Abstract
We describe the design and construction of a miniaturized multichannel near infrared (NIR) endoscopic imaging system developed for high-resolution imaging of mice. The device allows for simultaneous real-time video images in white light and two independent NIR channels. Testing demonstrated independent acquisition of nanomolar concentrations of fluorochromes Cy5.5 and Cy7. Cross-talk between the NIR channels, partially a result of broad tails in the spectra of commonly used organic fluorochromes, was assessed, modeled for the linear range of the concentration/signal intensity function, and compensated. The calculated compensation was 5.5% and 22% of the total signal intensity in the two channels NIR700 and NIR780, respectively, at equal concentrations of the two fluorochromes. Using a mouse model of colonic adenomatosis, we show that both perfusion and protease activity can be detected simultaneously, independently, and repeatedly in live mice. The developed device should be useful for in vivo imaging of diverse molecular targets.
Introduction
Near-infrared fluorescence (NIRF) imaging is an emerging imaging modality, currently successfully implemented in vivo on the micro-and macroscopic scale [1,2]. In the mouse model, both epifluorescent [3] (i.e., surface-weighted) and tomographic [4] methods have been employed, and recently, catheter-based NIRF imaging has been proposed [5]. Two distinct advantages set optical imaging apart from other modalities [6]: First, “smart” fluorescent probes with high target-to-background and activation ratios are available [7,8]. Second, NIR light penetrates biologic tissue more easily than visible light, and the spectral window of the NIR region (as other regions of the optical spectrum) allows for the simultaneous excitation and recording of several independent channels enabling diverse biological applications: (a) colocalization of multiple targets, (b) determination of expression/activity ratios between targets, (c) disease characterization based on multiple attributes, and (d) better quantitation by the introduction of a reference channel which can compensate for differences in probe delivery and tissue absorption in heterogeneous disease states often seen in humans [9,10]. Additionally, in endoscopic and laparoscopic applications [11], the reference channel may also compensate for differences in target distance and illumination angle.
The purpose of this study was to develop a miniaturized multichannel NIRF detection system for micro-endoscopy in mouse models of disease. The criteria used in the development included: the need for simultaneous white-light, and two or more infrared channels; the availability of images in white light and real-time (video mode) to allow for anatomic orientation; image integration times which are short enough to avoid motion artifacts; the adaptability of the system to miniaturized fiber bundles, allowing use in mouse models; and image acquisition integrated with custom software (CMIR Image) to allow simultaneous display and storage of still and video frames in all channels and the generation of calculated images that may combine information from the independent channels.
Materials and Methods
Optical System
The individual components of the system were assembled on a breadboard (Figure 1). Illumination for the visible spectrum and excitation for the lower NIR spectrum (NIR70o channel) was provided by a 300-W Xenon lamp (Minimally Invasive Surgical Technologies, Smithfield, NC). Since the Xe lamp alone provided insufficient photon flux to excite the high NIR spectrum (NIR780), a diode laser (Ceralas PDT, Ceramoptec, Bonn, Germany) was introduced to excite the NIR780 channel. The Xe lamp was modified by exchanging the built-in IR-cutoff filters, which have a transmission of 350–600 nm, with a hot mirror (Edmund Optics, Barrington, NJ) mounted at 45° to the beam to provide an effective transmission range of 350–720 nm. The light was then transmitted through a glass fiber bundle, filtered through a 690-nm lowpass filter (Omega Optical, Brattleboro, VT), and propagated to a dichroic mirror which was used to superimpose the beam of a GaAs laser diode at 738 nm for the excitation in the high NIR spectrum. The beam was then refocused in to the excitation fibers of the endoscope.

(a) Photograph of the device, including: dichroic mirror used to combine the light output fom Xenon lamp with the NIR laser diode; imaging beamsplitter array for the separation of signal into visible light channel and two NIR fluorescence channels; visible light video camera; and NIR video camera, (b) Schematic diagram of light path on the excitation and the emission side, (c) Transmission spectra of dichroic mirror 2 at various angles of incidence. Approximately 25-nm red-shift is observed when the angle of incidence is reduced from 45° to 23°. (d) Emission spectra of Cy5.5 (gray) and Cy7 (black) demonstrate partial overlap, especially from the Cy5.5 tail into the Cy7 region.
