Abstract
Magnetic resonance imaging (MRI) has evolved into a sophisticated, noninvasive imaging modality capable of high-resolution anatomical and functional characterization of transgenic animals. To expand the capabilities MRI, we have developed a novel MR signal amplification (MRamp) strategy based on enzyme-mediated polymerization of paramagnetic substrates into oligomers of higher magnetic relaxivity. The substrates consist of chelated gadolinium covalently bound to phenols, which then serve as electron donors during enzymatic hydrogen peroxide reduction by peroxidase. The converted monomers undergo rapid condensation into paramagnetic oligomers leading to a threefold increase in atomic relaxivity (R1/Gd). The observed relaxivity changes are largely due to an increase in the rotational correlation time τr of the lanthanide. Three applications of the developed system are demonstrated: (1) imaging of nanomolar amounts of an oxidoreductase (peroxidase); (2) detection of a model ligand using an enzyme-linked immunoadsorbent assay format; and (3) imaging of E-selectin on the surface of endothelial cells probed for with an anti-E-selectin – peroxidase conjugate. The development of “enzyme sensing” probes is expected to have utility for a number of applications including in vivo detection of specific molecular targets. One particular advantage of the MRamp technique is that the same paramagnetic substrate can be potentially used to identify different molecular targets by attaching enzymes to various antibodies or other target-seeking molecules.
Introduction
Noninvasive imaging of molecular expression in vitro and in vivo at high resolution is becoming an important adjunct to biomedical research and drug discovery effort [1–4]. In particular, magnetic resonance imaging (MRI) is exceptionally suited for the above purpose due to superb anatomical resolution [5–7]. New applications of MRI range from mouse genetics and developmental biology [8–13] to experimental pharmaceutics [14]. However, while expression profiling and high throughput screening have become relatively common-place in identifying suitable molecular targets, in vivo testing of target gene expression in animals has been more challenging. For example, molecular specificity in MRI has been largely lacking. In molecular MRI, it is assumed that antibodies, receptor ligands, or other “target-seeking” molecules tagged with magnetic reporters bind to the target and produce local increases in water proton relaxation rates that are detectable by MRI. This molecular detection strategy is hampered by the inherently low sensitivity of MRI to the presence of reporter molecules (the detectability limit for commonly used chelated paramagnetic gadolinium is estimated as 100 μmol Gd/g tissue [15],[16]). Therefore, several types of MR signal amplification (MRamp) have been previously described. The first approach includes labeling target-seeking molecules (e.g., transferrin [17],[18]) with a plurality of paramagnetic metal cations by attaching chelating polymers [17,19–22], colloids [18],[23], or liposomes prepared using paramagnetic lipids [24]. The second approach uses β-galactosidase-mediated catalytic cleavage of paramagnetic quasisubstrate [11–25],[26]. In this case, MR signal enhancement is caused by a relaxation effect resulting from an increase in the number of water molecules coordinated directly by a paramagnetic metal [25], namely, “inner-sphere” relaxation effects (reviewed in Lauffer [27] and Koenig [28]).
To facilitate wider studies of molecular target assessment using MRI, we have now developed a generic method of signal amplification that relies on enzyme-mediated substrate conversion into magnetically active oligomers (MRamp; Figure 1B). The major advantage of the novel strategy is that a single substrate potentially can be used as a reporter for thousands of targets, using an antibody-enzyme conjugate as the primary reporter, similar to that of a traditional enzyme-linked immunoassay. We assumed that the oxidoreductases (e.g., peroxidases, E) would catalyze reduction of peroxide using a low-relaxivity paramagnetic substrate (AH) as a donor of electrons (reaction 1). Oxidized substrate molecules (*A) would then self-polymerize (oligomerize) into the larger, high-relaxivity paramagnetic oligomers (reaction 2):
Three applications for the above system were demonstrated: screening of endogenous peroxidase activity, determining specific antigens in microtiter plates by imaging, and identifying cell surface expressed proteins in cell culture.

(A) Scheme of paramagnetic substrate synthesis. (1) t-Butoxycarbonyl-protected precursor of hydroxytyraminyl-glycylmethylDOTA (D-DOTA); (2) gadolinium salt of hydroxytyraminyl-glycylmethylDOTA [D-DOTA(Gd)]. (B) Schema illustrating the principle of MRamp as a consequence of substrate polymerization.
Materials and Methods
Substrate Synthesis
10-(3-Aza-4-carboxy-2-oxo-1-methyl-butane-1-yl)-1,4,7,10-tetraazacyclododecane-1,4,7-tris-acetic acid tert-butyl ester (Gly-MetDOTA, tri tBu) was provided by Dr. B. Raduechel (Schering, Berlin, Germany). Linking of glycine carboxyl to the amino group of dopamine (hydroxytyramine) was accomplished by reacting equimolar amounts (0.25 mmol) in the presence of 1.1-fold molar excess of dicyclohexylcarbodii-mide (Figure 1A). After the removal of dicyclohexylurea, the product was recovered by vacuum evaporation and treated with 50% TFA for 1 hr. Diethyl ether-washed, crude D-DOTA(Gd) was dissolved in a solution of equimolar amount of Gd citrate (pH 4.5), heated at 75°C for 1 hr under argon and purified by using a Vydac C-18 HPLC column eluted by a gradient of acetonitrile in 0.1% TFA. The major peak with absorbance at 280 nm was collected, passed through Chelex-100 resin (Bio-Rad), and dried. Analysis of the free acid by MALDI-TOF gave a mass (m/z) of 594 (593 calculated). Analysis of the purified gadolinium complex gave an m/z of 748 corresponding to a monogadolinium salt [D-DOTA(Gd)].
