitted with a low-energy high-resolution parallel-hole collimator. The SPECT data were reconstructed with various algorithms including OSEM with collimator spatial response (CR), and OSEM with attenuation correction (AC) and CR. The regions of interests were defined in the microCT images, and the counts were measured in the co-registered SPECT images. OSEM-CR generated the higher resolution SPECT images than those without CR. For a 4-mm lesion, 80% of activities were recovered from imaging. AC did not improve quantitative accuracy significantly, since the fraction of attenuation and scatter is small in this object size. This study indicates that the radiotracer concentration is quantifiable in small animal SPECT imaging. Higher resolution pinhole SPECT imaging will improve quantification to smaller lesions.
K. Iwata, None.
Abstract ID: 129
Bone density is often chosen as a biomarker or end-point for a number of studies. A non-invasive means to measure the density of certain bones of interest in a rodent and to repeat the measurements along a time course is very important and much needed. In this study, microCT was used to scan certain mose models and CT numbers were converted into densities. The conversion was acomplished by imaging a group of mimirods of know densities. The materials (and their densities) of these rods include PVC (1.3750), acrylic (1.0964), magnesium (1.4622), aluminum (2.1221). We also use water (1.0) and air (0.0). A microCT scan was done using the CT componenet of XSPECT (GammaMedica) on these mimirods put in a tube filled with water. CT number for each material was obtained by averaging imaging values of the voxels from a volume region inside each material. The data poins were used to create a relationship between CT number and density. The result is a mapping scheme consists of three linear segments along CT numbers: −1024~200, 200~2916, 2916~. The shape of this three line-segments conversion function was comfirmed by clinical scanner with lower spatial resolution. As a initial testing, dead transgenic as well as wild type mice were scanned and the images were reconstructed into voxels of 50 micron size. The CT number of each voxel was converted into density using the conversion established. By viewing regional imaging data using interactive imagre procesing procedures, density values at 1.15 was chosen as the aboundry between soft tissues and bony structures. To compare the results for the same bones at similar positions between transgenic and wild type mice, image registration was used to aligned the bones from two types of mice. Challenges, main from motion blurr, remain when we use this method for live animals.
Z. Lee, None.
Abstract ID: 130
Intracellular sodium contributes significantly to triple quantum (TQ) sodium signals, providing a valuable cellular parameter on several molecular mechanisms maintaining sodium homeostasis. In many cases, the extra-cellular sodium concentration remains unchanged, thus TQ sodium signals can impart direct information about changes in intracellular Na. The main disadvantage of the TQ signal is that it is only a fraction (~6%) of the total single quantum (SQ) sodium signal.
Dramatic improvement in efficiency of the TQ signal detection (up to 5 times) can be achieved by avoiding filtration and acquiring a total TQ signal. The pulse sequence 90-t-90-90 commonly used to detect triple quantum filtered (TQF) sodium signals was modified by implementing a simultaneous increment of inter-pulse delay (t = n*Δ) and phase (φ = n*90). Detection of the TQ signals using time proportional phase increment (TQTPPI) does not cancel the SQ signal and acquire TQ and SQ signals concurrently as separate peaks. There is no need to perform an optimization of inter-pulse delay, the durations of 90 pulses are not crucial and a small tissue RF power deposition relative to the spin-lock version of the pulse sequence is a valuable advantage.
The pulse sequence was tested in a 9L rat glioma model. After BCNU chemotherapy treatment, the tumor sodium SQ signals increased 30% relative to the non-treated tumor. Sodium TQ/SQ signals from the whole head at the same time decreased from 32% in the normal brain to ~20% in the tumored brain. The opposite changes of TQ and SQ signal during tumor growth and treatment make the ratio of TQ/SQ a practically convenient and sensitive measure of the molecular changes taking place in tumor. A decrease in the ratio TQ/SQ ratio correlates with a corresponding increase in tumor ADC, supporting the predictive power of a sodium MR biomarker for anticancer therapy.
V.D. Schepkin, None.
Abstract ID: 131
One feature that makes in vivo molecular imaging particularly alluring is the ability to quantify physiologic function so that changes due to disease and treatment may be objectively measured and tracked. One challenge in this is the blood volume of small rodents is limited. Herein we demonstrate that it is feasible to measure the whole blood arterial input in rats given a high-sensitivity continuous flow detector.

C.A. Salinas, None.
Abstract ID: 132
The purpose of the study was to evaluate a special X-SPECT, a commercial SPECT/CT system from Gamma Medica, Inc., for small animal imaging. Different from the standard X-SPECT, the special system has two 12.5cm × 12.5cm modular cameras with 80×80 instead of 57×57 pixellated NaI(Tl) elements. The system is also equipped with a CT unit with a 12cm × 12cm x-ray detector that provides anatomical CT images correlated with the functional SPECT images. The performance characteristics of the special system using single and multiple pinholes were analyzed using theoretical formulations and Monte Carlo simulations. The results were compared with experimental measurements using point and line sources and physical phantoms. Also, the performance of the system was demonstrated through small animal imaging studies. We found the performance of the system agreed with predictions from theoretical and simulation studies. With an 80×80 pixel element array, ~1mm resolution can be achieved with the special system using a single 1mm pinhole aperture and a magnification factor of 4 when imaging a ~2.5cm diameter mouse. This highest resolution was unattainable with the 57×57 image matrix even with smaller aperture sizes. In multi-pinhole SPECT, the projections from the pinhole apertures at each projection view shared the image array for increased detection efficiency. With a 4-pinhole aperture collimator, we demonstrated that the highest resolution was ~<2mm despite the use of 1mm pinhole apertures. High resolution single and 4-pinhole bone SPECT images of mice confirmed the findings. In conclusion, the larger pixellated arrays of the modular cameras in the special X-SPECT system allow microSPECT imaging of mice with a highest resolution of ~1mm using a single pinhole collimator. The large image acquisition matrix also allows multi-pinhole SPECT imaging with higher detection efficiency and a resolution limit of ~<2mm.
B.M. Tsui, None.
Abstract ID: 133
This paper presents an ultra high resolution molecular imaging system - Cell-vizio - based on its fibered confocal fluorescence microscopy (FCFM) technology, specifically designed to visualize cellular structures in living animals and quantify molecular events.
This unique imaging system is the result of technological breakthroughs in different fields leading to the three main elements of the technology: i) an ultra fast laser scanning unit; ii) a range of fiber bundle micro systems allowing in situ imaging; iii) a dedicated software performing real-time control and image processing. The Cell-vizio provides images with typical characteristics (varying with the fiber bundle micro system) as follows: image lateral resolution: 2.5 micrometers; axial resolution: 20 micrometers; field of view: 400×280 micrometers; optical imaging depth: 80 micrometers (deeper in transparent tissue); data acquisition frame rate: 12 Hz. With fiber bundle micro system as small as 300 micrometers in diameter, Cell-vizio offers unique access capabilities.
In vivo and in situ imaging are two real challenges for the observation of undisturbed physiological processes. We will present the Cell-vizio and its unprecedented applications for small animal imaging such as: neuroscience, cancer, microvascular, or pre-clinical research, and gene expression imaging. FCFM can also be envisioned for many potential clinical applications in conjunction with future appropriate molecular biomarkers. The included image shows in vivo and in situ observation of the mouse colonic crypts with an endoscopic access (field of view: 400 × 280, axial resolution: 15 micrometers; lateral resolution: 5 micrometers). The Cell-vizio is the first ever FCFM integrated product dedicated to small animal imaging.

S. Loiseau, Mauna Kea Technologies 5.
Abstract ID: 134
Micro-CT has the potential to non-invasively study the structures of organs in rodent models with high resolution and fast image acquisition. However, motion artefacts associated with the normal respiratory motion of the animal may arise when imaging the abdomen or chest cavity. To reduce these artefacts and the accompanying loss of spatial resolution, we propose a prospective respiratory gating technique for use with anesthetized, free-breathing rodents.
A custom-made bed with an embedded pressure chamber was connected to a pressure transducer. Anesthetized animals were placed in a prone position on the bed with their abdomens located over the chamber. Inhalation motion caused an increase in the chamber pressure, which was converted into a voltage signal by the transducer. An output voltage was used to trigger an image acquisition at any point in the respiratory cycle.
Fluoroscopy images were acquired of anesthetized, free-breathing rats with a Siemens Multistar digital radiography system to correlate the respiratory waveform with organ motion. The respiratory waveform was monitored, along with the fluoroscopy radiation pulses, and a temporal imaging window was defined. Micro-CT images were acquired with a GE Medical Systems eXploreRS in vivo MicroCT in the regular ungated acquisition mode, and using the respiratory cycle-based trigger for the respiratory gating mode. Gated-images were acquired during expiration when the organ motion was minimized. Gated-images were acquired with an isotropic spatial resolution of 46 mm in 13 minutes (80 kV, 85 mAs, 300 ms per projection).
In this work, we have characterized the respiratory waveform of free-breathing, anesthetized rats and correlated organ motion to the respiratory cycle. We have used this information to define an imaging window, and used our respiratory gating apparatus to prospectively gate a micro-CT image acquisition of the thorax.
N.L. Ford, None.
Abstract ID: 136
To assess whether PET may be used in a particular application, the lowest detectable activity must be known. We examine two situations: 1) object of interest is larger than the scanner resolution (e.g. imaging reporter gene expression in the liver). 2) object is smaller than the resolution (e.g. imaging of metastasis); One limitation of PET scanners based on LSO detectors is that LSO is itself radioactive and generates background events. This background depends on the lower energy threshold, ranging from 690 cps to 70 cps for a 150 and 350 keV threshold respectively. A phantom 10 mm in diameter by 50 mm long was filled with 1 μCi/cc 18F and scanned for 30 minutes on microPET II. Data corresponding to 30 minute scans with concentrations ranging from 0.00005-1 μCi/cc were generated. Studies were repeated using a 1.58 μCi 22Na point source to address detection of small objects. All images were reconstructed with OSEM and contrast to noise ratio (CNR) was calculated (Figure). An object with a CNR exceeding 4 is considered detectable. For energy windows of 350–750 keV and 150–750 keV, the detection limits for a large object are 0.005 μCi/cc and 0.015 μCi/cc respectively. For objects smaller than the resolution, a source with a total activity of 0.5 nCi can be detected with a 150–750 keV window. These results provide an estimate of the lower detection limits using microPET II. Future work will compare reconstruction algorithms and assess detection in the presence of background activity in the animal.

Y. Yang, None.
Abstract ID: 137
This paper quantifies relationship between concentration of iron particles and MRI parameters (T2, T2′, T2*) and evaluates reproducibility of the measurements. We constructed 22 phantoms using mixture of gelatin and various concentrations of Feridex (ferumoxides), ranging from 2.24 μg/ml to 112 μg/ml. In one of the studies, we added Bovine Serum Albumin to generate an environment similar to in vivo. After solutions solidified, we acquired MRI of multiple slices through the phantoms using a 3T whole body and a 7T small bore magnets. The imaging parameters were 1) 3T; T2*: TR = 2s, TE = 10,40ms; T2: TR = 2s, TE=20,60ms 2) 7T; T2*: TR = 5s, TE = 7.5,15,22.5,30ms; T2: TR = 5s, TE = 7.5,15,22.5,30msec. MRI data were transferred to our image analysis workstation and MRI parameters estimated. The results were analyzed by SAS before and after deleting the outliers, which were found in heterogeneous parts of phantoms. Statistical analysis included estimation of correlation coefficients between each MRI parameter and iron particle concentration as well as between different MRI parameters. To evaluate reproducibility, new phantoms with the same specifications were made, imaged, and MRI parameters estimated and analyzed. Experimental results show linear correlations between concentration of iron particles and MRI parameters. The correlation coefficients were between 84% and 98%, confirming linear correlation between our variables. R-squares or coefficient of determinants were between 67% and 98%, showing a strong ability to predict Feridex concentrations using MRI parameters. The p values were less than 0.05 (0.02-0.04) for T2 and T2*. The largest p values occurred for T2′(0.04-0.12) when the outliers were not deleted. Reproducibility of the measurements improved as the concentration of Feridex increased. This may be attributed to lower MRI contrast-to-noise-ratios and inaccuracies in making exactly the same solutions when iron particle concentrations are low. Using MRI and image analysis methods, concentration of iron particles can be accurately and reproducibly measured.
H. Soltanian-Zadeh, None.
Abstract ID: 138
Visual-Servoing Optical Microscopy (VSOM) instrumentation and protocols are being developed that will assist in the development of fluorescently-labeled peptide nucleic acids (PNAs) suitable for imaging specific mRNA transcripts in living cells. Streptolysin-O (SLO) is being used to gently and reversibly permeabilize cells so that the intracellular accumulation and wash-out of Texas Red (TR) labeled PNAs can be imaged. Cells that expressed high levels of uterocalin (UTC) mRNA and low levels of glial fibrillary acidic protein (GFAP) mRNA were imaged using an inverted fluorescence microscope. A TR-labeled PNA targeted to UTC mRNA was synthesized together with a control TR-PNA targeted to GFAP mRNA. Cells were imaged in three channels (transmitted light, green fluorescence, and red fluorescence) as computer-controlled pumps perfused Ca+2-free medium containing SLO (500ng/mL) into a cell chamber (15min, 37°C). This was followed by a 20min perfusion of Ca+2 -containing medium which also contained YOPRO-1 (1μM), and a TR-PNA (20nM). A 1hr wash-out with Ca+2-containing medium completed these experiments. Cells were grown on the surface of the chamber and remained attached during perfusions. Two phases of SLO-generated pore formation were observed in the green YOPRO-1 channel. In the first phase, formation of pores large enough for YOPRO-1 to enter the cell was observed, but these pores were not large enough for TR-PNAs (15mers) to enter. In a second phase, (indicated by a 10-fold increase in the cytoplasmic intensity of YOPRO-1) accumulation and wash-out of TR-PNAs was observed in the red channel. However, the use of a fixed pump schedule (and fixed SLO exposure time) has not been reliable. In future VSOM experiments, perfusion of TR-PNAs will not be triggered until analysis of YOPRO-1 accumulation indicates that the second phase of pore opening has occurred. This will facilitate comparisons of the intracellular accumulation and retention of targeted and control TR-PNAs. D.E. Callahan, VSOM, Inc. 1.
Abstract ID: 139
4 pixels in increments of 1. The α factors were calculated for the resulting seven ROIs and plotted versus their shrinkage/expansion steps.
Abstract ID: 140
Water phantom containing SPIO (Resovist, Schering AG) Top: without correction, Bottom: with correction

T2*map of brain slice. Left: without correction. Right: with correction

T. Schaeffter, Philips Research 5.
Abstract ID: 141
A critical component for characterization and validation of new synthetic probes for direct in vivo imaging of gene expression is the kinetics of uptake and washout profiles. A precursor to the in vivo translational model entails in vitro screening of synthetic compounds in living cells where toxicity, in- and out-fluxes, and signal-to-noise ratio are quantified. The current model system are cells grown in a monolayer to characterize new assays, and to initiate development of new algorithms and bioinformatics tools for quantitative analysis. Ultimately, the model cell system will be grown in a colony, which will be closer to the in vivo models. The major computational and informatics challenges are (1) delineation of individual living cells imaged through bright field microscopy, and (2) the imaging bioinformatics framework for maintaining time-lapsed images (approximately 60Mbytes per 30 minutes per experiment), information about the images, and the protocol used in each experiment.
Automated delineation of cells imaged through transmission light microscopy is complex as a result of lack of contrast, presence of perceptual boundaries, and non-uniformities in the intensity distribution. A method, based on iterative voting and perceptual grouping, has been developed to recover individual cells in a confluent state that are imaged with a phase contrast objective. The concept of geometric voting can be applied on the gradient or curvature image. Voting can be applied either in the gradient direction to localizes each cell corresponding to its center of mass, or in the normal direction to reveal cellular boundaries. Once each cell is delineated, its individual uptake or washout is quantified as the fluorescently labeled synthetic compound is perfused into or washed out from the micro-incubator. The informatics system captures time-lapsed images and their quantitative representation, instrumentation parameters, and parameters associated with the living cell assay. Preliminary results will be presented.
B. Parvin, None.
Abstract ID: 142
In vivo 13C magnetic resonance (MR) wedded to stable isotope administration techniques, generally referred to as isotopomer analysis, has the potential to serve as a powerful molecular imaging tool. In order to take advantage of this methodology, robust data processing methods must be established that optimally extract the information present in the magnetic resonance data. We have paired Bayesian Probability Theory with Markov Chain Monte Carlo (MCMC) simulations to not only make estimates of metabolic fluxes but also quantify the uncertainty associated with each measurement.
As a model system, we have chosen to study acetate perfused rat hearts that utilize a variety of different 13C labeling schemes. The 13C MR spectrum of glutamate extracts manifests strong spin-spin coupling that is indicative of the rates of TCA cycle turnover in the heart. Since we monitor glutamate in the hearts, there is an abundance of prior information regarding chemical shifts, etc. This prior information can be used as constraints for the Bayesian MCMC simulations and ultimately removed from the estimates of the metabolic flux measurements.
As the signal-to-noise decreases, the simulation still manages to make good estimates of the influx of 13C label into position two of acetate, Fc2, and the anaplerotic contribution to the TCA cycle, y.
In summary, the Bayesian/MCMC method for reducing 13C MR data to metabolic information has the potential to allow molecular imaging of metabolism in the low signal to noise environment that will always be present in in vivo 13C MR.

Bayesian MCMC simulations accurately reconstruct the carbon spectra even as the signal-to noise decreases in the spectrum.
M.E. Merritt, None.
Advances in PET and SPECT Radiochemistry
Abstract ID: 143

C.A. Foss, None.
Abstract ID: 144
Overexpression of epidermal growth factor receptor (EGFR) is common in many human solid tumors, such as colorectal, breast, lung carcinomas, and cancers of the head and neck. It correlates with increased metastasis, decreased survival and a poor prognosis. Therefore, EGFR represents an attractive target for the design of novel anticancer diagnostic and therapeutic agents. Cetuximab (C225, Im-Clone Systems Inc.) is a FDA approved drug to treat patients with EGFR-expressing, metastatic colorectal cancer. C225 specifically binds to the extracellular domain of EGFR on tumor cells, thereby inhibiting receptor-associated tyrosine kinase. The goal of this study is to image the over-expression of EGFR in tumors by Positron Emission Tomography (PET) with radiolabeled C225. C225 was radiolabeled either with 64Cu by conjugating a bifunctional chelator DOTA (64Cu-DOTA-C225) or 124I (124I-C225). A specificity binding assay demonstrated that both 64Cu-DOTA-C225 and 124I-C225 showed great binding affinity to EGFR-positive A 431 cells (IC50's of 2.70 nM, 2.22 nM, respectively). MicroPET images of 64Cu-DOTA-C225 in A431 tumor xenografts delineated that significant tumor uptake was observed, with a standard uptake value (SUV) of 2.10 ± 0.59 after injection 20 h, and this tumor localization remained constant out to 46 h post-injection. Liver uptake with a 2.06 ± 0.28 of SUV at 20 h post injection was also determined. A reduction of tumor uptake (1.02 ± 0.10 of SUV) was observed in the A 431 mice injected 1 mg of C225 20 h before administration of 64Cu-DOTA-C225. However, tumors in the EGFR-negative MDA-MB-435 xenografts were not visualized with 64Cu-DOTA-C225. These data suggest that the 64Cu was residualized into the cells and the potential of radiolabeled C225 for imaging EGFR overexpression in tumors. MicroPET imaging studies of 124I-C225 will be performed in A431 and MDA-MB-435 tumor xenografts. It is hoped that similar tumor uptake will be observed with 124I-C225.
W. Li, None.
Abstract ID: 145
‘Peripheral benzodiazepine receptors’ (PBR) are located on the mitochondrial outer membrane particularly in the central nervous system. Their density increases in injured brain. Therefore, imaging of brain PBR is relevant to the investigation of several neurodegenerative disorders. We aimed to develop a sensitive PET radioligand for imaging brain PBR, based on high affinity N-acetyl-N-(2-carboxybenzyl)-2-phenoxyaniline
Treatment of the precursor
Within 15 min of administering [11C]
[11C]
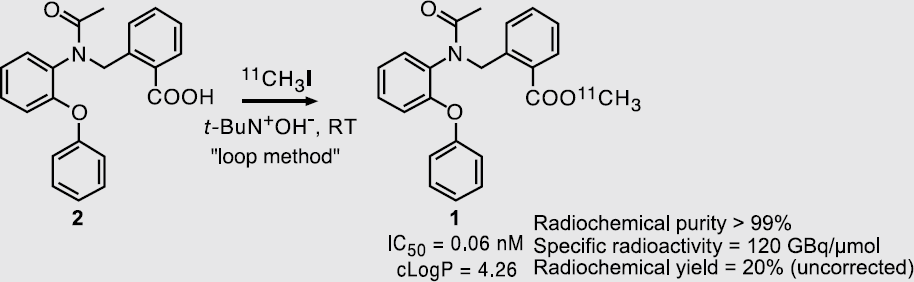
Abstract ID: 146
[18F]Fluoromisonidazole ([18F]FMISO) uptake in ta C3H mouse squamous cell carcinoma treated with mild hyperthermia.
S. Lee, None.
Abstract ID: 147
Several pyridopyrimidinone derivatives are potent inhibitors of variety of tyrosine kinases, which are overexpressed in many malignant cancers such as AML or ALL. Labeling these novel molecules with radionuclides such as 124I or 18F or other imageable nuclides can aid in the detection of the overexpression of the tyrosine kinase activity. In our effort to develop novel imaging agents we report the synthesis of radiolabeled derviatives of pyridopyrimidinones. A high yielding method for the synthesis of [124,125,131I]-iodopyridopyrimidinone (
M. Namavari, None.
Abstract ID: 148
A series of monomeric and dimeric RGD peptide antagonists of integrin αvβ3 have been developed for radionuclide and optical imaging of tumor angiogenesis and metastasis, but little has been studied with the suitability of radiolabeled peptidomimetic integrin αvβ3 antagonists for integrin targeting in vivo. We recently developed a quinolone-based vitronectin receptor antagonist TA138 and labeled the DOTA conjugate with 111In and 90Y for tumor imaging and therapy. In this study we would like to extend this effort and label TA138 with 64Cu (t1/2 = 12.8 h) for brain tumor targeting. TA138 revealed high integrin αvβ3 binding affinity (IC50 = 0.54 nM) for U87MG cells. 64Cu-TA138 indicated significantly slower blood clearance than RGD peptides. Circulation blood levels of 64Cu-TA138 were 1.38 and 0.95% ID/g at 0.5 and 4 hrs, respectively. The excretion was slow through both hepatic and renal routes. Direct tissue sampling found that tumor-to-background ratio remained constant at the time points examined (tumor/muscle ~11, tumor/blood ~6, tumor/liver 0.5-0.6, and tumor/kidney 1.3-1.5). Blocking experiment confirmed the receptor specificity of TA138 complex in vivo. At 1 h p.i., the tumor uptake decreased from 6.95 % ID/g in the control mice to 1.94 %ID/g in the blocking mice. Significant decrease of uptake in non-target organs was also observed. 2D projection microPET images (10 min static single frame beginning at 1 h after intravenous injection of 500 μCi of 64Cu-TA138 with (right) or without (left) 10 mg/kg of c(RGDyK), OSEM reconstruction) of mice bearing subcutaneous U87MG glioma tumor were (Fig 1). Further modification of peptidomimetics for improved in vivo kinetics is currently in progress (Funding support: NIBIB EB001785, DOD BCRP DAMD17-03-1-072 and BC030012, 64Cu via R24 CA86307).

X. Chen, None.
Abstract ID: 149
Functional and molecular in-vivo imaging of apoptosis is rapidly evolving as a non-invasive tool for evaluation of pathological alterations associated with numerous medical disorders. Positron emission tomography (PET), is regarded as a modality of choice for future molecular functional imaging, defining the need for small-molecule probes, amenable to rapid radiolabel attachment, and manifesting satisfactory selective accumulation in the target tissues, while having rapid clearance from non-target organs. To date, such PET-imaging agent for apoptosis is not available.
Intravenous administration of
Molecular Imaging of apoptosis-related processes in cancer patients, by the novel
A. Shirvan, NST 5.
Abstract ID: 150
Because of their low abundance, imaging mRNA in tissues with radiolabeled antisense oligonucleotide probes requires using probes that have a high specific activity. In this project we developed a novel and effective approach for the rapid radiolabeling of peptide nucleic acids and oligonucleotides to high specific activity with radioactive iodine. The approach was additionally designed to be compatible for radiolabeling with fluorine-18 for in vivo PET studies to image gene expression. For the radiolabeling procedure, we first synthesized a radiosynthon that consisted of a 3-[I-125]iodobenzoyl moiety conjugated to biotinyl-L-lysine. The radiosynthon also contained either an activated ester moiety or a bromoacetyl group, for coupling to a peptide nucleic acid probe or an oligonucleotide probe terminated with a 5′thiophosphate, respectively. Following the coupling, the biotin moiety in the radiosynthon enabled the radiolabeled probe to be efficiently separated out from the large excess of unradiolabeled probe in the reaction mixture (which is not biotinylated) by selective absorption of the radiolabeled probe onto agarose beads coated with immobilized streptavidin. A variation of this procedure in which the agarose beads coated with an antibody against the 3-iodobenozyl moiety of the radiosynthon was also examined. Following washing, the radiolabeled probe was cleaved off the agarose beads using a small amount glacial acetic acid. Using this selective absorption strategy we were able to obtain radiolabeled peptide nucleic acid probes and oligonucleotide probes that had a very high level of specific activity, without the need for a final HPLC purification step. The high specific activity will make these probes suitable for imaging the distribution of even low-abundance mRNA in tissues in vitro and/or in vivo. Supported by DOE OBER
A.N. Gifford, None.
Abstract ID: 151
J.V. Frangioni, None.
Abstract ID: 152
A spectrum of thymidine analogs have been radiolabeled with SPECT (123I or 131I) or PET (11C, 18F or 124I) isotopes and are currently being evaluated as potential radiopharmaceutical probes for imaging Herpes Simplex Virus 1 thymidine kinase (HSV1-tk) gene expression in vivo. Wiebe and co-workers (J Med Chem. 40, 2184-2190,

N. Pillarsetty, None.
Abstract ID: 153
H.R. Maecke, None.
Abstract ID: 154
A. Khanamiryan, None.
Abstract ID: 155
The emerging pivotal role of neuroprotective drugs in future therapy for cerebrovascular injury emphasizes the need for functional imaging technologies, capable of defining the extent of neuronal death induced by cerebral insults and monitoring efficacy of potential neuroprotective therapeutic strategies.
A. Shirvan, NST Ltd. 5.
Abstract ID: 156
Imaging Claudin 4 and PSCA in Pancreatic Cancer Xenografts
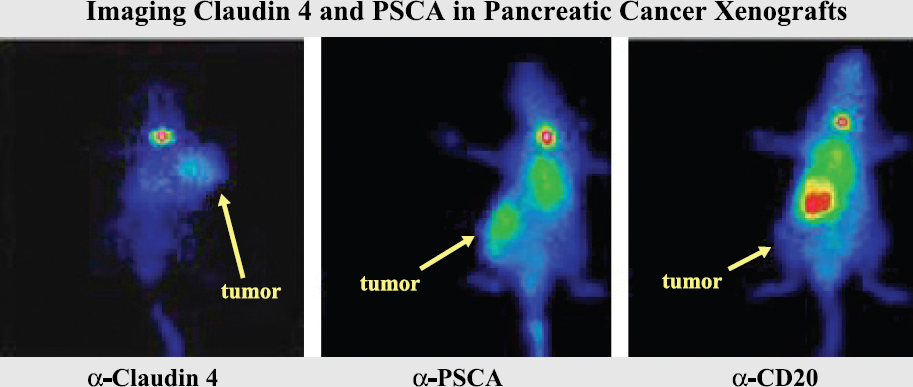
C.A. Foss, None.
Abstract ID: 157
DOTA-Tyr3-octroetide has been synthesized using the 9-fluorenyl-methoxycarbonyl method of solid phase peptide synthesis. The Fmoc-Thr(ol)(tBu)-2-chlorotrityl resin was coupled with various selected amino acids to form a straight chain of peptide-resin DOTA(tBu)3-D-Phe-Cys(Trt)-Tyr(tBu) -D-Trp(Boc)-Lys(Boc)-Thr(tBu)-Cys(Trt)-Thr(ol)-(tBu)-2-chlorotrityl resin. Cleavage of the peptide from the resin was achieved by trifluoroacetic acid (TFA). The cleavaged peptide reacted with charcol to give disulfide-containing peptide DOTA-D-Phe-Cys-Tyr-D-Trp-Lys-Thr-Cys-Thr(ol) (DOTA-Tyr3-octroetide). The final product was obtained by the reverse-phase HPLC purification. The mass analysis of DOTA-Tyr3-octroetide is 1421.3Da by Fab-MS. The 67Ga labeling of DOTA-Tyr3-octreotide was prepared by addition of DOTA-Tyr3-octreotide and 67Ga-acetylacetone. The radiolabeling procedure gave labeling yield ranged from 85% to 95%. In-vitro competitive binding curves indicates that DOTA-Tyr3-octreotide showed an effective displacement of 125I labeled Tyr1-somatostatin (IC50 = 4.39±0.83 nM). MicroSPECT imaging of 67Ga-DOTA-Tyr3-octreotide in AR42J tumor bearing Lewis rats revealed a significant uptake in the thigh tumor in the rat model. These results demonstrate that 67Ga-DOTA-Tyr3-octreotide is a suitable radiopharmaceutical for imaging of somatostatin receptor (SSTR) positive tumors.
W. Hsu, None.
Abstract ID: 158
It is well known that the bonding interactions of technetium (and rhenium) with phosphines result in in vivo stable radiometal-phosphine compounds. Therefore, the design of phosphines functionalized with tumor-avid peptides could be pivotal for the development of radiometal-essential tumor specific diagnostic/therapeutic agents. Inspired by the unmatched molecular imaging potential of the d6 organometallic [99mTc(CO)3(H2O)3]+ (and [Re(CO)3(H2O)3]+) cores, considerable effort has been focused toward the design and development of facially spanning tridentate ligand frameworks for achieving in vivo stability for such organometallic cores. However, the precise topology of ligand frameworks based on chain length, chemical architecture, and combination of electron donors, required to impart kinetic/thermodynamic stability to specific radio metals (e.g., Re(I)/Tc(I)) has eluded scientists to date. As part of our ongoing investigations focused on the design and development of novel chemical vectors that display optimum chemical, pharmacokinetic and in vivo specific properties, we have hypothesized that compact tripodal chemical frameworks with combinations of p-acid phosphine (P) and soft donor thioethers (S) would provide ideal bonding propensities toward the production of in vivo stable Tc/Re compounds. This presentation reports our successful organic chemistry for the synthesis of phosphino dithioether (PS2) tripodal ligand backbone, its bioconjugation with a tumor-avid peptide and the “proof of principle” that such PS2-peptide conjugates will produce in vivo stable cancer seeking 99mTc-based imaging agents.
R. Kannan, None.
Tracking Infectious Agents & Imaging Host Response
Abstract ID: 159
Bioluminescence provides a sensitive and simple detection method for the study of biological process in living animal models of human diseases. The most commonly used reporters for such investigations are firefly (luc) and bacterial (lux) luciferases. Since oxygen is considered as one of the co-factors that are required for either type of luciferase reaction to occur, we investigated the effect of low oxygen concentrations on bioluminescence reaction. Bacterial strains that were engineered with either lux or luc were monitored for bioluminescence under microaerophilic to anaerobic growth conditions, using Xenogen's IVIS® 100 Series Imaging System. Under anaerobic conditions that permitted strict anaerobe Bacteroides fragilis to grow, bioluminescent signals (photons/sec/cell) from a strain of Staphylococcus aureus harboring a lux operon or firefly luc were reduced by 1 log and 2–3 logs respectively, compared to the same strains grown under aerobic conditions for 24 hours. The bioluminescence of bacterial luciferase from both S. aureus and Pseudomonas aeruginosa growing anaerobically remained stable up to 30 days. In contrast, the bioluminescence from firefly luciferase diminished after only 2 days. Upon exposure to air, bioluminescent signals instantly increased 10–100 folds for bacterial lux and 1000 folds for firefly luc, indicating that the biolumi–nescent reaction of the latter enzyme was more dependent on environmental oxygen. Under a microaerophilic environment (2-3% oxygen), neither luciferase appeared to be adversely affected in its bioluminescence capacity. In summary, the bioluminescence generated from bacterial lux operon could be monitored over extended periods of time under strict anaerobic conditions. It suggests that the bacterial bioluminescence system in particular might play an important role in monitoring the metabolic state of pathogens infecting biological tissues at low oxygen environment in vivo.
J. Yu, Xenogen Corporation 5.
Abstract ID: 160
The implantation into animals of sponge matrices carrying mammalian cells represents a means to study different aspects of immunity and tolerance. Immune responses against implants are generally evaluated using histological examination and other techniques that require removal of the sponge matrix. Using bioluminescent cells to load sponges would allow an investigator to follow the kinetics of destruction of implanted cells and to study the effects of a given immunosuppressant in the context of a specific disease. Here our goal was to determine to what extent the bioluminescence activity of mammalian cells expressing a Renilla luciferase cDNA and grown in vitro in a matrix would be affected by different matrices and multiple injections of luciferase substrate, and to observe immune rejection in mice bases on loss of light emission. A human stable cell line expressing a Renilla luciferase cDNA was used to seed polyurethane, collagen, and gelatin sponge matrices coated with PBS, polylysine, fibronectin, collagen, or gelatin. Sponges were then incubated in tissue culture plates for 3 weeks and recieved 2, 9, or 15 injections of the luciferin did not affect light emission significantly. Histological analysis of sponge matrices showed that cells formed islet-like structures in the sponge and that the highest number of these structures were found in the gelatin matrix. As anticipated, visualization of implanted bioluminescent sponges in Balb/C and Balb/C-scid mice showed accelerated loss of light emission in Balb/C mice. These results indicate that gelatin sponges containing mammalian cells synthesizing Renilla luciferase are valid candidates to be used as implants for studying immune rejection in an animal model system.
O.N. Ojogho, None.
Abstract ID: 163
Aptamers are short single stranded nucleic acids that, like antibodies have high affinities and specificities for their target molecules. Many aptamer targets are small molecules that could serve as a contrast agents. An aptamer that recognizes such a contrast agent and retains it in or over the cell with which it is associated could be used to image the associated cell. As part of an RNA molecule synthesized from a defined promoter, an aptamer can be used as a reporter to measure the relative activity in the cell of the promoter from which it is expressed. The aptamer could also be expressed from a constitutive promoter that would allow it to be used to track the cells that express it. We have obtained preliminary evidence that aptamers can be used for imaging cells. Multiple copies of the malachite green aptamer were incorporated into the sequence of a longer RNA sequence that was expressed from the U6 snRNA promoter in chinese hamster ovary cells. The control plasmid was identical with the same RNA sequence except it contained a centrally located QPZ aptamer in place of the malachite green aptamers. Binding of malachite green by the aptamer results in increased fluorescence from malachite green. When exposed to malachite green, cells that expressed the malachite green aptamer showed more fluorescent emission than the control cells that expressed the QPZ aptamer. Cells are imaged in bright field and delineated from background using geometric grouping of spatial features based on continuity proximity. The delineated cells, in the bright field, are used as masks to compute the mean responses in the corresponding fluorescence channel for quantification. The results suggest that aptamers have the potential for use as a means of tracking stem cells or measuring promoter in vivo.
Supported by DOE OBER.
M. Nilsen-Hamilton, None.
Abstract ID: 164
Recently, cancer vaccine using plasmid DNAs encoding tumor-associated antigens has been emerged as an immunotherapy method for cancer patient. In this study, we visualized the migration of DNA vaccine in immunizing mice with a plasmid DNA encoding Firefly Luciferase gene.
pcDNA (control group, 300g/50μl) as well as pcDNA-luciferase (experimental group, 300μg/50μl) were injected intradermally on the hind limbs of Balb/c mice. The bioluminescence images of mice were serially obtained using IVIS system (Xenogen Technology) at 2, 6, 10, 14, 24 hr, 2, 10 days after DNA injection. After then, we sacrificed and skinned the mice. Following the skinning of the mice, we again obtained the bioluminescence images of the lymph nodes in the skin.
At 2 hr image, there was no significant image signal from both injection regions and proximal lymph nodes. However, significant bioluminescence intensities were obtained from either injection regions or proximal lymph nodes between 10 hr and 10 days. Particularly, we could see the significant intensity in both injection sites and lymphatic lymphoid organ at the 24 hr. Also, we could observe the discrete intensities from injection site and proximal lymph nodes in the skin.
Optical image using firefly luciferase gene could be useful in monitoring the expression of plasmid DNA cancer vaccine. We suggest that this monitoring system is also appropriate in deciding a dose, frequency of administration and adjuvant supplement before the administration of actual DNA cancer vaccine.
Y. Jeon, None.
Abstract ID: 165
Cell tracking studies have been performed with various modalities, mainly MRI, bioluminescence and PET imaging, however only few have utilized SPECT/CT. The latter offers the advantage of using readily available and clinically approved isotopes for cell labeling (e.g. Indium-111-Oxine), and exact anatomical co-localization of the radioactivity in 3 dimensions.
Here we show that experimental SPECT/CT (X-SPECT, GammaMedica) can be used to image in vivo the trafficking of various cell types throughout the body with high sensitivity. Phantom studies revealed a resolution of the SPECT system at or lower than 1 mm (depending on the pinhole-diameter). Cell phantom studies showed the ability to image 100,000 cells / 64 nCi total, with the ability to even distinguish 10,000 cells from the background. We were able to discern the distribution of systemically injected cells (> 5 Mio.) containing low amounts of radioactivity (as low as 30 microCi total) in vivo. Serial in vivo imaging showed the distribution of cells over time and their accumulation in various organs in a time dependent fashion. Leukocyte trafficking in mice showed initial accumulation in the lungs, followed by redistribution to liver and spleen as well as lymph nodes. After clearance from the blood within 3 to 5 days accumulation of cells even in the aortic wall at sides of plaques could be shown.
Imaging of cell trafficking by SPECT/CT is sensitive enough to enable a noninvasive, quantitative and dynamic in vivo assessment of the biodistribution of labeled cells and allows investigation of fundamental biological questions in oncology, stem cell and vascular research. This approach could be translated to clinical applications using existing human scanners and approved labeling protocols. J. Grimm, None.
Abstract ID: 166
P.A. Erba, Theseus Imaging Division, North American Scientific 1.
Imaging Markers of Cardiovascular Disease
Abstract ID: 167
Direct visualization of trafficking iron oxide in atherosclerosis using a near infrared fluorescent iron oxide particle (CLIO-Cy 5.5). Example light (A) and NIRF (B) image of an apo E mouse injected with CLIO-Cy 5.5. Note focal NIRF signal in the aortic root (AR), aortic arch (AA), and great vessels (GV). Histological light (C) and NIRF (D, high power E) images confirming focal NIR signal within atherosclerotic lesions (PLQ). Note that the normal vascular wall is without significant NIR signal.