The system was designed to accept standard-sized endoscope heads. For mouse studies, we used a fiber-optic endoscope (Baxter, Irvine, CA) with a total diameter of 1.4 mm and 100 cm working length. The catheter consisted of an imaging fiber bundle of 15,000 fibers with 0.5 mm diameter, of two illumination fiber bundles with 0.4 mm and of a working channel of 0.6 mm for irrigation, insufflation, and guidewire insertion.
After the emitted fluorescent and reflected white light passes through the endoscope, it enters a beam splitter. The beam splitter array, schematized in Figure 1b, is designed to provide separation, for simultaneous independent recording, of visible light, short-wavelength NIR band (690–720 nm, NIR700), and the long-wavelength NIR band (760–800 nm, NIR780) with a minimal number of reflections. The first dichroic mirror (all dichroics and filters: Omega Optical) transmits the visible light from 400 to 670 nm, and reflects the entire NIR region from 700 to 900 nm. A second dichroic and a fully reflecting mirror are then used to separate the NIR emissions. This second dichroic transmits the NIR780 band (760–800 nm), and reflects the remaining NIR700 band (680–720 nm), which is then reflected by a third, fully reflecting mirror. The two last mirrors are aimed at the objective lens of the NIR camera, and the angle between them (seen from the objective lens) is selected such that two separate (nonoverlapping) images of the low and high emission band are created next to each other on the CCD chip (ICX 248AL, Sony Instruments) of the NIR video camera (StellaCam EX, Adirondack Video, NY) after passing through sharp cutoff bandpass filters. The visible light passes through the first dichroic mirror and is recorded on a color video camera (Series 8290, Cohu, San Diego, CA).
To assess the ability to shift the frequency bands that are transmitted and reflected by the second dichroic mirror (Figure 1c) via changes in angle of incident light, spectral shifts were recorded on a spectrophotometer (U3000, Hitachi, Tokyo, Japan) at incident angles of 45°, 22°, and 0° from normal. This ability to fine tune the spectrum of NIR light that enters the NIR700 and NIR780 bandpass filters aides in optimization of both spectral separation and percent of true signal in the respective channels that is not cutoff by the bandpass filters and may thus be recorded by the NIR CCD. Subsequent experiments below were performed based upon the determined optimized angle of dichroic 2. The spectral throughput of the system for the two NIR channels was calculated using the transmission and reflection curves for each of the optical components as measured spectrophotometrically.
Sensitivity and Cross-talk
Sensitivity was tested by imaging serial dilutions of Cy5.5 and Cy7 (Amersham, Uppsala, Sweden) in phosphate-buffered saline, with concentrations from 30 nM to 10 μM in a 96-well plate. The tip of the endoscope was immersed at an angle of 45° to avoid reflection of excitation light from the bottom of the wells. The IR camera was set to integrate 30 video frames at 1/60 sec each. The signal intensity (SI) of the central region both in the NIR700 and in the NIR780 channels were measured using custom software (CMIR Image), and normalized to percentages of the maximum saturation values.
To improve the accuracy of quantitative measurements of fluorochrome concentrations, the changes in SI that each fluorochrome contributes to the respective other channel (interchannel cross-talk) at a given concentration were determined experimentally. Regression lines were fit to the linear parts of the SI versus concentration plots using the least-squares method, and compensation terms for interchannel cross-talk were formulated.
Intravital Testing
Colonoscopy was performed by two operators (H.A., M.A.F.) according to previous descriptions [12] on ApcMin +/− mice (age 20–30 weeks) obtained from the Jackson Laboratories (Bar Harbor, ME). These mice have a heterozygous deletion in the APC gene, which results in intestinal polyposis that mimics human disease [13]. All animal procedures were approved by the institution's animal care committee. The scope was lubricated with water and was introduced rectally into the anesthetized mouse. The colon was gently insufflated with air, while keeping the mean pressure less than 10 mmHg to avoid overinsufflation of the entire bowel, which could lead to perforation of the bowel or regurgitation of fluid through the esophagus. As the scope was gently advanced into the colon, the abdomen was observed both to localize the tip of the scope with transillumination and to monitor for overinflation. The average length of insertion was 4 cm, and each examination required 10 to 15 minutes to perform.