F.A. Jaffer, None.
Abstract ID: 168
Imaging of endothelial-specific markers holds promise for noninvasive assessment of inflammation, atherosclerosis, angiogenesis and early assessment of anti-angiogenesic therapy in cancer. Several alternative models enabling testing of targeted human vascular endothelium cell (HVEC)- specific imaging probes are currently under development. The aims of the current study were: 1) to develop a mouse model of HVEC adoptive transfer; 2) to test in vivo a MR-detectable reporter with high affinity to human E-selectin, a proinflammatory marker involved in inflammation and HVEC proliferation. To induce differentiation of HVEC into vessel-like endotubes in vivo, cell suspension in Matrigel was injected s.q. in athymic mice. Two-to-four weeks after the implantation animals were studied using MRI (7T whole body system, Siemens, using T1w, T2w SE and T2* GE sequences). Imaging probes consisted of cross-linked iron oxide (CLIO) nanoparticles conjugated either to anti-human E-selectin F(ab′)2 (H18/7 [1–2]) or to control, non-specific antibody fragments. To induce E-selectin expression, mice were treated with IL-1b i.p.
First, we demonstrated that HVEC treated with IL-1b expressed E-selectin and had a 100–200 times higher binding of CLIO particles than control cells resulting in T2 decrease to 30–40 ms compared to 1500 ms in control. Second, HVEC-containing Matrigel implants in mice injected with specific conjugates appeared darker than controls both after visual inspection and on MRI SE and GE images (Fig.1 B). MR images of mice implanted with Matrigel containing HVEC showed highly specific T2* signal decrease after the IL-1b treatment (Fig.1 B). There was no signal change in control implants (Fig. 1. A,C,D). Therefore, our experiments provide preliminary evidence of HVEC imaging feasibility in vivo and justify further refinement of HVEC-targeted agents.
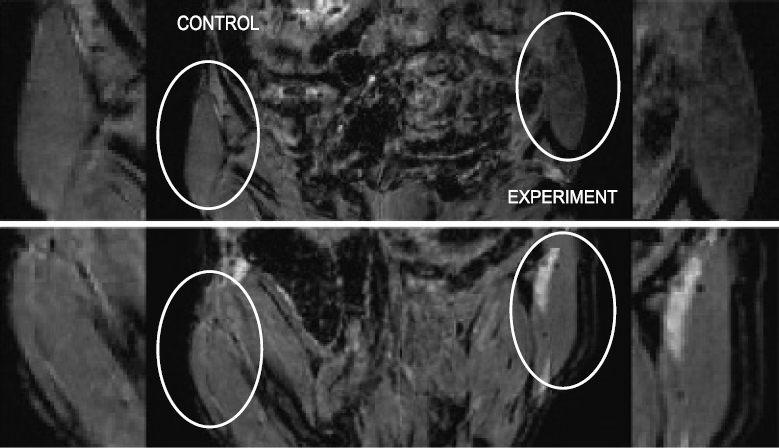
T2 -weighted GEMR images obtained in mice implanted with control (A,C) or HUVEC-containing (C,D) Matrigel plugs after anti-E-selection (top images) or control antibody-CLIO conjugates (bottom images) were injected.
A.A. Bogdanov, None.
Abstract ID: 169
Imaging of vascular gene expression is important for successful implementation of gene therapy for cardiovascular disease. Optical imaging has the advantages of being sensitive, cost-effective and rapid because of which it is ideally suited to molecular imaging. The feasibility of using digital optical imaging for the detection of green fluorescent proteins acting as markers for host cells in vitro has been proved. Since then, surface optical imaging has been successfully used to monitor fluorescent gene expression from gene-transfected vessels in various animal models. Optical imaging is limited in its ability to send/receive light through the different layers of tissue primarily because of the absorption and scattering. In order to circumvent this problem, we decided to develop a system based on a minimally invasive percutaneous approach. The system comprises a halogen light source, a CCD camera, and a dual port semi-rigid optical imaging probe (diameter - 1.5 mm and length - 5 mm). The system design was optimized using Monte Carlo simulations. The system's performance has been tested over a series of experiments involving detection of fluorescence from green fluorescent protein in human tissue-like phantoms, porcine meat slabs and finally under in vivo settings on live pigs. In the in vivo experiments, the blood vessel was localized using color Doppler imaging and the percutaneous imaging probe was positioned close to the vessel wall by ultrasound imaging for optical imaging. In all of the experiments, the system has shown significantly higher signal intensity from GFP-tagged cells compared to saline, which was used as control. In the in vivo studies, conducted on GFP-transfected femoral arteries of live pigs, the optical signal intensity from the gene-transfected arterial segments was significantly higher (p<0.05) than the signal from saline treated control segments: (GFP+ / saline=: 215.7±8.9 a.u./157.4±44.8 a.u.). This was further confirmed by performing surgery-based optical imaging.
X. Yang, None.
Abstract ID: 170

C.H. Cunningham, None.
Abstract ID: 171
Endothelial vascular adhesion molecule (VCAM-1), a critical component of the leukocyte-endothelial adhesion cascade, is upregulated on the endothelium under inflammatory conditions including atherosclerosis and cardiac allograft rejection. The strict temporal and spatial regulation of VCAM-1 make it an ideal target for imaging and therapy. Unfortunately, the development of targeted cardiovascular imaging agents is frequently hampered by poor target-to-background ratios resulting from non-selective distribution, low target accumulation, and delivery hurdles. The goal of this study was to develop a VCAM-1 targeted peptide capable of cell mediated internalization that could be harnessed as an amplification strategy to chaperone imaging agents across the endothelial plasma membrane. Thus, we developed a modified phage display approach to screen for peptide sequences that would be internalized by VCAM-1 expressing endothelial cells under physiological flow conditions. A peptide sequence with homology to very late antigen (VLA)4, was shown to bind to VCAM-1, to be rapidly internalized, and to block leukocyte-endothelial interactions. Using the peptide, we generated a multivalent nanoparticle with improved pharmacokinetics that is detectable by both fluorescence and magnetic resonance. In vivo, the agent successfully identified VCAM-1 expressing endothelial cells in a murine TNF-α-induced inflammatory model. Furthermore, atherosclerotic lesions in cholesterol-fed apoE-/- mice, which are known to have upregulated VCAM-1 levels, were readily detected by magnetic resonance imaging following administration of the agent.
The data presented here demonstrate that phage display derived VCAM-1 mediated cell internalizing peptides are capable of shuttling nanoparticles across the endothelial plasma membrane and provide a means of trapping such payloads at the intended target site. The described approach is useful for direct imaging of VCAM-1 expression in vivo and provides a novel system for the development of targeted therapies.
K. Kelly, None.
Abstract ID: 172
Ferumoxtran-10 signal in the anterior infarct region as shown by a signal void (white arrows).
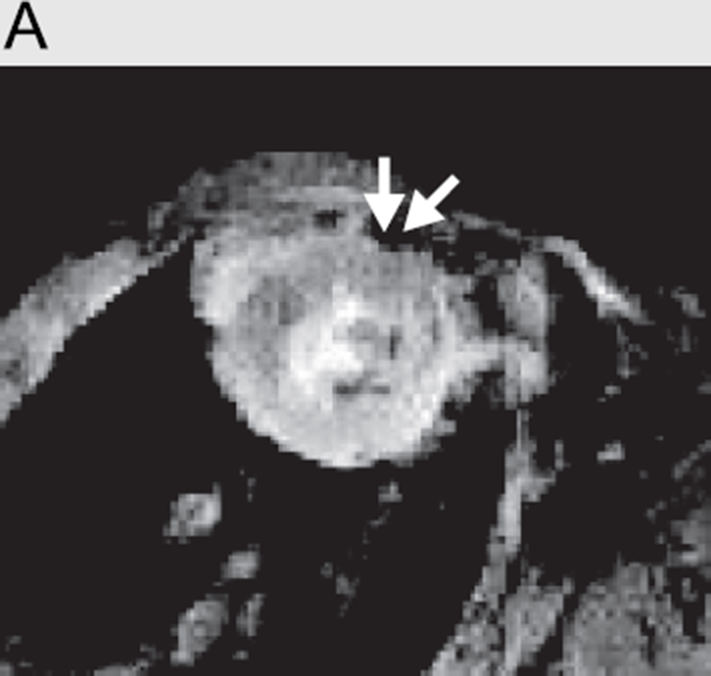
High-power image (15x) of the infarct region demonstrates macrophage-specific stain.

Ferumoxtran-10 in the anterior infarct territory (blue box) shown by Prussian Blue stain (black arrow).

High-power image (20x) of ferumoxtran-10 in the infract region (black arrow).

T. Arai, None.
Abstract ID: 173
Alteration in myocardial energy metabolism is believed to be one mechanism involved in the progression of heart failure. Imaging studies have shown that myocardial glucose uptake rates are lower in heart failure and myocardial free fatty acid uptake rates are higher than in the normal heart. Imaging studies have also shown that myocardial fatty acid oxidation begins to be inhibited and that washout of fatty acid increases in the compensated stage of left ventricular dysfunction. In the advanced stage of heart failure the myocardial extraction and retention of fatty acid are further impaired. Therefore, shifts in myocardial glucose and fatty acid use may be an important mechanism for the impaired efficiency of the failing heart and a target for specific therapies designed to decrease overall energy requirements. This study was designed to follow the change in glucose metabolism with the progression of hypertrophy in the spontaneously hypertensive rat (SHR). Dynamic PET data were acquired using the microPET II. A dose of 1 mCi of F-18-FDG was injected into a control Wistar Kyoto (WKY) rat and the same dose was injected into a SHR rat. Each rat was imaged using a dynamic acquisition for 80 minutes acquiring list mode data with cardiac gating of approximately 900 million total counts. Both rats were imaged at 10 week intervals for a total of five imaging sessions. The input function was calculated both using factor analysis of dynamic structures (FADS) and fitting splines from the list mode data. The dynamic data for myocardial tissue regions were fit to a 2-compartment model. Using a single blood sample, the metabolic rate of glucose was evaluated. The study is being used to test the hypothesis that the SHR rat models congestive heart failure and that this model demonstrates changes in metabolism with progression of cardiac hypertrophy.
G.T. Gullberg, None.
Abstract ID: 174
Determining regional and temporal gene expression in vivo has become an important focus in biological research. Here we present a novel generic method to detect gene expression in vivo using MRI based magnetic-antigen-gene imaging contrast (MAGIC). The method involves a combination of MRI and the use of a surface antigen of choice to which a specific antibody recognises, the antibody being conjugated to a dextran-coated iron oxide core. The antigen of choice was a truncated form of the H2kk antigen, the antigen is not expressed in commonly used strains of experimental mice, other rodents or man. HeLa cells, a human cervical epithelial cell line, does not normally express the H2kk antigen, was transiently transfected to express the H2kk antigen. HeLa cells transiently and mock transfected were harvested 44h after transfection and MRI performed. The transverse (T2) relaxation times of the cells were determined to be 57.6± 17.0ms and 424.0±38.7ms for the H2kk transfected and mock transfected cells, respectively. The lower T2 relaxation time of the former cells arises from the binding of the iron-conjugated antibody to the H2kk antigen. Surface expression of the H2kk antigen in the transiently transfected cells was confirmed by fluorescence cytometry, fluorescence microscopy and electron microscopy. The MAGIC methodology presented here is fast, reliable and has the unique advantage of being readily accessible to all researchers whilst having the flexibility to be tailored to individual researchers needs.
P. So, None.
Abstract ID: 175
Electrocardiographic (ECG) gating of nuclear cardiac images has become standard practice in evaluation of patients with suspected ischemic heart disease, and can provide information about regional and global function in addition to myocardial perfusion. Evaluation of these regional and global indices or perfusion and cardiac function are also important in preclinical rodent studies. We have implemented ECG-gated cardiac perfusion imaging to a hybrid microSPECT/CT system, which provides sub-millimeter resolution in SPECT and co-registered high resolution CT anatomical maps. Implementation of cardiac gating in rodents is challenging due to the extremely fast heart rate compared to that of humans. Radionuclide detection is synchronized with the R-wave of the cardiac cycle and separated into 16 time bins using an ECG monitor and triggering device for gating. Images were acquired with a 5 inch by 5 inch small field of view pixilated NaI(Tl) detector, using a 2 mm pinhole collimator with 9 cm focal length. ECG-gated images were acquired in anesthetized rats following intravenous injection of 99mTc-Sestamibi. Scans were obtained in both normal (control) rats, and rats after surgical induction of myocardial infarct (MI) by ligation of anterior coronary artery. 4-D images (3D plus timing) were reconstructed using cone-beam reconstruction software, and quantitative abnormalities in regional perfusion were related to changes in regional wall motion and global ejection fraction. Anteroapical hypokinesis was observed in corresponding regions of hypoperfusion in those rats with MI. These findings matched results obtained using echocardiography. The simultaneous assessment of both regional perfusion and function with ECG-gated microSPECT/CT offers a new noninvasive approach for phenotyping rodents or assessing changes in cardiac physiology in rodents before and after therapeutic interventions.
T.J. Vandehei, Gamma Medica, Inc. 5.
Abstract ID: 176
We previously used bioluminescence imaging (BLI) to investigate interferon signaling on pathogenesis of Herpes simplex virus type-1 (HSV1). Localized HSV1 infection progresses to limited systemic disease in mice lacking type I interferon receptors (IFN I R−/−), while deficiency of both type I and II IFN receptors (IFN I/II R−/−) permits disseminated infection to progress to lethality. To investigate differential effects of IFN signaling on HSV1, we identified infected immune cells and analyzed effects of depleting these cells on pathogenesis in mice. Wild-type, IFN I R−/−, and IFN I/II R−/− mice were infected in footpads with a recombinant strain KOS virus expressing GFP or firefly luciferase. Flow cytometry of draining lymph nodes and spleen showed that neutrophils were the predominant cell type infected with KOS/GFP in IFN I/II R−/− mice during a 3-day course of infection. Compared with IFN I/II R−/− mice, 50% fewer neutrophils were infected in spleen of IFN I R−/− mice, while virus did not spread beyond footpads in wild-type mice. Neutrophils from IFN I R−/− and IFN I/II R−/−mice were permissive for in vitro infection and expression of luciferase from the early gene ICP8 promoter in KOS/dlux, but the reporter was not expressed in wild-type neutrophils. However, none of the neutrophil genotypes permitted complete HSV1 replication in vitro as quantified by viral titers. In IFN I R−/− mice, antibody-mediated depletion of neutrophils increased HSV1 replication by 10-fold as quantified by BLI without affecting local viral replication in either footpad or corneal models of infection. Limited systemic dissemination of KOS/dlux from footpad infection also was detected in IFN-competent mice that lack neutrophil enzymes neutrophil elastase and/or myeloperoxidase. Using this novel combination of fluorescent and bioluminescent reporter viruses, we conclude that interferons regulate susceptibility of neutrophils to HSV1 infection and that neutrophils limit systemic spread of HSV1.
G. Luker, None.
Optical Tomography & Low Light Imaging
Abstract ID: 179
Non-invasive visualization of cell adhesion molecule αvβ3 inetgrin expression has been well studied by means of radionuclide imaging modalities such as PET and SPECT in various preclinical tumor models. The use of cyanine dyes as contrast agents for optical detection of tumor integrin is not known. We report here the integrin receptor specificity of novel peptide-dye conjugate RGD-Cy5.5 as a contrast agent both in vitro and in vivo. The RGD-Cy5.5 indicated intermediate affinity for αvβ3 integrin (IC50 = 58.1±5.6 nM) and the conjugate led to elevated cell-associated fluorescence on integrin-expressing tumor cells and endothelial cells and the produced minimal cell fluorescence when co-incubated with c(RGDyK). Intracellar localization could be visualized with prolonged incubation time. The subcutaneous U87MG tumor was clearly visualized with a broad range of concentration of fluorescent probe administered. Intermediate dose (e.g. 0.5 nmol) produces better tumor contrast that high dose (e.g. 3 nmol) and low does (e.g. 01. nmol) during 30 min to 24 h postinjection, because of partial self-inhibition of receptor specific tumor uptake at high dose and the presence of significant amount of background fluorescence at low dose, respectively. The tumor contrast was also dependent on the mouse mounting angles ascribed to different skin thickness at different locations on the tumor surface. Tumor contrast was best visualized at 60 degree mounting angle where minimal skin scattering and NIR fluorescence intensity attenuation were observed. Moreover, tumor uptake of RGD-Cy5.5 was blocked by c(RGDyK). This study suggests that combination of the specificity of RGD peptide/integrin interaction with near-infrared (NIR) fluorescence detection, may be applied to noninvasive imaging of integrin expression and monitoring anti-integrin treatment efficacy (Funding support: NIBIB EB001785, DAMD17-03-752 and BC030012).
X. Chen, None.
Abstract ID: 180
Bioluminescent molecular probes have proven to be a valuable tool for monitoring expression of targeted genes in vivo. While basic 2D bioluminescent imaging is useful for monitoring relative bioluminescent emission levels in vivo, improved measurement of biologic activity at the cellular level can achieved with three-dimensional localization of photon source distribution, and quantification of source intensity. Three-dimensional localization and quantification of photon sources embedded in tissue can be achieved with diffuse tomography reconstructions based upon single-view 2D images of the surface topography and photon emission from an animal subject. Resolution is refined when multiple wavelength data are incorporated into the tomographic inversion.
As the Green's function kernel relating photon sources to photon density signal at the surface decreases monotonically as a function of depth in tissue with homogeneous optical properties, solutions to this tomographic problem gives preferential weight to shallow source depths for single-view, single-wavelength data. As the degree to which photons are scattered and absorbed in media is wavelength dependent, the Green's function kernel sensitivity at a fixed depth differs for different wavelengths. Thus, inclusion of multiple wavelengths in the Green's function kernel matrix and consequently, in the data, improves conditioning of the inverse problem.
The improvement of resolution for source location and quantification afforded to single-view diffuse luminescent imaging tomography by including multiple wavelength data is demonstrated with evaluation of a single fiber optic source embedded in tissue phantoms. We will also show photon source localization and quantification from in vivo bioluminescent images of mice with subcutaneously implanted B16F10 tumor cells, and mice presenting metastatic progression of PC3M-luc cells which were tail-vein injected five weeks prior to imaging.

C. Kuo, Xenogen Corporation 5.
Abstract ID: 181
Fluorescence-enhanced optical imaging of in vivo nude mouse melanoma xenografts is explored and compared to planar gamma imaging. A peptide labeled with both 111In and indocyanine green was developed for direct targeting to melanoma cell integrin receptors. Optical imaging (integration time=800 msec) with an NIR, intensified CCD camera system demonstrated successful targeting of the tumor evidenced by the [300 × 300] multi-pixel image. The detection of the targeted tumor was less visible by the conventional gamma scintigram (integration time= 900 sec M.CAM, Siemens). Quantitatively, the average tumor to background ratios for optical (1.6 +/− 0.3) and nuclear (1.8 +/− 0.9) were statistically similar, indicating the equivalent detection by each modality of NIR photons and gamma photons emitted from the tumor location. However, increased signal to noise was achieved with the ICCD system (7.9 dB) over the nuclear M.CAM system (1.4 dB) suggesting a reduced noise floor in optical images, and the weighting of optical modality detection to shallow and sub-surface photons for non-tomographic images. Optical imaging with fluorescent contrast agents must be evaluated if desired for functional imaging in the clinic. Optical imaging has promising temporal resolution with dynamically favorable integration times, increased spatial resolution, high sensitivity to emitted photons, and the potential for tomographic techniques which allow the identification of photons depths. Our results demonstrate the great potential of optical imaging as a diagnostic tool, and show that the complementary imaging of optical and nuclear provide a means of assessing image quality figures of merit.
J.P. Houston, None.
Abstract ID: 182
Quantitative, wide-field imaging of tissue fluorescence in small animals requires accurate knowledge of the background optical properties and the ability to resolve surface (skin) fluorescence from that of subsurface tissues (molecular targets in tumors, for example). Here we present a fast, non-contact and scan-free method that enables quantitative, depth-resolved imaging of tissue fluorescence, absorption, and scattering optical properties over a wide field-of-view. This method measures the modulation transfer function (MTF) of the tissue reflectance in the spatial frequency domain using spatially modulated illumination.
The tissue MTF encodes both optical property and depth information. This is analogous to the frequency-domain photon migration technique, where the temporal frequency of the photon density waves is related to the spatial frequency through the speed of photon density wave propagation in the medium of interest. Through an analytical diffusion model in the spatial frequency domain, one can quantitatively sample the optical properties of tissue. Furthermore, by varying the spatial frequency content of the illumination, one can control the depth profile of the fluorescence excitation light inside the tissue. Tissue fluorescence emission can therefore be resolved in depth, enabling images of surface and subsurface fluorescence.
A tissue-simulating phantom with surface and subsurface inclusions of fluorescent dye was imaged to demonstrate this technique. Forty spatial frequency images, ranging from 0/mm to 0.3/mm, were acquired at both excitation and emission wavelengths. The data demonstrate the two main features of the technique. First, quantitative maps of the absorption and scattering properties are demonstrated at the excitation wavelength. Second, depth-selectivity of the excited fluorescence is illustrated, enabling discrimination of surface and subsurface fluorescence features.
D.J. Cuccia, None.
Abstract ID: 183
Photoproteins have played a major role in advancing our understanding of biological processes. Yet, a more extended array of biocompatible, non-toxic and novel reporters would be highly desirable. We report the use of two novel luciferases identified from copepod marine organisms, Gaussia princeps and Pleuromamma xiphias. Both coelenterazine luciferases were efficiently expressed in mammalian cells, non-toxic, naturally secreted and showed unique flash characteristics. The humanized form of Gaussia luciferase (hGLuc) was up to 1 × 103-fold brighter compared to currently used luciferases, and had a somewhat broader emission spectrum. These novel luciferases provide a powerful new tool to imaging biologic processes.
B.A. Tannous, None.
Abstract ID: 184
The ability to image and quantitate fluorescent protein-labeled tumors and other fluorescently labeled markers in-vivo has generally been limited by the autofluorescence of the tissue. Skin, in particular, has a strong autofluorescence signal when excited around 490 nm (the appropriate excitation wavelength range for green fluorescent protein) and while longer wavelengths are more amenable to deep-tissue imaging, GFP-labeled tumor models and other transgenic systems are still used extensively for revealing details of tumor growth, angiogenesis, and metastatic spread, for example. RFP and other longer wavelength labels are being employed more frequently, and while tissue autofluorescence decreases with increasing excitation wavelength, the presence of any autofluorescence reduces the sensitivity of detection and accuracy of quantitation. A solution to signal degradation imposed by autofluorescence is to use multispectral imaging techniques. The CRI Maestro system is a complete multi-spectral imaging system that uses novel tunable filters optimized for in-vivo imaging in the emission range 500 to 950 nm. Additional features include high-quality optics, bright and flexible illumination and innovative software capabilities. These allow the user to spectrally characterize and computationally eliminate autofluorescence, revealing otherwise invisible labeled targets, which now appear bright and readily quantifiable against a near-black background. This improvement in signal-to-noise can increase sensitivity many-fold, allowing much smaller targets to be detected. In addition to autofluorescence removal, spectral tuning allows for the use of multiple fluorescent signaling molecules (multiplexing) as well as the detection and display of tissue components, including vascular structures.
R. Levenson, CRI 5.
Abstract ID: 185
The more heat resistant cells are the longer it takes until HSP70 gets expressed at increasing temperatures.
W. Hundt, None.
Abstract ID: 186
Fluorescent proteins (FP) have become essential reporter systems in biomedical research. FP are commonly detected by intravital microscopy (confocal or multiphoton) or reflectance imaging at the macroscopic whole animal level. The latter has been useful in detecting and following tumors in vivo, particularly those implanted near the surface or in surgically exposed organs. However, reflectance imaging has inherent limitations, as it does not retrieve depth information nor allow absolute quantification of fluorescence.
Here we present a set of technologies developed for tomographic imaging and quantization of FP expressing cells in vivo. We were particularly interested in FP in the visible range and employed a multi-projection illumination scheme and a highly sensitive CCD camera. Light propagation was modeled using a normalized Born field and a modified solution of the diffusion equation employing an absorption-dependent diffusion coefficient.
We show that detection of FP expression is feasible in deep tissues quantitatively (current detection threshold of 50 × 103 GFP cells in the center of a mouse) and that the method can be used for in vivo real time imaging (Figure 1). We have applied the technique to imaging and monitoring of GFP expressing lung cancers and for multichannel detection of viral transfection and gene delivery in a subcutaneous tumor model. We were able to image lung tumors as small as ~2mm in diameter, situated 0.8 cm deep and to simultaneously detect tumor and virus fluorescence using YFP and GFP expression respectively. In conclusion, the technique is useful for imaging and quantization of FP covering a broad wavelength range. This enables the use of different proteins (including red shifted proteins such as DsRed and HcRed) in a myriad of mouse models.

G. Zacharakis, None.
Abstract ID: 187
A. Wall, None.
Abstract ID: 188
Bioluminescent imaging of luciferase-expressing cells in living animals is a powerful technique for investigating molecular events and tumor growth and metastasis. Current commercial instruments support planar imaging, an established method for evaluation of tumor dynamics and response to therapy. A three-dimensional imaging approach would greatly enhance applications in biomedicine since light-emitting cell populations would be better associated with specific organs and tissues and because depth-dependent corrections for quantitative light output would be included in the technique.
We report a novel approach using multiple rotating CCD cameras, together with image reconstruction software to provide a 3D representation of light emitting cells within a living mouse. Multiple high sensitivity CCD cameras (at present three, with future expansion to five) simultaneously capture low-intensity light from the animal, and a computer-controlled rotation mechanism allows imaging at multiple angular positions, as required for three dimensional reconstructions. The support electronics allow for the simultaneous control of light exposure, image readout and preprocessing, temperature, and vacuum. Each camera in the system is calibrated so that the resulting images can be combined. A horizontal bed, made of sparse mesh material to reduce interference with light, is used to support and immobilize the anesthetized animal during imaging.
We present two experimental tumor models grown orthotopically in the mouse: the first produced by A549-LUC human tumor cells growing in the lung after tail-vein injection, and the second by human pancreatic Mia-LUC tumor cells implanted orthotopically in the pancreas. Both reveal growth patterns confirmed at necropsy.

E. Richer, None.
Abstract ID: 189
Luciferases provide the signals for in vivo bioluminescence imaging (BLI), and their properties are key to advancing this imaging modality. The intensity and spectral properties of firefly luciferase (Fluc+), two variants of click beetle luciferase (CBGr68 and CBRed) and Renilla luciferase (hRLuc), all optimized for mammalian expression, were determined in vitro and in vivo expressed in cultured mammalian cells, cells implanted in animals, or expressed hepatically in mice. Spectral analyses on the light emitted by cells expressing each luciferase in culture using a spectrometer correlated well to the spectra obtained from a cooled CCD camera with 20nm band pass filters (IVIS 200 Imaging System, Xenogen). Each luciferase revealed its unique spectrum with λmax at 578nm (Fluc+), 543nm (CBGr68), 615nm (CBRed) and 480nm (hRLuc) at 25°C. Luciferase activity increased with elevated temperature, but in addition a spectral red shift was uniquely observed with Fluc+, when the λmax at 37°C was 612nm. The influence of tissue on spectral profiles was assessed for tissue types and depths by expressing each enzyme in liver, lung or skin models and measuring surface-emitted bioluminescence. For Fluc+, CBGr68 and CBRed, only light above 600nm was detectable from liver or lung, whereas the spectral pattern of hRluc consisted of peaks at 500nm and 620nm. Modest effects of tissue on the emission spectra were observed subcutaneously expressed enzymes. The spectral shifts due to tissue results from the hemoglobin absorption, which has a strong absorption peak at 560nm. Greater attenuation was measured from deep-tissue expression models, due to scattering as well as hemoglobin absorption. These results have implications for luciferase selection for use in various animal models.
H. Zhao, None.
Abstract ID: 190
In vivo fluorescence optical imaging (OI) of mice is an emerging field in biomedical research. While qualitative detection of light emitted by fluorochromes is easily feasible, quantification is a challenge.
Limiting factors studied in this work are the homogeneity of the excitation light-source, autofluorescence sources in rodent food, and variations of in vitro cell labeling (important for cell trafficking studies). This work uses a hyperspectral imaging system, based on a V10E spectrometer (Specim), which maps one spatial dimension and wavelength onto a VersArray:512F CCD-camera (Roper Scientific). In contrast to filter-based OI systems, we acquire spectral information at 2.8nm resolution which allows removal of autofluorescence background.
For excitation, a Lasiris SNF-501 line-laser (640nm, StockerYale) was used. This laser illuminates 65 × 1mm2 with a homogeneity of 85%. To image the entire mouse, the laser and spectrometer slit are scanned across the surface to produce a 3-D dataset that gives spectral information (λ) for every point (x,y) on the mouse surface.
To study variations of cell labeling, melanoma cells were incubated in vitro with a lipophilic fluorescence dye (Cy5, Molecular Probes). Emission signals from the cells were analyzed comparing linearity and intensity for different labeling methods. As an example for 0.2 × 106−4.5 × 106 cells labeled by the same method, the normalized signal intensity varied ~77%. This large variation makes accurate quantitation and comparison across studies difficult.
In vivo detection linearity was tested with 0.5×106, 1.5×106 and 2.0×106 labeled cells from one batch injected subcutaneously in a mouse. The emitted signals showed a linearity of about 70% (670-760nm). The in vivo imaging study showed significant autofluorescence caused by mouse food interfering with the signals from the fluorochrome. 105001 Lab-Chow-Meal (Deans Animal Feed) was compared with manganese-free food (EF-1/81, ssniff) showing a 1.6 times higher autofluorescence signal at 650-750nm. Using manganese-free food can significantly reduce background signals.
U. Weisser, None.
Abstract ID: 191
Fluorescence enhanced optical tomography is a promising approach for in vivo imaging of the biomolecular environment in living tissues. For fluorescence optical tomography to emerge as a leading molecular imaging technique, we need to demonstrate its ability to interrogate functional and metabolic states of tissue at high resolution. The achievable resolution in current tomographic approaches is restricted by a priori fixed discretization. Adaptive mesh refinement may improve the quality of tomographic reconstructions in fluorescence optical imaging but it is yet to be deployed in medical imaging. In this contribution we present a novel fluorescence tomographic algorithm which utilizes dual adaptive finite element meshes for the three dimensional reconstruction of fluorescent targets beneath the simulated tissue surface illuminated with excitation light modulated at 100 MHz. Simulation results are presented for tissue phantoms. Frequency domain fluorescence measurements taken on the illumination surface are used to reconstruct the buried fluorescence target. Figures 1(a, b) depict the fluorescence amplitude solutions for two 1.95 mm3 targets placed 2 mm apart. High resolution adaptive image reconstruction is demonstrated in Figure 2 which depicts a 2 mm radius target reconstructed from surface fluorescence measurements. The algorithm will be applied for reconstruction of fluorescence yield images from the data collected at the photon migration laboratories on tissue phantoms filled with 1% Liposyn with fluorescent target containing 1 micro-molar Indocyanine Green solution.

Initial coarse mesh solution for two closely spaced 1.25 × 1.25 × 1.25 mm targets

Fluorescence amplitude after three adaptive mesh refinements
Image of the reconstructed fluorescent target
A. Joshi, None.
Abstract ID: 192
We present the first transport-theory-based, three-dimensional, tomographic reconstruction algorithm for fluorescence molecular imaging (FMT). Unlike currently existing codes, this algorithm is using a light propagation model based on the equation of radiative transfer (ERT) within a nonlinear optimization scheme to determine the spatial distribution of fluorophore concentrations inside highly scattering media. The ERT-based model is particularly well-suited to take small geometries and small source-detector separations into account as they are encountered in FMT of small animals. We have tested the performance of the code with an in vivo mouse model for Lewis Lung Carcinoma (LLC). The animal was injected with a Cy5.5-based fluorescent probe with high sensitivity to cathepsins. For the data aquisition the mouse was immersed into an imaging chamber containing a scattering matching fluid. One side of the chamber was illuminated with light (wavelength = 674 nm) emerging from 46 fibers arranged in a symmetric pattern [red crosses in Figure 1a)]. A CCD camera captured the excitation and fluorescence light (wavelength = 694nm) on the side opposite to the illuminating fibers [blue circles in Figure 1a)]. The measured light intensities became input to the image reconstruction algorithm. An example is shown in Figure 1. Figure 1a) shows a surface-weighted fluorescence image of the mouse model with a LLC on the left side, but no spatial information of the fluorophore concentration is available. Figures 1b - f) show tomographic images of the fluorophore concentration at different depths: b) 1 mm, c) 2 mm, d) 3 mm, e) 4 mm, and f) 5 mm.
Image of the reconstructed fluorescent target
A.D. Klose, None.
Abstract ID: 195
Holographic optical coherence imaging (OCI) can record full-frame depth-resolved images from biological tissue, without computed tomography, allowing fast video display under the interactive control of an operator [1]. The key component in the system is a photorefractive multiple quantum well (PRQW) that acts as a coherence filter passing back full-frame coherent image bearing light while rejecting the scattered background [1]. We propose the use of an InGaAs/GaAs PRQW as a coherent domain functional imaging platform based on multi-spectral imaging for application to both in vitro and in vivo biomedical imaging. The InGaAs/GaAs PRQW allows image holograms to be written by light that is at or above its band-gap (~1.06 μm). This realizes the potential to look at optically active biomarkers in tissue and also at tissue response to optical stimuli over a large spectral region by using a single holographic film. The use of biomarkers enhances the image contrast and allows one to differentiate between different types of tissue. By using genetic tags on the biomarkers, one can also look at the progression of disease in a specific area of tissue and also observe tissue response to chemical stimuli. Enzymatic pathways identified can be modulated to increase the production of reactive metabolites and selected biomarkers can be used to examine their toxic effects. This coherent domain functional imaging gives a lateral resolution of around 10 μm and a longitudinal resolution of around 15 μm. This can be further improved by a suitable choice of spectral bandwidth and spatial coherence of the LED.
Abstract ID: 196
The ability to sense more than one fluorescent color at a time is at a premium because of the explosion of probes made available by the genetics revolution. Traditional approaches to multicolor imaging generally employ multiple filter cubes, multiband dichroic filter sets or filter wheels, all of which lack spectral finesse and whose use can be complicated by slow switching, mechanical complexity, image shift due to changes in the optical path, and vibration.
True multispectral imaging can generate precise optical spectra at every pixel. Furthermore, once complete optical spectra are known for a particular sample, an efficient subset of wavelengths can be identified for use in subsequent imaging. The Nuance&tradesr; multispectral imaging system can transmit in a number of wavelength ranges (e.g., 400 to 720 nm or 500 to 950 nm) and when mounted on a microscope can be used for a variety of samples, from chromogenically stained slides in brightfield to a wide variety of light-emitting dyes in fluorescence mode.
Multispectral imaging has proved useful for multicolor FISH, and in particular, for resolving multiple species of GFP with overlapping emission spectra. It is helpful for the identification and elimination of interfering autofluorescence. This capability has also shown great utility in imaging tumor models in live animals. The excellent match between multispectral imaging technology and the new quantum-dot-based fluorescent labels is emphasized. The ability to detect accurately the spectral qualities of a dye in-situ proves to be particularly valuable. Multispectral imaging in brightfield can also be performed: it is possible to detect and quantitate the simultaneous presence 3–4 overlapping chromogens (deposited as part of immunohistochemical or in-situ hybridization procedures).
J. Mansfield, CRI, Inc. 5.
Probe Design: Novel Activation Strategies
Abstract ID: 197
Non-invasive imaging with transgenic reporter genes have been used for studying gene expression and regulation, and for assessing cell migration patterns in laboratory animals. BLI offers the advantages of rapid sensitive assays with little or no background, and coupling this approach with small molecule sensors will expand the current capabilities. Bioluminescence reactions involve emission of photons upon enzymatic (luciferase) oxidation of substrates, such as luciferin and aminoluciferin, into their oxy-luciferyl counterparts in the presence of ATP, 02 and Mg2+. These substrates can be modified and adapted for sensing other enzymatic activities, and we have modified the luciferase substrates by incorporating them into peptides for the purpose of developing in vivo protease assays. We utilize the amine group in aminoluciferin to attach unique, cleavable peptide sequences that can be recognized by specific proteases. Aminoluciferin is a substrate for luciferase only in its unattached form. Hence, in the presence of the targeted protease, aminoluciferin is released from the peptide-substrate for further catalysis by luciferase resulting in light production. We have synthesized aminoluciferyl probes for prostate specific antigen (PSA), a clinical marker for prostate cancer (PCa). Here we demonstrate that this approach can be used for in-vitro and in-vivo PSA assays. The peptide-substrate sequence is specific to PSA and does not react to any of its homologues. Such high degree of specificity will be useful in developing drug delivery systems and studying protease activity. This is a versatile approach and can be targeted to other proteases. Successful results from the work and the understanding thus gained may lead to improved in-vitro assays and refined animal models that will increase our understanding of disease mechanisms and enhance drug discovery and development.
R. Shinde, None.
Abstract ID: 198
Elastin and elastin fragments are being actively investigated for their role in tumor cell-extracellular matrix interactions, tumor growth, and matrix metalloproteinase activation and regulation. Elastin derived peptides and recombinant elastin-like proteins have been used experimentally as targeting agents. However, elastin has not been as extensively evaluated. This is in part due to the fact that purified elastin remains insoluble in solution, severely limiting its application to biological systems. Here we report the development of elastin nanospheres for diagnosis and therapy, specifically addressing the molecular targeting of elastin receptor positive tumor cells for targeted gene delivery. Elastin nanospheres were synthesized using sonochemical techniques and characterized according to concentration, size, shape, zeta potential and solubility. Production of thermally responsive nanospheres potentially capable of retaining the unique thermosensitivity of elastin-derived peptides is described. Elastin receptor positive M21 human melanoma cells were used for both in vitro receptor mediated uptake and in vivo studies via systemic administration of targeted contrast particles in the M21 Balb/c nu/nu subcutaneous tumor model. Cellular uptake of FITC labeled elastin nanospheres was studied in vitro via fluorescence and confocal microscopy. Data on the biodistribution and toxicity of elastin nanospheres in the mouse tumor model will be presented. Encapsulation of plasmid DNA encoding GFP within elastin nanospheres was utilized for ligand/receptor targeted gene delivery and HIFU mediated gene delivery studies. The effect of encapsulation on the plasmid vector was studied via PAGE and agarose gel electrophoresis, TEM and SEM studies using gold labeled plasmid DNA. Transfection efficiency was evaluated via comparison with standard lipofectamine assays. A molecular targeting approach utilizing elastin nanospheres may potentially increase the specificity and efficacy of tumor targeting and HIFU mediated gene delivery. In addition, this approach may lead to a better understanding of the role of elastin in tumor angiogenesis, invasion and dissemination.
M.A. McDonald, None.
Abstract ID: 199
There is increasing evidence for the importance of tissue oxygenation in development, progression, and response to cancer therapy. Thus, the opportunity to measure tissue oxygen tension (pO2) non-invasively may be significant in understanding mechanisms of tissue function and in clinical prognosis. The linear dependence of the spin lattice relaxation rate, R1, of the 19F NMR resonances of fluorocarbons on pO2 is well known and has been studied extensively. However, proton MRI is far more readily available. We now present hexamethyldisiloxane (HMDSO) as a 1H NMR based pO2 reporter molecule.
HMDSO has a single proton NMR resonance, which is ideal for imaging. It is highly hydrophobic interacting with dissolved gases, but not metal ions. We have found a linear response: R1 = 0.13 + 0.00175*pO2[torr] at 37 oC and 4.7 T. 1H MRI of reporter molecules is subject to potential interference from the large water and fat resonances, but the chemical shift of −5 ppm relative to water allows chemical shift selective imaging. We have implemented a spin-echo EPI based pulse sequence for imaging and measuring T1 values. The sequence comprises a train of 20 non-selective 90o pulses for saturation of signal followed by a delay t for magnetization recovery. Three CHESS pulses provide frequency selective saturation of water and fat immediately followed by spin-echo EPI detection with a slice selective 90o pulse and a frequency selective 180o pulse. pO2 maps were obtained in vivo in rat tumors in 21/2 mins following direct intra tumoral injection of 50 μl. Most significantly dynamic changes in pO2 were assessed with respect to interventions (e.g., respiratory challenge). Given the minimal toxicity and ready availability of HMDSO, we believe it has great potential for ultimate application as a pO2 reporter molecule in the clinic.
This work was supported by NCI Pre-ICMIC P20 CA086354 and DOD DAMD17-03-1-0101
R. Mason, None.
Abstract ID: 200
Gene-based therapy has been stimulated by remarkable progress in understanding molecular biology. However, progress and clinical trials would be enhanced by the ability to determine the location, the degree of activity and change in magnitude over time of the expression of the therapeutic genes. The development of non-invasive reporter techniques based on appropriate molecules and imaging modalities may help to assay gene expression. While many nuclear and optical imaging approaches have been presented as gene reporter methods, MR has lagged behind. LacZ, which encodes b-galactosidase, has been the primary choice of reporter gene to verify effective transfection in biochemistry for many years, and due to its promiscuous activity many reporter molecules are available for biological and histological analysis. A recent addition to the detective toolkit is S-GalTM 3,4-cyclohexenoesculetin-b-D-galactopyranoside). Upon cleavage by b-galactosidase in the presence of ferric ions (Fe3+), the aglycone chelates iron to produce an intense black stain. In the spirit of multi modality approaches to imaging, it occurred to us that the iron (Fe3 +) complex is not only visible, but also paramagnetic. We will present data demonstrating this novel approach to detecting gene activity.
S-GalTM is commercially available and readily enters cells. Action of b-gal rapidly generates an intense black precipitate, which induces strong T2 relaxation and intense MRI contrast. We believe this holds great promise as a novel MRI approach for imaging gene activity and detecting function.
Supported by NCI P20CA86354
R.P. Mason, None.
Abstract ID: 201
Unstable plaque rupture causes thrombosis, embolism and is a major factor in the pathogenesis of the acute events in cardiovascular events. It has previously been shown that myeloperoxidase (MPO), is a) secreted in abundance by activated neutrophils and macrophages in unstable plaque and b) serves as a prognostic indicator of inflammation. We hypothesized that by using paramagnetic electron donor compounds that rapidly oxidize and polymerize in the presence of MPO, the presence of MPO in a lesion can be spatially localized. We synthesized three potential paramagnetic substrates for MPO and tested them by utilizing magnetic resonance (MR) and MR imaging techniques. One of the compounds, a covalent conjugate of GdDOTA analog with serotonin (3-(2-aminoethyl)-5-hydroxyindole, 5-HT-DOTA(Gd) showed rapid polymerization in the presence of human neutrophil MPO resulting in a 1.7-2 fold increase in proton relaxivity (R1 and R2). The presence of myeloperoxidase activity in solution or in tissue-like gel phantoms could be directly imaged at 1.5T. Using a Matrigel&tradesr; tissue model system we were able to delineate the interface between the gel and the substrate solution suggesting the accumulation of MPO-converted polymerized 5-HT-DOTA(Gd). Therefore, MPO activity in tissues such as unstable plaques can potentially be visualized using MPO-sensitive “smart” molecular imaging probes detectable by MR imaging.
MPO activity imaging using SE T1-weighted MR image at 1.5 T (TR 350, TE 11) in the presence of 5-HT-DOTA(Gd). 100 μM. a) substrate only, b) substrate + 650 U MPO, c) substrate + 1300 U MPO, d) substrate + 2000 U MPO, e) substrate + 1400 U HRP (positive control), and f) control, substrate + 650 U MPO without H2O2 (negative control).
J. Chen, None.
Abstract ID: 202
In order to image overexpressed mRNAs noninvasively in tumors, we have developed methods for synthesis of PNA probes targeting oncogene mRNAs overexpressed in human cancer cells. The probes carry a small cyclized peptide analog specific for the insulin-like growth factor 1 receptor overexpressed on malignant cells, and a chelator to bind radioactive metal ions or a fluorophore. We synthesized chelator-PNA-cyclic peptide chimeras specific for CCND1, p53, MYC and KRAS mRNAs, along with several control sequences. Human ER+ MCF7 breast cancer cells overexpress cyclin D1 protein and insulin-like growth factor 1 receptor. The MYC chelator-PNA-peptide chimeras blocked reverse transcription of MYC from MCF7 RNA in vitro. Fluorescent confocal imaging revealed that fluorescein-labeled CCND1 PNA-peptides were internalized by MCF7 cells, and lowered cyclin D1 protein MCF7 xenografts in nude mice after intratumoral injection. The presence of CCND1 and MYC Tc-99m-PNA-peptides concentrated in MCF7 cells as a result of receptor internalization and mRNA hybridization was detected by scintigraphic imaging of γ-particles released by Tc-99m decay. Peptide and PNA mismatch sequences were inactive. Supported by DOE ER63055, NIH CA42960, and HL59769.
X. Tian, None.
Abstract ID: 203
A continuing challenge in the use of imaging for biochemical and physiological information is one of interpretation. Our approach has been to verify biochemical information obtained by MRS/MRI with self-quenching reporter probes that release fluorescent moieties upon enzymatic phospholipase cleavage, with the goal of correlating specific processes with MR-observable metabolic changes.
Phospholipases are a series of enzymes that catalyze phospholipid breakdown and perform a number of critical regulatory functions within cells and tissues. Phospholipase A2 (PLA2) levels are increased during inflammation, hyperproliferation and apoptosis. PLA2 has been implicated in a number of diseases including cancer, arthritis and Alzheimer's disease.
NMR studies of cells and tumors have shown increases in glycerophosphocholine (GPC) levels associated with chemo- or radiotherapy that has been proposed to arise from increased phospholipid catabolism and phospholipase activation. In this study, DU145 prostate cancer cells were treated with the differentiating agent phenylbutyrate (PB) and examined using 31P and diffusion-weighted 1H NMR spectroscopy. The self-quenching fluorescent phospholipid analogue, PED-6, was used to detect PLA2 activity. PB caused a dramatic time-dependent increase in 1H mobile fatty acyl chain, total choline and 31P GPC resonances. This was accompanied by significant increases in fluorescence released from PED-6 liposomes in PB-treated cells relative to controls. The combination of these two imaging modalities provides strong evidence that metabolic changes detected by NMR can be attributed to PLA2 activation. Given the importance of PLA2 to a wide range of illnesses, this study has significant implications for in-vivo imaging of disease processes and treatment regimens.
J. Delikatny, None.
Abstract ID: 204
Transcriptional changes are some of the earliest events that signal the presence of disease or a change in cell differentiation. Consequently there is great interest in developing probes that can be used to identify altered gene expression. We describe a novel nucleic acid probe design called a TRAP (Targeted Reversibly Attenuated Probe) in which aptamer activity is regulated by hybridization with a complementary nucleic acid. Aptamers are short single stranded nucleic acids that, like antibodies have high affinities and specificities for their target molecules. Many aptamer targets are small molecules that could serve as a contrast agents. An aptamer that recognizes such a contrast agent and retains it in or over the cell with which it is associated could be used to image the associated cell. If the aptamer activity is regulated by another intracellular molecule, such as a specific nucleic sequence, it becomes the basis of a method for imaging gene expression in vivo. Here we demonstrate an aptamer-based biosensor in which hybridization of a complementary regulatory nucleic acid controls the ability of the aptamer to bind its target molecule. Because the sequence of the portion of the TRAP that is regulated by the complementary nucleic acid is distinct from the aptamer sequence, alternative TRAPs can be created with different nucleic acid sequences to regulate the same or different aptamers. In the absence of the complementary nucleic acid, the aptamer in the TRAP is inactive due to its hybridization with an attenuator region at the opposite end of the molecule. Activation of the aptamer in the TRAP by the complementary nucleic acid at physiological temperatures is sensitive to single base mismatches. Thus a TRAP, optimized for intracellular function, could be applied as a probe for imaging gene expression in vivo.
Supported by DOE OBER
M. Nilsen-Hamilton, None.
Abstract ID: 205
Monocrystalline iron oxide nanoparticles (MION) have been explored as switchable magnetic “nanosensors” for magnetic resonance imaging where particle clustering can have a significant effect on R2. Here we present numerical simulations studying the effect of clustering of magnetic nanoparticles.
Monte Carlo simulations were performed in which particles were allowed to come together in groups of 2, 3 and 4 of varying inter-particle distances. Clusters are randomly distributed in space, and protons take random walks through the particle fields. Magnetic parameters represented MION-46L (2064 Fe atoms/particle, 65emu/g Fe, average diameter 4.6 nm).
Additional simulation parameters include binding efficiency (representing the percentage of successful clustering) and hydrodynamic radius (accounting for coating size surrounding the particle core).
Figure 1a shows a binding efficiency as low as 25% can be distinguished from the case of no binding. Figure 1d shows that for a given core size, R2 effects significantly decrease with increasing hydrodynamic radius.

Monte Carlo simulation results: a) R2 vs. Binding Efficiency, b) R2 vs. Number of Particles per Cluster with binding efficiency of 1.0, c) R2 vs. Binding Distance for binding efficiency 1.0, d) R2 vs. Hydrodynamic Radius for fixed core size.
Our results indicate that significant changes in R2 can be achieved in cases of limited clustering as may be expected in-vivo. Such situations require identification of binding against a background of unbound particles. The results also indicate that clusters involving small numbers of particles can be distinguished, as may be desired in “molecular beacon” type applications. Additionally, the results demonstrate the importance of binding distance and maintaining small coating thickness for a given core size.
These results are useful for designing switchable magnetic nanosensors and in applications where a limited number of particles may be involved in clustering.
O. Zurkiya, None.
Abstract ID: 206
We previously reporter the development of a dual, IRES-linked reporter gene containing the human sodium-iodide symporter (hNIS) and enhanced green fluorescent protein (EGFP) cDNA for both nuclear and optical imaging. Constitutive expression of the reporter gene was successfully assessed by FACS and fluorescence microscopy, as well as imaged by gamma camera ([99mTc]-pertechnetate) and PET ([124I]-iodide). Now we show that this dual reporter can be used to monitor the response of endogenous heat-shock protein (HSP) genes.

R.G. Blasberg, None.
Abstract ID: 207
Gene therapy shows promise for the treatment various disorders and clinical trials are underway. However, It is thought that, non-invasive detection of transgene expression in vivo would be of considerable value for assessing the location, magnitude and persistence of transgene expression. Because the induction of lacZ gene encoding the enzyme β-galactosidase(β-gal) has become a standard means of assaying clonal insertion, transcriptional activation, protein expression, and protein interaction, lacZ gene has been the most attractive reporter gene for detection of β-gal activity. Many chromogenic or fluorogenic substrates are well-established, but they are generally limited to histology or in vitro assays. We recently demonstrated that introduction of a fluorine atom into the traditional biochemical substrate ortho-nitro-phenyl galactopyranoside, could provide a novel enzyme activity sensor (viz. gene reporter) with minimal perturbation to a well-proven substrate. We have now developed a novel approach incorporating the 19F NMR pH reporter (6-FPOL). We will present the design, synthesis and evaluation of some new fluorinated vitamin B6 β-D-galactosides as exciting substrates for lacZ gene expression using the 19F chemical shift imaging.
J. Yu, None.
Abstract ID: 208
The most frequently used radiopeptides in the clinic are analogs of somatostatin for neuroendocrine tumor imaging and radiotherapy.
The need for higher accumulation is of importance for MRI contrast enhanced and gamma imaging and for targeted radiotherapy. This can be obtained by the grafting of several Gd(III)/In-111/Y-90 chelates onto the peptide carrier.
H.R. Maecke, None.
Abstract ID: 209
Two novel gadolinium-based magnetic resonance (MR) contrast agents for molecular imaging were designed and synthesized. They have the potential to trace physiological changes of extracellular pH, calcium concentration, and enzymatic activity.
DO3A-EP; 1,4,7 tris(carboxymethyl)-10-(2-phosphono-ethyl)-1,4,7, 10-tetraazacyclododecane (

For initial MR results, the gadolinium complex of DO3A-EP was prepared in aqueous conditions at pH 6.3. In vitro MR relaxivity studies were performed at 300 MHz to probe relaxivity changes with different pH. For Gd-DO3A-EP at 21°C, r1 increased by 70% from 2.3 to 3.9 s−1 mM−1(pH 7.5 to pH 5.5) and r2 increased by 57% from 2.8 to 4.4 s−1 mM−1(pH 7.5 to pH 5.5).
A.K. Mishra, None.
Abstract ID: 210
We report a new concept for photodynamic therapy (PDT) based on incorporating the photosensitizer (PS) and a singlet-oxygen (1O2) quenching/scavenging molecule onto a disease-targeting carrier, such that the PS becomes activatable by light only when targeting has occurred. This has the potential to give very high disease specificity in PDT treatment. As proof-of-principle of this concept, we designed the target-triggered PDT agent as depicted in Figure 1. This consists of a short peptide sequence specific to a protease that is over-expressed in cancer cells. The PS and a 1O2 quencher/scavenger (Q) are conjugated to the opposite ends of this sequence. Proximity of PS and Q can either quench 1O2 generation through PS triplet-state energy transfer and/or can scavenge the 1O2 that has been generated. We hypothesize that, in the presence of tumor-specific enzyme, the substrate sequence will be cleaved and PS and Q will separate so that the photosensitizer can be photo-activated. We have now synthesized a model compound in high yield and excellent purity. This agent contains 1) a pyropheophorbide as the PS; 2) a carotenoid as the 1O2 quencher; and 3) the PS and Q in this molecule were kept in close proximity by the self-folding of a caspase-3 specific peptide sequence (GDEVDGSGK). Upon caspase-3-induced cleavage, the 1O2 production increased markedly that was observed using direct 1O2 luminescence and lifetime measurements, providing proof-of-concept of this PDT ‘beacon’.

G. Zheng, None.
Abstract ID: 211
Gold nanoparticles have attracted the interest of investigators seeking to provide increased contrast in optoacoustic imaging applications. Bioconjugated gold nanparticles may be useful in providing tumor specific contrast in such applications. However, to the best of our knowledge, there have been no experimental studies that measure basic acoustic properties of these materials, namely, sound speed, attenuation and backscatter coefficients.
The work represented here will report on such a study. Experimental steps in this study include the synthesis of gold nanoparticles, casting gold nanoparticles in agarose gel phantoms, and measurements of the acoustic properties in a well-characterized laboratory ultrasound system. In addition, atomic force microscopy and light absorption spectra are used to evaluate the size and concentration of gold nanoparticles in the agarose phantoms.
The summary of the presentation will provide comparison between experimental results and theoretical models of acoustic scattering and how acoustic scattering is affected by the size and concentration of nanoparticles. Discussion of the potential of bioconjugated gold nanoparticles to provide ultrasound image contrast will be included.
E.J. Boote, None.
Abstract ID: 212
In Vivo visualization of gene expression is one of the goals of molecular imaging. In the present imaging techniques, MRI provides higher resolution than SPECT and PET and a deeper vision than the optic images. In this abstract, we designed a new enzymatic activity specific MR contrast agent, Gd-TTDA-GP [(3,6,10-di(carboxymethyl)-6-β-galactopyranosyl-3,6,10-triazadodecanedioic acid) gadolinium(III)], being sensitive to the present of beta-galactosidase which has a high turnover rate and is absent in mammalian cells.
The beta-galacyopyranose moiety as attached served to block the Gd(III) ion in the chelate from interaction with water and thereby maintained the complex in its silent form with respect to MR contrast enhancement. When the beta-galacyopyranose was removed, the agent would provide a positive contrast-enhancement signal. The enzymatic activity of beta-galactosidase to Gd-TTDA-GP in different buffers and conditions was tested in order to find the best reaction condition of the new contrast agent. Different concentrations of Gd-TTDA-GP are performed in Citrate Phosphate buffer (pH 4.2) and TBS buffer (PH 7.5) incubated in the presence of the serial dilutions of beta-galactosidase and heat-inactive beta-galactosidase at 37.0±1°C. The effect of Gd-TTDA-GP in T1 upon enzymatic cleavage for Gd-TTDA-GP is determined by NTU Bruker 3T MRI. Our results indicate that the Gd-TTDA-GP had better activity and better linear proportional performance in the TBS buffer and 30 units of beta-galactosidase after six hours. This preliminary result (Figure 1.) showed that Gd-TTDA-GP could be effectively cleaved by beta-galactosidase and has a highly correlation. It suggests that Gd-TTDA-GP would be a useful enzymatic activity specific MR contrast agent for visualization of gene expression in living animals by MRI.
The effect of Gd-TTDA-GP observed in T1 upon beta-gal-catalyzed cleavage after reaction for 6 hours at 37 °C. Where the definition of beta-gal unit is that one unit will hydrolyze 1.0 umole of o-nitrophenyl β-D-galactoside to o-nitrophenol and D galactose per min.
C. Chang, None.
Abstract ID: 213
Early detection is one of the main goals for treating malignancy. Using magnetic resonance imaging (MRI), we can now detect tumor size as small as 1 cm in diameter. However, detection of smaller primary or metastatic tumor is still challenging. The αvβ3 integrin plays an important role in human metastasis and tumor-induced angiogenesis. Targeting this receptor by RGD-peptide may provide information about the tumor invasion and metastatic status. In order to monitor the small lesion, we employed targeted liposomes encapsulating MR contrast agent for tumor imaging. The liposomes containing MR contrast agent gadolinium-DTPA (Gd-liposomes), fluorescent marker hydroxypyrene trisulfonic acid (HPTS-liposomes), or both, were prepared by a reversed phase method. The composition of liposomes were DPPC: cholesterol: PEG-DOPE: RGD-PEG-DOPE = 11:9: 0.01: 0.0001. In the flow cytometry experiment, RGD-liposomes showed better binding capability to human PC-3 prostate cancer cells as compared to the control liposomes without RGD-PEG-DOPE. Further, the T1 relaxation signal still can be detected when Gd-DTPA (100 mM) was encapsulated in the liposomes. Next we will test the RGD-liposomes in the mouse tumor models. We hope this system can provide not only the early tumor detection but also tumor-targeted therapy.
J. Chen, None.
Abstract ID: 214
The use of fluorescent reporters to study biological activity in live laboratory animals is a well-established technology with a wide range of applications in biomedical research. Fluorescent reporters include genetically encoded proteins, fluorescent dyes and microspheres, and newly developed semiconductor nanocrystals known as quantum dots (QDs). The unique optical properties of QDs make them attractive for in vivo measurements. These particles come in a wide range of colors, can be excited over a broad wavelength range, and have narrow emission bands at wavelengths that are directly related to the particle size. QDs have high quantum yields, are highly photostable, and due to their engineered surface chemistry, are biocompatible.
In the work presented here, three sets of QDs have been evaluated for in vivo applications. Emission peaks in the red to near-infrared range (605, 655, 705 nm) were selected for improved transmission through tissue. The excitation/emission spectra were measured in vitro using a spectrofluorometer. Images of QDs in suspension and implanted in mice were acquired using an IVIS® Imaging System 200 Series.
Both subcutaneous and intraperitoneal implantations of encapsulated QDs in silastic tubing as well as IV injections of small QD concentrations were investigated. These studies indicated that the longer wavelength QDs (705 nm) give improved sensitivity in vivo. A kinetic study of the IV injections was also conducted with results showing that the non-coated QDs localize in the liver and lymph nodes within 20 min of injection and remain there at least 24 hours post injection. Surface modification of the quantum dots with polyethylene glycol resulted in substantially longer retention in the circulation. Finally, a sequence of images taken with narrow band emission filters was used to demonstrate the subtraction of autofluorescent backgrounds. The narrow fluorescent emission peak of QDs is advantageous for implementing such tissue autofluorescence subtraction algorithms.
T. Troy, None.
Abstract ID: 215
The optical signal generated by Renilla Luciferase (RLuc) allows its use as a biomolecular tag in addition to being a reporter gene. Use of RLuc as a tag requires high stability and activity in a variety of environments including mammalian serum. For this purpose, we have generated a mutant RLuc with vastly improved stability in murine serum in addition to increased light output.
Using PredictProtein (http://cubic.bioc.columbia.edu/predictprotein), RLuc was found to have high homology (34-56% similar) to a number of bacterial Haloalkane Dehalogenases; this may indicate a lateral gene transfer event. From the aligned sequences, candidate mutations were chosen at positions where RLuc clearly diverged from the consensus. These candidates were prioritized using a homology model of the RLuc structure generated using SWISS-MODEL (v3.5, http://www.expasy.org/swissmod/) and crystal structures of the haloalkane dehalogenase LinB from Sphingomonas Paucimobilis.
Mutations were introduced using a site-directed mutagenesis kit on a template containing hRLuc (Promega) that already incorporated a C124Amutation (Liu and Escher, Gene, 1999). His-tagged proteins were produced by periplasmic expression in E. coli followed by Ni-NTA affinity chromatography. Purified mutant protein was tested for both light output per mass of protein and stability of luciferase activity in mouse serum at 37C. The 7 most favorable mutations which did not lead to significant shifts in the emission spectra were combined into a single protein.
The result of combining these 7 mutations with the C124A mutation was a mutant RLuc that, compared to native RLuc, had a 150-fold longer serum half-life (>105 hrs versus 0.7 hrs), a light output level 10-fold greater, and an emission spectrum that was red shifted 5 nm. This optimized Renilla Luciferase represents a significant improvement that will allow its use for tagging and monitoring biomolecules in vivo.
A.M. Loening, None.
Abstract ID: 216
The lacZ gene, encoding the enzyme β-galactosidase (β-gal), has been the most attractive reporter gene for detection of β-gal activity. Many chromogenic or fluorogenic substrates are well-established, but they are generally limited to histology or in vitro assays. 2-Fluoro-4-nitrophenol-β-D-galactopyranoside (OFPNPG) belongs to a novel class of NMR active molecules (fluorophenyl-β-D-galactopyranosides), which are highly responsive to the action of β-gal. OFPNPG has a single 19F peak with chemical shift of 55 ppm. It is cleaved by β-gal to OFPNP, which has a pH sensitive chemical shift of 59–61 ppm. The large change in the chemical shift allows us to image β-gal activity with magnetic resonance chemical shift imaging (CSI). We will present the results of 19F CSI studies of enzyme activity and lacZ gene expression in 9L glioma cells. A standard spin-echo CSI sequence was used at 4.7 T for these studies. Sodium trifluoroacetate was used as a standard for quantifying dynamic changes on a voxel by voxel basis. Our results indicate that OFPNPG is a promising gene-reporter molecule for future in vivo studies.
V.D. Kodibagkar, None.
Abstract ID: 217
Her-2 is a growth factor receptor protein that expressed on the cytoplasmic membrane of a cell, which was found to be differentially upregulated in a significant proportion of cancers with clinical prognostic significance. We have previously developed a novel non-polymer coated yet well dispersed iron-oxide nanoparticle with varied biocompatibility. Through a sandwich conjugation strategy, the anti-Her-2 monoclonal antibody produced from a hybridoma cell line was able to be tagged on the nanoparticle. The antibody-nanoparticle complex recognized the hepatoma cell line GP7TB that overexpress Her-2 and allowed a differential collection of the cancer cells from the control NIH3T3 cells without Her-2 expression by an externally applied magnetic field. Furthermore, the complex was able to inhibit cancer cell growth in Her-2 expressing tumors according to the WST-1 assay. The MRI contrast effect of the nanoparticle dispersion alone and antibody conjugated nanoparticles were analyzed in a 3T Bruker MR imager using TrueFISP and FSE sequences. The results indicate the nanoparticle displayed extremely low T1 and T2 relaxation time.
D. Shieh, None.
Abstract ID: 218
Ion-oxide particles have been used for MRI negative contrast agent due to its distinctive contrast effect in the T2 sequences. In this study, we have synthesized monodispersive ion-oxide nanoparticles about 4~6 nm size in water phase without polymer coatings. The nanoparticles presented excellent biocompatibility in both in vitro and in vitro analysis. In the NMR characterization, the nanoparticles presented extremely shortened T1 and T2 relaxation time in both water and serum. Both T2 and T2* contrast enhancement was obviously observed above a critical concentration using an arrays of concentration gradients of the nanoparticles. The MR contrast effect of the newly synthesized nanoparticle was evaluated in the healthy rat using a Clinical 1.5T GE MR imager. Significant negative contrast enhancement was observed in the parenchyma of liver immediately after IV administration of the particles. The nanoparticles was predominantly distributed in the Kepffer cells of the liver as indicated by the iron staining in the paraffin embedded tissue sections and the TEM ultra-structural observations.
D. Shieh, None.
Abstract ID: 219
Herein we report the development of selective fluorogenic reporter substrates for imaging hydroxysteroid dehydrogenase activity. Guided by a simple chemical concept, that an enzyme's reaction promiscuity may be exploited to develop agents for molecular imaging, a struc-turally diverse fluorogenic substrate library was synthesized and evaluated for favorable photophysical characteristics. Upon enzymatic screening for activity, a fluorogenic reporter was found to be selective for 3a-hydroxysteroid dehydrogenases. The dehydrogenases play essential roles in redox metabolism and have been implicated to be involved in prostate cancer. A second generation fluorogenic substrate library has been synthesized and evaluated for activity with human aldoketoreductases. These fluorogenic substrates are non-fluorescent until reduced, when they generate highly fluorescent products. The fluorescent products may be excited at 450 nm and have high quantum yields in biological media (>0.4). We would like to detail this thorough study which provides a promising new lead for optical imaging of redox metabolism.
D.J. Yee, None.
Abstract ID: 220
Elastin fragments play an important role in angiogenesis and tumor invasion. Elastin derived proteins (EDP) and recombinant elastin-like proteins have been evaluated as drug carriers for delivery of radionuclides and hydrophobic chemotherapeutics. Their role in tumor progression and control of extracellular matrix degradation via regulation of matrix metalloproteinases is currently an area of intensive research. Here we report synthesis and characterization of a dual labeled probe, cy5.5 conjugated to an EDP/dextran coated gadolinium oxide platform, developed for MR imaging and whole body optical imaging. Endocytosis secondary to elastin receptor binding provides a molecular target that was evaluated for its efficiency in delivering genes to M21 human melanoma cells. Electrostatic interactions between cationic gadolinium oxide nanoparticles and plasmid DNA containing a GFP vector was exploited for coupling DNA to the EDP-dextran Gd2O3 bioconjugate. A thermosensitive gene carrier system was developed using HIFU to induce the inverse temperature phase transition of EDP for targeted gene delivery. In aqueous solution at physiological temperature EDP are soluble but at temperatures greater than their transition temperature they hydrophobically collapse and aggregate, becoming insoluble. Accumulation of the DNA containing bioconjugate within tumor cells of the Balb/c nu/nu subcutaneous tumor model was evaluated via MR and optical imaging, confocal microscopy and immunohistology. Data on animal biodistribution and toxicity studies using radiolabeled (153-Gd) gadolinium oxide will be presented. Relaxometric characterization of the paramagnetic metal nanoparticle and confocal microscopy studies of FITC labeled EDP-dextran Gd2O3 were completed. Size, morphology, charge and conformational status of the plasmid DNA vector were analyzed via TEM, SEM, gel electrophoresis, photon correlation spectroscopy and zeta analysis. Transfection efficiency was evaluated using standard lipofectamine assays. It is anticipated that a molecular targeting scheme utilizing the temperature responsiveness of EDP for gene delivery will provide an attractive and efficacious new approach to tumor imaging and therapy.
M.A. McDonald, None.
Abstract ID: 221
The ability to visualize in real-time the expression level and localization of specific endogenous RNAs in living cells can offer tremendous opportunities for biological and disease studies. Here we demonstrate such a capability using two molecular beacon based methodologies. Molecular beacons are dual-labeled oligonucleotide hairpin probes with a reporter fluorophore at one end and a quencher at the other; they become fluoresce upon hybridization with a complementary target. The first method utilizes a pair of molecular beacons, one with a donor and the other with an acceptor fluorophore that hybridize to adjacent regions on the same mRNA target, resulting in fluorescence resonance energy transfer (FRET). Detection of the FRET signal significantly reduced false-positives, leading to sensitive imaging of K-ras and survivin mRNAs in live HDF and MIAPaCa-2 cells. FRET detection gave a ratio of 2.25 of K-ras mRNA expression in stimulated and unstimulated HDF, comparable to the ratio of 1.95 using RT-PCR, and in contrast to single-beacon result of 1.2. As the second approach, we developed peptide-linked molecular beacons that possess self-delivery, targeting and reporting functions, and demonstrated the sensitive detection of specific cytoplasmic and nuclear RNA in living cells. We conjugated the cell-penetrating peptide TAT to molecular beacons designed to target GAPDH mRNA, or U3 snRNA respectively, performed live-cell delivery of these probes, and detected fluorescently the RNA expression in HeLa and HDF cells. We found that the TAT peptide can deliver probes into cell cytoplasm and nucleus very fast (within 30 min), and the peptide-linked molecular beacons can specifically target cytoplasmic and nuclear RNA with high sensitivity. We further revealed intriguing subcellular localization patterns of RNA. We believe that the both the dual FRET molecular beacons and the peptide-linked molecular beacons approaches promise to open new and exciting opportunities for sensitive RNA detection and quantification in vivo.
P.J. Santangelo, None.
Imaging Advanced Animal Models of Disease
Abstract ID: 222
Evaluation and optimization of dosage schedules using orthotopic tumor models has traditionally relied upon surgical inspection of lesions or survival as the quantitative endpoint. Diffusion MRI (DMRI) has been shown to be a sensitive surrogate marker for tumor cellularity. Successful therapeutic intervention results in tumor cells dying, and thus a decrease in cellularity over time which results in an increase in the apparent diffusion coefficient (ADC) of water within the tumor. This study compared the effects of single (26.6 mg/kg body wt) and fractionated (2 mg/kg body wt daily, 10 doses total) doses of BCNU on tumor ADC values in intracerebral 9L tumors.
Tumor ADC values were quantified over pre-, during and post-treatment using a DMRI sequence. MRI was used to calculated tumor growth, delay, and log cell kill for each treatment group.
The fractionated treatment resulted in a significant retardation of tumor growth with an increase in ADC values (top panel). However, the diffusion values began to decline during the second dosage week which was interpreted to be due to the tumor becoming resistant to BCNU. Treatment of the tumor with a single dose of BCNU (bottom panel) resulted in both tumor regression and a greater increase in ADC values, compared to the fractionated schedule. MRI-based volume measurements revealed that the tumor slowed dramatically for the fractionated group but the ADC measurements revealed that the tumor began to undergo repopulation of the tumor mass prior to the end of the second week of therapy which could not have been detected by traditional anatomical imaging. These studies clearly reveal the power of DMRI in pre-clinical optimization of dosage schedules for anti-neoplastic agents.

D.E. Hall, None.
Abstract ID: 223
Bone marrow-derived cells (BMCs) have been found to play an important role in the formation of neointimal hyperplasia in different types of atherosclerosis, such as transplant-related graft vasculopathy, post-angioplasty restenosis, and hyperlipidemia-induced athero-sclerosis. Several studies have demonstrated the presence of BM-derived smooth muscle cells (SMCs) at the site of neointima formation in atherosclerotic disorders using tissue-staining methods. We attempted to develop a method using MR imaging (MRI) to track the biodistribution of BMCs in mouse models of atherosclerosis.
The study was divided into two experimental groups. First, to confirm the contribution of BM progenitor cells to vascular remodeling, BMCs were extracted from green fluorescent protein (GFP)-engineered mice, and then transplanted into Balb/c nude mice, in which the intima of the unilateral iliac arteries were injured via a wire-mediated endovascular approach. Second, to evaluate the possibility of using MRI to track BMC's, we extracted BMCs from LacZ mice, labeled them with an MR contrast agent, ‘Feridex,’ and transplanted the labeled BMCs into ApoE−/− mice, which were then fed an atherogenic diet to induce arterial plaque. Two months post-atherogenic diet, T2-weighted 3-D, ex-vivo MR imagingof the aorta of ApoE−/− mice was obtained using an 11.7 T MR scanner. In the first group, fluorescent microscopy confirmed the presence of “green” cells in the region of neointimal hyperplasia of the injured artery segments. In the second group, ex vivo MRI demonstrated multiple MR signal voids along the aortic wall of the ApoE−/− mice, associated with positive staining of Feridex and LacZ of the aorta tissue sections. These preliminary results provide encouraging evidence to support the potential of using MRI to track the migration of magnetically-labeled BMCs to atherosclerotic lesions, which could help to investigate the role of BM progenitor cells in the pathogenesis of atherosclerosis, and target them for therapeutic interventions.
X. Yang, None.
Abstract ID: 224
S. Biswal, None.
Abstract ID: 225
Quantitation of the number of cells residing in a certain organ/region of the body is critical for the assessment of the efficacy of the cell transfer associated applications including adoptive cell therapy and BMT. It is also important for the assessment of the proliferation potential and persistanceof the transfered cells. Previously, we demonstrated several applications of PET reporter gene imaging for generation of quantitative parametric assessments of the viral transfection. Here we test the feasibility of use of direct intratumoral administration of reporter expressing T-cells followed by MicroPET and bioluminescent imaging (BLI) to build a cell number(dose)/image intensity correlation.
M.M. Doubrovin, None.
Abstract ID: 226
Activation of the tyrosine kinase receptor Met by its ligand HGF/SF leads to diverse biological outcomes, proliferation, scattering, invasion, angiogenesis, and cell survival. Mimp, a Met-signaling induced mitochondrial protein, is homologous to the family of mitochondrial carrier proteins that localized in mitochondria, and leads to mitochondrial depolarization. Mimp induction leads to growth arrest, in response to HGF/SF in a dose-dependent manner, but not to apoptosis, in-vitro and significantly decreases tumor growth rate in-vivo.
GFP-Mimp contract was used for cell transfections and to generate transgenic mice. GFP-Mimp localizes to the mitochondria and induces accumulation of fat vesicles in transfected cells. The distribution of GFP-Mimp and alteration of mitochondrial activity were determined using a Zeiss LSM510 confocal microscope, spectral analysis with a Meta detector and image analysis.
These analyses revealed GFP-Mimp expression in mitochondria. This expression was coupled to a dramatic reduction of mitochondrial potential at the resolution of a single mitochondrion. Intravital imaging of GFP-Mimp expression in the transgenic mice revealed expression in adipose bundles and blood vessels. Scanning of exteriorized GFP-Mimp transgenic mice organs showed expression in the liver, kidney, heart, lung, and brain, and this expression was confirmed by Western blot analyses. Exteriorized brains showed high GFP-Mimp expression in the mitochondria of Purkinje cells. Ultrasound scanning of GFP-Mimp transgenic mice revealed a high occurrence of fatty livers (hepatosteatosis) in 62.5% of the mice relative to 25% of control, age-matched mice. About 71% of GFP-Mimp mice developed fatty kidneys, compared with 33% of age-matched control mice. The glucose levels in the serum of GFP-Mimp transgenic mice on a high fat diet were significantly higher compare to age matched mice. These results show the power of using transgenic mice, and molecular imaging, to study alteration of mitochondrial-potential and metabolic disorders.
This work was supported in part by NIH research grant (P50CA93990).
I. Tsarfaty, None.
Abstract ID: 227
Transgenic mice expressing SV40 T-antigen (Tag) under the control of the rat insulin promoter (RIP) develop carcinomas in pancreatic islets. Tumors develop via a multi-step process; at 8 weeks RIP1-Tag2 mice develop adenomas and at 10 weeks carcinomas; they die at an age of ~15 weeks from hypoglycemia. In preliminary studies, we found that weekly treatment of RIP1-Tag2 mice with intra-peritoneally (i.p.) injected interferon-γ producing Tag specific Th1 cells prolong life about two-fold. While little is known about the mode of Th1 cell trafficking, the focus of this work was to visualize Th1 cell trafficking, homing, and accumulation by in vivo non-invasive high resolution positron emission tomography (PET) followed by ex vivo autoradiography and histology. Tag-Th1 cells (107) cells were incubated in vitro with 400 μCi [64Cu]PTSM in 2.5 ml cell culture medium. After 3 hours incubation and wash cycles the cells incorporated ~20% 64Cu. Trypan staining confirmed that >90% cells were viable after labeling, and 60% after 24 hours. The labeled cells were i.p. injected into RIP1-Tag2 and naïve C3H mice. PET scans were repeated 30 minutes, 3, 8, and 24 hours after injection and clearly showed the time course of Th1 cell homing close to the pancreas, while solid tumors showed only faint uptake at this early time. After each PET scan the mice were imaged in a 7T magnetic resonance imaging scanner and the images were fused for better anatomic localization. Autoradiography and H&E histology confirmed the PET findings and revealed intensive accumulation of Th1 cells in pancreas lymph nodes. The uptake ratio of lymph node to pancreas tissue was ~22-25. Dynamic non invasive PET imaging over 24 hours showed clearly Th1 cell trafficking and sites of accumulation. These findings were confirmed by autoradiography and histology. Further studies will show potential tumor uptake at later time points.
B.J. Pichler, None.
Abstract ID: 228
The early detection of bone metastasis resulting from breast and prostate cancer continues to be a signifcant clinical challenge requiring new approaches to monitoring and treating disease progression. Herein, we report the characterization of two bone metastasis models using noninvasive Micro-CT imaging correlated with histopathology. Using an intracardiac injection technique, either 5×105 PC-3 androgen independent human prostate cancer cells or 5×105 T47D estrogen receptor positive human breast cancer cells were inoculated into 6–8 week old female scid mice. Estradiol pellets were subcutaneously implanted into the T47D inoculated mice one week prior to inoculation. Mice were then serially imaged weekly under anesthesia using an ImTek MicroCAT II Micro-CT scanner. Mice were euthanized at the development of adverse clinical signs. The development and progression of bone metastases were followed using Micro-CT imaging. Bone metastases were confirmed by histopathology. Approximately 60% of the PC-3 inoculated mice and 66% of T47D inoculated mice developed bone metastatic lesions. Bone metastatic sites included femur, tibia, humerus, ulna, radius, and mandible. Bone lesions less than 1 mm were able to be discerned on the Micro-CT images. Other soft tissue metastatic sites included adrenal gland, heart, subcutaneous tissue, and muscle. These studies demonstrate the utility of using Micro-CT imaging to follow bone lesion development in mouse models of prostate and breast cancer bone metastasis. This work was supported by NIH Grants DHHS-2R01-CA72942, DHHS-1P50-CA13013, DHHS-T32 RR07004, and American Cancer Society Grant RSG-99-331-04-CDD.
C.T. Winkelmann, None.
Abstract ID: 229
There is currently no reliable method for differentiating normal, malignant and non-malignant lesions in the mammary gland in vivo and thus no method for studying the stages of carcinogenesis in vivo in an animal model. Optical spectroscopy in the ultraviolet to visible (UV-VIS) wavelength region offers promise for the diagnosis of cancer because it is fast, low cost, and sensitive to changes in the structural and biochemical state of tissue. Meaningful physiological parameters, such as hemoglobin concentration (oxygenated and deoxygenated) and beta-carotene concentration can be obtained from diffuse reflectance spectra. Optical properties extracted from diffuse reflectance spectra measured in vivo in the 400–600 nm range were used to differentiate hyperplasias (n=7), tumors (n=14) and normal regions (n=23) of the exposed mammary glands of 17 ENU-treated FVBxB6 Apc Min /+ mice. Statistically significant differences between tumor and normal were found for total hemoglobin concentration (p<0.005), oxygenated hemoglobin concentration (p<0.005), deoxygenated hemoglobin concentration (p<0.005) and beta-carotene concentration (p<0.05). Statistically significant differences were also found between tumor and hyperplasia for beta-carotene concentration (p<0.05). The results of this study indicate that normal, malignant and non-malignant lesions in the mouse mammary gland can be differentiated with optical spectroscopy in vivo based on endogenous physiological parameters.
M. Skala, None.
Abstract ID: 230
Myelin damage, as seen in various neurological disorders, impairs axonal conduction and is associated with axonal degeneration. Therapeutic interventions targeting the cessation of disease progression, promotion of remyelination and inhibition of axonal destruction require an effective diagnosis to monitor the efficacy of new treatments. However, a diagnostic modality capable of non-invasively and effectively evaluating myelin integrity is currently not available. MR diffusion tensor imaging (DTI) has been widely applied to characterize underlying mechanisms of development and pathology in the central nervous system (CNS) by detailed analysis of water diffusion. An analytical approach for interpreting DTI parameters that takes into consideration white matter structure and the underlying pathology has been proposed. Evidence suggests that axial and radial diffusivities derived from DTI are directly linked to axon and myelin integrity. To further test this hypothesis, we examined a well-established mouse model of reversible demyelination in the brain using in vivo DTI. The model involves the addition of cuprizone to the animal feed. Six weeks of cuprizone feeding results in severe to complete demyelination of the corpus callosum in the mouse brain. This cuprizone induced demyelination is readily reversed by returning the animal to normal feed. Our MR results demonstrate that radial dif-fusivity accurately predicts the course of cuprizone induced demyelination and the subsequent remyelination after removal of cuprizone in the animal feed non-invasively. Thus, MR diffusion analysis provides a noninvasive, nondestructive window on the molecular events in myelin pathology.
S. Song, None.
Abstract ID: 231
Corneal fibrosis (condition characterized by opacity and/or scarring) is the final outcome to a variety of corneal diseases and a leading cause of blindness. We are using endogenous cellular fluorescence and second harmonic signals from collagen fibrils to determine the extent of corneal damage in K14-DN-Clim mice. The dominant negative (DN) Clim (Co-factor of LIM - a diverse group of transcription factors) is expressed in the epithelial tissues in mice using keratin 14 (K14) promoter resulting in a disrupted epithelial transcriptional complex. Eighty percent of mice are blind due to corneal opacities. In severe cases, the effected mice exhibit abnormal overgrowths of the stratified squamous epithelium, which are visually detectable and confirmed by histology. In moderately effected individuals, endogenous cellular fluorescence and second harmonic signals from collagen show displasia in the squamous epithelium, irregular collagen arrays in the stroma, and a compromised posterior endothelium. An intrinsic cellular emission allied with second harmonic signals are promising in-vivo clinical diagnostic tools for corneal abnormalities.
J.G. Lyubovitsky, None.
Abstract ID: 232
Crosstalk between the androgen receptor (AR) and mitogen activated protein kinase (MAPK) is hypothesized to play a role in prostate cancer progression from the androgen-dependent (AD) to independent (AI) state. We developed optical imaging constructs that can non-invasively monitor AR and MAPK activity in AD and AI tumors within xenograft mice. The constructs are based on the concept of Two-Step Transcriptional Amplification (TSTA). In TSTA, a prostate-specific regulatory region drives the potent activator GAL4-VP16, which in turn activates firefly luciferase (Fluc) to high levels. The TSTA approach greatly amplifies reporter gene activity facilitating optical imaging in dense tissues like tumors. Fluc activity in TSTA-adenovirus infected tumors is monitored using the Xenogen CCD camera following injection of D-luciferin into tumor-bearing mice. The second construct, TSTA-ELK, monitors both AR and MAPK activity in AD to AI prostate cancer progression. In TSTA-ELK, GAL4-ELK is synthesized in the presence of AR but only activated upon phosphorylation by MAPK. Transient transfections with TSTA-ELK in LNCaP cell lines show that TSTA-ELK responds to both androgen activation by AR and MAPK activation via the EGF pathway. Additionally, inhibitors of AR and EGF block TSTA-ELK optimal activity. Ex vivo and adenovirus tumor infection studies in xenograft models will be reported along with the effects of drug intervention in the various pathways. The TSTA and TSTA-ELK systems represent new transcription based approaches for optical imaging of specific pathways in live animal models of cancer.
R.S. Ilagan, None.
Abstract ID: 233
Transgenic line carrying a specific tissue tagged by green-fluorescence-protein (GFP) live marker has been a powerful tool to study developmental biology because they encapsulate the expression of endogenous genes. Traditionally with two-photon optical microscopy based on a femtosecond Ti:sapphire laser (with a wavelength between 700-980nm), green fluorescence can be excited by simultaneous absorption of two photons for high-resolution three-dimensional (3D) optical imaging. However for future in vivo biolo-gical applications, Ti:sapphire-laser based optical technology still presents several limitations including finite penetration depth, strong on-focus cell damage, and phototoxicity. For high optical penetration and minimized photodamages, two-photon molecular imaging based on light sources with an optical wavelength located around the biological penetration window (~1300nm) is desired, where unwanted light-tissue interactions including scattering, absorption, and photodamages can all be minimized. Previous experiments around the optical penetration window indicated inefficient green fluorescence excitation of GFP through three-photon absorption. Red fluo-rescence protein with an emission wavelength close to near-IR is thus highly desired for future noninvasive two-photon molecular imaging in vivo. Screening from embryos injected with DNA fragment containing a heart-specific regulatory element of zebrafish cardiac myosin light chain 2 gene (cmlc2) fused with HC-Red gene, we generate a zebrafish line that only has strong two-photon red fluorescence expressed in the heart based on a 1230nm femtosecond light source working in the biological penetration window. Combined with its nonlinearity, high penetration depth, minimized photodamages, and the capability to combine with multiharmonic optical microscopy, this new method provides superb imaging capability compared with the traditional GFP based two-photon micros-copy, offering deep insight into the noninvasive in vivo studies of gene expression in vertebrate embryos.
T. Tsai, None.
Abstract ID: 234
The relationship between the over-expression of the multidrug resistance-1 (MDR1) gene and clinical drug resistance in human cancers remains tentative. Good models to study the links between drug resistance and MDR1 expression have not been available. Using small animal imaging tools we now have designed a mouse model that will allow us to examine mdr1a (a homologue of human MDR1) expression in vivo, in real time, and in response to developmental, physiological, and environmental signals. Through homologous recombination we have inserted the Renilla luciferase (LUC) gene into the genomic locus placing it under the control of the mdr1a promoter. This model has been designed to allow us to examine tissue-specific expression of mdr1a through use of Cre-loxP technologies. To our knowledge our model is unique and therefore, if successful, will be the first system of its kind to examine locus controlled, regulated reporter gene expression in specific tissues in vivo.
The following milestones have been accomplished: 1) we have engineered a targeting vector to insert LUC into the genomic locus of the mouse mdr1a gene in a way that makes in-frame expression of the reporter conditional on Cre-mediated recombination. 2) we have created mouse embryonic stem (ES) cells and demonstrated homologous recombination into the mouse mdr1a genomic locus. 3) we have shown that Cre transfection into these cells mediates LUC recombination into the first coding exon of mdr1a. 4) mdr1a/LUC ES cells have been used to generate the corresponding knock-in mice. 5) mdr1a/LUC mice have been crossed with ubiquitous Cre-donator mice and resulting off-spring have been screened for Cre-recombination; positives have been obtained, and analysis of these mice is in progress.
D.A. Brown, None.
Abstract ID: 235
Apoptosis is an important pathogenic mechanism in acute renal failure. Currently there are no available methods for real-time detection of renal cell death in vivo that would permit early assessment of evolving injury in acute tubular necrosis (ATN), a common cause of acute renal failure. Positron emission tomography (PET), is regarded as a modality of choice for future molecular functional imaging, therefore emphasizing the need for an apoptosis PET-imaging tool.
A. Shirvan, NST Ltd. 5.
Abstract ID: 236
Recent studies have shown the ability for bone marrow-derived cells to differentiate into glomerular mesangial cells and to modify the phenotype of normal glomeruli. We performed MR cell tracking of IV-injected Feridex-labeled mesenchymal stem cells (MSCs) in order to assess their homing potential for repair of mesangial glomerular cells in a rat model of mesangiolysis (by co-injection of OX-7 moab and PAN). Rat MSCs were magnetically labeled with Feridex and Superfect. Fourteen rats were used: two pathologic groups (n=5) in which 1E7 MSCs were injected i.v. 4 and 8 days after the induction of the nephropathy; and two control groups (n=2 each). For the in vivo MR imaging, performed at 4.7T with 1 mm slice thickness, SI measurements of pathologic kidneys at 6 days post MSC injection did not show any signal drop, including the cortex (p=0.7). On the contrary, a significant signal decrease was noted in the liver (p<.01) indicating the trapping of labeled cells by the liver, acting as a filter. For the ex vivo MR imaging, performed at 9.4 T (50 μm resolution), also at 6 days post injection, all pathologic kidneys showed focal cortical areas of signal loss, corresponding to the anatomical location of glomeruli, that was absent in the control groups. Anti-alpha-actin immunostaining showed a focal nephropathy with areas of glomerular damage, that matched with the localization of DiI positive MSCs, and that was absent in control kidneys. These results show that labeled MSCs injected intravenously can specifically localize in damaged components (glomeruli) of pathological kidneys and that these cells can be detected ex vivo by MR imaging. The lack of signal changes on the in vivo MR images can be explained by the much lower label sensitivity due to the lower resolution that was used.
J.W. Bulte, None.
Abstract ID: 237
The exquisite specificity of the immune system makes adoptive immunotherapy an attractive component of the anti-tumor arsenal. The ultimate goal is to use T-cells and antibodies to eliminate residual or metastatic tumors after treatment with surgery and/or chemotherapy. Rodent tumor models have been instrumental in tumor immunology. We have a murine CD8+ TCR transgenic (DUC18)/CMS5 tumor model to study adoptive immunotherapy (Hanson et. al., Immunity, 2000, Matsui et. al., Int. Immunol., 2003). DUC18 T-cells specifically recognize the antigen/MHC complex on CMS5 fibrosarcoma cells and the antigen recognized by DUC18 T-cells arises from a mutated ERK2 kinase (Ikeda et. al., PNAS, 1997). In this system, day-8 established CMS5 tumors are eliminated by 20×106 activated DUC18 T-cells.
We have previously utilized microPET and MRI to monitor the T-cell distribution patterns, tumor growth and ablation processes in the DUC18/CMS5 system. The use of high-resolution imaging technologies, and in particular MR, has the potential to significantly impact and advance this field. The use of MRI to monitor cell uptake and -trafficking in vivo has been recently described (Kircher, Cancer Research, 2003; Anderson, Ann Neurol, 2004). The MRI method relies upon the shortening of the T2* of water that results from paramagnetic iron in Fe-labeled cells. Here, we demonstrate the use of MRI to track the distribution of Fe-labeled tumor-specific CD8+ T-cells in our tumor model. In this study, we labeled DUC18 T-cells with poly-
J.R. Garbow, None.
Abstract ID: 238