By white-light endoscopy, four animals carrying spontaneous polyps in the descending colon or sigmoid were identified. The preinjection (unenhanced) fluorescence intensities of each lesion were recorded. These four animals underwent intravenous injection of 2 nmol/mouse of a protease imaging probe fluorescent in NIR780. Colonoscopy was repeated 24 hr later, the time at which the peak fluorescence intensity of the protease probe occurs. During this second investigation, a perfusion imaging agent fluorescent in NIR700, indocyanine dye Cy5.5 conjugated to a crosslinked iron oxide nanoparticle, was injected at a dose of 1 nmol/mouse. The fluorescence intensity of the adenomas and of normal bowel wall were recorded in both NIR channels before and after injection of the second contrast agent, to demonstrate the ability to separate these two different activities. The synthesis of the protease agent [14,15] and the perfusion agent [12] has been described previously. After colonoscopy, the animals were sacrificed and the colon was inspected in situ on a macroscopic scale using a custom-built epifluorescence imaging system prototype (Siemens, Erlangen, Germany). The localization and spectral distribution of the respective fluorescence signals that were detected at colonoscopy were recorded. In addition, the polyps were excised together with the adjacent normal colon, and tissue sections were processed for histology by staining with hematoxylin and eosin.
Results
A precise separation of the two NIR emission channels could be obtained by fine-tuning the cutoff wavelength of the second dichroic mirror which is strongly dependent on the incident angle of the light that the mirror reflects (Figure 1c). Changing the angle of incidence from 0° to 45° results in a shift of 23 nm of the cutoff wavelength. By adjusting the angle to 22° and placing the other optical elements accordingly, optimal channel separation with minimal light loss could be obtained.
The image created by the reflected white light on the color camera CCD, and the separate images of the NIR700 and the NIR780 channel created next to each other on the CCD of the NIR camera were simultaneously displayed on the computer screen. The resolution of the imaging fiber bundle is approximately 7 line pairs per mm. The three video endoscopic images were recorded at a resolution of 130 pixels in diameter each.
To assess the fidelity of spectral separation in the two NIR channels, total system throughput was calculated based on the emission spectra of Cy5.5 and Cy7 (Figure 1d) and the spectral properties of all components. Assuming 100% input at both the maximum emission wavelength of Cy5.5 and Cy7, the spectral throughput of the imaging system progressively removes unwanted fluorescence at each optical component as follows: after the first dichroic, the relative intensities are 83% and 97% for Cy5.5 and Cy7 fluorescence, respectively, in the single NIR beam. After the second dichroic at which point the NIR700 and NIR780 channels separate, 82% and 18% in the NIR700 channel, and 17% and 85% in the NIR780 channel of the input signal of Cy5.5 and Cy7 fluorescence, respectively, remain. After the emission bandpass filters: 68% and 0% in the NIR700 channel, and 12% and 72% in the NIR780 channel, of the Cy5.5 and Cy7 fluorescence, respectively are present.
To determine interchannel cross-talk compensation, the linear part of the curve defining the relation between fluorochrome concentration and SI for each channel was used. The total SI in each channel SINIR700, SINIR780 is defined by:
From the SI versus concentration plots (Figure 2), regression was performed in the linear region of the curves between 30 nM and 1 μM for NIR700 and 30 nM and 3 μM for NIR780. The respective coefficients were calculated to be: a = 1.00 μM−1, b = 0.16 μM−1, c = 0.056 μM−1, and d = 0.751 μM−1. The proper compensations for interchannel cross-talk are therefore:
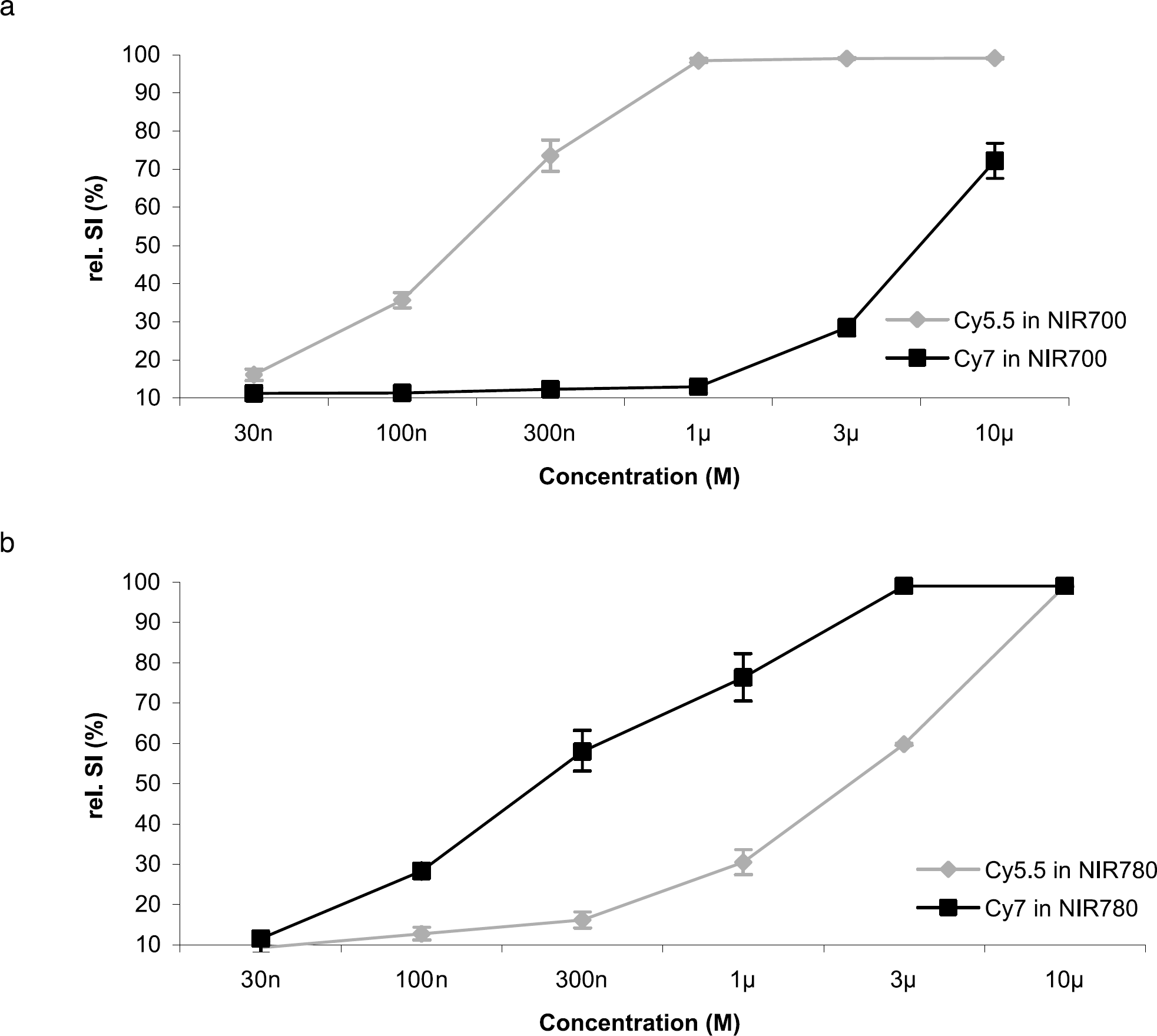
Relative signal intensity versus fluorochrome concentration: (a) NIR700l (b) NIR780, for Cy5.5 (gray line) and Cy7 (black line). Cy5.5 results in a signal intensity of approximately 40% in the NIR780 channel of its signal intensity in NIR700, while Cy7 produces less than 10% cross-talk into the NIR700 channel relative to its NIR780 signal intensity. Error bars represent standard deviation.
Cross-talk compensation (i.e., the coefficients calculated above) was implemented in the displayed fusion images (Figure 3d and e). The correction introduced into the NIR700 channel was 5.5%, whereas a larger correction of 22% was introduced into the NIR780 channel. This asymmetry is due to the overlap and asymmetry of the tails of the Cy5.5 and Cy7 emission spectra (Figure 1d). While there is almost no emission light from a fluorochrome emitting primarily in the NIR780 channel recorded in NIR700, the spectrum of a typical NIR700 fluorochrome is still recorded to a moderate extent in NIR780. The corrections were validated and shown to hold true in phantoms doped with known concentrations of fluorochrome mixtures (data not shown).

(a–c) Colonoscopy images acquired simultaneously in the white light (a), the NIR700 (b), and the NIR780 (c) channel, after intravenous injection of an intravascular contrast agent fluorescent in NIR700, and a protease-activatible probe fluorescent in NIR780. Two different fluorescence patterns are visible. (d,e) Fusion image between white light and NIR700 (d), and NIR780 (e), respectively, with cross-talk compensation, (f,g) Histologic correlation: elevated submucosal vascularity (f) in the region fluorescent in NIR700, and colonic adenoma (g) correlating to the fluorescence in NIR780. (h) Normal control colon, with normal submucosal vascularity, (f,h) Acquired at ×200 magnification; (g) Acquired at ×20 magnification. Epithelial detachment in (f) reflects processing artifact.