Current progresses in harmony with stem cell biology and tissue engineering techniques have extremely contributed to the clinical applications. Transplantation of cells into patients will require techniques that can monitor their tissue biodistribution noninvasively. For this purpose, firstly, human bone marrow-derived mesenchymal stem cells (hMSCs) were labeled with superparamagneit iron oxide (Feridex). Histological study and MTT assay showed that cell labeling with MR contrast agent did not give harm in the cell viability. Also, Feridex-labeled hMSCs showed significant decrease in T2 signal intensity, even within the gelatin sponge in vitro. Lastly, there was in accordance with in vivo MR imaging and histological study. As a result, MR imaging played significant roles not only in visualizing the implanted tissue engineered, but in monitoring the biodistribution of implanted cells in vivo, which illustrates the potential of new approach proposed here for in vivo monitoring of implanted cell-based tissue engineered product.
Figure 1. MR imaging of the nude mouse four weeks after the transplantation of tissue engineered SPIO labeled hMSC. Susceptibility artifact extended over the estimated boundary of the scaffold implanted subcutaneously (arrows). MRI was performed in 1.5T clinical magnet with 47mm micro surface coil with scan parameter of TR 183ms, TE 14ms, flip angle 30°, matrix 256×256, FOV 60mm.

Figure 2. Prussian blue staining of the implanted tissue engineered scaffold and surrounding host tissue en block at 4 weeks after the transplatation. Implanted hMSC was identified within and outside of the scaffold, subcutaneous tissue, and dermal layer of the skin.
H. Song, None.
Abstract ID: 239
Systemically administered transplants of stem and immune cells have been used for many types of cell tracking studies where tracking of cells to a site of interest by natural signaling and migration is desirable, including T-cells in autoimmune disease and stem cells in cancer. Intravenously administered cells pass through the lung, liver and spleen soon after administration. We report persistent MRI effects in liver for over three weeks along with very distinct SI and pattern changes after transplants of non-stimulated lymphocytes or splenocytes in mice with direct-induction experimental autoimmune encephalomyelitis. 20 million lymphocytes or splenocytes were magnetically labeled with Ferumoxides-Protamine sulfate complexes (1) and administered via tail vein. Mice were imaged serially at 10, 17 and 24 days at 7 T, and euthanized at each time point. MR microscopy was performed at 7 T on fixed liver at 50–60 micron isotropic resolution. The liver was strongly hypointense at day 10, and had a marked mottled pattern at day 17, evident on both in vivo and ex vivo MRI, that was consistent with contrast effects concentrated outside the hepatic lobule and around portal triads. By 24 days the liver had attained a uniform appearance that was hypointense relative to livers from mice receiving unlabeled cells. In comparison rats that received intravenous FE-Pro labeled stem cells (2) had homogenously hypointense liver compared to kidney and returned to baseline control over a period of 28 days. MRIs of lymphocyte and splenocyte infusions were similar and may be correlated to the distribution of cells in the liver. Histopathology was performed and compared to the MRI to provide a correlation between MRI features and the cell density and distribution of magnetically labeled cells in liver.
S.A. Anderson, None.
Abstract ID: 240
We have developed a portfolio of agents for the purpose of imaging molecular activities known to be associated with multiple aspects of disease pathology such as inflammation in arthritis models and vascular volume in angiogenesis models. Some of these agents are designed to be activated by disease-associated enzymes. We describe the characterization and use of these agents in defined systems in vitro and in corollary models of disease pathology in vivo. We then demonstrate the utility of these agents in measuring disease progression and in quantitating therapeutic responses to drugs targeting diverse molecular pathways.
D. Falb, VisEn Medical 5.
Abstract ID: 241
We describe the characterization of a protease-activatable probe, which we refer to as ProSense, and its use for imaging disease onset and response to treatment in a mouse model of arthritis. Correlation between clinical score and ProSense activation will be presented. The activities of a number of proteases (Cathepsins B, C, D, G, K, L and S, Plasmin, Plasma Kallikrein, uPA and CD10) known to be associated with the development and maintenance of arthritis were measured in paw homogenates from healthy and arthritic mice. Protease expression and activity, as measured by immunoassays and the ability to cleave synthetic substrates, respectively, were significantly higher in arthritic paw homogenates than in healthy ones. The increase in protease activity correlated positively with increases in imaging intensity and clinical score. With the goal of better characterizing the protease activities measured in paw homogenates, we used a combination of synthetic substrates and protease inhibitors. The specificity profile of protease activities in paw homogenates against these substrates and inhibitors was compared to that of recombinant enzymes from the panel described above. The data obtained showed a significant increase in a cathepsin B-like cysteine protease activity in arthritic paws when compared to healthy ones. A complete analysis of the results from these studies will be discussed.
Based on our data, it is evident that ProSense activation closely reflects the disease status in arthritic paws. The use of this imaging agent could be valuable when screening compounds for anti-arthritic effects in small animal models.
M. Alaoui-Ismaili, None.
Abstract ID: 242
The purpose of this study is to demonstrate the ability of functional X-ray CT to follow autosomal recessive polycystic kidney disease (ARPKD) progression in pcy mice.
ARPKD is the childhood form of the polycystic kidney disease (PKD). PKD is a genetic disorder causing renal failure and death in adults and children. The pcy mouse is a model of human ARPKD. The mouse model is marked by an inability to concentrate urine in the early stages of the disease and end stage renal failure. PKD mice have higher levels of Adenosine-3′, 5′-cyclic monophosphate (cAMP) in the kidney compared to normal wild type mice and cAMP has a major role in proliferation and enlargement of renal cysts formed mainly in the collecting ducts. This is accompanied by changes in blood flow, vascular and interstitial volumes and progressive loss of renal function. Functional X-ray computed tomography (CT) could quantify tissue physiologic parameters by observing and modeling the passage of iodinated radio-opaque contrast agent through tissue. We hypothesize that renal physiologic parameters obtained from functional X-ray CT namely perfusion, permeability, and intravascular and interstitial volume fractions, would capture the changes in kidney physiology brought about by changes in renal cAMP content and progression of PKD. There are two experimental groups of 4 mice each. The first group consists of pcy mice and the second group consists of normal wild type mice. The mice are scanned with dynamic contrast enhanced X-ray CT protocol and later sacrificed for immunohistochemical analysis. Following injection of 0.2ml of radio-opaque contrast agent images are acquired every 0.95 seconds for 4 minutes and the every 5 seconds for the next 16 minutes. The difference between the renal physiologic parameters of normal and PKD mice will be reported and their correlation with results from immunohistochemical studies will be presented.
G. Krishnamurthi, None.
Abstract ID: 243
The last few years has seen an explosion in the development of antiangiogenic pharmaceuticals for tumor therapy. Currently the gold standard for measuring their efficacy is cessation of growth during a specific time period. However, the heterogeneity of tumor type, tumor location, and tumor morphology means that the results of such measurements are often inconclusive. Clearly then, there is a need to develop better surrogate markers of antiangiogenic efficacy. Significant effort over the last few years has been made to understand the effects antiangiogenic pharmaceuticals on tumor physiology in preclinical animal models through various types of imaging. It was the hypothesis of this study that blood flow imaging of rat brain glioma models could provide complimentary information regarding the effect VEGF on tumor physiology. To test this hypothesis stably transfected 9L cell lines with wild type (VEGF-0), sense (VEGF++) and anti-sense (VEGF–) levels of VEGF were grown in cell culture and then transplanted intra-cerebrally into Fischer 344 rats (6 animals per cell line). Tumor diffusion, growth rate and tumor perfusion were then measured using an MRI protocol that included, T2 weighted imaging, diffusion weighted imaging and CASL-MRI. The results of this imaging were then correlated to both histology and VEGF levels as measured by Northen blot analysis.
The use of molecular biology to genetically engineer glioma cells with 3 different vascular phenotypes provides an exciting opportunity to evaluate the effects of VEGF expression levels on perfusion MRI. Unexpected findings include tumor perfusion values which were higher in VEGF– clones relative to wild type. These results indicate the probability that the tumor is recruiting neovasculature via a different mechanism. This will be further investigated using gene expression array technology. Finally, these results indicate that anti-angiogenic therapies targeting VEGF may not prove entirely beneficial due to alternative molecular pathways of tumor angiogenesis.
B.A. Moffat, None.
Abstract ID: 244
J.P. Weichert, None.
Abstract ID: 245
Advances in laboratory animal imaging have provided new biomedical resources in the discovery of radiopharmaceuticals. Among these technologies is micro-computed tomography (Micro-CT) which is widely used to obtain high resolution anatomic images. Because Micro-CT depends on ionizing radiation for image formation, radiation exposure during imaging has raised justifiable concerns. The objective of this study is to quantify the radiation exposure a mouse receives during a Micro-CT scan and to test for potential therapeutic effects associated with this radiation dose in a murine cancer model. Radiation exposure was measured using thermoluminescent dosimeters (TLD) irradiated during a typical scan protocol (80 kVp X-ray source with fixed exposure times of 300 msec/step, 360° of rotation in 360 steps with a beam current of 500 μA). Results indicated a single scan dose of 7.695±0.456 R when conducted in a lucite anesthesia support module and a dose of 8.025±0.850 R with out the use of the anesthesia module. The data was validated using a Keithley 35050A ion chamber with a 1.5 cc probe, providing measured exposures of 6.858±0.036 R and 8.625±0.049 R, respectively. Potential in-vivo therapeutic effects due to radiation exposure were assessed using a rodent lung tumor model derived by IV injection of B16F10 mouse melanoma cells in C57BL6 mice. Survival analysis demonstrated a mean survival of nontreated control animals of 28.7±4.2 days, with animals receiving up to five sequential Micro-CT studies surviving a mean of 30.6±3.4 days. This data suggests that there is negligible therapeutic effect from the radiation exposure delivered during Micro-CT analysis in the animal model investigated.
S. Daibes, None.
Abstract ID: 246
To design customised nanoparticles for in vivo applications in relation to certain brain diseases require a basic knowledge of the physical and physiological properties in the interaction between the nanoparticles and the brain tissue. We have therefor studied the interaction between the brain tissue and two well-characterised iron oxide/dextran based nanoparticles. We have used a rat model (male Sprague-Dawley rats, 200–220 g) and magnetic resonance imaging (MRI) at a 4.7 T field strength to trace the intracranial localisation and distribution of nanoparticles.
The distribution of nanoparticles was observed at 0.5 h 24 h and 10 days after injection using both a Rapid Acquisition with Relaxation Enhancement RARE protocol and a gradient echo sequence utilising the susceptibility effect of the iron oxide. Two groups with 8 rats were injected with Endorem (Guerbet, France 0.1 mg/0.5 ml, 0.5 ml), or MION (Harvard, USA 0.15 mg/0.5 ml, 0.5 ml) and 4 were injected with artificial mock cerebrospinal fluid (mCSF) as controls. The rats were anaesthetised with Isoflurane in air before the nanoparticles were injection into the right striatum.
At 0.5 h and 24 h the nanoparticles were detected in both groups while at 10 days only Endorem was possible to detect. In both groups a spherical distribution around the injection site was observed but also a distribution along the external capsule.
This study reflects upon the different sized nanoparticles and their distribution in the neuropil and active transport systems of the brain. Previously reported facilitated transport of dextran along myelinated fiber tracts can be traced using preferentially the nanoparticles with larger diameter. The smaller nanoparticles had a shorter lifetime in the tissue probably due to a more efficient transport into the venous system draining the brain.
X. Wu, None.
Abstract ID: 247
The advent of small animal PET imaging instrumentation has allowed the detailed measurement of time-activity data in rodent models of human disease. These instruments also allow longitudinal studies and the use of dual-tracer techniques for measuring multiple processes within the same animal. To investigate limitations of these methods, we performed dynamic FDG scans on a group of athymic mice bearing tumor xenografts of various sizes (50-500 mg). Tumor-bearing animals were anesthetized (isoflurane), weighed, fitted with a tail vein catheter and placed on a microPET R4 (Concorde Microsystems, Inc.) small animal tomograph. FDG was injected via the catheter (0.7-3 mCi in 0.2 uL) and dynamic emission data were collected over 60 minutes (five 2 minute frames, four 5 minute frames and three 10 minutes frames). Volumes of interest (VOI) were drawn manually over the entire tumor and time-activity curves (TAC), calibrated in nCi/cc, were generated. The TAC for each animal was then normalized to the injected dose and animal weight to provide a standardized uptake value (SUV). Comparison of dynamic SUV measurements for mice bearing A-431 human epidermoid carcinomas displayed large variability in both kinetics and magnitude of FDG accumulation. Maximum SUV values ranged from 0.7 to 2.3. This variability in the radioactivity concentration measurement was independent of tumor size (r2=0.01). Results were similar for all tumor cell lines examined. Manual VOI placement was investigated as a possible source of this variability, however multiple VOIs drawn for a single tumor yielded only a 5% variability in SUV data. The results of this study indicate that significant limitations exist when comparing small animal PET tumor data among groups of animals. Factors such as injection rate, scanner dead-time, blood flow or the intrinsic variability of a particular radiotracer may contribute to this phenomenon and should be addressed in the experimental design.
S. Snyder, None.
Abstract ID: 248
Imaging of cerebral perfusion by SPECT is an important approach in evaluating neurological and psychiatric diseases. A lipophilic blood flow marker 99mTc-hexamethyl propyleneamine oxime (99mTc-HMPAO) has been employed to image the cerebral blood flow in human. However, investigating the cerebral perfusion of rodents by in vivo imaging is still a challenge. The purpose of this study was to establish a technique to measure the regional cerebral blood flow (rCBF) in ischemia stroke rats using [99mTc] HMPAO in conjunction with microSPECT, a dedicated SPECT designed for imaging small animals.
Sprague-Dawley rats were underwent bilateral carotids occlusion for 60 min followed by reperfusion. Subsequently, the rats were administrated [99mTc] HMPAO via tail vein. The images of brain were obtained about 75 min after administration of [99mTc] HMPAO with a GAMMA Medica XSPECT (microSPECT). An x-ray computed tomography (CT) was acquired to visual the brain contour. To assess the long-term recovery for surgical rats, the [99mTc] HMPAO microSPECT imaging was performed at 2, 7, and 14 days after surgery. In addition, behavior tests for the rats were performed at the same days. SPECT images displayed significant [99mTc] HMPAO uptake in the cortex region in normal rats, whereas the distribution of [99mTc] HMPAO activity in ischemic stroke rats was significantly decreased in the same region. Longitudinal imaging studies of the surgical rat revealed that the rat was recovered evidently, which was consistent with the behavior findings for the model. The [99mTc] HMPAO microSPECT imaging in modeled animals provides new opportunities in pathological investigations associated with the alteration in cerebral perfusion.
W. Hsu, None.
Abstract ID: 249
[18F]Fluorodeoxyglucose (FDG) positron emission tomography (PET) imaging has been extensively exploited to evaluate glucose metabolism in various cerebral abnormalities in human. In the field of CNS drug research, small animal model is promising for evaluating novel therapeutic or neuroprotecting drugs. Altered energy demand in brain can be measured by FDG high resolution PET scanner (microPET), which is designed specifically for small animal imaging. To provide an approach to study the effect of drugs on CNS, we established a technique to assess the brain metabolic activity by FDG microPET imaging in an acute cerebral ischemia rat model with bilateral carotid occlusion. Sprague-Dawley rats were subjected to bilateral carotid occlusion for 1 hr followed by reperfusion. And then the rats were administrated FDG via tail vein immediately. The images were obtained from 10~60 min after administration of FDG with a Concord R4 microPET. For long-term recovery study, the FDG microPET imaging was performed at 7, 17 and 22 days after surgery.PET images displayed significant FDG uptake in the cortex, striatum and thalamus region in normal rats, whereas the distribution of FDG activity in cerebral ischemic rats was decreased significantly (p<0.01), which was consistent with the behavior findings for the model. Longitudinal studies of the recovery for bilateral carotid occlusion rats revealed that the rats recovered around 80 % of glucose utilization at 22 days after bilateral occlusion. The modeled animals with microPET imaging provide new opportunities in CNS drug development for assessment of alteration in neuronal activity.

Assessment of glucose metabolism in rat brain by using FDG imaging. A. Normal rat; B. Rat with bilateral carotid occlusion. The images were acquired at the first day after surgery.
W. Hsu, None.
Abstract ID: 252
Genetically engineered mouse models offer the opportunity to study how specific molecular lesions collaborate in the development of the mammary glands, mammary preneoplasia and cancer. Ultrasound can demonstrate the normal mammary gland structures, detect preneoplastic hyperplastic alveolar nodules, and mammary cancers. The lead author's laboratory has demonstrated that gain of ERα (references), gain of AIB1 (reference) or loss of Stat5a function (reference) alter mammary cancer progression in mouse models. Twenty female mice from four different oncogenic transgenic backgrounds and 4 control mice were studied. For ultrasonography hair overlying all ten mammary glands and the abdomen was removed by depilatory treatment and the mice were anesthetized. Digital ultrasound images of all ten mammary glands and liver were obtained at 55MHz. One group of mice was euthanized immediately following ultrasound examination and lesions found on ultrasonography correlated with histological studies. In a second group of mice, serial imaging and physical examination were used to follow the lesions. To date we found that preneoplastic mammary lesions as small as 0.3 mm3 were detectable by ultrasonography, growth of mammary adenocarcinomas can be followed, lesions with necrosis can be discriminated from those without, and liver metastases identified.
These images will be incorporated into a mouse mammary gland ultrasound atlas of normal and abnormal structures.
M. Freedman, None.
Abstract ID: 253
Although it is commonly assumed that intestinal carcinomas arise from adenomas, it is uncertain when adenomas appear or how long they persist before transformation. A non-invasive imaging technique could help answer these questions through serial examinations of the same mouse. We are evaluating small-animal PET and F-18 fluorodeoxyglucose (FDG) for that purpose using mice with heterozygous mutations in the Apc gene (Min mice) that develop adenomas in the small intestine.
J.R. Bading, None.
Oncogenesis, Angiogenesis & Disease Progression
Abstract ID: 255
The tyrosine kinase receptor Met and its ligand hepatocyte growth factor/scatter factor (HGF/SF) play important roles in normal processes, tumorigenicity, and metastasis. We have generated transgenic mouse lines expressing functional GFP-Met. The distribution of GFP-Met was determined using a Zeiss LSM510 confocal microscope, spectral analysis with a Meta detector and image analysis.
In Vitro subcellular localization of GFP-Met demonstrated cell membrane localizion. Intravital imaging of GFP-Met molecules in the transgenic mice revealed expression in epithelial and endothelial tissues. Fluorescence was enhanced in sebaceous glands. Single cells within the sebaceous gland expressing GFP-Met on their surface as well as the 3D patterns of GFP-Met expression were imaged in live mice. HGF/SF injection into live animals showed rapid GFP-Met internalization in sebaceous glands. Intravital FRAP analysis of GFP-Met in sebaceous glands showed membrane mobility rates similar to the GFP-Met mobility observed in 293T cells. Male GFP-Met transgenic mice spontaneously develop sebaceous gland tumors in their lower abdominal area adjacent to the gonads. Tumor transplantations into nude mice were successful only when transplanted adjacent to the gonads in male mice, suggesting hormonal dependency. Intravital imaging of the primary sebaceous tumors reveals that single epithelial cells that express high GFP-Met levels surround the tumor. These cells may be precursors from which new local metastasis develop. Functional molecular imaging of these tumors using ultrasound contrast media demonstrated increasing signal intensity upon HGF/SF injection which correlated with high levels of GFP-Met expression.
High-resolution intravital molecular imaging of GFP-Met enables us to image primary events of tumorigenesis and metastasis in a single cell resolution, showing that molecular imaging is a powerful tool in cancer research and diagnosis. This work was supported in part by NIH research grant (P50CA93990).
S. Moshkovitz, None.
Abstract ID: 256
The chemokine receptor CXCR4 and its cognate ligand CXCL12 recently have been proposed to regulate characteristic patterns of breast cancer metastases to organs including lung, liver, lymph nodes, and bone marrow. However, effects of CXCR4 on growth of primary breast cancer tumors and established metastases, as well as overall survival, have not been determined. We used RNA interference to reduce CXCR4 expression and signaling in murine 4T1 cells, a highly metastatic mammary cancer cell line that is a model for stage IV human breast cancer. Using bioluminescence and magnetic resonance imaging to monitor tumor progression, we showed that reduced expression of CXCR4 significantly limited growth of orthotopically-transplanted breast cancer cells. Parental 4T1 cells produced progressively enlarging primary tumors that spontaneously metastasized to multiple organs, invariably resulting in death of mice from metastatic disease. By comparison, RNA interference of CXCR4 decreased primary tumor growth rates in all mice and prevented primary tumor formation in some recipient mice. Furthermore, all mice transplanted with CXCR RNA interference cells survived without developing macroscopic metastases. To analyze effects of CXCR4 on lung metastases, an organ commonly affected by metastatic breast cancer, we produced experimental lung metastases by intravenous injection of tumor cells and monitored disease progression with bioluminescence imaging. RNA interference targeted to CXCR4 or treatment with the specific CXCR4 antagonist AMD3100 significantly delayed initial proliferation of 4T1 cells in lung, although sustained inhibition of tumor cell growth only occurred with RNA interference. These data indicate that CXCR4 is essential for initial proliferation of breast cancer cells in vivo and suggest that CXCR4 inhibitors may improve treatment of patients with primary and metastatic breast cancer.
K.E. Luker, None.
Abstract ID: 259
Cell adhesion receptors of the integrin family, which are responsible for a wide range of cell-extracellular matrix (ECM) and cell-cell interactions, have been well studied in many tumor types, but the function of integrins in lung cancer growth and metastasis is not well understood. In this study we would like to visualize integrin αvβ3 expression in an orthotopic non-small cell lung cancer (NSCLC) tumor xenograft model using microPET and 64Cu-labeled RGD peptide antagonist of αvβ3. The radiotracer 64Cu-DOTA-PEG-E[c(RGDyK)]2 with a heterofunctional poly(ethylene glycol) (PEG, M.W. = 3,400) linker demonstrated intermediate receptor binding affinity for αvβ3 integrin expressed on NCI-H1975 NSCLC lung cancer cells (IC50 = 84.9 nM). The distribution of radioactivity in subcutaneous tumor model revealed rapid blood clearance and excellent tumor-to-background ratios at all time points examined. The receptor specific tumor uptake reached maximum at 1h p.i. (2.64±0.14 %ID/g) with some washout with time elapsed (e.g. 1.15±0.07 %ID/g at 24h p.i.). We also applied the same radiotracer to orthotopic lung cancer model (6 wk-old SCID-bg mice implanted with 2 million NCI-H1975 cells in the left lung upper lobe) for tumor visualization. The primary tumor was clearly delineated by both FDG and 64Cu-DOTA-PEG-E[c(RGDyK)]2, but the RGD peptide tracer showed significantly higher tumor-to-lung contrast as that of FDG. In particular, RGD was also able to detect mediastinal metastasis and the invasion the lung on the right, FDG uptake in these areas was masked by intense cardiac uptake. Coronal slices and corresponding autoradiography clearly visualized both primary tumor and metastases to the mediastinum, the other lung, and diaphragm with high tumor-to-lung contrast (funding support: ALA California, NIBIB EB001785, DOD BCRP DAMD17-03-0752 and BC030012).

X. Chen, None.
Abstract ID: 260
The growth and metastasis of the majority of tumors depends on the formation of new blood vessels or angiogenesis. Emerging evidence indicates that bone marrow derived circulating endothelial progenitor cells (EPC) can contribute to the angiogenesis. Until recently there was no direct method to image the incorporation of endothelial cells into the neovasculature of tumors by MRI. In this study we attempted to image angiogenesis in a subcutaneous mouse flank tumor model using ferumoxides labeled human EPC (AC133+). AC133+ cells, a subpopulation of CD34+ hematopoietic stem cells obtained from peripheral blood leukapheresis, were labeled with ferumoxides-protamine sulfate (FePro) complex, washed, and injected intravenously (IV) in C6 tumor bearing nude mice. One group of mice received simultaneous implantation of tumor and IV administration of labeled cells (group 1), and other group received IV administration of labeled cells when the implanted tumor grew to 0.2 cm in size (Group 2). Both groups had control mice with injected unlabeled cells. MR imaging was performed at different stages of tumor growth from 0.5 cm to 1.5 cm in sizes. After in-vivo MRI, mice were euthanized and perfused with 4% paraformaldehyde, and tumors were collected for high-resolution ex-vivo MRI followed by Prussian blue and immunohistological staining. Some mice in both groups were also injected with FePro-Quantum Dot double-labeled cells for confocal microscopy. Both MRI and histology showed incorporation of iron positive cells at the periphery of the tumors by 3–5 day and most of the incorporation was observed at tumor size of 1 cm. Confocal microscopy showed incorporation of labeled cells in the vessels of the tumors. Immunohistochemistry showed expression of endothelial markers in the iron positive cells. Detection of magnetically labeled EPC in implanted flank tumor vasculature was observed on in vivo and ex vivo MRI.
A.S. Arbab, None.
Abstract ID: 261
VEGF is an essential angiogenic factor in tumor vascularization. Increased expression of VEGF is related to cancer aggressiveness and metastasis. To investigate the role of VEGF in cancer cell invasion and metastasis, we generated stable clones of VEGF-A overexpressing human MDA-MB-231 breast cancer cells. 1H and 31P nuclear magnetic resonance (NMR) spectroscopy were employed to determine the effects of VEGF-A overexpression on choline phospholipid metabolism, since increased phosphocholine (PC) and total choline (tCho) are associated with increased malignancy.
MDA-MB-231 human breast cancer cells were stably transfected with VEGF-A construct or empty-vector as control. MDA-MB-231 wild type (231-wt), MDA-MB-231 clones overexpressing VEGF-A (231-VEGF) or empty-vector controls (231-vector) were inoculated into SCID mice and perchloric acid extracts were obtained from those tumors. 1H and 31P NMR spectroscopy of the tumor extracts were performed on a Bruker Avance 500 NMR spectrometer. Enzyme-linked immunosorbent assays (ELISA) were performed to determine the expression level of human VEGF in the solid tumors.
ELISA of the solid tumors revealed increased VEGF levels in 231-VEGF tumors compared to 231-vector tumors and 231-wt tumors (2.7 fold and 1.9 fold, respectively). Solid 231-VEGF tumors exhibited significantly increased PC (35% increase) and significantly decreased GPC levels (36% decrease) compared to 231-vector tumors. tCho levels remained unchanged by VEGF overexpression in solid MDA-MB-231 tumors. 31P NMR spectra also showed higher level of PC in 231-VEGF tumors compared to 231-wt tumors. In 31P NMR spectra, GPC levels were reduced in 231-VEGF tumors compared to 231-vector tumors, but not in 231-wt tumors. Our data demonstrate that an increase in VEGF-A levels in solid breast tumors may, in part, cause the elevated PC and decreased GPC levels typically detected in breast tumors. This work was supported by P50 CA103175. We thank Genentech for the VEGF-A construct and Mr. Cromwell and Mrs. Wildes for technical support.
N. Mori, None.
Abstract ID: 262
J. Wolter, None.
Abstract ID: 263
T. Persigehl, None.
Abstract ID: 264
K. Licha, None.
Abstract ID: 265
Cancer growth, invasion and metastasis are highly related to tumor-associated neovasculature. Angiogenic molecular targeting is clinically relevant and an ideal goal for cancer treatment. Due to continuous tumor growth and high interstitial pressure, the presence and progression of angiogenic endothelial cells in cancer is chaotic, unorganized, and the angiogenic vessels are less functional. Therefore, not all markers appearing on the chaotic endothelial cells are assessable if the drug was given intravenously. Identifying endothelial cell markers from those functional cancer angiogenic vessels will indicate the accessibility and efficacy of vascular targeted therapies. In order to quickly and effectively identify endothelial cell markers on the accessible and functional tumor vessels, here we developed a novel technique by which tumor angiogenic vessel systemic in vivo labeling was combined with Laser Capture Microdissection of microscopically isolated endothelial cells from a heterogeneous matrix for genomic and proteomic screening. Female C3H mice with established SCC-7 tumors were treated with Rhodamine-RCA by tail vein injection, and after the fluorescence microscope showed a successful vasculature staining, LCM was performed on frozen section tissue using the PixCell II instrument with CapSure HS caps under the Rhodamine filter. By this approach, the fluorescent angiogenic endothelial cells were successfully picked up. The presence of lectin labeled endothelial cells on the “cap” image and the absence of the targeted cells in the “after” microdissection image was overlapped. RNA was extracted from approximately 250 micro-dissected endothelial cells and followed by two-round amplification. The BioAnalyser spectra clearly indicated the broad amplification with high RNA quality. RT-PCR and gene electrophoresis successfully detected CD31 and β-Actin molecules without Keratin 19 expression, which served as the negative control. Thus, in vivo Rhodamine RCA angiogenic vessel labeling provided a practical approach to effectively guide functional endothelium epithelial cell isolation by laser capture microdissection for genomic and proteomic assay.
F.K. Hunter, None.
Abstract ID: 266
Plasminogen activator inhibitor-1 (PAI-1) regulates urokinase PA activity and, thereby, pericellular plasmin generation. PAI-1 affects cell motility in vivo and in vitro by controlling both the extent/localization of matrix barrier proteolysis and cell-to-matrix adhesion mediated by integrins and the urokinase receptor. To evaluate relationships between PAI-1 expression and cellular migratory activity, 806 bp of the human PAI-1 promoter were cloned upstream of an insert encoding a PAI-1-GFP chimeric protein and induced PAI-1-GFP visualized in situ by fluorescence microscopy. Gene activation and subsequent deposition of PAI-1-GFP into keratinocyte migration tracks was assessed using a planar (2-D) scrape wound assay to initiate PAI-1 transcription and cell motility. Comparisons of PAI-1 chimera expression with that of the endogenous PAI-1 gene (at the mRNA/protein levels) revealed similar inductive kinetics indicating that the PAI-1-GFP construct provided an easily accessible “reporter” system to evaluate PAI-1 promoter function within the real-time of gene activation. PAI-1 synthesizing cells were highly motile in the both the scrape wound and 3-D Matrigel invasion models of induced migration. Stimulated PAI-1 transcription reflected recruitment of helix-loop-helix factors of the USF family to a defined E box (CACGTG) motif in the proximal promoter of the PAI-1 gene. Motile rates of PAI-1-GFP transfectants were significantly greater than control GFP-only transfectants under the respective culture conditions. Both responses could be attenuated by incubation with PAI-1 neutralizing antibodies or by secondary transfection with PAI-1 antisense constructs confirming the importance of PAI-1 in cell locomotion. PAI-1 protein synthesis and 3-D migratory activity declined significantly in keratinocytes transfected with dominant-negative USF constructs. These findings illustrate the usefulness of this approach to image transcriptional and invasive responses in single living epithelial cells using GFP reporters under control of promoter sequences from genes implicated in migratory activity. (Supported by grants from the NIH and the Department of the Army).
P.J. Higgins, None.
Abstract ID: 267
Endothelial cells in the angiogenic vessels, surrounding the tumor tissue, are known to express several makers, being either barely detectable or entirely absent in normal blood vessels. Amongst them, αvβ3 integrin has received significant interest since its level of expression in the tumor vasculature correlates with the grade of malignancy, which may provide the opportunity to destroy tumor vessels by drug targeting without affecting the microvasculature of normal tissues. In recent years, it has been demonstrated by in vivo studies that peptide antagonists of αvβ3 integrin inhibit the tumor growth by promoting apoptosis of the angiogenic vascular cells. In an attempt to develop the effective peptide delivery system for the inhibition of angiogenesis, we herein prepared HGC-based self-aggregates loaded with peptides containing RGD sequence, specifically binding to αvβ3 integrin expressed on endothelial cells. HGC capable of forming nano-sized self-aggregates was prepared by the chemical conjugation of 5β-cholanic acid to the main backbone of glycol chitosan. The RGD peptide labeled with fluoresein isothiocyanate (FITC-GRGDS) was loaded into self-aggregates in three different conditions: simple mixing, sonication, and solvent evaporation methods. The release behavior of FITC-GRGDS in physiological solution was observed. The biological activity of FITC-GRGDS was evaluated from cell adhesion assay using human umbilical vein endothelial cells (HUVECs). To verify whether FITC labeling of peptides affects biological activity or not, the cell adhesion assay was performed. Overall, there were no evidences that biological activity of peptides was significantly deteriorated by FITC-labeling. In other words, the number of adhered cells on the surface coated with βig-h3 proteins, being known to mediate endothelial cell adhesion by the specific interaction with αvβ3 integrin, decreased by the treatment with FITC-GRGDS in a dose-dependent manner. Self-aggregates loaded with FITC-GRGDS might be useful for monitoring, imaging, or destroying the angiogenic vessels surrounding the tumor tissue.
R.V. Jacko, None.
I. Kwon, None.
Abstract ID: 268
Most Amp, KEP, and KEL values varied 24%-92%, 21%-140%, and 38%-210% in average, respectively, across tumor layers. Amp and KEP have generally decreased after treatment for the entire tumor, the inner layers, and outer layers. While assessing response to therapy, a reduction in heterogeneity coincides with biologic effectiveness.
Abstract ID: 269
Multidrug resistance (MDR) remains a major obstacle to successful chemotherapeutic treatment of cancer and can be caused by overexpression of P-glycoprotein (Pgp), the MDR1 gene product. To further explore a molecular genetic approach circumventing MDR, we developed a Pgp inhibition strategy using retroviral-mediated RNA interference (RNAi). Two of eight tested RNAi constructs targeted against MDR1 mRNA inhibited expression of Pgp by >95%, whereas control RNAi had no effect. Ablation of Pgp was documented by Western blot analysis and substantially increased sensitivity toward the cytotoxic drugs vincristine, paclitaxel and doxorubicin. Pgp transport activity can be non-invasively monitored with bioluminescence, because coelenterazine, the imidazolopyrazine substrate for Renilla luciferase (Rluc), shares structural properties of other compounds transported out of cells by Pgp. Using Rluc transfected HCT-8 cells and bioluminescence, we directly monitored RNAi-mediated downregulation of MDR1 in Pgp-expressing cells and in living mice. Results show that HCT-8 cells with RNAi against MDR1 mRNA have ~80% increased light output when compared to control cells. Similar effects were also detected in mice with tumors of HCT-8 control and HCT-8 RNAi cells. Thus, coelenterazine can be used to image RNAi-mediated downregulation of Pgp in intact cells and living animals.
A. Pichler, None.
Abstract ID: 270
Vascular endothelial growth factor (VEGF) is frequently upregulated in hypoxic tumors, and has been linked to increased cancer metastases [1]. We investigated the effects of VEGF overexpression on invasion and metabolism of PC-3 cells co-cultured with human umbilical vein endothelial cells (HUVECs) under oxygenation or hypoxia, using our Metabolic Boyden Chamber assay [2]. PC-3 cells overexpressing human VEGF165 were generated using human pHuVEGF.21 (Genentech) ligated into the expression vector pCR3.1. VEGF overexpression did not affect invasion under well-oxygenated conditions. Under continuous hypoxia, the decreased invasive capacity of parental PC-3 cells was reversed by VEGF overexpression leading to invasion comparable to well-oxygenated cancer cells. These results suggest that in regions of vascular collapse characterized by hypoxia in the presence of endothelial cells, VEGF may increase invasion of cancer cells.
Under hypoxia, nucleoside triphosphates remained constant and intracellular phosphocreatine increased in VEGF overexpressing as well as wild-type cells, while intracellular mobile lipids decreased only in overexpressing cells. These results suggest a shutdown of energy-consuming processes in favor of maintaining nucleoside triphosphate levels under hypoxia [3]. References: [1] L.M. Ellis, I.J. Fidler; EurJCancer 32A(14):2451-60, 1996; [2] U. Pilatus, et al.; Neoplasia 2(3):273-79, 2000; [3] R.G. Boutillier; JExpBiol 204:3171-81, 2001

Invasion Index (mean±SE) of wild-type PC-3 and the VEGF-overexpressing clone PC-3VEGFc2, in the presence of HUVECs under oxygenation or hypoxia.
E. Ackerstaff, None.
Abstract ID: 271
Y. Tang, None.
Abstract ID: 272
Vascular endothelial growth factor (VEGF) mediated angiogenesis has been shown to be important for growth of solid tumors and is mediated by VEGF binding to VEGFR on vascular endothelial cells. Development of molecular imaging agents able to probe the extent of tumour angiogenesis could be valuable to assess the efficacy of antiangiogenic therapy. Our goal was to construct, express and purify a novel fusion protein: hnTf-VEGF165, with which human VEGF165 isoform was fused through a flexible peptide linker [GGGGS]3 to N-lobe of human transferrin. N-lobe hnTf allows direct high-affinity complexation of 111In at a site remote from the VEGFR-binding domain without the need to introduce metal chelators. DNA coding for hnTf and VEGF165 were cloned into Pichia pastoris expression vector pPICZαB which contains α-factor peptide secretion signal sequence at N-end of the hnTf-VEGF fusion protein to direct secretion of fusion protein into medium, and a 6× His tag at the C-terminal of the fusion protein for detection and purification. The pPICZαB-hnTf-VEGF plasmid was transformed into Pichia pastoris yeast KM71H and protein expression was induced by methanol. The fusion protein was purified from medium under native conditions using Co2+ metal affinity resin. SDS-PAGE gel under reducing and non-denaturing conditions showed the size of hnTf-VEGF was about 65 kDa and 130 kDa respectively, indicating the fusion protein existed as a native dimer (Mr 130 kDa) in solution. In vitro studies including flow cytometry analysis and endothelial cell proliferation assay showed biological activities of VEGF domain of the fusion protein were retained while the fusion protein was able to bind 111 In up to a specific activity of 2.65 GBq/mmol. Biodistribution and imaging studies in athymic mice implanted with s.c. U87MG glioblastoma xenografts revealed specific tumour uptake (6.68% i.d./g) which was decreased 15-fold by administration of an excess of unlabeled VEGF.
C. Chan, None.
Abstract ID: 273
High resolution micro-imaging tools have enabled non invasive visualization of tissue remodelling in longitudinal studies of the mouse. In mice, the hyaloid vasculature of the eye degenerates from a well defined structure at birth via progressive loss of branching structure and parallels retinal vascular maturation. Until the present it has not been possible to study ocular vascular patterning noninvasively, to which high frequency ultrasound microimaging (UBM) is well suited. In this study, flow waveforms were measured in the hyaloid artery (HA), vasa hyaloidea propria (VHP), tunica vasculosa lentis (TVL), and retina in CD-1 mice for the first 16 days postnatally. The LHbTAg mouse model of retinoblastoma was also assessed from birth to 4.5 months. Ultrasound imaging and Doppler were performed with a Vevo 660 (VisualSonics, Toronto) mouse scanner. Contrast enhanced MicroCT was used to image vessel structure (MS8 scanner, EVS, London Ontario). Normal mice showed marked remodeling of the hyaloid vasculature over the first 16 days after birth. Peak flow velocities in the hyaloid artery dropped from 3.0 ± 1.0 cm/s to 0 between P0 and P13 with similar regression patterns in the VHP and TVL. No flow or hyaloid vascular structure was observed in CD-1 mice after P13. Flow in the retinal vasculature showed an inversely proportional increase from 0.3 ± 0.05 cm/s to 4.1 ± 0.6 cm/s over the same period. In the retinoblastoma model, intraocular tumours were visible at 2 months and by 4.5 months had filled the vitreous cavity. The blood supply for these tumours appears to be derived from a persistent and significantly augmented hyaloid vasculature. Functional flow patterns of the hyaloid vasculature appear to be inversely proportional to flow in the retinal circulation. Ocular tumours in a spontaneous model of retinoblastoma appear to recruit and expand the hyaloid vasculature to sustain aggressive growth.
A.S. Duckett, None.
Abstract ID: 274
Breast cancer cells invade the extracellular matrix and metastasize by utilizing different types of proteases. Lysosomes may play a major role in breast cancer invasion and metastasis, because they mediate protease routing, regulation, and secretion. The pathophysiological microenvironment of tumors, frequently characterized by extracellular acidity, may affect lysosomes. Therefore, we characterized the total lysosomal volume in highly invasive/metastatic (MDA-MB-231, MDA-MB-435) and rarely invasive/metastatic (MCF-7) human breast cancer cells, as well as normal (MCF-12A) human mammary epithelial cells (HMECs) under acidic extracellular conditions. Immunofluorescence staining of lysosomes was performed using anti-lysosome-associated-membrane-protein antibody and confocal laser-scanning fluorescence microscopy. Image analysis software was used to detect lysosome boundaries and counterstained nuclei using a boundary detection algorithm. This software produced datasets containing a histogram of the lysosome-to-nucleus distance for all lysosomes in a cell, the lysosome diameter distribution per cell, and the total number of lysosomes per cell. Lysosome diameters and numbers were utilized to calculate total lysosomal volume per cell as shown in the figure.
Extracellular acidification resulted in a significant increase in total lysosomal volume in both highly invasive/metastatic breast cancer cell lines (Fig.). In rarely invasive/metastatic breast cancer cells and HMECs, a significant decrease in total lysosomal volume was detected following extracellular acidification (Fig.). Thus, the parameter ‘total lysosomal volume’ was able to discriminate non-invasive and highly invasive/metastatic breast cancer cells at low extracellular pH. This is a promising parameter that can be exploited for developing imaging techniques to predict metastatic disease in breast cancer. The extracellular acidity typically found in tumor microenvironments may affect invasion and metastasis. Our research may also open up new avenues for treating metastatic disease by disrupting lysosomal trafficking. Support: P50CA103175.

Total lysosomal volume per cell in cubic micrometer at pH 7.4 (control), pH 6.8, and pH 6.4 in an HMEC line (MCF-12A) and three breast cancer cell lines representing different stages of breast carcinogenesis (MCF-7, MDA-MB-231, MDA-MB-435). Data were obtained by analyzing immunofluorescence stainings using our in house software.
K. Glunde, None.
Abstract ID: 275
Here, we report the effects of VEGF-A165 expression on the tumor vasculature and lymphangiogenesis, using MRI and fluorescent microscopy. Overexpression of VEGF-A165 in a human breast cancer model (MCF-7) resulted in drastic alterations of its vascular phenotype, i.e. there was a significant increase in vascular volume (

A.P. Pathak, None.
Abstract ID: 276
B. Shen, None.
Abstract ID: 277
Urokinase plasminogen activator (uPA) plays an important role in tumor invasion and metastasis. Direct monitoring of uPA activity in a whole animal can provide valuable information for tumor biology and treatment development. We previously have developed an optical probe which is sensitive and specific to urokinase in vitro. In currect study, we extended its application for in vivo enzyme activity imaging using human fibrosarcoma (HT-1080) xenographic model. The animals were imaged using a reflactance near-infrared (NIR) fluorescent imaging system at different time points. As early as 3 hours after administration of the probe, all animals (n = 8) showed significant fluorescence contrast at the tumor sites. The observed fluorescence emissions are significantly higher (p<0.01) when compared with others (n = 7) receiving the scrambled control probe. And the NIR fluorescence singal was found correlated well with urokinase expression by histological analysis. We conclude that tumor-accociated urokinase activity can be detected in vivo sensitively and specifically by using this novel molecular probe.
J. Hsiao, None.
Abstract ID: 278
Many different assays have been developed and used by researchers to understand angiogenic mechanisms and to evaluate the efficacy of novel angiogenesis inhibitors. However, many in vivo angiogenesis assays are expensive and technically difficult to perform. Even minimally invasive optical windows can result in tissue injury and alter the tissue microenvironment. The aim of our experiments was to perform non-invasive, yet simple and reproducible system for direct optical imaging assessment of angiogenesis in small developing tumor over time.
GFP fluorescence showing RG2TG xenograft and developing vessels

White light image of the same xenograft.
T.A. Beresten, None.
Abstract ID: 279
For successful antiangiogenic therapy, it is essential to understand the endothelial cell response to angiogenic factors. We describe an assay to observe the effects of angiogenic factors secreted by MDA-MB-231 cells on the network formation, invasion and migration of Human Umbilical Vein Endothelial Cells (HUVECs) labeled with a T2 MR contrast agent (FeridexI.V.™).
HUVEC labeling: Prior to seeding, HUVECs (Clonetics, USA) were incubated for 24 h with 9 μg FeridexI.V.™ and 0.125 μg Poly-L-lysine per ml of EGM-2. Chamber preparation: The assays were housed in non-tissue-culture inserts with 0.4 μm membrane pore size, and layered with i) 120 μl of ECM gel, ii) 1.5 × 105 MDA-MB-231 cells suspended in 100 μl EGM-2; 100 μl EGM-2 was added to the control chambers, iii) 200 μl of ECM gel, and iv) 1.5 × 105 labeled HUVECs. MR Imaging: MR images were obtained on an 11.74 T system with a Bruker Avance spectrometer, using a T2-weighted spin-echo sequence.

Representative axial Images of a 0.5 mm slice containing MDA-MB-231 cells (grey; central), showing increased presence of FeridexI.V.™-labeled HUVECs (dark; peripheral), evident from the deepening signal loss on days 4 and 6 compared to day 2 post seeding. The images were obtained using a T2.weighted spin-echo sequence with a field-of-view = 1.6 cm, acquisition matrix = 256 × 256, TR = 857 msec, TE = 90 msec, number of averages = 2. FerldexI.V.™ labeling appeared to have no detrimental effect on the survival or morphogenesis of HUVECs over the period of observation, as was concluded from phase contrast light micrographs, MR Images, and verified by MTT assay. The chambers were checked for cell presence and structure by phase contrast light microscopy and by fluorescence microscopy of HUVECs stained with a fluorescent CD31 monoclonal antibody.
HUVECs in chambers containing MDA-MB-231 breast cancer cells traversed the ECM gel toward the cancer cells for up to 6 days whereas HUVECs in control chambers did not traverse the gel. We have developed an assay to study endothelial cell response to paracrine factors secreted by cancer cells. This assay will be useful to study the effects of angiogenic and growth factors on endothelial cell tubulogenesis, migration, and invasion.
We thank Dr. Dmitri Artemov for technical assistance.
We acknowledge support from NIH 2RO1 CA 82337, NS045062 and NIH 1P50 CA 103175.
B. Gimi, None.
Abstract ID: 280
Our previous work in gene therapy of lung cancer allowed us to identify how important it is to follow in real time and in a non-invasive manner the biodistribution and the therapeutic activity of gene therapy, the presence of deep tumours like lung metastasis but also the activity of anti-angiogenic treatments.
Here, we report the in vitro and in vivo features of a new molecular conjugate vector leading to hitherto unprecedented properties for neo-vascular targeting, imaging and therapy of tumours. This vector is composed of two independent functional domains tethered by a cyclodecapeptide platform: a cell targeting domain based on a clustered RGD-ligand for αVÎ23 integrin recognition, and a labelling or drug delivery domain. In addition to a detailed analysis of RAFT(cRGD)4 properties in vitro, we demonstrate that compared to the cognate cRGD peptide, RAFT(cRGD)4 dramatically improves the targeting specificity of subcutaneous tumour masses as well as disseminated metastasis after intravenous injections. This was performed using an in house 2D fluorescent imaging system, coupled to a bioluminescence apparatus.

J. Coll, None.
Abstract ID: 281
Paola A. Erba1, Rossella Di Stefano2, Elena Lazzeri1, Daniele Barone2, Giampiero Giovacchini1, Tatiana Santoni2, Paola Collecchi3, Alberto Balbarini2, Giuliano Mariani1. 1University of Pisa Medical School, Nuclear Medicine Department, Italy; 2University of Pisa Medical School, Angiology Unit, Cardiothoracic Departmen, Italy; 3University of Pisa Medical School, Pathology Department, Italy. Contact e-mail:
Endothelial progenitor cells (EPCs) can contribute to neovascularization of ischemic tissue. Current techniques evaluating EPCs biodistribution require tissue biopsies or animal sacrifice for immunohistochemical analysis, with obvious limitations. Therefore, noninvasive imaging approaches for monitoring EPCs distribution are desirable. Aim of the work is the evaluation of 111In-oxine labelling and biodistribution in normal mice and in animal models of induced peripheral muscle ischemia and inflammation. Human EPCs were isolated from peripheral blood, characterized and labeled with 111In-oxine and tested for cell viability before i.v. injection into male balb-c mice (3.7 MBq, groups of 5 animals). EPCs biodistribution was evaluated 2, 8 and 24 hr after injection into normal mice, at 8 and 96 hr in ischemic and inflammed mice (72 hr after ischemia/inflammation induction). Animals were sacrificed and organs counted for radioactivity. Results are expressed as %ID/g and as ischemic or inflammed to contralateral muscle ratios (I/C). High radioacctivity accumulation was found in lungs, spleen, liver and bladder soon after injection. Radioactivity in the kidneys, thyroid and stomach increased slowly reaching a maximum at 96 hr. No radioactivity was detectable in bone marrow. A consistent pattern of overall tissue biodistribution was observed in normal, ischemic and inflamed mice up to 8 hr, with an average 1.32 %ID/g in the normal muscle. Both the ischemic and the inflammed muscle accumulated significantly more radioactivity (2.21 and 2.70 %ID/g, respectively). Histologic sections were analysed showing high expression of endothelial markers in both ischemic and inflammed muscles. These results confirm that tissue ischemia is a major determinant for local homing of EPCs. Inflammation seems to reproduce the same pattern of EPCs homing, probably because of a similar pathophysiologic mechanism in the early induction of ischemia. Radioactive labelling of EPCs provides a noninvasive approach suitable to assess tissue biodistribution of EPCs and a possible basis for imaging.
P.A. Erba, None.
Abstract ID: 282
We have developed a novel tumor targeting fusion protein consisting of the anti-carcinoembryonic (CEA) diabody fused to the bioluminescence protein Renilla luciferase (hRLuc). The genetically engineered anti-CEA T84.66 diabody, a dimer of the single-chain Fv, has been previously radiolabeled and used for positron emission tomography (PET) imaging of athymic mice bearing CEA-positive LS174T xenografts. We will perform similar in vivo experiments with our fusion protein, Db-hRLuc, using optical imaging with the cooled charge-coupled device (CCD) camera. Genes encoding the 25 kDa anti-CEA diabody and the 36 kDa hRLuc were fused via a 5 or 18 amino acid linker. Due to the unstable nature of hRLuc at 37°C, we explored two bacterial expression systems, a variety of induction conditions, codon optimized host bacteria, as well as mammalian expression. Ultimately, using e.coli BL21 cells with lactose as an inducer has produced the greatest amount of intact Db-hRLuc. In addition, a cysteine to alanine substitution resulted in an increase in the stability similar to that seen by previous investigators. Purification was performed using metal affinity chromatography. Bio-luminescence ELISA assays have demonstrated the fusion protein can simultaneously bind to recombinant CEA and oxidize coelenterazine to emit light. Western blots using antibodies specific for both the anti-CEA diabody and hRLuc identified the same protein band corresponding to a molecular weight of 61 kDa, as well as smaller bands representing partially clipped protein. Stability studies demonstrate Db-hRLuc retains 43% of its bioluminescence activity in mouse serum after 4 hours at 37°C. Mammalian secretion also yielded 61 kDa fusion protein visualized by western blotting. This work demonstrates the feasibility of moving this novel tumor targeting fusion protein into an in vivo animal model using athymic mice with CEA-positive xenografts.
K. Venisnik, None.
Abstract ID: 283
Long and short term effects resulting from exposure to ionization radiation (IR) continue to be documented in the scientific literature. Our laboratory has identified clusterin (CLU) as an IR inducible protein/transcript. Secretory clusterin (sCLU), the fully processed, glycosylated form of this protein has been shown to be induced at IR doses as low as 2cGy in in-vitro experiments. This protein is known to provide cytoprotection after various cell stresses, including IR, due to its role as molecular chaperone. However, sCLU is a late-induced gene after IR, with weak promoter activity, and having transcript and protein levels occurring 48–72 hours after exposure. To monitor promoter activity in-vivo, we have developed a transgenic mouse model that incorporates 1403 base pairs of the sCLU promoter tagged with the firefly luciferase reporter gene. This model enables us to perform real-time bioluminescent imaging on the animal after injecting them intraperitoneally (i.p.) with the substrate, luciferin. Basal level promoter activity is negligible in these mice. Whole-body radiation resulted in the upregulation of clusterin levels in the intestinal tract and the femur, 72h post IR treatment. To ensure all the light emitted has been captured, the mice were shaved before imaging. Furthermore, to examine clusterin activity ex-vivo, the animals were sacrificed and their individual organs were then isolated in well plates. After treatment with luciferin, bioluminescent activity was observed in the colon, small intestine and the bone marrow. Western blot analyses of tissue samples also confirmed these findings. These results correspond with the first phase of radiation exposure damage. Our data suggests that sCLU can aid as a biological marker of genetic instability caused by exposure to low levels of low or high Linear Energy Transfer (LET) IR.
L. Sampath, None.
Abstract ID: 284
Elevated phosphocholine (PC) and total choline (tCho) levels, typically detected by 1H magnetic resonance spectroscopy (MRS) of cancer cells, are linked to malignant transformation, invasion, and metastasis. Increased choline kinase (ChK) expression and activity may cause the increased PC and tCho levels in breast cancers, since a correlation between increased ChK activity and high tumor grade has been demonstrated. Thus, ChK may be a unique target in breast cancer cells. To understand the role of ChK expression and PC levels in breast cancer invasion and metastasis, we generated stable clones of highly metastatic human MDA-MB-231 breast cancer cells expressing small interfering RNA specific for ChK (siRNA-chk) to permanently reduce their ChK levels. ChK mRNA and protein levels were characterized by quantitative reverse transcription-polymerase chain reaction (Q-RT-PCR) and Western Blot analysis using anti-ChK antibody, respectively. PC and tCho levels were measured using 1H MRS. MDA-MB-231 clones chk23 and chk25 stably expressing siRNA-chk contained substantially reduced ChK levels as demonstrated by Q-RT-PCR and Western Blotting shown in Figure 1B. 1H MRS revealed that these clones also displayed the most pronounced decrease in cellular PC and tCho levels (Fig. 1A). Cell growth was reduced in clones chk23 and chk25, but not in con5 and chk27, which displayed high ChK expression (Fig. 1C). ChK expression levels were directly correlated to the cellular PC and tCho levels detected by 1H MRS in breast cancer cells. Reduced ChK expression decreased cell proliferation in breast cancer cells. 1H MRS proved useful in delineating the functional outcome of ChK knock-down clones of human breast cancer cells. ChK knock-down may prove useful as target specific cancer therapy that can be monitored by 1H MRS.
Characterization of choline kinase knock-down clones derived from parental MDA-MB-231 breast cancer cells: (A) Expanded 1H MR spectra of the empty-vector control clones con5, con7, and choline kinase knock-down clones chk23, chk25, chk27 demonstrating levels of glycerophosphocholine (GPC), phosphocholine (PC), and free choline (Cho). (B) Western Blots of a panel of breast cancer cells representing different stages of breast carcinogenesis (MCF-12A, MCF-7, MDA-MB-231, MDA-MB-435) and clones con5, chk23, chk25, and chk27 using choline kinase antibody. (C) Growth curves and cell doubling times of clones con5, chk23, chk25, and chk27.
K. Glunde, None.
Abstract ID: 285
Endothelin (ET) receptors are potential targets for diagnostic imaging and treatment of breast cancer, but information about their expression in human breast cancer is limited. The goal of these experiments was to quantify the ET receptor A and B (ETAR and ETBR) proteins and mRNA expression in normal and neoplastic human breast tissue.
Tissue specimens were obtained at the time of surgery from 10 women, aged 31 - 89 yrs, (1 with ductal carcinoma in situ, 8 with invasive ductal carcinoma, and 1 with basaloid carcinoma). The samples contained both normal and tumor-affected regions. A method was developed for quantification of ETAR and ETBR mRNA that included reverse transcription, PCR and real time PCR. Anti-ETAR and ETBR antibodies were used for immunostaining of cryostat and formalin-fixed, paraffin-embedded tissues.
ETAR protein was detected in 7 of 10 samples and ETBR in 10 of 10. The receptors were located predominantly in the plasma membranes, although there was some positive staining of the cytoplasm. ETBR immunohistochemistry demonstrated strong immunostaining of all tumor tissues. Moderate staining was seen for ETAR. Normal breast tissue did not stain significantly. ETBR protein expression by tumor cells was particularly high in invasive ductal carcinoma.
PCR showed bands at the expected location for ETAR and ETBR in most of the tested human breast tumor samples. No bands were found in normal tissues. ETBR mRNA levels were significantly higher in tumors compared with normal breast. ETAR mRNA was detected in breast cancer tissue with less intensity and was not detected in normal breast tissue.
These results provide evidence that the ET receptors are strongly expressed in human breast cancer. This may further motivate development of PET/SPECT techniques for imaging the endothelin receptors in breast cancer.
J. Xia, None.
Abstract ID: 286
The galectins, a family of carbohydrate-binding animal lectins, are likely involved in the metastatic process. Galectin-3 is a well-characterized multifunctional oncogenic galectin. In addition to regulating the common apoptosis commitment pathways intracellularly, studies indicate that galectin-3 is also involved in carbohydrate-mediated metastatic events and interacts with the tumor specific disaccharide Thomsen-Friedenreich antigen (TFAg). Given that increased carcinoma cell adhesion is associated with increased metastatic potential, we hypothesize that inhibition of the galectin-3-TFAg interaction will reduce homotypic (between carcinoma cells) and heterotypic (between carcinoma cells and endothelium) adhesion. Furthermore, this inhibition would suppress malignant cell survival and tumor growth. To test this, we identified peptide antagonists of galectin-3 using combinatorial bacteriophage display technology. The peptides bound to the purified galectin-3 protein with high affinity (Kd bcong; 17-80nM) and to cell surface galectin-3. Experiments using truncated galectin-3 mutants indicated that the selected peptides bound to the carbohydrate-recognition domain (CRD) of galectin-3. The galectin-3 CRD-specific peptides blocked galectin-3-TFAg interaction significantly and inhibited homotypic adhesion between human MDA-MB-435 breast carcinoma cells and heterotypic adhesion of MDA-MB-435 cells to endothelial cells under conditions of flow. In addition, to explore the role of anti-galectin-3 peptides in facilitating apoptosis of carcinoma cells, we initiated peptide internalization experiments. To the best of our knowledge, this is the first time that peptide modulators of galectin functions have been isolated. The anti-galectin-3 peptides possess superior binding characteristics (affinity, specificity) and are easy to synthesize relative to carbohydrate modulators of galectin-3. Consequently, small galectin-3-specific peptides will facilitate the investigation of galectin-3 functions and the role of carbohydrate-lectin interactions in hematogenous cancer metastasis.
J. Zou, None.
Abstract ID: 287
Tumor angiogenesis is an essential step in tumor progression and is an important target for anti-cancer therapies. There have been a number of methods reported that are designed to monitor the theraputic effects. The purpose of this study is to investigate the change in transverse relaxivity following contrast-enhanced MRI for assessing tumor response to AdExtek in a mouse melanoma model. Following Tropes, we assume average vessel size (VSI) is proportional to the ratio of deltaR2*/deltaR2 and that fractional blood volume is proportional to deltaR2* (Tropres MRM45:397-408). Images were acquired at 3T. GE and SE images were acquired pre and post injection of ferumoxytol (Advanced Magnetics, Inc.) via a jugular vein catheter. These images were used to produce parametric maps of deltaR2*, deltaR2 and deltaR2*/deltaR2. Figure 1A illustrates a T2 weighted anatomical image and the corresponding deltaR2* (figure 1B) and deltaR2 (figure 1C) maps. The tumor exhibited significant heterogeneity in both maps. In the example below, the mean fBV for the tumor center, periphery, and the contralateral muscle are, respectively, 13%, 22%, and 8% while the estimated VSI relative to muscle for the tumor center and peripheral tumor areas are 4.7 and 12.0, respectively. The tumor periphery has a higher relative VSI than the tumor center, which in turn is higher relative VSI than reference muscle. Additionally, the fractional blood volume is higher in the tumor periphery and in the tumor center, as compared to the reference muscle. This technique is a promising method for monitoring efficacy of anti-angiogenic treatment modalities.

B. GreenJarvis, None.
Abstract ID: 288
By standard molecular cloning techniques, a construct was made in which the tri-fusion gene is under the control of a minimal promoter and 8 Gli transcription factor binding sites. The Gli-trireporter construct was introduced into different tumor cell lines by transient transfection and its expression was studied under different conditions by luciferase assay. The results show that the expression of the trifusion gene is indeed upregulated by Gli, and inhibited by KAAD-cyclopamine, a HH signal pathway specific inhibitor. Next, Gli-trireporter stably transfeced U87 and SNB19 cell lines were made and injected into nude mice to generate tumors, and the tumors were successfully imaged by Xenogen bioluminescence imaging. Fluorescence imaging and XSPECT imaging will also be perfomed in the near future. Reagents that can specifically inhibit HH signal pathway will be tested in vitro and in vivo to see if the imaging can serve as a good marker of HH signal pathway and to monitor therapy application.
Y. Zhang, None.
Abstract ID: 289
Recent reports from our lab have shown that ITG, an iodinated hepatocyte-selective contrast agent, used in conjunction with microCT is an effective method for identifying and roughly quantifying hepatomas in murine tumor models. This study aims to improve upon the method of quantifying net tumor volume and also to track tumor growth (and eventually treatment) serially in living mice.
In studies of live mice without contrast, a histogram of voxel densities consistently generates four identifiable, normally distributed peaks. Analysis by selective regions of interest (ROI's) matches these peaks to air, lungs, soft tissue, and bone. A retrospective study of 12 mice has shown that in each case there is an additional peak when ITG was present.
Six mice undergo direct splenic tumor cell inoculation to induce diffuse liver tumors. The mice are scanned by microCT (GE eXplore RS, 80 kVp) following administration of ITG contrast (0.3 ml/20g bw) at days 0, 3, 6, 9, and 12. At day 12 the mice will be euthanized for gross pathological inspection. The livers of each mouse will be analyzed by isolating their respective ITG peaks for volume (+/- SD) calculations. As the metastases progress, it is expected that the viable liver volume will decrease, and the standard deviation of the entire liver will increase.
Preliminary studies in a single mouse have shown that liver volume can accurately be assessed by calculating the area under the ITG peak by integration, and multiplying the result by voxel size. A comparison of two such calculations (in-vivo and ex-vivo liver) shows good precision (<10% error) in this method.
B. Durkee, None.
Abstract ID: 290
M.A. Longino, None.
Abstract ID: 291
A highly specific and effective form of cancer therapy may be to target the repair, replacement or inactivation of specific oncogenes. We have utilized the tetracycline-dependent regulatory system to generate conditional models of oncogene induced tumorigenesis. Previously, we have shown that inactivation of the MYC oncogene is sufficient to induce sustained tumor regression. To further examine the mechanism of tumor regression, we have utilized bioluminescent imaging. We have confirmed that we can stably label tumor cells with luciferase. Luciferase activity was independent of that state of MYC activation. The labeled cells can be detected at a sensitivity of as low as 1000 cells. Labeled tumor cells can be detected over a dynamic range over 5 logs. Using this imaging approach, MYC inactivation was shown to result in tumor cells being rapidly eliminated from a host. Now, we will use this approach to study how genetic and host contexts influence the ability of oncogene inactivation to induce tumor regression. The results of these studies will have important implications for the mechanisms by which oncogene inactivation induces tumor regression and will provide a powerful preclinical model to examine potential new therapies for cancer.
P. Bachireddy, None.
Abstract ID: 292
Mesenchymal stem cells can be stimulated to differentiate into specific somatic lineage cells in vitro, but little is known about their in vivo response to malignant tumor formation. We studied this aspect in immunodeficient mice transplanted with human cancer cells subcutaneously and 3–4 days later with dual gene-marked human mesenchymal stem cells (tg-hMSC) through tail vein. Tumors formed in 3–4 weeks were detectable by positron emission tomography for nucleoside analogue retention catalyzed by the tg-hMSC marker herpes simplex virus thymidine kinase. Differentiated tg-hMSC progeny cells with green fluorescent protein marker were found in internal capillaries, peripheral blood vessel inner layer, dermal base and hair follicles at the tumor site. Numbers of the marked cells, some with human endothelial antigens, in the tumor were more than initially inoculated tg-hMSC. Our results indicate that mesenchymal stem cells can target, proliferate and differentiate at the tumor site and thus be exploited for noninvasive image tracing, in addition to therapeutic control, of cancer development in the body.
W. Deng, None.
Abstract ID: 293
STAT transcription factors are activated by JAK1-mediated phosphorylation on Y701 in response to IFN-γ and thought to signal through phosphorylation-dependent dimerization of STAT proteins with subsequent translocation of the active dimer to the nucleus. However, several studies have suggested the existence of a non-phosphorylated pool of STAT dimers. We have optimized firefly luciferase protein fragment complementation imaging (LCI) by screening incremental truncation libraries of N- and C-terminal fragments of luciferase and applied LCI to directly test for preassociation of non-phosphorylated homodimers of p91 STAT1 in intact cells. The STAT1-NLuc/CLuc-STAT1 LCI pair produced strong IFN-γ-independent bioluminescence in HEK-293 cells that was specific for the matched STAT1 LCI pair and not observed with mismatched STAT1 LCI pairs. In addition, Y701F mutation of the STAT1 reporters resulted in bioluminescence identical to cells transfected with wild-type STAT1 reporters, whether these were expressed as homodimeric mutant pairs or as heterodimers with wild-type STAT1 fusions. We also constructed separate lentiviruses expressing STAT1-NLuc or CLuc-STAT1 fusions driven by CMV promotors and co-expressing EGFP or mRFP from an IRES, respectively. Infected, FACS sorted, but untreated HEK-293 transfectants, showed high bioluminescence and Western blotting confirmed expression of both STAT1-NLuc and CLuc-STAT1 proteins, each unphosphorylated on Y701. Treatment with IFN-γ resulted in phosphorylation of both STAT1-NLuc and CLuc-STAT1 as well as endogenous p91 and p84 STAT1. To confirm that phosphorylated STAT1 LCI pairs were biologically functional, we measured the IFN-γ-inducible nuclear accumulation of bioluminescence activity. Bioluminescence (photon flux/μg protein) was specifically increased in nuclear extracts prepared from IFN-γ-treated cells, but not in extracts of cells transfected with the mutant STAT1 LCI pair or intact luciferase. Therefore, these data showed that the STAT1-NLuc/CLuc-STAT1 LCI pair associated constitutively and specifically in live cells, was phosphorylated in response to IFN-γ treatment, and that bioluminescent dimers translocated to the nucleus in an IFN-γ-dependent manner.
K.E. Luker, None.
Methods of Multimodality Imaging: Fusion Instruments & Software Solutions
Abstract ID: 294
J.V. Frangioni, None.
Abstract ID: 296
Micro PET and SPECT are essential tools for development of new pharmaceuticals, testing new therapeutic approaches, and imaging of gene expression, etc. They can be used noninvasively for the detection and the quantification. However, functional images provide little information about the detailed anatomy, and this leads to the mislocalization of the lesion. The image fusion techniques can be employed to map the functional images to structural images (such as x-ray CT) to aid in target identification and to make functional images easier to interpret. Furthermore, fusing two functional images of SPECT and PET can be beneficial for studies that require two biological processes to be monitored. Thus far, image registration algorithms effective in human studies have not been characterized in small animals. The purpose of this research is to develop an automatic fusion technique especially for small animal imaging. A special designed calibration phantom and a holder were employed to obtain the relation of three-dimension field of views of independent modalities. While the holder was aligned at fixed positions on the couches of different modalities, the spatial transformation matrix between the modalities keeps unchanged. Therefore, acquired tomograms from different modalities can be registered automatically by the predetermined matrix, independent of the imaged object, as long as the object were scanned with the holder together. The holder we designed was effortless to align for each scan, and the spatial three-dimension transformation matrix with 21 parameters can be created easily by the aid of calibration phantom. In this work, the Concorde's microPET R4 and the Gamma Medica's X-SPECT/CT scanners were used to acquire PET, SPECT, and CT data sets. Phantom and in vivo animal imaging studies have been successfully performed and will be presented. Preliminary experiments show the maximum registration errors were 0.22 mm × 0.28 mm × 0.48mm (axial).
M. Jan, None.
Abstract ID: 297
Exploration of dual-modality instrumentation for simultaneous tomographic imaging of optical and radiolabeled distributions has become a desirable task as both imaging methods have separately shown their potential to be highly sensitive in identifying location, magnitude, and time variation of molecular processes in vivo. At present, it is common practice to acquire projection data of optical and radiolabeled markers one after another and thus issues such as image registration, time-resolved concurrent data analysis, and animal handling become crucial. In order to investigate and evaluate innovative instrumentation design prototypes for dual-modality optical/PET micro-tomographic systems we have developed a fully integrated Monte Carlo simulation framework in which an unlimited number of optical and PET detector elements can be integrated without geometrical restrictions using a variety of animal phantom representation models. We exemplify the effectiveness of this approach by simulating a newly proposed combined tomographic small animal imaging system. A radially assembled array of micro-lens equipped CCD detectors is mounted in front of a scintillator crystal ring array. While these thin detector elements strip optical photons from the multi-energetic photon flux, isotopic (511keV) photons continue traveling and will eventually release their energy within the scintillator crystal. Integrated into this design is a network of optical fiber bundles which guides laser excitation light from an external multi-wavelength laser to the imaged object. The fibers have their exit points at the cross sections of the CCD offsets at fixed spacing. Individual fibers can be activated arbitrarily, allowing for a variety of laser excitation patterns. Integrated into the system are also axially movable optical filters for wavelength separation. System performance has been studied for a variety of CCD and scintillator crystal geometries. Special point of interest was the investigation of attenuation and scattering effects of the optical elements imposed on isotopic photons.
J. Peter, None.
Abstract ID: 298
P.L. Chow, None.
Abstract ID: 299
Histological methods are used to monitor the growth and response to various treatments of lung carcinoma in mice. The objective was to describe a noninvasive, quantitative and tomographic method to monitor metastatic progression by microPET and CT in mice in vivo. The dual-modality could be applied in the study of metastatic mechanism, and the response of chemotherapy and radiotherapy for lung tumors. In this study, male C57BL/6 mice were inoculated intravenously with 1 × 10 Lewis lung carcinoma cells (LLC1). [18F]FDG were injected intravenously for microPET and CT scans on day 0, 5, 9, 13, 18 and 26 to monitor the growth of tumor. At completion of imaging, both naïve and moribound tumored mice were sacrificed for autoradiography. MicroPET and CT images were further fused to monitor the localization and metastasis of lung carcinoma in lung and liver. The results of microPET and CT images showed the growth of tumor could be monitored longitudinally. MicroPET could detect tumor as early as 9 day after inoculation, where as CT couldn't detect tumor until 13 day after inoculation. The tumors located in lung and liver were visualized by fusion of microPET and CT images, and further confirmed by whole body autoradiography. The SUV of [18F]FDG increases from 0.51 at day 0 to 1.27 at day 26 following the growth of tumor in mice. In conclusion, the increase of SUV could be an alternative for assessing the growth of tumor in vivo. The metastasis of lung carcinoma could be identified using the microPET/CT dual-modality in mice longitudinally.
C. Chang, None.
Abstract ID: 300
J.V. Frangioni, General Electric 1.
Abstract ID: 301
J.V. Frangioni, None.
Abstract ID: 302
Small animals such as mice and rats are widely used in biological studies and drug developments. SPECT/CT dual modality imaging system dedicated to small animals provides a noninvasive method of assessing both biological functions and anatomical structures for these studies.
We are developing a dedicated 4-head SPECT/CT system for small animal imaging. A benchtop microCT subsystem have been implemented and tested. The microCT subsystem consists of a microfocus x-ray tube (60 kV, 1 mA) and a high-resolution CCD camera with a gadolinium oxysulfide scintillator (GOS). The GOS is coupled to the CCD camera with a reducing fiber optic taper. The camera provides low noise and dark current characteristics, with 70 μm pixel size and 7×7 cm2 active area.
Standard method with line and point was used to align the subsystem and to calibrate the geometry. Severe image error due to camera tilt was found but could not be reasonably adjusted. A series of tests indicated the error came from the optic taper. Further exam showed an asymmetric distortion pattern in the taper and a displacement between mismatched pixels as large as 1.12 mm. It resulted great degradation in projection data and severe artifacts in reconstructed images, and brought large difficulty in alignment and even made alignment arbitrary.
The distortion pattern was studied and a few key parameters were extracted to efficiently model the distortion. Corrections based on these parameters were performed and results were assessed. Phantom study showed that artifacts were eliminated and spatial resolution was significantly restored after correction.
M. Sun, None.
Abstract ID: 303
C. Lartizien, None.
Abstract ID: 304
Knowing the exact loalization of adoptively transfered cytotoxic T-cells (CTLs) is essential for understanding the biology of their action. In order to distinguish signal coming from the reporter gene-transduced CTLs in the body, we assessed a combination of imaging techniques showing anatomical structure of the organs.
M.M. Doubrovin, None.
Abstract ID: 305
Molecular imaging presents a new challenge for information management. Merging of in-vivo and in-vitro diagnostics is driven by the increased diagnostic certainty and as yet unrecognised potential that the combination of these complimentary data sets provides. Data acquired from new multi-modality imaging platforms is being merged with other clinical datasets, including a broad range of information derived from histology, chemistry, molecular biology, genomics, proteomics and other newly emerging diagnostic methodologies. Traditional clinical information systems are currently not able to provide the integration of such diverse laboratory diagnostics with standard clinical imaging. Information is scattered over multiple databases with no systematic structure that enables data mining across datasets. Additionally, data processing and analysis algorithms require quantitative measurements to be captured along with imaging datasets; this capability is currently not available on clinical PACS systems. To anticipate the challenges of this new diagnostics environment, a new platform is under development between Siemens Medical Solutions and the Center for Molecular Imaging Research. This new system extends the common PACS functionality beyond radiologic imaging. The flexible architecture allows for the integration of new modules and provides capabilities for cross-modular information management including access control and security, searching and data mining. The proof of concept system is currently implemented as an experimental platform for small animal molecular imaging. The software environment is based on the Siemens Syngo framework utilizing web technologies for cross platform user access and is implemented in a heterogeneous, academic research environment serving Windows, Unix, and Macintosh clients. The system provides access to images acquired from various imaging modalities along with other experimental data sets. Information is organised into studies with secure access based on user privileges.
M. Pivovarov, None.
Abstract ID: 306
Since its invention in 1990, two-photon-fluorescence-microscopy (2PFM) has made a strong impact to various research fields, especially to bio-imaging due to the reduced photodamage and optical sectioning power. However, compared to the commonly used Ti:sapphire laser in 2PFM, we have previously demonstrated that with a light source around 1200–1300 nm, the attenuation in biological tissues can be greatly reduced, resulting in enhanced penetration capability and diminished photodamage possibility. With the longer wavelength excitation, three-photon-fluorescence (3PF) would be necessary for efficient use of intrinsic and extrinsic visible fluorophores. The three-photon process can provide much better spatial resolution than 2PFM due to the cubic power dependency. On the other hand, third-harmonic-generation (THG), another intrinsic three-photon process, is interface-sensitive and can be used as a general structural imaging modality to show the exact location of cellular membranes. The virtual-transition characteristic of THG prevents any excess energy from releasing in bio-tissues and thus THG acts as a truly noninvasive imaging tool. Here we demonstrated the first combined 3PF and THG microscopy, which can provide three-dimensional high-resolution images with both functional molecule specificity and sub-micrometer structural mapping capability. The simultaneously acquired 3PF and THG images based on a 1230-nm Cr:forsterite femtosecond laser are shown below with a Hoechst-labeled hepatic cell sample. Strong 3PF around 450-nm from DNA-bounded Hoechst-33258 can be observed inside each nucleus while THG reveals the location of plasma membranes and other membrane-based organelles such as mitochondria. Considering the maximum-allowable laser power in common 2PFM is less than 10-mW, it is remarkable that even with a 100-mW incident power, there is no damage on the cells, demonstrating the noninvasiveness of this novel microscopy technique.