Colonoscopy was feasible in all animals. The colon could fully be inspected up to the splenic flexure. The polyps were easily identifiable after administration of the protease-activatible probe in the NIR780 channel. The simultaneous application of the intravascular contrast agent, in contrast, showed an entirely different spatial pattern, as demonstrated in Figure 3a–e. While the polyps showed only moderate and incomplete enhancement, the brightest regions in the NIR700 channel were the hyperemic parts of the intestine, identifiable in the white-light channel as red-hued parts of the mucosa. Fluorescence recorded with the endoscope correlated both in spectral quality as well as in localization in all cases with the fluorescence observed ex vivo in the epifluorescence system. Subsequent histologic examination of the tissues (Figure 3f and g) compared with normal colon (Figure 3h) confirmed the correlation of intestinal adenomas with high SI in NIR780, and high vascular density with high SI in NIR700 (Figure 3d and e, respectively).
Discussion
We designed an NIR fluorescence endoscope that simultaneously records a white light and two NIR channels, suitable for the independent probing of two molecular or physiologic parameters at the same time and in the same sample, in both video and still capture modes. The experiments described in this study demonstrate the feasibility of imaging perfusion and enzyme activity in a spontaneous colon tumor model. The proposed method offers the potential of repeated, nondestructive access of the same regions, and for minimally invasive bioptic verification of the tissues under investigation.
The main issue in the design of a miniaturized multichannel NIR fluorescence endoscope is its sensitivity to collecting fluorescent light through miniaturized optics and fibers. On the illumination side, the total photon flux is limited by the product of diameter and numeric aperture of the illumination fiber bundles (Helmholtz invariant [16]) and the luminous flux of available broadband light sources. A Xenon lamp with a superimposed laser source at 738 nm represented the best compromise for our specific purpose between spectral intensity distribution and photon density.
The concentration and quantum efficiency of the fluorochromes in the target region is an additional limiting factor. In the future, this limitation may be decreased as fluorochromes are developed with improved quantum efficiency, as well as with the use of less-quenching fluorochromes to allow greater perfusion imaging signal. Moreover, as fluorochromes with narrower excitation and emission spectra are produced, broader bandpass filters may be employed which allow the recording of a greater percentage of the emission photons [17]. Of note, a number of fluorochromes from a variety of manufacturers are currently available for both NIR700 and NIR780, providing additional versatility to the system.
Because of the inherent limitations of excitation photon flux detailed above, we focused on optimizing light detection. When the eyepiece of the endoscope is correctly focused, the light coming from the image on the fiber face is collimated. However, this holds true only for image points along the optical axis. Since the entire area of the fiber bundle is imaged, the emerging rays will be divergent, as determined by the diameter of the fiber bundle and the focal length of the eyepiece. Thus, the relay lenses that refocus the collimated light onto the respective CCD chips have to be of sufficient size not to create an aperture that limits throughput. The CCD chip used for NIR imaging in the present setup is a state-of-the-art design with three improvements over previous CCD chips: improved IR light response, larger on-chip microlenses, thus reducing the inefficient area of the chip, and improved overall quantum efficiency of 62% at 700 nm, and 45% at 750 nm.
The capability for the independent assessment of two channels can potentially be used not only for investigating two independent molecular parameters, but also to compensate for effects that could skew the quantitation in a typical one-channel setup (e.g., variability in illumination angle of the target and distance to target from the endoscope tip). The effects of inhomogenous probe distribution may also be corrected for by the coinjection of a reference nonactivatable contrast agent which may be recorded in the second NIR channel.
In conclusion, we have designed and tested a miniaturized multichannel endoscope usable in NIRF imaging in mouse models of human disease. The simultaneous acquisition of two distinct parameters, namely, vascularization and local enzyme activity, was feasible. This device can potentially be used in repeated, nondestructive optical imaging of a multitude of molecular targets.
Footnotes
Acknowledgments
This work was supported in part by NIH grant EB001872. UM was supported in part by an American Roentgen Ray Society Scholarship. MAF was supported by a stipend from the Max Kade Foundation. KK supported in part by the National Colorectal Cancer Research Alliance. We thank Ted Graves for development of CMIR Image and the new routines he created to implement the correction schemes detailed here.