S. Chu, None.
Abstract ID: 307
J.V. Frangioni, General Electric 1.
Imaging in Drug Discovery and Development
Abstract ID: 308
Complex signaling circuits govern cellular growth and their perturbation is key to tumor development. The 14-3-3 small adapter proteins have emerged as central regulators of members of these signaling networks as their protein expression and activity is altered in many human cancers. 14-3-3's bind to multiple proteins and modulate their function predominantly through altering their subcellular localization. Specifically, 14-3-3 proteins bind to multiple ligands in the nucleus, such as FOXO1a, and promote their transport into the cytoplasm.
Our group has previously utilized a chemical genetics strategy and used a cell-based high throughput approach to isolate compounds that inhibit the transport of FOXO1a out of the nucleus. Further characterization of these compounds revealed new insights on FOXO1a regulation. A similar strategy aims to decipher the regulatory pathways that control 14-3-3 nucleocytoplasmic transport as a means to understand the role of 14-3-3 in tumor development. Specifically, we are using 14-3-3 sigma nuclear export as a readout in a phenotypic cell-based assay to screen chemical libraries for small molecules that inhibit 14-3-3 nucleocytoplasmic transport. The screen utilizes automated microscopy strategies that ensure efficient throughput. To date we have identified a number of chemical compounds that inhibit 14-3-3 export from the nucleus and are currently performing a series of secondary assays to determine the mode of action of these compounds. Among others, we anticipate isolating classes of molecules that interfere with the general nuclear export machinery, 14-3-3-ligand binding, 14-3-3 dimerization, and 14-3-3 phosphorylation. Interestingly, our preliminary results suggest that 14-3-3 sigma utilizes CRM1 independent pathways to exit the nucleus.
Together, the assays proposed herein will elucidate the regulatory networks that control 14-3-3 subcellular localization and will also identify potential cancer therapeutic candidates.
G.G. Toby, None.
Abstract ID: 309
99mTc-glucarate (GLA), a six-carbon dicarboxylic acid sugar labeled with 99mTc, has shown high tumor uptake in human breast cancer xenografts. It is still unclear if GLA will localize in other solid tumors. The purpose of this study was to determine the properties of GLA in detecting xenografted human A549 lung cancer in mice using high-resolution SPECT imaging. We compared the kinetic characteristics of GLA in A549 cancer to those in MCF7 breast cancer xenografts.
Z. Liu, Molecular Targeting Technologies Inc 6.
Abstract ID: 310
M. Rusckowski, None.
Abstract ID: 311
Delta opioid receptors are promising therapeutic targets for pain modulation, mood disorders and Parkinson's disease. In drug discovery, characterized preclinical tools are critical for understanding biological actions of novel compounds. Naltriben (NTB) is a selective, nonpeptide delta opioid (δ) receptor antagonist. [3H]-NTB has been characterized in mouse brain as a δ selective radioligand (Lever and Scheffel, 1998). We established [3H]-NTB (Tocris, 21 Ci/mmol) for δ receptor binding in adult rat brain using in vitro and in vivo binding techniques.
In vitro autoradiography: Fresh frozen rat brain sections (20 μm) were mounted on slides and incubated in buffer with [3H]-NTB or with [3H]-NTB plus competing compounds for non-specific binding. [3H]-NTB binding was high in δ -rich regions (such as striatum and cortical layers), low in regions containing few δ receptors (such as cerebellum, thalamus and hippocampus) and blocked by competing compounds.
Ex vivo autoradiography: Rats received [3H]-NTB, i.v., and were euthanized at 30 min. Slide mounted brain sections were analyzed by β-Imager [3H]-NTB occupied δ receptor-rich brain regions and had low non-specific binding.
In vivo receptor binding: Dose response and time course for [3H]-NTB, i.v., uptake in the rat brain established optimal conditions for displacement studies. Saline, naltrindole (δ-specific antagonist), BW373U86 (δ-specific agonist), naloxone (pan-opioid antagonist) or U50488 (κ, agonist) were injected prior [3H]-NTB (~0.153nmol/ml/rat). Rats were euthanized at 30 min; brain regions were weighed, solubilized and counted (DPM, fmol/mg). [3H]-NTB rapidly accumulated in δ -specific regions (prefrontal cortex, striatum, olfactory tubercle). High specific binding was maintained for several hours. [3H]-NTB binding was selectively displaced by compounds occupying delta opioid receptor sites. [3H]-NTB penetrates the rat brain after systemic administration, has high affinity for the δ-receptor site, has low non-specific binding and is displaced by δ-specific ligands. Thus, [3H]-NTB is a useful radioligand for preclinical investigation of novel δ-specific compounds.
D.L. Maier, None.
Abstract ID: 312
G. Jia, None.
Abstract ID: 313
99mTc-labeled sestamibi (MIBI), tetrofosmin (TF) and furifosmin (Q12) are monocationic agents and substrates recognized by the multidrug resistance (MDR) P-glycoprotein (Pgp). The purpose of this study was to compare the properties of these agents in assessing inhibition of Pgp function with a chemosensitizer, Amdray (PSC833), in mice bearing human breast tumors using a small-animal SPECT system, FASTSPECT, with the capability of fast dynamic acquisition.
Radioactive washout (%) of the MCF7 Tumors
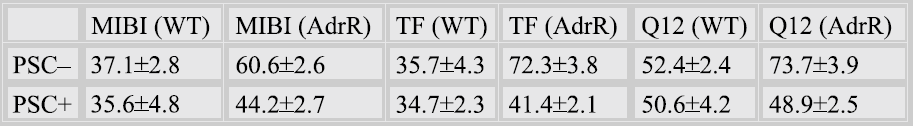
Z. Liu, Novartis Pharma 6; GE Healthcare (Amersham) 6; Mallinckrodt Medical Inc 6.
Abstract ID: 314
Most pre-clinical studies for evaluating radiolabeled somatostatin analogs have used rat pancreatic carcinoma cells. The goal of this study was to characterize human tumor cells expressing various levels of somatostatin receptor subtype 2 (SSTR2) that we believe will have added utility for evaluation of somatostatin analogs. A-427 human non-small cell lung cancer cells were stably transfected with an expression plasmid containing SSTR2 with a hemagglutinin (HA) tag on the N-terminus of SSTR2 (HASSTR2). Four G418 resistant, stable clones (#2, 4, 5, and 7) were isolated for further characterization. The expression in the four clones was analyzed both in vitro and in vivo. Membrane preparations of the four clones were analyzed via a competitive binding assay which showed the order of expression being 4<5<2<7. Analysis of the clones by FACS using an anti-HA antibody showed a similar expression profile. A-427-7 was then screened for internalization of HASSTR2 using a copper-64-labeled octreotide analog (64Cu-TETA-octreotide) at 15, 60, 120, and 240 min. Internalization of the 64Cu-TETA-octreotide in A-427-7 increased from 1.5% of the total radioactivity added at 15 min to 14.2% at 240 min. Biodistribution of 64Cu-TETA-octreotide in nude mice bearing subcutaneous tumors of the clones showed variable uptake that ranged from 0.8% to 6.5% ID/g 1 h after i.v. injection that followed the same rank order (4<5<2<7) found in the binding and FACS assays. MicroPET imaging with clones 4 and 7 showed good uptake in 7 and background uptake in 4 with clearance of the peptide through the kidneys. Thus, stable transfection of HASSTR2 into human A-427 cells has been positively demonstrated to be both functional and effective for imaging of tumors using radiolabeled somatostatin analogs. These cells should have advantages over the rat pancreatic carcinoma cells for future evaluation of radiolabeled somatostatin analogs.
B. Rogers, None.
Abstract ID: 315
In the recently released NIH Roadmap for future research, it is stated that “NIH will support development of high-specificity/high-sensitivity probes with the goal of improving detection sensitivity 10- to 100-fold within five years.” Though laudable, this goal as stated is rather imprecise. No rigorous definition of detection sensitivity is suggested, and no consideration is given to the factors that limit detectability in different imaging modalities. Moreover, it seems to be assumed that detectability is limited solely by the properties of the molecular probe, when in fact properties of the background object, the imaging system, the reconstruction algorithm and the detection algorithm or observer all play key roles. Detectability is a property of the whole imaging chain, not just the probe.
The purpose of this presentation is to summarize the current state of knowledge of signal-detection theory as applied to imaging, provide a rigorous definition of detectability, and identify the factors that can limit detectability with different modalities. From this perspective we shall identify the leverage points in each modality and assess opportunities for improvement. One conclusion that will emerge from the analysis is that improvements in spatial resolution can lead to large gains in detectability, especially when the target of interest must be imaged against a spatially inhomogeneous background. By the same token, changes in probe design or data acquisition that reduce the level or the inhomogeneity of the background can also increase detectability significantly. Signal-detection theory allows us to make these statements quantitative.
Though we do not propose to answer the question in the title (yet), we shall use the viewpoint of signal-detection theory to suggest future directions for research and technology development.
H.H. Barrett, None.
Abstract ID: 316
Figure 1. Gamma images of Balb/C mice. A, Treated with 99mTc-HYNIC Chitosan. B, Treated with 99mTc-HYNIC-GC. C, Coinjected with 99mTc-HYNIC-GC and free galacotse 50 mM. D, Coinjected with 99mTc-HYNIC-GC and free galacotse 80 mM.

H.J. Jeong, None.
Abstract ID: 317
Retinoic acid (RA) can inhibit tumor outgrowth by suppressing cell proliferation, promoting cellular differentiation, or by inducing apoptotic cell death. To monitor the intranuclear response of retinoic acid receptors, we developed a cis-imaging reporter system using luciferase gene under the control of enhancer regulatory elements that are responsive to RA-specific activators (So et. al., SMI, 2003). However, this system has a drawback with relatively low expression of imaging reporter genes by the poor transcriptional activity of a weak promoter. To improve in vivo image acquisition, it is essential to enhance the transcriptional activity of the RA-targeted promoter. We applied an amplification approach using a two-step transcriptional amplification (TSTA) system.
Reporter plasmid was constructed using TSTA system (kindly provided Dr. Gambhir from Stanford university) with artificial cis-acting retinoic acid enhancer (RARE) and named as pTRL. The pTRL was transfected to SK-HEP1 cells (human hepatocellular carcinoma) with lipofectamine. After treatment of all-trans RA for 48 hours, luminescence intensity of the cells was measured. For in vivo imaging, a cooled charge-coupled device optical imaging system (LAS3000, Fuji, Japan) was used to visualize the luciferase expression in living mice bearing transfected SK-HEP1 tumor.
We found approximately 7-fold higher luciferase activity by using the TSTA system approach compared to the one step system in vitro. In vivo imaging experiments revealed approximately 3-fold gain in imaging signal by using the TSTA system over the one-step system. Bioluminescence images showed higher intensity in SK-HEP1 transfected vector using the two step system with RA than those without RA treatment.
The application of TSTA system to cis-enhanced reporter showed higher reporter gene expression, and was helpful for monitoring gene expression in vivo as well as in vitro. This amplified imaging reporter system can be useful for the evaluation of RA response of cancer such as cellular differentiation and chemoprevention.
M. So, None.
Abstract ID: 318
[18F]FP-TZTP is a M2 muscarinic subtype selective radioligand whose selectivity has been validated using knock-out mice. The biodistribution of [18F]FP-TZTP was compared in rats both awake and under isoflurane using either a bolus injection or a constant infusion method. Brain concentration was obtained 30 min after injection by ex vivo autoradiography (Fuji Phosphorimager) or by imaging (ATLAS, Advanced Technology Laboratory Animal Scanner).
Because of the fast clearance and metabolism, more blood samples are required to estimate the input function. In order to minimize the amount of blood removed from the animal, analysis of whole blood for metabolites was investigated. Blood and plasma were analyzed for metabolites using an extraction method. The distribution ratio was determined from the brain radioactivity divided by the average activity (bolus injection method) or final activity (constant infusion method) in the metabolite-corrected plasma or whole blood. Because there was no constant relationship between input functions determined from plasma and whole blood, the bolus injection was not a suitable method for this compound. By comparison the constant infusion method requires fewer blood samples to define the input function, thus plasma can be analyzed for metabolites.
During a constant infusion of ~ 4 mCi of [18F]FP-TZTP, the plasma input function in rats under isoflurane was greater than in awake rats and the volume of distribution of [18F]FP-TZTP in 8 brain regions was greater in awake rats compared to anesthetized rats, although not significantly. The parent fraction in whole blood was always greater than in the plasma and the plasma input function reaches equilibrium faster than the whole blood input function.
The constant infusion method enables the calculation of equilibrium distribution ratios.
L. Ravasi, None.
Abstract ID: 319
Treatment of cells with antifolates such as methotrexate (MTX) results in “induction” of DHFR. This induction is cycloheximide sensitive but actinomycin D insensitive suggesting that it occurs at the translational/post translational level. Our studies have shown that DHFR binds its cognate RNA and regulates its own translation. MTX relieves this “translational control” allowing resumption of DHFR synthesis. Fusion proteins of DHFR are similarly regulated and can be induced upon antifolate treatment. Concerns regarding antigenicity of fusion proteins of DHFR, particularly as applied to gene transfer studies in vivo, prompted us to examine whether proteins separated from DHFR by an IRES element could also be regulated in a similar manner as the fusion proteins. We hypothesized that DHFR, by binding the bicistronic message can regulate translation of both itself and the partner protein. Antifolate treatment of cells, expressing these two proteins from a bicistronic construct, would thus lead to induction of both DHFR and the partner protein. We demonstrate induction of reporter proteins such as EGFP or Luc, both as a fusion partner and separated from DHFR by an IRES element upon MTX treatment of cells transfected to express these cDNAs. Using Western blotting as well as imaging, in vitro and in vivo, we demonstrate antifolate mediated induction of the reporter. While the fusion protein is induced within 24h of antifolate treatment, induction appears to be delayed for the reporter when present as part of the bicistronic construct DHFR-IRES-Reporter but is sustained over a period of 72h. This suggests that levels of desired proteins can be modulated by antifolate treatment even when the protein is expressed from a bicistronic construct containing DHFR rather than a fusion protein of DHFR. This is particularly important for gene transfer strategies utilizing DHFR and a second protein for purposes of myeloprotection or suicide gene therapy.
L.G. Menon, None.
Abstract ID: 320
Hsp90 is the most abundant protein of the eukaryotic cytosol, which in cooperation with Hsc70, p60, and other factors, prevents protein aggregation and assists protein folding. Hsp90 can bind partially unfolded proteins, including firefly luciferase, and prevent their irreversible aggregation. Unfolded luciferase bound by the multichaperone complex is either released in an ATP-dependent manner for refolding or is presented for degradation (Schneider C. et al., 1996). Binding of the ansamycins antibiotics, such as geldanamycin, to Hsp90 shifts this chaperone system from protein refolding to proteasomal degradation. To image degradation of Hsp90-dependent proteins by the geldanamycin derivative 17-AAG, we transduced C6 glioma cells with the triple fusion reporter system (Ponomarev et al., 2004). This reporter system produces a single fusion protein with three functional subunits, herpes simplex virus thymidine kinase, green fluorescence protein and firefly luciferase (HSV-TK/GFP/FLuc). The C6/TGL cell line was treated with 17-AAG and the activity FLuc was monitored. After 4h of treatment, luciferase activity decreased in a 17-AAG concentration dependent manner. The FACS analysis revealed that the mean GFP fluorescence of the triple fusion also decreased in a concentration dependent manner. Radiotracer assay for HSV-TK correlated well with the decreased activity of the FLuc and GFP components. These results suggest that integrity of the triple fusion depends on Hsp90, and degradation of the reporter protein is caused by inhibition of Hsp90 activity. To prove this assumption we performed Western blotting. We found a decreased amount of the fusion reporter protein in the treated cells which correlated with a decrease phospho-Akt protein, whose degradation is known to be dependant on Hsp90. In vivo bioluminescence and microPET imaging also demonstrates that inhibition of Hsp90 by 17-AAG can be imaged using an artificial fusion protein as a reporter for Hsp90-dependent degradation.
I.S. Serganova, None.
Abstract ID: 321
Over the last few years, our group has been involved in product development and preclinical and clinical evaluation of rh-Annexin V derivatives for in vivo imaging of apoptosis. Major emphasis has been placed on 99mTc-Hynic-rh-Annexin V, which is by far the most extensively investigated and best characterized phosphatidylserine apoptosis marker. This molecule is currently in Phase 2 clinical studies for prediction of response to cancer treatment and has shown promise for evaluation of cell death in stroke, Crohn's disease, rheumatoid arthritis and vulnerable atherosclerotic plaque. Preliminary observations suggest that baseline uptake of this agent may be a predictive biomarker in advanced non-small cell lung cancer patients undergoing platinum chemotherapy, and that early post-treatment changes in localization may predict improved survival. Recently, we have developed new rh-Annexin V based agents including near infrared prototypes as well as nuclear agents that carry both a chelate and a biotin for faster blood clearance. These agents will permit evaluation of promising new strategies for the development of the ideal cell death detecting imaging agent. We will present several aspects of product development of rh-Annexin V, the general strategy for derivatization, data on characterization of the derivatives by typical biochemical methods including mass spectroscopy, and our approach to the evaluation of the agents from animal imaging models to early human studies. We will compare the surface plasmon resonance (SPR) on doped phosphatidyl serine surfaces as a determination of retention of biopotency with other direct methods involving cells or phospholipid constructs. Special emphasis will be placed on the proper characterization of the new derivatives, the review of animal models and their translation to early human studies, and the issue of the timing of the injection of the imaging agent after the start of treatment.
J. Vanderheyden, Theseus Imaging Corporation 5.
Advances in Clinical Molecular Imaging
Abstract ID: 323
We aim to develop novel nanoparticle ultrasound contrast agents that will be capable of molecular targeting. Currently, microbubbles are the only FDA-approved ultrasound contrast agents. These agents have limited use for tissue-specific contrast enhancement due to their relatively large size (above micron), short half-lives in circulation and limited potential for surface modification. Solid nanoparticles may provide advantages in these areas that are crucial for molecular targeting and selective enhancement of tissue. Previous studies have demonstrated the contrast potential of solid particles of the size of a few microns. In this study, we tested the contrast feasibility of submicron solid particles of various material compositions. For in vitro studies, the particles were homogeneously suspended in agarose gel at various concentrations. High frequency ultrasound probes (35-55MHz) were used to image the gel with and without particle suspensions using the high-resolution ultrasound imaging system Vevo 660 (VisualSonics). For in vivo studies, the livers of Balb/C mice were imaged before and after intravenous administration of nanoparticle suspension in saline. Our results showed that submicron particles generated contrast enhancement in agarose gel, and the enhancement increased with increased particle concentration and size. The contrast enhancement was also dependent on particle material composition. Our in vivo data showed a steady increase in contrast enhancement of the liver immediately following particle administration for about half an hour. After that, it was observed that the contrast stayed unchanged for at least two hours. These studies demonstrated the feasibility of nanoparticles to increase ultrasound contrast in vitro and in vivo. It will be important in the future to develop tissue specificity of the nanoparticles with surface modifications.
J. Liu, None.
Abstract ID: 324
Encouraging results have been reported when using bone marrow-derived mesenchymal stem cells (MSCs) for tissue repair. For MR tracking of SPIO-labeled MSCs, it is important to determine if magnetic labeling affects the differentiation capacity of labeled MSCs. We therefore assayed the differentiation capacity of human MSCs (using three different donors) after labeling for 24h with 375 ng/ml poly-L-lysine (PLL) and 25 μg Fe/ml Feridex or with PLL only as control. The proliferation of human MSCs was unaffected by PLL-Feridex treatment. Adipogenesis (induced by treatment with 1-methyl-3-isobutylxanthine, dexamethasone, indomethacin, and insulin) was assessed by Oil Red O staining for neutral lipids. Labeled cells differentiated normally. Osteogenesis (induced by treatment with dexamethasone, b-glycerol phosphate, and ascorbic acid 2-phosphate) was visualized using von Kossa stain (mineralized matrix) and immunohistochemistry (Type I collagen), and a quantitative colorimetric assay for deposition of calcium phosphate. All three osteogenic assays demonstrated normal differentation into osteocytes. For the chondrogenic assay, MSC micromass pellets were treated with transforming growth factor-b3 and stained with Safranin O (proteoglycans) or immunostained for Type II collagen. Surpisingly, although cells proliferated normally, the differentiation into chondrocytes was completely blocke. This was further demonstrated by experiments using 50:50 mixtures of Feridex-labeled and unlabeled MSCs: only the areas without PB staining (i.e. unlabeled cells) were positive for Safranin O and Collagen II. Chondrogenesis was unaffected by treatment of MSCs with PLL alone; the blocking of chondrogenic activity is thus mediated by the Feridex, and not by the transfection agent. The nature of Feridex interference with this mesenchymal pathway is unknown. This is the first study reporting Feridex blocking of the differentation down a specific pathway of a specific cell type, while not affecting viability and proliferation, and indicates the need for careful titration and lineage studies when considering clinical trials using Feridex-labeled stem cells.
J.W. Bulte, Osiris Therapeutics, Inc. 1.
Abstract ID: 325
M. Larabi, None.
Abstract ID: 326
Ferumoxides (FeridexTM, Berlex, Wayne, NJ) an FDA-approved superparamagnetic iron oxide (SPIO) nanoparticle suspension and protamine sulfate (USP), an FDA-approved anti-heparin drug, can be complexed (forming FePro) and used to magnetically label cells in vitro. Labeling stem cells with FePro allows for non-invasive monitoring by MRI. However, in order for stem cell trials or therapies to be effective, this labeling technique must not inhibit the ability of cells to differentiate. In this study, we examined the effect of FeProlabeling on human CD34+ hematopoietic (HSC) and mesenchymal (MSC) stem cell differentiation. Colony-forming unit (CFU) assays showed FeProlabeled HSC's capacity to differentiate was equal to unlabeled control. Furthermore, flow cytometry revealed that labeling did not alter HSC expression of phenotypic markers (CD34, CD31, CXCR4, CD20, CD3 and CD14). SDF-1-induced HSC migration was also unaffected.
Both FePro-labeled and unlabeled MSC were cultured in chondrogenesis-inducing conditions using materials and methods obtained from the supplier (Cambrex, Walkersville, MD). Frozen sections of the resultant cell pellets from both the labeled and unlabeled conditions underwent histological and immunohistochemical analyses. Prussian blue staining confirmed cells in labeled pellets contained iron oxide, while cells in unlabeled pellets did not. Alcian blue staining for proteoglycans revealed similar chondrogenic differentiation for both FePro labeled and unlabeled cells. Furthermore, collagen II and collagen X, indicators of cartilage formation, were detected at similar levels in both labeled and unlabeled cell pellets. Interestingly, collagen expression seemed to be locally inhibited in a few cells within the labeled pellet that were near large, extracellular clumps of FePro complexes. FePro labeling generally leads to small intracellular iron particles, and the addition of heparin sulfate to the wash results in disassociation of the FePro complex, leaving few, if any, extracellular iron oxide nanoparticles. This may explain why this technique does not inhibit chondrogenesis.
G.T. Yocum, None.
Abstract ID: 327
J.V. Frangioni, None.
Abstract ID: 328

C. Menard, None.
Abstract ID: 329
J.V. Frangioni, None.
Abstract ID: 330
J.V. Frangioni, None.
Abstract ID: 331
For MRI measurements agarose-gel phantoms were prepared containing (a) 1×105 CLL-185 cells with increasing amounts of cellular SPIO load (1.0-5.0 mg Fe/ml in the medium), (b) different amounts of identically labelled cells (5×103−2.5×105 cells/ml agarose) and (c) free SPIO at identical concentration as measured in (a) and (b). All measurements were performed using multi-echo readout sequences at a 3T scanner (Philips Intera) Cellular iron-uptake was qualitatively analyzed by light-microscopy (LM) and quantified by Atomic Emission Spectroscopy (AES).
MR-phantom measurements showed a strong, dose dependent decrease of T2 *-relaxation time, while the decrease in T2-relaxation time was more subtle (e.g: [0.6-27.5 μg Fe/100,000 cells: T2*: 46.9-10.6 ms; T2: 60.6-44.0 ms).
T2* effects were significantly higher (p<0.01) and T2 effects significantly lower (p<0.05) for iron labelled cells compared to phantoms containing free SPIO (c).
Cellular iron load (a; r = 0.99; p<0.0001) and cell number (b; r = 0.99; p<0.0001) were linearly correlated with R2* values.
Further studies are warranted to test the applicability of this method for non-invasive cell quantification in-vivo.
L. Matuszewski, None.
Abstract ID: 332
J.V. Frangioni, None.
Abstract ID: 333
Traditional thinking about PACS is finally being replaced, giving way to new perspectives about the key elements of PACS. In the process, the radiology modality and film vendors who installed the early systems are learning a difficult but important lesson: that Radiology PACS is a departmental solution that needs to extend beyond Radiology into the enterprise.
The old PACS paradigm defined four distinct elements of a Radiology PACS. #1. Image acquisition, #2. Image management, #3 Image display, #4. Image storage. But today, many of these same vendors continue to ignore the fact that a Radiology PACS is but one clinical system in an increasingly integrated world of information systems. It is, of course, critical that Radiology manage the clinical aspects of their PACS. Diagnostic workstations must be easy to use and fast. But what about the other images outside of Radiology? What about clinician access to a molecular medical record? Many departments deal with high-resolution imaging and require a PACS as well.
Thankfully, information systems vendors are just now realizing that integration of a scalable patient-centric, multimedia enterprise infrastructure is the key to efficient care. Using RAID and other hi-speed archives, improves all the elements of the imaging chain. As a result of this realization, a fifth element of PACS has emerged. One that is secure and can achieve complete integration with a well founded enterprise infrastructure. The result is powerful and compelling, with patient-centric multimedia access for clinicians who log on once, identify the patient once and get access to all of the information they need instantly.
Radiology truly will not get the chance to go filmless until Radiology's customers are comfortable in that filmless environment. Until vendors come to terms with that reality, their customers are locked into a self-limiting, Radio-centric universe.
K. Waldbillig, EMC 5.
Abstract ID: 334
The non-invasive quantification of beta-amyloid in Alzheimer's Disease (AD) may enable early diagnosis and therapeutic efficacy monitoring. In previous work(1) we simulated the preclinical imaging of beta amyloid species in a transgenic mouse. The current study aims to extend our previous work to humans by developing a prototype PET image simulator with specific applications to beta-amyloid imaging.
A virtual phantom of human brain(2) was modified by merging regions and allocating regions to virtual pharmacokinetic compartments. Each compartment was assumed to include both specific and non-specific binding. Pharmacokinetic parameters were extracted from published measurements of one candidate amyloid tracer(3). Nonspecific binding was estimated from matching a kinetic model to both AD and human control data. Specific binding to beta-amyloid is simulated over a range of binding strengths and density of binding sites. The scanner simulation represents a clinical PET scanner (GE DST) and the simulated acquisition protocols are based upon clinical PET practices.
First generation simulated images show differential uptake of the amyloid tracer in cortical regions versus regions of expected low amyloid and are comparable to published data. Our results demonstrate the feasibility of constructing a comprehensive PET simulator that includes brain region geometry, amyloid biology, pharmacokinetics and image hardware. This model can be used to simultaneously explore complicated multivariate imaging inputs and complicated imaging metric outputs.
M. Simmons, None.
Abstract ID: 336
Imaging technology plays an important role in modern biology and medicine. For example, in vitro cell imaging data can be acquired using bio-nanoimaging acquisition technology to study cell behavior for cancer diagnosis and drug responses. The important next step after acquisition is to identify histological and anatomical features from these images. These images usually contain various shapes that require robust segmentation techniques. Thus, level set technique becomes attractive for processing these images due to its topological independence. However, its application to large-sized images is constrained by its high computational cost. This is because a set of numerical simulations has to be performed repeatedly on every pixel of an image and the general purpose central processing unit (CPU), that has single execution core and limited memory bandwidth, is far from optimal for this type of tasks. In comparison to CPU, modern programmable graphics processing units (GPUs) are designed to perform large amount of repeated tasks effectively thanks to the multiple processing pipelines and wide memory bus. As a result, we designed and developed a graphics processing unit (GPU) based level set algorithm to process large size of molecular and cell images.
M. Wang, None.
Abstract ID; 337
J.P. Weichert, Cellectar, LLC 2.
Abstract ID: 338
Breast cancer is one of the most common cancers and is the second leading cause of death in women. After lumpectomy, if cancer cells are found in the margins of the excised tumor, additional excision surgery is performed. Met and its ligand, hepatocyte growth factor/scatter factor (HGF/SF), mediate mitogenicity, tumorigenicity, and angiogenesis in human cancer. Met is inappropriately overexpressed in ~30% of human breast cancers and its high expression correlates with poor prognosis. Met activation by HGF/SF induces hemodynamic changes that can be measured by BOLD MRI. Based on these changes Met functional imaging was developed. Mice bearing DA3-mouse and human breast tumors were injected i.v. with saline and Ultrasound contrast media (CM) as a baseline, followed by HGF/SF or saline i.v. injection, and then re-injections of CM after 20 min. HGF/SF increases the area of blood vessels with significant blood flow as well as increasing the blood volume in the tumor by opening new blood vessels with slower blood flow.
Functional molecular imaging analysis based on HGF/SF induced hemodynamics changes in mice bearing tumors expressing Met was used for mapping tumor margins. Differences in the hemodynamics in tumor borders were monitored before and after HGF/SF injection using ultrasound CM. Signal intensity before and after HGF/SF injection was mapped, and the differences were determined. Histopathology of fine needle biopsies taken from marked areas with enhanced signals in the tumor margins detected areas that contain local metastasis.
This work demonstrates a use of this novel molecular imaging modality to image tumor borders and local invasion. This imaging modality might reduce the need for re-excision surgery and enable better tailoring of the treatment. This work was supported in part by NIH research grant (P50CA93990).
G. Tsarfaty, None.
Abstract ID: 339
J.V. Frangioni, None.
Abstract ID: 340
MR is not clinically established for lymph node staging. However, preliminary studies at CMIR have shown that MR MION imaging can dramatically improve lymph node staging, which is an important step in cancer treatment planning. Lymphectomy (and histology) is the gold standard. In some instances PET has been utilized as imaging modality, while MR imaging is not yet clinically established. Imaging technologies are generally seen as an inadequate diagnostic choice if compared to other methods applied. However, clinical studies have been conducted at the CMIR on MR based detection of clinically occult lymph node metastases in prostate cancer using MION (ferumoxtran-10). MR sensitivity dramatically increases from 45,4 percent up to 100% (with a high specificity of 95,7%) by using ferumoxtran-10 during the diagnostic process. Not surprisingly today, MION imaging is requested by MGH surgeons prior to prostatectomy. Although this new technique provides a significant improvement, manual interpretation of MR datasets is complex, time consuming and dedicated analysis software is needed. Our objective was to develop MR specific oncology packages within the Siemens Syngo user interface and to test its applicability clinically on patients. By utilizing ferumoxtran-10 as agent, the current environment enables pre- and post MION image generation in as well as semi-automatic lymph node recognition. In addition, lymph nodes of interest can be categorized and displayed (benign + malignant) in 3D vessel MIP mode. Three-dimensional reconstruction of the prostate, vessels, and lymph nodes can assist in the surgical planning of radiotherapy.
A. Hengerer, None.
Enhancing Delivery and Traversing Barriers
Abstract ID: 341
The virus concentration for optimum locally-injected suicide gene therapy efficacy is not known, but depends on the viral dose, the more the better within the limits of accepted toxicity, and the injection volume, too little restricts the local spread of virus while too much reduces the multiplicity of infection. Experimental evidence is presented suggesting that the ideal virus concentration is determinable using the reporter gene, the sodium-iodide symporter, NIS. Exogenous gene expression for various viral concentrations was quantified experimentally at sub-millimeter resolution using a replication-competent adenovirus that incorporated suicide genes in addition to NIS, and localized the radioactive anion, pertechnetate, 99mTcO4-. Cells infected with adenovirus incorporating NIS acquired the ability to concentrate 99mTcO4- and allowed for thin-tissue autoradiographs in which optical density was a function of gene expression. Digitized autoradiographs were analyzed to yield frequency histograms of gene-expression pixels. The resultant gene expression histograms following a range of viral doses at fixed injection volume and a range of injection volumes at a fixed viral dose were measured in dog prostates. Under sterile surgical conditions, each prostate lobe was administered virus under controlled experimental conditions. One day after viral delivery, dogs were administered radioisotope and following at least 2 hours of uptake, prostates were excised, frozen and processed for autoradiographs. The results were two-fold. First, increased viral dose at fixed injection volumes resulted in increased gene expression magnitude and volume. Second, the greater the injection volume at a fixed viral dose, the greater the gene expression coverage measured as more pixels of optical density above the threshold corresponding to viral DNA. However, a concomitant decrease in the magnitude of gene expression was observed. Consequently, our results suggest that an ideal injection volume exists that represents a compromise between maximum viral spread and sufficient gene expression magnitude to be therapeutic.
S. Brown, None.
Abstract ID: 342
The development of hypoxia-regulated reporter genes presents an exciting possibility to observe hypoxia inducible factor (HIF) mediated gene expression in vivo and study its role in tumor progression, metastasis, and therapeutic resistance. However, the behavior of reporter genes in low oxygen conditions, in terms of gene transcription, translation, and activity of the resulting protein product, has not been rigorously assessed. These factors may adversely affect the ability of reporter genes regulated by promoters containing hypoxia response elements (HREs) to provide quantitative information on HIF-induced gene expression and correspondingly hypoxia. To evaluate the sensitivity of reporter gene function to hypoxia, we exposed 293T cells stably expressing a triple fusion reporter consisting of renilla luciferase (hRL), monomeric red fluorescent protein (mRFP), and truncated HSV1-sr39 thymidine kinase (ttk) under a CMV promoter to environments containing 21% (normoxic), 2% (hypoxic), or <0.02% (anoxic) O2. After 24 hours the cells were lysed and while still under controlled oxygen conditions, assayed for bioluminescence. The measurements were then repeated five minutes after exposing the lysates to normoxia. In addition, western blotting was performed to assess variations in protein levels induced by exposure to varying oxygen levels. It was observed that a modest decrease in luminescence occurs in reoxygenated lysates from cells exposed to decreasing oxygen levels. However, our results suggest that exposure to low oxygen concentrations during light collection results in an appreciable reduction in the amount of light produced per reporter protein. This implies a disconnect between the signal expected from in vivo imaging of severely hypoxic tumors and the actual amount of gene expression. This finding merits consideration when analyzing in vivo imaging data collected from hypoxic tumor models.
E.E. Graves, None.
Abstract ID: 343
R. Liu, None.
Abstract ID: 344
Molecular imaging strategies using non-invasive imaging techniques such as MRI, are currently being developed in mouse model systems where these strategies can be optimized. Imaging strategies based on endogenous receptors can suffer from a number of potential drawbacks. Our goal is to develop a non-mammalian receptor/ligand imaging system, to overcome such potential difficulties. We are evaluating fusion constructs of plasma membrane receptors. Our receptors have been engineered to have a native mammalian transmembrane (MT) type II receptor signal/anchor domain fused to the ligand binding-site (BS) of a non-mammalian type II receptor Er-1mem. The latter is from the marine protozoan cilate Euplotes raikovi.

The Er-1mem-Binding-Site/Er-1-Ligand system will be useful for wide-ranging applications in detecting gene expression, or gene expression driven by micro-environmental conditions such ashypoxia. In combination with the functional imaging capabilities of PET and MRI, the develop-ment of this system will allow a molecular/functional imaging approach to understanding thefunctional impact of genes of interest.
This work was supported by NIH P50 CA103175 (JHU ICMIC Program) through a research component to Venu Raman.
P.T. Winnard Jr, None.
Abstract ID: 345
Paramagnetic contrast agents such as gadolinium chelates have been used to enhance MR image contrast by disturbing the local magnetic field and the magnetization of surrounding proton spins. The uptake of the contrast agent into the tumor interstitial space is based upon permeation of contrast agent through arterioles and its diffusion within intercellular space. The uptake process is not specific. We have made use of a cationic immunoliposome system that employs a single-chain antibody variable fragment (scFv) against the human transferrin receptor (TfR) to target a liposome-contrast agent complex preferentially to cancer cells. Cationic liposomes are composed of positively charged lipid bilayers that are complexed to gadolinium chelates, Magnevist, by simple mixing. The resulting complex has a net positive charge that facilitates transfection of the cells, and a small size that allows penetration through the capillaries to the tumor cells. Magnevist is diluted in water and added to TfRscFv-Lip at a defined ratio. The equivalent dose of Magnevist is 0.3 mM/kg. Human leukemic cells K562 were transfected with the solution containing TfRscFv-Lip-Magnevist complex, or Magnevist only. After transfection, the cells were cultured for additional 48 hours. The cells then were pelleted for MRI study. T1-weighted MR images (SE TR/TE, 1000ms/13ms) of cell pellets show an intensity increase of 6 folds for TfRscFv-Lip-Magnevist complex compared to Magnevist only. Also, a study of transfection time shows the intensity change reaches a plateau after 60 minutes exposure. The advantages of targeted liposome as contrast agent delivery vehicle are its low toxicity, simplicity of preparation, its relative stability, and most significantly its ability to preferentially target tumor cells and efficiently transfer the constrast agent into them. Therefore use of this deliver system has the potential to enhance the sensitivity of contrast agents in patient.
P.C. Wang, None.
Abstract ID: 346
J.E. Bugaj, None.
Abstract ID: 347
Molecular diffusion occurs more slowly throughout biological tissue than it does in temperature-matched dilute aqueous solution. It is currently unknown to what extent reductions in brain apparent diffusion coefficient (ADC) values relative to free media are due to “static” structural restrictions, such as cell membranes; versus “dynamic” hindrances to diffusion, such as viscosity effects and collisions with mobile, molecular-scale cytoplasmic entities. Herein, we domonstrate that the N-acetylaspartate (NAA) magnetic resonance (MR) signal decay versus b value can be quantitatively explained by a theoretical model of NAA confined to an ensemple of differently oriented neuronal fibers (i.e. axons and dendrites). On the microscopic scale, NAA diffusion is found to be strongly anisotropic, with displacements occurring almost exclusively parallel to the local fiber axis. This parallel diffusivity, ADCparallel, is 0.36 ± 0.01 μm2/ms, and ADCperpendicular is essentially zero. From ADCparallel, the apparent viscosity of the neuron cytoplasm is estimated to be twice as large as that of a temperature-matched dilute aqueous solution.
C.D. Kroenke, None.
Abstract ID: 348
Ultrasonic molecular imaging entails coating microbubbles with ligands specific to receptors expressed on target cells located within the vasculature. Concentration of these highly echogenic contrast agents at the target enhances the ultrasound signal at that site, promoting diagnosis of disease states and delivery of therapeutic agents. Ultrasonic radiation force deflects freely flowing microbubbles from the center of flow to the vessel wall and enhances adhesion. The presence of the surface grafted polymer brush layer, which provides a steric barrier against coalescence and immune recognition, necessitates the use of a polymeric spacer between the ligand and microbubble shell. In this study, we interrogated the influence of microbubble surface architecture (i.e., the ligand location with respect to the polymer brush layer) on binding efficiency in static and flow conditions with and without the presence of ultrasonic radiation force. We engineered the surface architecture by incrementally changing the thickness of the steric brush layer with a constant spacer length. We show that the surface architecture can be modulated to facilitate binding under ultrasonic radiation force while minimizing ligand exposure to the milieu. These results indicate that surface architecture can be tailored to yield properties that are ideal for each targeting modality.
M.A. Borden, None.
Abstract ID: 349
Our goal is to create a strategy for simultaneous molecular imaging and localized drug delivery using engineered delivery vehicles and ultrasound energy. These drug delivery vehicles, referred to as acoustically active lipospheres, consist of a small gas bubble surrounded by a thick oil shell and are enclosed by an outermost lipid layer. A variety of hydrophobic drugs can be solubilized in this lipid-oil complex. Ultrasound pressure, termed radiation force, can be used to localize these vehicles near the blood vessel wall, and this can be followed by a higher intensity pulse to fragment the vehicle and transfer the contents to the endothelium. The delivery vehicles are displaced a distance on the same order of magnitude as typical microbubble contrast agents when exposed to ultrasound pulses. Blood velocity ranges from 1 to 10 mm/s in the microvasculature, therefore, an agent in a capillary can be insonified by a transducer with a 1 mm focal beam width for 0.1 to 1 seconds. With this exposure time, the agent can be displaced 1 to 2 mm, depending on the ultrasound parameters. Delivery vehicles containing either Sudan Black or 5-dodecanoylaminofluorescein within the oil layer are exposed to ultrasound in cell chambers or flowing vessels. Fluorescent intensity greater than the background level increased more than 10-fold with the application of radiation force and fragmentation, both over no exposure and over fragmentation pulses alone (both P < 0.001), implying that radiation force is necessary to bring the AALs into the proximity of the cell monolayer before its destruction in order for the fragments to remain adherent to the cell layer. In flowing vessels, molecularly-targeted agents are displaced towards the vessel wall by radiation force and remain adherent to the vessel wall until a fragmentation pulse is applied. Upon fragmentation, AAL contents are dispersed over the endothelium.
M.J. Shortencarier, None.
Abstract ID: 350
Off-resonance (OR) image 1 Day after labeling (number of the cells: 0.45 million)

OR image 2 days after labeling (number of the cells: 0.9 million)

OR image 4 days after labeling (number of the cells: 3.2 million)

Relationship between the area of signal enhancement and the number of the labeled cells. The solid line connects average areas in each cell population.

T. Arai, None.
Abstract ID: 351
Cationic liposomes are effective vector systems for gene therapy in animal models of benign or malignant diseases. This nonviral gene transfer system has been shown to be safe and nontoxic in human melanoma model. In this study, we visualize gene transfer with cationic liposome using luciferase and sodium iodide symporter as an imaging reporter gene.
pcDNA-Luc and pcDNA-NIS, vectors having the luciferase (Luc) and human sodium iodide symporter (NIS) gene were established. The expression of reporter gene was controlled by the cytomegalovirus (CMV) immediate-early promoter and the bovine growth hormone (BGH) polyadenylation signal. Cationic liposomes and DNA plasmid were mixed with molar ratio of plasmid DNA to cationic liposome of 4: 1. Liposome-plasmid DNA complex was injected via tail vein or directly into the liver using a 30 gauge needle. The scintigraphic images for NIS expression were obtained after injection of Tc-99m pertechnetate with a gamma camera. The optical images were acquired with an IVIS system (Xenogen, Co.) after injection of luciferin on 2 days postinjection of liposome-plasmid DNA complex. The luciferase and NIS gene expression were evaluated by RT-PCR in the organs including the liver, spleen, lung and muscle.
In optical image study, the high intensity was observed in the liver and spleen of pcDNA-Luc injected mice through tail vein. In case of direct injection into the liver, luciferase gene was locally expressed at the injected site on the liver. But in scintigraphic image, radioactivity of the liver was lower than optical intensity in luciferase images. We confirmed to the expression of NIS and luciferase genes in the liver and spleen by RT-PCR.
This dual imaging reporter gene using NIS and luciferase gene can be used for monitoring the liposome mediated gene transfer.
Y. Lee, None.
Abstract ID: 352
D.S. Wang, None.
Abstract ID: 353
J.P. Weichert, None.
Abstract ID: 354
Molecular imaging with ultrasound employs contrast agents such as microbubbles, nanoparticles, or liposomes coated with ligands specific for receptors expressed on cells at target sites such as angiogenesis, inflammation, or thrombus. The concentration of contrast agents at a target site enhances the ultrasound signal received from that site, promoting ultrasonic detection and assessment of the target. Acoustic radiation force has been shown to cause localization of contrast agents in-vitro and in-vivo, and we show that acoustic radiation force can be used to push targeted agents to a vessel wall, greatly increasing the number of agents binding to available surface receptors. A theoretical evaluation of the magnitude of acoustic radiation force demonstrates that it is possible to displace micron-sized agents physiologically-relevant distances. Experiments demonstrate that the number of biotinylated microbubbles adherent to a synthetic vessel coated with avidin increases as much as 20-fold when acoustic radiation force is applied. Additionally, the adhesion of contrast agents targeted to αvβ3 expressed on human umbilical vein endothelial cells (HUVEC) increases 27-fold within a mimetic vessel when radiation force is applied. The image signal-to-noise ratio in a phantom vessel is observed to increase substantially using a combination of radiation force and a targeted contrast agent, over use of a targeted contrast agent alone.
P. Dayton, None.
Chemistry of Multifunctional Probes
Abstract ID: 355
A new diethylenetriaminepentaacetic acid (DTPA) derivative (p-succinic amido-benzyl-DTPA, or DTPA-Bz-NH-SA) was synthesized and applied to solid phase peptide synthesis. Two DTPA derivatives were successfully conjugated to the cyclic peptides, DTPA-Bz-NH-SA-c(KRGDf) and DTPA-Bz-c(ERGDf), on solid support with high efficiency. The approaches can be readily applied to the synthesis of DTPA-conjugated peptides containing free amino or carboxyl or hydroxyl group. Compared to the conventional cyclic DTPA dianhydride method, the technique reported here had all five acetic acid residues in DTPA available for chelation with raidometal ions. Furthermore, the conjugation reaction can be carried out on either N-terminal or side chain employing solid phase peptide synthesis. Thus, the reported procedures overcomes the problem encountered with p-SCN-Bz-DTPA, which is not suitable for the introduction of DTPA moiety to peptide using solid phase peptide chemistry because the thiourea bond formed between SCN and a-NH2 is susceptible to Edman degradation in acidic cleavage conditions. The reported method should be applicable to the conjugation of DTPA to a variety of peptides directly on solid support.
W. Wang, None.
Abstract ID: 356
The investigation of cyanine dyes as contrast agents in optical imaging has attracted considerable interests recently. For in vivo biomedical imaging applications, fluorophores should emit fluorescence signal between 700–900 nm, ideally towards longer wavenumber, should have high quantum yield, contains functional groups for derivatization, and should be stable under a diverse reaction conditions. Here we describe facile synthesis of two new amine-reactive indocyanine NIR reagents with excellent optical and physicochemical properties. The carboxylic acid groups of the fluorophores could react with amines in biomolecules by means of N-hydroxysuccinimide activated ester. Four sulfonic groups introduced to the aromatic rings of the benzindocyanine moiety afforded enhanced water solubility. The tetramethinine moiety gave increased excitation and emission wavenumber for the fluorescent signal, being 763 nm and 793 nm, respectively. The fluorophores were used to synthesize conjugates containing one and two cyclic RGD pentapeptide. Both conjugates demonstrated successful imaging of integrins in melanoma xenograft models. Furthermore, the dimeric conjugate appeared to have greater receptor binding affinity than the monomeric conjugate. The fluorophores reported here may find diverse applications in labeling of biomolecules (Supported by NIH grants R01 EB00174, Prostate Cancer SPORE Career Development Award CA90271 and John S. Dunn Foundation).
W. Wang, None.
Abstract ID: 357
Bone imaging is one of the most frequently performed radionuclide procedures. It can be used to diagnose osteomyelitis, septic arthritis, multiple myeloma, and bone metastases. Contrast agents for bone imaging used in the clinics include 99mTc-labeled sulfur colloid, 99mTc-methylene diphosphonate, 18F-fluoride etc. In this study, we report the use of poly (L-glutamic acid) (PG) as a bone-seeking imaging agent. PG polymers of different molecular weights were labeled with a near-infrared dye ICG or with 111In-chelated dimethylenetriaminepentaacetic acid. In vitro, PG having molecular weight of 6,000 bound tightly to hydroxyapatite. Both in vivo optical imaging and gamma scintigraphy studies of labeled PG revealed high level of accumulation of the polymer in the bone of mice after its intravenous injection. Biodistribution studies confirmed the imaging findings. Our data suggest that PG polymer may be useful as a carrier for targeted delivery of both therapeutic and diagnostic agents to the bone (Supported by John S. Dunn Foundation and Prostate Cancer SPORE Career Development Award CA90271).
X. Cao, None.
Abstract ID: 358
Recently we demonstrated the use of MRI in detecting HER-2/neu receptor expression in an animal breast cancer model using a novel two -step avidin-biotin system. Here we have extended this concept to a three-step approach to increase sensitivity of detection. We performed MRI studies of human HER-2/neu expressing breast cancer BT-474 cells grown as xenografts in SCID female mice. HER-2/neu receptors were pre-targeted with biotinylated anti-HER-2/neu mAb Herceptin (6mg/kg). After 24h mAb were chased by avidin (20 mg/ml) that (i) cleared circulating mAb from the vasculature and (ii) labeled mAb attached to the tumor receptors. Pre-labeled receptors were imaged with biotinylated albumin-GdDTPA contrast agent (300mg/kg) using quantitative T1 MRI. Specific accumulation of the contrast agent in HER-2/neu expressing tumors pre-targeted with the specific biotinylated antibody was demonstrated (Fig.1). Pharmacokinetics of the individual components was also monitored with micro-PET using 64Cu-labeled avidin, and with micro-SPECT imaging using 125I-labeled Herceptin. We have demonstrated efficient clearing of circulating biotinylated mAb's with avidin chase and its feasibility for generation of MR contrast using an appropriate biotinylated MR contrast agent. Quantitative T1 maps allow determination of the accessible receptor density that can be an important parameter for planning anti-cancer immunotherapy. In comparison with MRI, nuclear imaging modalities PET and SPECT provided dramatically higher detection sensitivity, however the spatial resolution was significantly lower than for quantitative T1 MR imaging. This three-step approach can be extended to other biotinylated imaging platforms with higher T1 or T2 relaxivity.

T1 maps of BT-474 tumors imaged with (a) biotinylated and (b) non-biotinylated Herceptin
D. Artemov, None.
Abstract ID: 359
Optical coherence tomography (OCT) is a real-time cellular resolution imaging modality providing superior depth penetration (typically 2mm) over light microscopies through heterodyne detection. This technology may be particularly suited for understanding tissue transport and extravasation of targeted drugs. However, the development of agents exhibiting high OCT contrast cannot rely on incoherent (e.g. fluorescent) light reporters. We have investigated several classes of OCT contrast agents including plasmon-resonant gold nanorods, near-infrared (NIR) absorbing dyes, magnetically-inducible nanoparticles, ferrofluid-filled microspheres, and nanoliposomes. A 3×5mm resolution, 129dB SNR, Ti:sapphire laser-based OCT imaging system centered at 800nm with single-mode fiber interferometer has been employed. Plasmon-resonant nanorods and NIR dyes are detected using spectroscopic OCT to measure the agent-specific absorption. Cross-sectional imaging of dye with characteristic absorption in the shorter wavelengths within the OCT light source spectrum (nominally <800nm) has been demonstrated within a botanical sample by computing the centroid shift of the spectrum of the backscattered light.
Magnetically-induced scattering changes in ferromagnetic agents allow magnetic-specific OCT imaging. Modulation of an external magnetic field is implemented through the addition of a small electromagnet to the imaging arm of the OCT system. With this system we demonstrate tracking of magnetically-labeled macrophage cells in a tissue scaffold, imaging of agent diffusion after topical application of ferrofluidic protein microspheres to an in vivo Xenopus laevis tadpole, and imaging of interstitially injected nanoparticle iron oxide ferrofluid within a mouse.
Additionally, we have investigated the optical backscattering properties of liposomes and synthesized magnetoliposomes with potential toward magnetic-specific contrast in OCT. Molecular targeting of fluorescent nanoliposomes in vitro to HT29 cells is achieved through surface functionalization with the IGF-R1 Mab. Known bioconjugation techniques can be used to provide molecular specificity to any of these OCT contrast agents which will comprise a powerful new tool for molecular OCT imaging in real-time.
A.L. Oldenburg, None.
Abstract ID: 360
Thiol specific reagents provide a unique avenue for the attachment of radiometric and fluorometric probes to proteins and peptides. A new family of bifunctional linkers containing a tridentate donor set for the {M(CO)3}+ (M = Tc, Re) core and a thiol-reactive maleimide group have been prepared conveniently and in good yield from the reductive alkylation of two equivalents of aldehyde with a series of amino alcohols followed by the introduction of the maleimide under Mitsunobu reaction conditions. The tridentate donor groups are generally aromatic heterocycles, such as pyridyl, quinoline etc. and carboxylates. All the rhenium complexes were characterized spectroscopically by IR, NMR and ESIMS and structurally by X-ray crystallography. The bisquinoline derivatives of rhenium complexes show excellent fluorescence properties with long emission life times and high anisotropy. Initial labeling experiments were carried out using a thiol containing tripeptide glutathion (GSH), a thiolated oligonucleotide, bovine serum albumin and human serum albumin. The corresponding 99mTc complexes were also prepared in good yield as expected. The facile synthesis of these new set of hetero bifunctional linkers coupled with their high affinities towards thiol containing biomolecules and the complexation with {M(CO)3}+(M = Tc, Re) cores make them ideal candidates for biomedical imaging applications.
S. Ray Banerjee, None.
Abstract ID: 361
The use of bifunctional chelators has become commonplace in nuclear medicine and MRI since the introduction of DTPA-bisanhydride. A much richer variety of bifunctional chelators offering differing conjugation chemistry and differing coordination chemistries are now available. One of the more effective functional groups for conjugating a chelator to a second component is the aryl isothiocyanate. Reacting under mild conditions this group retains a resistance to hydrolysis not shared by its competitors, such as active esters. For many applications ligands that incorporate aryl isothiocyanates are now the state of the art of bifunctional chelators.
A second consideration is the proper choice of metal ion binding function. Different medical applications have different requirements for the ligating function of a bifunctional chelator. For instance, if the agent is to be used in PET imaging, the speed with which a suitable radionuclide could be incorporated into the ligand is critical. On the other hand, other features such as the rate of water exchange may be a more important consideration for MRI applications. This poster will discuss recent advances made in this field. New bifunctionals that offer both faster and slower water exchange rates will be presented. These are ideal for either conventional T1 shortening MR agents or the new PARACEST agents, respectively. Results describing in more detail some more established bifunctional chelators such as substituted DOTA and DTPA derivatives will also be presented.

M. Woods, None.
Abstract ID: 362
We have successfully developed a series of bifunctional chelates derived from single amino acids and various biomolecules for complexation of the {M(CO)3}1+ core (M = Tc, Re). In this work, we have coupled the amino acid chelators with nucleoside derivatives to provide novel potential inhibitors for nucleoside kinases and as well as thymidine analogs. These ligands form robust chelate complexes of both rhenium/technetium making them suitable for therapy and diagnostic applications. Furthermore, the nucleoside-quinoline derivatives provide luminescence enhnacing overall biochemical and diagnostic utility. These features make them ideal candidates for radiopharmaceuticals and fluorescent probes based upon the {M(CO)3}1+ (M = Re, Tc) core.
Thymidine and uridine were modified at the C2′, C5′ ribose positions to form amine analogues of the nucleosides. Following direct amination with NaBH(OAC)3 in DCE and standard coupling procedures with HOBT-EDCI in DMF, a new series of bifunctional chelators derived from nucleosides has been prepared and the rhenium tricarbonyl complexes have been isolated. The ligands and rhenium complexes were obtained in good yields and characterized by common spectroscopic techniques including 1D and 2D NMR, ESI-MS, IR and luminescence spectroscopy. All metal complexes are remarkably stable toward histidine and cysteine challenges. Syntheses and characterization of thymidine, uridine and adenosine at base positions and biodistribution studies are underway.
L. Wei, None.
Abstract ID: 363
We have developed a novel magnetic iron oxide nanoparticle (MION)-based probe for detection of oligonucleotide hybridization. A PEG-phospholipid micelle-based coating was used to achieve water solubility, uniform size distribution, and functionalized surface for conjugation (Nitin et al., JBIC, in press). In this design, DNA oligonucleotides of varying length were conjugated to the functionalized MIONs at both 5′ and 3′ ends using chemical crosslinkers to create a random-coil or stem-loop hairpin structure (see Figure 1). Hybridization to complementary oligonucleotides induced a conformational change from the closed structure (Fig. 1A) to the open, double-stranded structure (Fig. 1B), resulting in an increase in the T2 relaxation time as measured by a 0.47 T Bruker Minispec Analyzer MQ20. Control experiments were performed with single-MION conjugated oligonucleotides under the same conditions to determine their contribution to the background noise. Numerical simulations (using a Monte Carlo technique) were done to further understand the effect of the distance between MIONs on relaxation characteristics of such magnetic probes. Experimentally observed increases in T2 relaxation upon increase of distance between bound particles were consistent with the results of Monte Carlo simulations. Previous work has shown (e.g. Josephson et al., 2001 Angew. Chem. Int. Ed.) that clustering of magnetic nanoparticles is an effective method of detecting oligonucleotide hybridization. In this work, we have compared our detection method based on structural changes of the probe with clustering-based hybridization detection techniques. This direct comparison will help further elucidate the mechanisms and possible applications of molecular magnetic switches for molecular imaging using MRI.

N. Nitin, None.
Abstract ID: 364
We describe a new material for the use of labeling and tracking cells in vivo. We report here the construction and characterization of a dual-labeled Gadolinium-Rhodamine nanoparticle. These nanoparticles are constructed from lipid monomers - sonicated and photolyzed to form polymerized liposomes. Cells are efficiently labeled with these nanoparticles and show no signs of toxicity with their uptake. We have inoculated labeled tumor cells subcutaneouosly into the flanks of C3H mice, and have been able to image this tumor via MRI and optical imaging. Furthermore, the labeled tumor cells can be visualized via fluorescent microscopy after tissue biopsy. Our results suggest that these nanoparticles could be used to track cells in vivo, and by using this basic chemical platform, this nanoparticle can be modified with different fluorophores and targeting agents.
K. Vuu, None.
Abstract ID: 365
We have synthesized a novel PDT agent on which we have performed preliminary cellular studies. In addition to its original role in treating cancer, this agent is designed to directly assess PDT-induced apoptosis by monitoring the caspase-3 activity in the treated cancer cells.
In this novel PDT-BIAR design, the molecule contains 1) a photosensitizer (pyropheophorbide) that produces singlet oxygen upon irradiation and also emits fluorescence; 2) a fluorescence quencher (BHQ3) that is not able to quench the production of singlet oxygen; and 3) a linker that is a nine-amino acid caspase-3 substrate (GDEVDGSGK). Once the PDT-BIAR is localized inside the tumor, the light activated photosensitizer produces the cytotoxic singlet oxygen and triggers a series of apoptotic events. One of the early characteristics of apoptosis is the activation of caspase-3 enzyme. Thus, when PDT-induced apoptosis occurs, caspase-3 will cleave the peptide linker between the photosensitizer and the fluorescence quencher. Once separated, the quencher is no longer able to serve its purpose and apoptotic cells will be visible by the fluorescence of the photosensitizer (Fig. 1). We observed a more than 8-time fluorescence increase after the caspase-3 cleavage in vitro. We have also confirmed the intracellular localization of this new PDT agent in human hepatoblastoma G2 (HepG2) cells.
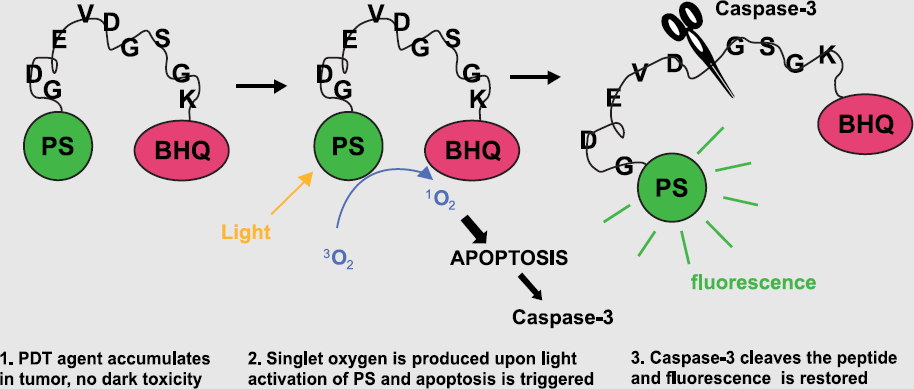
K. Stefflova, None.
Abstract ID: 366
We designed a new photodynamic therapy (PDT) beacon based on singlet oxygen quenching and activation. This PDT beacon consists of an antisense oligonucleotide (AS-ON) sequence complementary to mRNA specific for the target cancer cells (Fig. 1). This beacon contains a single-stranded DNA that forms a stem-loop structure. The loop sequence is an AS-ON. The stem consists of two complementary arm sequences that are on either side of the loop sequence. A dye (D) and a quencher (Q) are attached to the end of each arm sequence respectively. Proximity of the dye and the quencher quenches fluorescence and photoreactivity of the dye by energy transfer. In the presence of the tumor specific mRNA, the beacon hybridizes with the mRNA, disrupting the hydrogen bonds of the stem. The quencher is removed from the vicinity of the dye. Upon irradiating with light, the dye emits fluorescence and generates cytotoxic singlet oxygen. We have synthesized the first PDT beacon targeting c-raf-1 mRNA and have verified that the fluorescence and singlet oxygen production of this beacon was restored after hybridization with the target mRNA. This molecular beacon serves both as a directed photodynamic therapy agent and as a tumor specific diagnostic agent.

Schematic of molecular beacon PDT targeted at a specific mRNA in the tumor cells.
J. Chen, None.
Abstract ID: 367
Targeted fluorescent dyes, especially in the near infrared range, are among the most promising tools for molecular imaging of human disease. NIR fluorescent dyes have been successfully used in a number of novel imaging modalities in animal models. As a new development, fluorescent nanoparticles known as quantum dots have been introduced into imaging as a alternative, brighter and more stable fluorophore. We have explored the capabilities of quantum dots for imaging on a cellular level. As with other dye conjugates, efficient binding and high accumulation within the cell are prerequisites for sufficient signal/noise ratios. Therefore, we studied binding, endocytosis and trafficking of ligand-modified as well as unmodified quantum dots in human tumor cell lines and compared efficiency of uptake, persistance of the fluorescent signal and subcellular movement. As quantum dots are composed of potentially hazardous semiconductor and heavy metal compounds, toxicity is an issue with respect to potential imaging modalities in humans. We therefore evaluated the toxicity of several different quantum dots in a cell culture model. Given the high fluorescence intensity of these nanoparticles, doses required for efficient imaging are very low and toxicity in cell culture was low or undetectable in these concentrations. However, systematic studies in animal models have to be conducted to explore this in detail.
C. Grotzinger, None.
Abstract ID: 368
There is burgeoning interest in the generation of gold nanoparticles (AuNPs) and studies of their photo-physical properties due to their potential applications in biomedical optical imaging and bio-sensor design. Gold is capable of serving in the unoxidized state at the nano-level where as most of the less noble metals will be oxidized to a depth of several nanometers or more, often obliterating the nano scale component. In particular, the generation of AuNPs in biologically benign media is pivotal for in-vivo imaging of tumors. A recent discovery from our laboratory on the generation of highly fluorescent and photo stable gold nanoparticles, prepared using a novel peptide host in aqueous media, has allowed conjugation of nanoparticulate gold labels to a wide spectrum of bioactive molecules. The average size of the AuNPs is 15–20 nm (± 10%) as determined by transmission electron microscopy (TEM). The maximum of the plasmon absorption spectrum, λmax, is at ~ 553 nm. Bioconjugation of AuNPs involved complexation reactions with cysteine. Cysteine was chosen for the initial investigation since it is a common amino acid in several biologically relevant proteins and tumor avid synthetic peptides. A simple technique is employed to determine the amount of cysteine needed to conjugate and saturate the AuNPs. At low cysteine concentrations (~0.01mM), few AuNPs were conjugated with cysteine, therefore aggregation was observed upon addition of NaCl. At optimal cysteine concentrations (~ 8.5mM), all AuNP sites are saturated with cysteine. At this end point in the AuNP-cysteine titration, further additions of NaCl causes no significant aggregation. At very high cysteine concentrations (~0.5 M), self assembly of CAuNPs ensued, resulting in micro-sized particulates. These photo-physical properties of CAuNP are corroborated by TEM measurements. Detailed bioconjugation studies, photo-physical properties, in-vitro stability results and their implications in biomedical optical imaging will be discussed.
V. Kattumuri, None.
Abstract ID: 369
Gold and Silver nanoparticles have become the corner stones of nanoscience and nanotechnology. Powerful surface plasmon absorption of nanoparticulate gold and silver play critical roles in their applications in biomedical optical imaging and biosensor design. Development of smart materials which include: optical imaging agents, and bio implantable photo markers depend on photochromic properties of embedded nano materials. The high surface area coupled with biocydal (antimicrobial) properties of silver nanoparticles would be ideal for in vivo applications involved in surface enhanced optical signals for tumor/other diseases imaging. Despite numerous biomedical/biotechnological/societal applications of gold and silver nanoparticles, technologies that produce and stabilize gold and silver nanoparticles under mild, non toxic, renewable and economically feasible conditions are still lacking. For large scale nanotechnological applications and for new optical imaging technologies utilizing gold and silver nanoparticles, it is imperative to discover new technologies to produce and stabilize gold and silver nanoparticles under biologically benign media.
The current abstract provides details of a novel synthetic strategy for the production and stabilization of gold and silver nanoparticles in aqueous media. This new technology utilizes amino acid conjugate (MUNANO) that can be produced in large scales using industrially produced chemicals. The gold and silver nanoparticles are produced within 30 minutes by simple mixing of MUNANO with NaAuCl4 or AgNO3 at 25°C. MUNANO is non toxic and serves as a mild water soluble reducing agent to convert the gold or silver salts in to the corresponding nanoparticles at physiological pH in aqueous media. Details of our results on the development of uniform sized gold (20-25nm) and silver (15-20 nm) nanoparticles, their stabilization in aqueous media and potential biomedical imaging applications will be discussed.
K.V. Katti, None.
Abstract ID: 370
Malignant melanoma is the sixth most commonly diagnosed cancer with an increasing incidence in the United States. Recently, studies have shown α- melanocyte stimulating hormone (α-MSH) analogues as potential candidates of melanoma imaging and therapy agents. In previous studies, 99mTc and 188Re-(Arg11) CCMSH exhibited excellent tumor targeting and retention properties in both murine and human melanoma bearing mouse models. Understanding the structural basis and peptide-receptor binding mechanism is a crucial step towards developing novel agents for early detection or therapeutic treatment. In this study, we implied high resolution nuclear magnetic resonance spectroscopy (NMRS) to characterize the solution structure and binding surface property of Re-(Arg11)CCMSH and DOTA-Re(Arg11)CCMSH. Three-dimensional high resolution structures were determined by multidimensional 1H and 13C NMRS. The structures of Re-(Arg11) CCMSH showed that the molecule forms a globular stable body in aqueous solution. The interaction between the C-terminus Val13 and His6 forms a turn which both stabilizes the Arg11 and exposes its sidechain towards the binding surface. The intrinsic binding site (His6-Phe7-Arg8-Trp9) forms a convex shaped patch with the sidechains exposed on the surface. The site-specific substitutions on site Arg11 with Lys, Met, Glu, Gln, or Orn alter the biodistribution and decrease the tumor uptake. We found that charge distribution on the binding surface may play a crucial role to the tumor targeting property. The structures of Re-(Arg11) CCMSH were compared with structures of DOTA-Re (Arg11) CCMSH.
L. Ma, None.
Abstract ID: 372
The differential expression of the αvβ3 integrin on angiogenic vessels provides an opportunity for targeting and in vivo imaging. Strategies are being sought to incorporate optical or paramagnetic imaging beacons into αvβ3-targeted nanoparticle (NP) agents as molecular imaging tools to improve angiogenesis detection. C57/BL6 and nude mice were subcutaneously implanted with murine colon adenocarcinoma (MC38) and human melanoma (M21) cells, respectively. After tumors reached approximately 1 cm, we injected mice with either: 1) dual-labeled (FITC-Gadolinium DOTA) αvβ3-targeted NPs with RGD targeting ligands or 2) non-targeted NPs of similar composition without the RGD sequence. Dynamic MRI sequences (4.7T) were obtained for 15 minutes post-injection. T1-weighted multi-slice sequences were attained at 2,4,6,8,12, and 24hrs post-injection to assess time of maximum enhancement. Using an optical imaging station (Xenogen IVIS), we acquired post-injection dynamic sequences with follow-up imaging at similar time points. Some mice were injected with an endothelium-avid, red fluorescent lectin 15 minutes prior to tumor resection. Confocal microscopy, optical imaging, and MR microscopy (7T vertical) of isolated targeted and non-targeted NP treated tumors were attained and compared. Overall, MR enhancement of intact tumors was equivocal with, at best, a 20% differential signal increase at 6 hours in M21 tumors injected with 0.025mmol/kg of targeted NP. Dynamic MR sequences post-NP injection did not reach the quality seen with contrast agents such as Gd-DTPA. Optical imaging results demonstrated improved sensitivity versus MRI. Confocal and MR microscopy suggested preferential targeting to endothelium by targeted NP versus non-targeted. Overall, our results suggest that variables such as tumor line, animal model, and NP concentration influence detection capabilities. The low MR signals likely reflect insufficient tumor blood flow needed to accumulate significant contrast agent. Other challenges to the nanoparticle platform include increasing contrast loading in NPs, improving relaxivity, and seeking better, tumor-specific targets that could improve signal-to-noise ratio.
C.M. Castro, None.
Imaging Gene Therapy & Advanced Vector Design
Abstract ID: 374
There have been reported quite a few in vivo reporter gene systems. Each system has its own advantages and disadvantages and none can satisfy all the criteria required.
We have been developing a new PET reporter gene system with FES and human estrogen receptor ligand binding domain(hERL). The system was designed to be able to work in wide variety of tissues including the brain, minimized with the physiological effect brought by the reporter expression, and safe to be used in human. We constructed a plasmid, pTIER, to co-express a model therapeutic gene and hERL and evaluated the potential of the reporter gene system in gene therapy monitoring using cell culture and mouse models. Cos7 cells transfected with pTIER expressed hERL in parallel to the therapeutic gene and accumulated 3H-estradiol at the level of about 170,000 molecule/cell or 3 pmole/mg protein in 10 min incubation. The uptake of 18F-FES by pTIER transfected cells was about 80 times higher than that by the cells transfected with a control plasmid, and the concentration of 18F-FES in pTIER transfected cells was about 140 times higher than that in the media. When the pTIER transfected cells were injected into mouse abdominal cavity and radioligand, either 3H-estradiol or 18F-FES, was injected from the tail vein, the cells took up significantly higher amount of the radioligand than the control cells, which demonstrates that hERL expressing cells can accumulate radioligand at a high level in vivo. We are now trying in vivo transfection of the plasmid and imaging of the reporter gene expression by PET.
So far the results from our experiments look promising and we expect that this system will make a favorable addition to the choice of in vivo reporter gene imaging systems.
T. Furukawa, None.
Abstract ID: 375
We tried to analyze the increased radioactive iodine uptake of differentiated thyroid cancer cells (ARO cells) by adenovirus-mediated human Na+/I− symporter (hNIS) gene transfer.
Y. So, None.
Abstract ID: 377
Non-viral gene transfer lacks the efficacy of virus-mediated transduction while having potential advantages of lower immunogenicity and toxicity. The goal of our research was to investigate the potential of novel hyperbranched dendron (HD) polymers in cell trasnfection in vivo and in vitro using bioluminescent imaging. HD polymers were synthesized using low molecular weight polyethyleneimine core (BPEI) as a starting material. As a result of successive attachment of ethyleneimine moieties to PEI core, the relative ratio of linear-to-branched structures was lowered from 1.17 to 0.70. We found that the more extensive branching of PEI enables condensing plasmid DNA into nanostructures with a size of 70–100 nm. The obtained complexes were stable at least for 3 weeks at 4o C. The HD-DNA complexes prepared using secondary and tertiary amine containing dendrons exerted a very low cytotoxicity in vitro during a co-incubation with cells for 48 hours. Using firefly luciferase as a marker of protein expression, we established that HD complexes were efficient in transfecting cells in the presence of serum. Under optimized conditions the transfection activity at the nitrogen-to-phosphate (N/P) ratio of 6 was approximately six times higher than that of the commercially available polycationic transfection reagent. Bioluminescent imaging of in vivo gene expression using luciferase reporter gene showed the increase of the signal in the liver and in submandibular lymph nodes in live mice. In conclusion, in vivo gene expression imaging demonstrated the potential of HD polymers that could be potentially useful for lymph node gene delivery.
A.A. Bogdanov, None.
Abstract ID: 378
Strategies to increase the expression in vivo of a therapeutic transgene (Flk-Fc) on a non-replicating adenoviral vector, by combining this virus with a tumor-targeting, replicating adenovirus (dl922/947) were examined. In order to test transgene expression levels, a reporter vector (consisting of an E1A-deleted, non-replicating adenovirus expressing luciferase) was used in combination with the oncolytic vector (dl922/947) both in vitro on transformed and non-transformed cell lines and in vivo in the treatment of human tumor xenografts in immunodeficient mouse models. Because the tumor-targeting deletion within dl922/947 is within the CR2 region of the E1A gene, recombination events occurring between these two viruses cannot produce a new virus. In vitro studies initially demonstrated that dual infection with both viruses allows re-packaging of the non-replicating virus in transformed cell lines capable of supporting replication of the oncolytic vector. Further studies used in vivo bioluminescent imaging (BLI) to study the duration and levels of luciferase expression following combinations of these therapies. Treatment regimes using the viruses delivered together or by different routes and at different times were evaluated. We observed that combination therapy could be used to boost both the levels and duration of gene expression and that repeated treatment with dl922/947 could increase the levels of gene expression. Although this increased gene expression is likely caused by co-infection of a subset of cells followed by repackaging and spread of the non-replicating vector, under certain conditions the boost in gene expression was found to occur at times before viral replication could have occurred. Further studies, using the anti-angiogenic therapeutic gene therapy vector (Ad-Flk-Fc), in combination with the oncolytic vector (dl922/947) have shown that combining these therapies produces anti-tumor effects greater than either treatment by itself.
S.H. Thorne, None.
Abstract ID: 379
Accurate assessment of tumor response to anticancer treatment is crucial for optimization of the treatment and efficacious cancer therapy. Minimally invasive imaging approaches have a great potential to provide the accurate information on treatment response. Small interfering RNA (siRNA) has the ability to interfere biological processes. Anti-VEGF siRNA can induce destruction of the mRNA expressing vascular endothelial growth factor (VEGF) in tumor tissue, resulting in blockage of tumor angiogenesis and regression of tumor growth. We have investigated tumor response to the treatment with an anti-VEGF siRNA using contrast enhanced MRI with a biodegradable macromolecular contrast agent in an animal model.
Biodegradable macromolecular MR contrast agent (Gd-DTPA)-cystine diethyl ester copolymers (GDCEP) was prepared by the copolymerization of DTPA dianhydride and cystine diethyl ester, followed by the complexation with GdCl3. Anti-VEGF siRNA and a cationic polymeric gene carrier were provided by Nitto Denko Tech. Corp. (Oceanside, CA). Female athymic nude mice bearing xenografts of MDA-MB-231 human breast carcinoma were treated with siRNA or siRNA-polymer complex, polymeric carrier and saline by intratumoral injection at every other day for a period of 21 days. Tumor response to the treatment was evaluated by contrast enhanced MRI on a Siemens Trio 3T scanner. The contrast enhanced MRI resulted in clear visualization of the tumor boundary and tumor size. It revealed that the treatment with the siRNA-polymer complex gave rise to significant tumor regression as compared with the treatment with saline or polymer and siRNA alone. The animals treated with the siRNA-polymer complex had much smaller tumors than those treated other treatments. MRI results were validated by biopsy-based evaluation including the measurement of tumor size, tumor microvessel density and VEGF expression. MRI with the biodegradable macromolecular contrast agent is effective for non-invasive evaluation of tumor response to the treatment with anti-VEGF siRNA.
Z. Lu, None.
Abstract ID: 380
Recently, conditionally replicative adenovirus (CRAD) is being more used as a strategy to overcome low gene transfer rate in gene therapy. CRAD can selectively replicate and also complement replication of other adenoviruses in tumor cells. In these studies, we evaluated that the co-transduction of replication defective reporter viruses with CRAD enhances transgene expression in reporter viruses.
We used CRAD (Δ24RGD) that produces mutant E1 which will not attract Rb but retains viral replication competence in cancer cells with a defective pRb/p16. Ad-luc (luciferase) and ad-hNIS (human sodium iodide symporter) are E1 deleted replication defective adenoviruses. These two reporter viruses were co-transduced to human lung cancer cell lines (A549, NCI-H460) with CRAD, respectively. After incubation for 24 hours, luminescence imaging and 125I uptake assay were performed.
All the combination of CRAD and E1 deleted reporter viruses increased the transduction efficiency of luciferase and hNIS depending on the titer of CRAD in both cell lines, A549 and NCI-H460. Combination of CRAD with ad-luc and ad-hNIS in A549 showed increase with values up to 1.8 and 1.6 fold compared to ad-luc and ad-hNIS alone respectively. In NCI-H460, combination of CRAD with ad-luc and ad-hNIS each showed increase of 1.3 and 1.1 fold compared to ad-luc and ad-hNIS alone respectively.
Combination of CRAD with replication-defective reporter viruses induced replication of reporter viruses which eventually increased reporter gene expression significantly compared to reporter virus alone.
K. Kim, None.
Abstract ID: 381
Telomerase, an enzyme which lengthens telomeric DNA at chromosome ends, is expressed in the vast majority of human carcinomas and has been implicated as a key factor in tumorigenesis. Since transcription of telomerase reverse transcriptase (hTERT) is the major regulator of telomerase enzyme activity, measuring hTERT expression may be clinically valuable. Previously, the measurement of hTERT expression has only been possible by methods that destroy the cells under examination. Here we present methods of non-invasively measuring hTERT promoter activity. The hTERT promoter is used to drive the expression of a fusion reporter construct containing synthetic humanized Renilla luciferase (hrl), monomeric red fluorescence protein 1 (mrfp1), and a truncated herpes simplex virus type 1 thymidine kinase (ttk), allowing for visualization of hTERT activity by bioluminescence, fluorescence, and radionuclide imaging. When the fusion construct is introduced into cell lines which express hTERT (293T and MCF7), all three reporter genes are highly expressed. Cell lines lacking hTERT transcription (U-2 OS and Saos) show no significant reporter gene expression. Treatment of 293T cells with 5-fluorouracil, a common chemotherapeutic, decreases both hTERT expression and telomerase activity as measured by PCR. These decreases correspond with reduced signal from all three reporter genes, and could be non-invasively demonstrated by both fluorescent microscopy and bioluminescent imaging. Thus, hTERT promoter activity, a key factor in tumorigenesis, can be measured by three different methods using a single reporter construct. The use of this reporter construct will promote the study of telomerase expression in living subjects.
P. Padmanabhan, None.
Abstract ID: 382
Several reporter genes currently available include Aequorea-derived fluorescent proteins (AFPs), luciferases, and herpes simplex virus type 1 thymidine kinase (HSV1-tk) genes. These reporter genes have limited clinical applications because of poor tissue penetration and resolution (optical and bioluminescent) or non-specific background activity of endogenous enzyme (Thymidine Kinase) and the requirement for an expensive radio-labeled probe for imaging (HSV1-tk).
Tc99m O4− is widely used as a radiopharmaceutical in nuclear medicine because of its excellent imaging properties, low toxicity, low cost, and easy availability. Enzymatic reduction and intracellular trapping of Tc99m O4− has been reported in bacteria, including Escherichia coli (E. coli), sulfate-reducing bacteria Desulfovibrio desulfuricans (DD) and in eukaryotic microorganisms, including human parasite Trichomonas vaginalis (TV). In E. coli, the Tc(VII) reductase activity has been identified as hydrogenase 3 encoded by the HycE and HycG genes in the hyc operon. Two genes, HydA and HydB encode for the hydrogenase activity in DD and TV.
We hypothesized that microbial technetium reductases are able to mediate enzymatic reduction and intracellular trapping of Tc99m O4− in bacterial and mammalian cells, when this microbial enzyme is ectopically expressed. In order to further investigate the use of microbial reductases for enzymatic reduction and intracellular trapping of Technetium 99m pertechnetate in the bacteria and animal cells, we have developed a novel reporter gene-probe system for molecular imaging. The hydrogenase genes from the organisms D. sulfuricans, T. vaginalis and E. coli (MC4100 strain) were subcloned and overexpressed in bacterial and mammalian vectors. Bacteria overexpressing the DD HydA and HydB genes showed significantly higher trapping of Tc99m O4− in vitro as compared to control cells. These and other results of in vivo imaging using mouse models will be presented.
S.D. Pandit, None.
Abstract ID: 383
One of the challenges faced in gene-based therapy includes the lack of tissue specificity, which can inadvertently result in toxicity. On the other hand, the activities of tissue specific promoters are generally weak resulting in low-level signals, consequently, low efficacy. Here we report the use of an adenovirus-based two-step transcriptional activation (AdTSTA) system that is capable of boosting the activity of the weak promoter by nearly 1000-fold while maintaining tissue selectivity. To develop therapeutic applications for prostate cancer, we have created adenoviral vectors expressing the firefly luciferase gene (AdTSTA-fl) and HSV1-thymidine kinase gene (AdTSTA-tk), under the control of a prostate-specific TSTA system. Evaluation of luciferase expression after intratumoral injection in LAPC-4 prostate cancer xenografts, revealed optical signal only in the tumor of the TSTA group while liver signal was also detected in the CMV group. Concurring with the optical signals, microPET images also revealed TK expression by uptake of [18F]-FHBG reporter probe, in the tumor of the TSTA group while both tumor and liver expression was observed in CMV group. High TK expression in the liver resulted in elevated liver enzyme ALT post gancyclovir treatment, reflecting liver toxicity and damage. Histological analysis of the liver revealed extensive apoptosis in the CMV group while damage was not observed in the TSTA group by TUNEL staining. Detailed tumor histology using proliferating marker Ki-67, TUNEL and cleaved anti-caspase-3 as apoptotic markers, demonstrated extensive tumor necrosis and apoptosis in both TSTA and CMV group compared to saline controls. In summary, we have demonstrated that the TSTA system can mediate highly amplified and tissue-specific gene expression resulting in effective tumor cell killing and reduced toxicity. Finally, non-invasive imaging modalities prove to be invaluable tools in translational research to monitor gene expression and therapeutic response in gene therapy.
M. Johnson, None.
Abstract ID: 384
Effective gene therapy requires the ability to evaluate the magnitude and duration of regional transgene expression within the tissue of interest. Magnetic resonance imaging methods developed for this purpose have focused on susceptibility-based contrast enhancement that is difficult to quantify. Heteronuclear nuclear magnetic resonance (NMR) spectroscopy permits the absolute quantitation of gene expression as described previously1; however, the sensitivity level is often insufficient to acquire images of regional gene expression. We report on the development of a novel fusion gene reporter that enables the quantitative assessment of regional transgene expression by in vivo fluorine chemical shift imaging (19FCSI). Walker 256 carcinosarcoma (W256) cells were stably transfected with the S. cerevisiae cytosine deaminase-H. influenzae uracil phosphoribosyltransferase fusion gene (yCDiUPRT). Following intravenous (i.v.) injection of 150 mg/kg 5-fluorouracil (5FU), the anabolism of 5FU to fluoronucleotides (FNuc) was observed in mice bearing subcutaneous W256 wild type and W256yCDiUPRT tumors by 19FNMR spectroscopy (n=5) (Figure 1A and B, respectively). The resulting spectral peaks were quantitated and a compartmental model describing the kinetics of 5-FU and FNuc in W256wt and in W256yCDiUPRT tumors was derived there from. Subsequently, two-dimensional 19FCSI was performed on additional mice bearing W256yCDiUPRT tumors following i.v. 5FU injection. A single peak representing the FNuc resonance was observed (Figure 2) and quantitated in relevant voxels. The metabolite concentrations and rate constants determined from the 19FNMR spectra allowed for the evaluation of regional yCDiUPRT expression levels based on the CSI.

T. Gade, None.
Abstract ID: 385
In the past few years, bioluminescence imaging has become an increasingly popular method for monitoring tumor growth, visualizing viral infection and neuronal activities and evaluating progress of cancer treatments in living animals. Its sensitivity and noninvasiveness allow investigators to visualize the clonal expansion and dynamic engraftment of hematopoietic stem cells (HSC) over a long period of time. HSC has been used as an effective target for gene therapy. Obstacles associated with HSC gene therapy often refer to its limited quantity and engraftment efficiency. Understanding the clonal expansion pattern and finding a way to enhance HSC expansion and engraftment benefit greatly to patients involved in HSC gene therapy.
Here we propose that clonal expansion and engraftment of bone marrow cells after transplantation are enhanced under selection pressure. We generated a bicistronic lentiviral vector containing mutated MGMT(P140K) gene and firefly luciferase gene linked by 2A sequence of FMDV. We have shown that lentiviral transduced bone marrow cells provided protection against chemotherapeutic drugs, such as BCNU and TMZ, in vitro. Whole bone marrow cells was collected from BALB/c mice 4 days after 5-FU treatment and transduced with P140K-MGMT-luc lentiviruses at MOI of 1.4. Transduced bone marrow cells were transplanted into lethally irradiated or non-myeloablated congeneic recipient mice at different cell numbers. Initial bioluminescent signal emerged 6–8 days after transplantation in both lethally irradiated and non-myeloablated conditions. Most of the signal emitted from bone marrow, especially in femur, humeri and vertebrae. Intense signal appeared in spleen at days 12–14 after transplantation and disappeared by day 20. Clonal expansion and engraftment enhanced greatly after treatments with BG+TMZ. Overall body bioluminescence intensity increased and maintained compared to untreated recipients. This approach of drug selection indicates active migration, between bone marrow and spleen, expansion and selection of HSC over extended period of time after transplantation.
Y. Lin, None.
Abstract ID: 386
Conventional analyses such as tumor volume and mean survival times are relatively rudimentary and time-consuming techniques to evaluate anticancer therapeutic efficacy. In our studies using the cytosine deaminase/5FC gene therapy strategy wherein cytosine deaminase converts the non-toxic prodrug 5-Fluorocytosine (5FC) to the common chemotherapeutic agent 5-Fluorouracil (5FU), we have utilized non-invasive imaging. We engineered mutant forms of CD which were predicted to either have increased activity (CD-UPRT) or enhanced bystander killing (vp22CD). In culture both of these mutants led to enhanced cytotoxicity. Using 19F Magnetic Resonance Spectroscopy (MRS) and diffusion weight imaging (DWI), we evaluated these different gene therapy strategies in vivo. 19F MRS was used to evaluate CD enzyme kinetics by quantitatively measuring the conversion of 5FC to 5-FU following adenoviral transduction of 9L flank tumors. In addition, DWI was shown to more sensitively identify enhanced cytotoxicity in CD-UPRT expressing tumors compared to CD expressing tumors. This enhanced efficacy of the CD-UPRT construct was not apparent when tumor volumes were evaluated over time. More importantly, prolonged survival of the CD-UPRT treated animals compared to the CD treated animals validated the DWI and 19F MRS results. In conclusion, these results demonstrate the potential of MRS and DWI as powerful tools to provide early detection of therapeutic response and moreover provide a more sensitive reporter of therapeutic efficacy.
K. Lee, None.
Abstract ID: 387
We have developed 19F-MRS imaging techniques for in vivo and in vitro studies of gene-directed enzyme prodrug therapy (GDEPT) using adenovirus as vehicles to carry bifunctional yeast CD/UPRT fusion proteins into cancer cells. In this study a replication incompetent adenoviral vector carrying a cytomegalovirus (CMV)-driven transcription unit of the yeast CDUPRT gene was constructed. Here we report a successful application of 19F-MRS to real time monitor the gene expression and follow up the pharmacokinetics of prodrug 5FC conversion to 5FU and subsequently Fluorinated nucleotides and nucleosides (FNuc) in 20 minutes or less in human glioma D54MG solid tumors (n=15) after mimicking the clinical situation by multiple injections of Ad5.CMV.CD/UPRT virus for 3 days following with a dose of 0.5 mg/kg of 5FC via i.p. injection. The 19F-MRS data were acquired with home-made Faraday shielded solenoidal coils of various diameters 0.5-0.9 cm at a Bruker/Avance 4.7T/40cm animal system. Series of 19F-MRS spectra were typically acquired in 15 min each for approximately 2.5 hrs following the i.p. injection of prodrug 5FC. Some experiments had follow-up for 5–6 hrs to see the utilized 5FC disappeared and temporal increased of FNuc. Fig.1 is a typical example of such a finding. With this scheme of treatment, 5FU (−19 ppm) approached maximal level at approximately 1.5 hr and FNuc greatly increased at 4.5 hr post injection of 5FC. Most of these mouse tumors (n=15) at 24 hrs after a single treatment protocol, the FNuc signal maintained at high level with diminished 5FC and 5FU, the result is consistent with the concept that FNu remains intracellular. Bystander effect was also measured strongly by 19F-MRS in this cell line.

Real time monitoring of the kinetics of prodrug 5FC conversion to 5FU and anabolites FNuc (F-nucleotides and F-nucleosides) in a subcutaneous glioma D54MG after treated with Ad5.CMV.CD/UPRT virus for 3 days followed with 0.5 mg/kg of 5FC i.p. injection. (Ng et al. unpublished data).
T.C. Ng, None.
Abstract ID: 388
S. Ray, None.
Abstract ID: 389
P. Ray, None.
Abstract ID: 390
Cystic Fibrosis (CF), being an autosomal recessive genetic disorder with a single gene defect, seemed an ideal target for gene therapy. Targeted delivery of the therapeutic gene, CFTR to airway epithelium should restore normal function. It is crucial to monitor the gene therapy by tracing genes: how many are actually expressed; and how long the expression lasts. A dual reporter gene approach was adopted to image gene expression in the lungs of CF knockout mice. HSV1-tk and luc were compacted using ploy-K, and mixed in equal molar for delivery. Such compaction is also done for CFTR gene delivery. As controls, naked HSV1-tk and luc were also mixed in equal molar for delivery. In addition, saline was delivery to another group of mice, as a control. Two delivery routes were tested and compared here. In the first method, the gene complex solution was dripped into mouse's left nostril. In the second method, mice underwent tracheostomy so as to bypass nasal breathing, and then the gene transfer complexes were applied to the lung. [I-125]-FIAU was injected 24 hrs post gene delivery and mice were imaged by a gamma camera for five days. D-luciferine was used for bioluminiscent imaging at 24 and 48 hrs post gene delivery. After imaging, animals were sacrificed and lungs excised for both tk assay and luminometer reading. The results showed that intra-tracheal delivery yielded higher gene expression than nasal delivery. Compacted gene delivery produced significantly higher gene expression in the lungs than either naked gene or saline, and image intnesity peaked at about 48 hrs post gene delivery. In general, gamma scintigraphic data agreed with bioluminescent findings about transgene expression in the CF mouse's lungs. Hepatic trapping of radioiodide appeared in gamma images due to dehalogenation of FIAU in vivo. In contrast, luc imaging had low background signals.
A. Ziady, None.
Abstract ID: 391
It is interesting to see that the nucleic acids exhibit long range dependency and self similarity. The present work is wound around the outcome of this self similarity. The concept is multi disciplinary involving information processing, signal processing, genetics, molecular biology, Bio chemistry, control systems and a little bit of electronics.
Analysis shows that it exhibits self similarity to maximize the entropy. In order to maximize entropy, the sequence makes use of differential feedback of different degrees. They form various levels of abstraction and by and large carry redundant information. The large chunks of unused nucleic acid sequence found on the genes can be attributed to this redundant information. This useful property can be exploited in gene mapping where a portion of the missing sequence may be predicted with arbitrarily small errors using Bayesian decision.
Because of abstraction and redundancy, even if a portion of the gene gets broken or destroyed or malfunctioning, it can be repaired or resynthesised using the redundant nucleic acids. This can be induced externally through adsorbents and molecular self assembly or biologically with ‘self generating genes’ that would be sitting in the nucleic acid strand.
M. R. None.
Late-Breaking
Abstract ID: 393
Co-registration of anatomical (e.g. CT and MRI) and functional (e.g. SPET/PET) images is a crucial step in developing multimodality approaches for small animal imaging research. In this work, we present a surface-based image registration algorithm developed to co-register CT and MRI images obtained from a rat brain. The first step of the algorithm is to segment the rat's skull surface in both the CT and MRI images. A semi-automatic 3D region-growing algorithm is developed to detect the skull surface and the 3D Canny edge is used as the supplementary condition to stop region growing. To start the algorithm, the analyst needs to pick the best region seed from several automatically found seed candidates. In the second step, an Iterative Closest Point (ICP) algorithm is applied to register the CT and MRI images. The algorithm iteratively transforms one image until the error between the two images is smaller than a threshold value. The assumption of the ICP algorithm is that one 3D image is rotated and translated relative to the other, reference image, which is not always true for the CT/MRI modality images. The preliminary results are visually checked by superimposing the two images. Future work needs to deal with the non-rigid distortion between images from the two modalities, especially due to the small size of animal images.
Q. Xu, None.
Abstract ID: 394
Circadian rhythms regulate behavioral and physiological processes which allow animals to synchronize to daily changes in their environment. The suprachiasmatic nucleus (SCN), which expresses daily rhythms in utero, has been found to be the master circadian pacemaker in mammals. Transgenic rats carrying the Period1 gene promoter driving firefly luciferase expression (Per1::luc) were used to examine the ontogeny of circadian rhythmicity in real time. Per1-driven bioluminescence was detectable in fetuses in utero as early as embryonic day 10 (E10) and rhythms appeared in whole pups harvested on E12 and maintained in vitro. As early as E16, the isolated anlagen of the SCN expressed circadian rhythms of significantly lower amplitude than the mother, but with a similar period. Robust rhythms were apparent in the E17 liver. Lesions to the maternal SCN on E7 abolished her locomotor rhythms, but did not affect the phasing or amplitude of periodicity in the fetal SCN or liver assayed in vitro on E19. These results indicate that fetal circadian oscillations are detectable early in development and do not depend upon the maternal or fetal SCN for their initiation.
M.T. Saxena, None.
Abstract ID: 395
The successful presentation of MR contrast agents throughout the brain parenchyma would allow investigations of the physiological environment with high spatial and temporal resolution but is hampered by the presence of a blood brain barrier. The present study characterizes the distribution kinetics of charged and uncharged gadolinium contrast agents in the cerebrospinal and interstitial fluids of rat brain. Since the blood brain barrier precludes delivery by vascular routes, agents are infused directly into ventricular cerebrospinal fluid whereby useful quantities of agent occur throughout the brain parenchyma for up to 20 hours after infusion. Experiments are performed with Sprague-Dawley rats and agent distributions in the brain are obtained from MR T1 maps acquired at 9.4T. We demonstrate a linear response in T1-dependent signal changes over a 4-fold range of GdDOTP5− or GdDTPA2− infusate concentrations with maximum agent doses decreasing T1 throughout the brain more than two-fold. These studies provide a method of measuring the exchange properties between cerebrospinal, extracellular, and intracellular fluid compartments. Results are also presented demonstrating the potential of using the pH sensitive T1-relaxivity agent, GdDOTA-4AmP5−, as a reporter of brain pH. Decreases in brain T1 have been observed with GdDOTA-4AmP5− distributed in the brain that are clearly and specifically consistent with global and local pH changes that would occur with global elevated pCO2 challenges and local forepaw stimulation. In summary, the results presented demonstrate a systematic approach to delivering molecular imaging and contrast agents, such as the pH reporting compound, GdDOTA-4AmP, to the brain.

in vivo GdDOTP dose response plot and T1 maps with and w/out GdDOTP

Decrease in T1 due to hypercapnic challenge with pH reporter, GdD0TA-4AmP, distributed in brain
P.A. Schornack, None.
Abstract ID: 396
Assessment of myocardial intermediary metabolism using microPET® and kinetic modeling is currently being validated in rodent models of cardiac disease at our institution. Maintaining insulin and plasma substrate levels relatively constant over time is vital for the reliable assessment of cardiac metabolism of diseased models. Insulin (IN; μU/mL), Glucose (Gl; mmol/L), and Free Fatty Acid (FFA; μmol/L) plasma levels were measured in normal SD rats, either fed normal chow ad libitum (FED; n=4), after an overnight fast (FAS; n = 6), or after an intravenous infusion of intralipid (IL; n=6). Each rat was anesthetized, and catheters were placed for blood sampling (t = 0,1hr, 2hr, 3hr, 4hr) (D1). The rats were recovered and repeat measurements were obtained after 6 days (D2). The variability of plasma IN between all three study groups FED, FAS, and IL were consistently more variable from D1 to D2, but not by a great amount. However, the variability of plasma IN between FAS and IL were significantly different. For all three study groups, FED, FAS, and IL, plasma Gl variability showed no significant differences between study groups, nor between study D1 and D2. The variability of plasma FFA levels between day one and two were not significant. FED rats consistently showed the most variability of plasma FFA levels, while a decrease in variability is shown between FAS and IL study groups. As anticipated, the variability of FAS was consistently less, or did not vary, when compared to the other 2 study groups. Results of this study show that insulin and plasma substrate levels, as well as their variability during a 4 h period are consistent within the same animal after a 6 day wait. Furthermore, the stability of insulin, glucose and free fatty acid plasma levels during the study period varied depending on the perennial state.
L.L. Neff, None.
Abstract ID: 397
Primary bronchogenic carcinomas are thought to be supplied with nutrients via the bronchial circulation of the lung. The high demand of such tumors stimulates angiogenesis within the bronchial circulation. We have developed a method for imaging and quantifying bronchial angiogenesis by examining pulmonary perfusion changes that result from left pulmonary artery (LPA) ligation in rats. Structural and functional images of the animals were acquired post ligation surgery with a dual-modality micro-CT/SPECT system developed in house using two MAA injections. First an aortic injection of 99mTc labeled macroaggregated albumin (MAA) was made and resulted in MAA accumulation in the capillaries of the systemic circulation, including the bronchial circulation. After all activity decayed, MAA was injected intravenously via a femoral vein resulting in MAA accumulation in the lung via the pulmonary circulation. The reconstructed SPECT and micro-CT volumes were co-registered and the lungs in the SPECT volume were segmented using the micro-CT volume. MAA accumulation within each lung was then determined and the ratio between the aortic and intravenous injections was computed as a measure of bronchial flow. Bronchial flow as a percentage of cardiac output increased from ~2% for control rats to ~4%, 8% and 14% for LPA ligated rats at 10, 20 and 40 days post surgery, respectively (figure 1). A novel imaging method for quantification of bronchial circulation angiogenesis in small animals has been developed and used to quantify the time course of the angiogenesis. This model will be useful for evaluating antiangiogenic treatments in the future.

C. Wietholt, None.
Abstract ID: 398
Due to the increasing development of small animal imaging it has been necessary to investigate a variety of contrast agents for tumor and vascular identification. Many of the studies, carried out at our institution, require microPET and microCT co-registration for anatomical measurements. Moreover, the demand for contrast enhanced CT imaging has significantly increased, requiring us to optimize and make routine a protocol for these measurements. Most commercially available CT contrast agents possess blood clearance properties that are not suited to the timescale of small animal CT protocols. The selected imaging study was chosen to test a commercially available intravenous contrast agent Fenestra™ VC, using the ImTek Inc., microCAT-II scanner in combination with microPET imaging.
Balb/C mice were implanted with EMT-6 tumor cells (5 × 106 / 0.1mL). On the study day mice were anesthetized with inhalant anesthesia, and a central venous catheter was placed into the right jugular vein. Mice were IV injected with 18F-FDG (400μCi), placed in a special designed tray for co-registration, and microPET imaging was obtained at 1 hr post injection. PET data was reconstructed using OSEM. After the PET imaging was completed the animal was IV injected with Fenestra™ VC (400 μL) and CT imaging was obtained at 40 min post injection. MicroCT data was obtained in the high-resolution mode and displayed in the coronal plane.
Results of this study show that Fenestra™ VC given at the described dose permits improved tumor delineation with microCT imaging, furthermore it aids in the anatomical representation of the microPET image for anatomical identification and accurate co-registration. Additional use of this agent would be in cardiac ventricular volume measurements, as well as vascular volume and size measurements. Fenestra™ VC shows promising results for future in-vivo experiments utilizing both microPET and microCT imaging modalities.
L.A. Strong, None.
Abstract ID: 399
An understanding of the tumor microenvironment is essential in the design of new chemotherapies for the treatment of cancer. One area of focus is tumor hypoxia, which can influence the efficacy of drug treatment, potential for metastasis, and induction of an aggressive tumor phenotype. When a tumor becomes hypoxic, the hypoxia inducible factor subunit, HIF-1α, is translocated to the nucleus of the cell where it dimerizes with HIF-1β. This then acts as a transcription factor responsible for upregulation of angiogenic factors and glycolytic enzymes. PET imaging utilizes the Warburg Effect, or the increased glycolytic nature of tumor cells, even in the presence of oxygen. Elevated HIF-1α levels may lead to a glycolytic phenotype due to increased transcription of glycolytic enzymes. Glucose uptake experiments were performed on two metastatic breast cancer cell lines, MDA-MB-231 and MDA-MB-435, and one non-metastatic cell line, MCF-7. The metastatic cell lines exhibited high glucose uptake in the presence of oxygen, while the non-metastatic cells had high uptake only under hypoxic conditions. Immunocytochemistry and Western blots were done on these cell lines, showing higher levels of HIF-1α and nuclear localization in metastatic cells in the presence of oxygen compared to the non-metastatic cells. HIF-1α was induced in all cell lines after exposure to hypoxia. A series of primary metastatic breast cell lines (passage # < 10) were stained for HIF-1α and screened for glucose uptake. Confocal microscopy results show a correlation between high nuclear localization of HIF-1α and increased glucose uptake under normoxic conditions. This suggests that an increase of HIF-1α in the nucleus may be related to the glycolytic phenotype.

B. Baggett, None.
Abstract ID: 400
S.R. Bunker, None.
Abstract ID: 401
Relationship between Tc-99m-Annexin uptake and tumor response (66 evaluation).

N.D. Steinmetz, Theseus Imaging 4, 5.
Abstract ID: 402
V. Ponomarev, None.
Abstract ID: 403
S. Lim, None.
Abstract ID: 404
Mesenchymal stem cells can be stimulated to differentiate into specific somatic lineage cells in vitro, but little is known about their in vivo response to malignant tumor formation. We studied this aspect in immunodeficient mice transplanted with human cancer cells subcutaneously and 3–4 days later with dual gene-marked human mesenchymal stem cells (tg-hMSC) through tail vein. Tumors formed in 3–4 weeks were detectable by positron emission tomography for nucleoside analogue retention catalyzed by the tg-hMSC marker herpes simplex virus thymidine kinase. Differentiated tg-hMSC progeny cells with green fluorescent protein marker were found in internal capillaries, peripheral blood vessel inner layer, dermal base and hair follicles at the tumor site. Numbers of the marked cells, some with human endothelial antigens, in the tumor were more than initially inoculated tg-hMSC. Our results indicate that mesenchymal stem cells can target, proliferate and differentiate at the tumor site and thus be exploited for noninvasive image tracing, in addition to therapeutic control, of cancer development in the body.
W. Deng, None.
Abstract ID: 407
Genetically engineered mouse models offer the opportunity to study how specific molecular lesions collaborate in the development of the mammary glands, mammary preneoplasia and cancer. Modification of a commercially available confocal microscopy system (VivaCell 5000TM, Tiba, LLC Rochester, NY, USA) has enabled the researchers to image mouse mammary glands in situ. Both ductal and vascular structures can be imaged. An atlas is in preparation to demonstrate the patterns of duct development in wild type and in three transgenic models in which the frequency of development of mammary cancer is increased. Our laboratory has demonstrated that gain of ERα (xxx et al. 2003 Am J Path; xxx et al. 2004 FASEB J), gain of AIB1 (xxx et al. 2004 FASEB J) or loss of Stat5a function (xxx et al. 2002 Oncogene) alter mammary cancer progression in mouse models. Mice in each of these groups are being studied at different time points of mammary gland development. Histological correlation is being performed. Development of survival models is planned so that duct development can be repeatedly monitored over time and so that the effects of interventions can be monitored.

In situ confocal microscopy demonstrates differences in the branching patters of mammary ducts in wild type and transgenic MMTV-rtTA/Stat5a1/1 at age 8 weeks.
M. Freedman, None.
Abstract ID: 408
The expression of glial fibrillary acidic protein (GFAP), the major intermediate filament protein of mature astrocytes, is considered to be a reliable biomarker of glial and neuronal damage. Moreover, the expression of this protein has been shown to increase during pathological conditions that result in damage to the CNS. Rapid, direct, nondestructive, repetitive in vivo monitoring of this biomarker during pathological conditions might provide insights into the disease process and assist in related drug discovery programs. We have developed a transgenic mouse model, using the GFAP promoter (Gfap) to drive luciferase (luc) reporter gene expression, which can be used for in vivo analysis of neurological damage. In vivo Gfap-luc expression was examined by bioluminescent image analysis following Streptococcus pneumoniae infections in the CNS. Mice were infected with live 104 CFU of S. pneumoniae type III by either percutaneous lumbar puncture at the 4/5 vertebra or by intracisternally inoculation. Strong bioluminescent signals were detected in the brain and spinal cords of infected mice as early as 24h after infection. No change in luciferase signal was measured in the vehicle control animals. Regardless of the route of infection, the signal appeared to spread over the brain and spinal cord 30h post infection, suggesting the dissemination of bacteria or bacterial products along the CNS. The detection of luciferase activity in CNS with the progression of the infection demonstrates a possible role of GFAP in meningitis and will allow us to evaluate the effect of drug interventions in real-time in living transgenic animals.
S.J. Smith, Xenogen Corporation 5.
Abstract ID: 409
Small animal CT is a modality that provides high-resolution anatomical images. However, high-resolution CT imaging is accompanied by a relatively large radiation exposure. For longitudinal studies, extended radiation exposure could become a significant factor in oncological applications because tumor growth may be altered. In this experiment BALB/c mice were implanted with the aggressive EMT6 tumor line, and animals were imaged on the microCAT-II (Imtek Inc). Image resolution was controlled by the binning parameter and the number and duration of exposures. In Part I, four groups of animals were imaged with a low (6 cGy) or high resolution (22 cGy) CT protocol either every day or every third day. An additional group served as controls. In Part II, 4 groups of mice were prepared and two groups were imaged with a similar CT imaging protocol as Part I but with higher CT dosage (11cGy and 55cGy). Tumor sizes were measured daily with calipers starting on day 6 (palpable tumor). In both CT experiments, tumor growth inhibition was noted for all animals. At day 8, when control tumors reached 95 mm3, tumor sizes were 63% smaller in the group that received 6 cGy daily, and 73% smaller for the group of 22 cGy daily. Imaging every third day resulted in lesser tumor growth inhibition. Tumors in Part II took 4 days longer to reach 95 mm3 than the tumors in Part I. It is possible that this was due to differences in the cell growth phase at implantation. As a consequence these tumors did not demonstrate as much inhibition as those in Part I suggesting resistance to radiation. These results indicate that the use of small animal CT can lead to tumor growth inhibition and radiation dosage is therefore an important consideration in all longitudinal studies for oncological applications.
R. Laforest, None.
Abstract ID: 410
Monocytes play an important role in the pathogenesis of atherosclerosis but their spatial and temporal involvement is largely unknown. In this study we utilized high resolution SPECT/CT to quantitate monocyte recruitment in atherosclerotic lesions of live mice. Peripheral blood monocytes from C57BL/6 mice were isolated by negative selection, labeled with 6–25 μCi 111-Indium-oxyquinoline (spec activity/million cells) and injected i.v. to syngeneic 4 month old ApoE−/− mice (4×106 monocytes/recipient). Negative selection of monocytes yielded a pure population (>90%) that, upon labeling, was viable (as assessed by MTS assay) and functional (as assessed in a peritonitis model). Serial SPECT/CT imaging (Gammamedica, XSPECT) of recipient animals revealed that, 3–5 days following adoptive transfer, 1,000-10,000 monocytes had been recruited to the ascending aorta. No such recruitment was seen in WT C57BL/6 recipients or ApoE−/− mice receiving PBMC depleted of monocytes. These findings were corroborated with autoradiography, as well as molecular (RT-PCR) and histological (IHC) techniques. We present evidence that the combination of gamma and X-ray detection provides the necessary sensitivity and resolution to track monocyte accumulation specifically and non-invasively in atherosclerotic mice.
F.K. Swirski, None.
Abstract ID: 411
Annexin V is a protein that binds tightly to the phosphatidylserine which redistributes to the outer leaflet of the cell membrane early on in apoptosis. The utility of annexin V as a marker of acute cell death has been recently reviewed by Lahorte et al. in Eur J Nucl Med Mol Imaging (2004) 31:887–919. Here we demonstrate the utility of combining tomographic data sets from x-ray CT and pinhole SPECT in mice as a sensitive means of measuring annexin V uptake in experimental tumors. Colo205 xenografts were treated with a single dose of Apo2L/TRAIL. Mice were injected with 5μg/150μCi Tc-99m-Hynic-rh-Annexin V (Theseus Imaging, Boston) and imaged an hour later with consecutive pinhole SPECT and CT (ASPECT, Gamma Medica, Inc.). Preliminary experiments with excised tumors showed increasing annexin uptake with increasing Apo2L doses. Six mice were imaged the day before treatment and again at either 2 or 6 hours post-treatment (n=3 each) and then a third time at 24 and a fourth time at 48 hours. Regions of interest were delimited easily on the CT and quantified with good sensitivity in the SPECT data. This showed that annexin V uptake was significantly elevated at 6 hours post-treatment in Apo2L/TRAIL treated animals and not in vehicle-treated controls. At 24 and 48 hours, there was no significant difference between treated and untreated animals. These data were consistent with previous histological studies that showed active apoptosis (condensed nuclei on H&E) at early timepoints but effective macrophage clearance of cell debris by 24 hours.

Difference from baseline of annexin V uptake in tumors.
S. Williams, Genentech 5; Theseus Imaging 5.
Abstract ID: 412
Conventional laser scanning microscopes can be upgraded easily with lifetime capability by picosecond time resolved single photon counting. Our presented solution is opening the door to the combination of SMD, FLIM and complementary FCS.
The optical design of our presented system MicroTime 200 is based on a standard inverted microscope base equipped with a three dimensional piezo scanning device which allows either for sample or probe (objective) scanning to allow also investigations of heavy and/or bulky samples. We are using as excitation sources compact pulsed picosecond laser diodes which are now not only available in the red and blue wavelength region, but via frequency doubling also in the green. The fluorescence is detected on a single photon base with highly efficient avalanche diodes in a freely configurable detection scheme consisting of up to four detectors. The data acquisition mode, allowing to implement time-resolved imaging as well as photon correlation techniques, is the so called Time-Tagged Time-Resolved (TTTR) mode implemented in the TimeHarp 200 board. Essentially, it is an elegant extension of the classical TCSPC histogram preserving the full photon information content.
The reconstruction of the steady-state and time resolved images using the markers and picosecond time-tags is the most obvious task as demonstrated on NBD labeled HepG2 cells (hepatocytes). For the case of immobilised single Cy5 molecules we show how the inherently limited photon information can still be exploited in different ways to characterize the blinking behavior of these single emitters.
The TTTR acquisition mode allows also to perform FCS as demonstrated on Cy5 solutions, without the limitations and approximations inherent in classical hardware autocorrelators. On-line monitoring of the autocorrelation function is also possible by on-the-fly calculation during TTTR recording, however the ultimate strength of the TTTR based FCS is the combination of TCSPC timing and time-tags.
R. Erdmann, PicoQuant GmbH 4, 5.
Abstract ID: 413
Small animal PET imaging has become a widely-used modality for the imaging of animals facilitating in the development of new radiopharmaceuticals and therapeutic agents. Its main advantage over conventional techniques resides in the ability to obtain accurate quantitative measurements in animals. In this study, animal PET-imaging experiments were compared to conventional biodistribution studies. The first group of mice was injected with 18F-FDG (230 μCi/animal), while a second group received 64Cu-PTSM (200 μCi/animal). After 90 min uptake for 18F-FDG and 45 min for 64Cu-PTSM the animals were sacrificed and imaged on a microPET®-FOCUS (Knoxville, TN) for 10 min. A transmission scan was performed prior to the emission scan and attenuation sinograms were obtained by a segmentation algorithm. The scanner was calibrated with a glass bottle containing 200 μCi of either 64Cu or 18F. Computed tomography images of the animals were acquired in a microCAT-II (Imtek Inc) and registered to the microPET data using fiducial markers. Following PET and CT imaging the animals were then dissected and organs were harvested and counted. The PET images were then analyzed where activity concentrations were measured from ROIs drawn on the visible organs both on attenuation (AC) and non-attenuation (NAC) corrected images. Additional ROIs were generated by drawing the ROI on the coregistered CT images and applying them to the PET data. Imaging data were then compared to dissection-based biodistribution data. Activity concentration from ROI's drawn on PET images were generally higher than biodistribution data, while PET-NAC data were systematically 15-20% lower than PET-AC. Better agreement of tissue activity concentrations were obtained with CT-drawn ROI with a correction for partial volume. In conclusion, we have shown that more accurate quantification of small animal PET data is obtained with co-registration with anatomical images and by applying both partial volume and attenuation corrections to the PET images.
R. Laforest, None.
Abstract ID: 414
Permeation peptides based on the HIV-1 Tat basic domain have been shown to enhance uptake of impermeant substrates into cells. The retina is an attractive target for this strategy given the accessibility of the eye, the ease of intraocular delivery, and the possibility for optical imaging in vivo. We examined the uptake of permeation peptides constructs linked to a fluorescent marker in the rat following in vivo intraocular injection, with emphasis on the retina. The permeation peptide sequence used was based on the wildtype HIV-1 Tat sequence, consisted entirely of D-isomer amino acid peptides, and was conjugated at the C-terminus to Alexa Fluor-594 so that cellular uptake could be identified (Ac-RKKRRORRRGC-AF594). Five μl of a 250–1000 μM solution was injected into the vitreous of anesthetized rats. Animals were euthanized at timepoints ranging from 2 hours to 2 weeks post-injection. The eyes were enucleated, the tissue fixed with 4% paraformaldehyde, and then processed for flat mounts or ocular paraffin sections. Immunohistochemistry for various retinal neuronal and glial markers was performed to identify cell types exhibiting uptake. At earlier timepoints, uptake by retinal ganglion cells (RGC) as well as by a specific subset of inner nuclear layer neurons was noted. At later timepoints up to two weeks post injection, uptake remained highly specific to RGCs. Immunofluorescence was also noted in nerve fibers in the optic nerve (axons of RGCs), and was more extensive at later timepoints. Since various moieties can be conjugated to permeation peptides, they may be useful for delivery of therapeutic agents to RGCs or in the imaging of RGC apoptosis and may have utility in the diagnosis and treatment of glaucoma.
E.M. Barnett, None.
Abstract ID: 415
Noninvasive radiological imaging has recently gained considerable interest in basic and preclinical research. In this study, we introduce flat-panel Volumetric Computed Tomography (fpVCT) as a powerful new tool for noninvasive imaging in preclinical research. This technology facilitates three-dimensional visualization capability, which provides even more information than histology, allowing longitudinal studies of the same animal. The isotropic high-resolution data provided by fpVCT is superior to the resolution of current clinical Multislice Computed Tomography (MSCT). The image quality in mice with fpVCT is comparable to that of patient scans with MSCT. In our study this performance is illustrated for the skeleton, chest, abdominal organs and the brain of mice. fpVCT enabled display of bone structural details such as intervertebral foramina of the mouse skeleton and foramina of the cranial base. Using contrast-enhanced scans, we displayed the vascular trees of the brain, liver and kidney at the subsegmental level. Even the diffuse vasculature of small experimental tumors in mice could be noninvasively monitored during tumor development. High-resolution visualization of murine chest scans enabled the characterization of small lung nodules. In addition, bronchiectasis and fibrotic strength were shown in a bleomycin-induced fibrosis model. Beyond scanning of small animal models, fpVCT enables imaging of larger animals up to primates, and therefore is a promising technique for a broad range of in vivo preclinical applications and animal models.
Flat-panel Volumetric Computed Tomography resulting in an isotropic high-resolution dataset: three-dimensional rendering of a tumor bearing nude mouse focusing on the vascularization. The in vivo scan was performed after administration of iodine-containing contrast medium.
S. Greschus, None.
Abstract ID: 416
Dynamic fluorescence images were obtained from xenografts bearing a subcutaneous human Karposi's sarcoma (KS1767) and human melanoma tumor (M21) model immediately following the i.v injection of an integrin-receptor (αvβ3) targeting Cy5.5-c(RGDf) at an equivalent dose of 3 nanomoles of c(RGDf). Both M21 and KS1767 models were integrin expressing. The fluorescence images were acquired using an intensified charge-coupled device (ICCD) system and were analyzed with a pharmacokinetic (PK) model to determine short term PK parameters of each tumor model over a period of 20 minutes. Imaging of both KS1767 and M21 models was also conducted in the presence of a competitive binder, free c(RGDf) administered i.v at a dose of 600 nanomoles. Our results show that the KS1767 exhibited early and rapid uptake of Cy5.5-c(RGDf) that could be mediated by the prior administration of c(RGDf) as a competitive binder to (αvβ3). However, the M21 tumor model showed different PK values from the KS1716 model. Indeed, there was little difference between the PK uptake rate constants between normal and M21 tissue regions at early times. Moreover, short term PK uptake rates were unchanged by the introduction of c(RGDf) prior to Cy5.5-c(RGDf) administration in M21. Finally, though KS1767 was visible from early dynamic fluorescence images, M21 was not readily contrasted at early times following administration even though it was contrasted by fluorescent uptake after 24 hours following Cy5.5-c(RGDf) administration. In vivo PK parameters may be reflective of the difference in tumor biology and molecular target expressions between KS1767 and M21.
S. Kwon, None.