Cell Culture
Human endothelial cells (Vascular Research Division, Department of Pathology, Brigham and Women's Hospital, Boston, MA) were isolated as described [29],[30]. Cells were plated on gelatin-covered plastic plates and cultured in 10% FBS, EBM medium (Clonetics) with endothelial growth supplements. Treatment of cells with human recombinant IL-1β (10 pg/ml) to induce E-selectin expression was performed at 37°C for 4 hr. E-selectin expression on the surface of IL-1β3-treated cells has been proven by fluorescent microscopy using F(ab′)2 fragment of monoclonal antihuman E-selectin antibody (H18/7, Vascular Research Division, Department of Pathology, Brigham and Women's Hospital) [31] followed by the antimouse-rhodamine conjugate (Pierce).

(A) Size-exclusion HPLC analysis of D-DOTA(Gd) (peak 2) and peroxidase-mediated reaction products (peak 1). (B) Mass spectrometry of reaction products (peak 1 in A). Oligomers of oxidized D-DOTA(Gd) with degree of polymerization n = 2–13 were identified. (C) MRI of D-DOTA(Gd) at various gadolinium concentrations in the presence (+ Px) or in the absence of peroxidase (− Px) and hydrogen peroxide. S: 0.05 mM Gd3+ aqueous standard solution. (D) Gadolinium concentration dependence of MR signal intensity before (open circles) and after (closed circles) activation with peroxidase as described in the Materials and Methods.
Peroxidase Catalysis and Imaging
Substrate D-DOTA(Gd) (10–200 μM) were treated by peroxidase (0.1–100 nM) and the excess of hydrogen peroxide (3.5 mM) in 10 mM PBS or 0.05 M sodium phosphate pH 6.8. In some experiments, peroxide-generating system (5 mM glucose, 0.5 U/ml glucose oxidase) has been used instead of hydrogen peroxide. The reaction was monitored by spectrophotometry at 400 nm and by NMR spectrometry (Minispec 120, Bruker run at 0.47 T (20 MHz) at 40°C). To analyze the kinetics of relaxivity change, the reaction was stopped by adding 1 M sodium azide (0.1 M final concentration) and measuring T1. The obtained experimental data was fitted using the function: 1/T1 = A(1-exp (− (k1 + k2)t)) + B, and a sum of apparent kinetic constants, k = k1 + k2 (where k ≈ k1 as k1k2), was determined. Imaging experiments were performed within 1 hr after the substrate addition to peroxidase. MRI was performed by using 1.5 T Signa GE system (63 MHz, 25°C) and surface or knee coils. Inversion-recovery pulse sequences (TE 11 msec/TR 1000 msec/TI 50–600 msec) were used for T1 measurements. Spin-echo sequences (TE 11 msec/TR 500–100 msec/3 NEX, Matrix 512 × 224) were used in signal amplification experiments. MR signal intensity was measured using a region-of-interest approach applied to 16-bit TIFF images (IP Lab Spectrum). All signal intensities were normalized using a gadolinium-DTPA standard to correct for inherent magnetic field inhomogeneities. The statistical significance of changes in the ROI values was determined using Student's t test.
MRI of Peroxidase Conjugate-Mediated Catalysis
The F(ab′)2 fragment of H18/7 monoclonal antihuman E-selectin antibody was a generous gift from Jeanne-Marie Kiely (Brigham and Women's Hospital). The fragment has been labeled with digoxigenin HSE (Roche Molecular Diagnostics) as suggested by the manufacturer. Digoxigenin-labeled antibody fragment (F(ab′)2-DIG) was serially diluted with 0.01 M sodium carbonate (pH 9) in a 96-well plate (Nunc) and adsorbed at 4°C overnight (1–1000 ng protein/well). The plate was washed with PBS, 0.1% Tween-20, pH 7.4 blocked with 1% BSA/PBS, and incubated in the presence of anti-DIG antibody-peroxidase conjugate (Roche, diluted 1:1000) for 1 hr. Washed wells were filled with 200 μl of 100 μM D-DOTA(Gd) and hydrogen peroxide (3.5 mM) and incubated for 30 min before imaging.
MRI of E-Selectin Expression
Human umbilical cord endothelial cells (HUVEC, 2 × 106/sample) were treated sequentially with IL-1β (10 pg/ml), anti-E-selectin F(ab′)2-DIG, and anti-DIG antibody-peroxidase. Cell suspensions were prepared in PBS and D-DOTA(Gd) was applied as described above. Cells were pelleted in 0.5 ml polypropylene tubes and subjected to MRI as described above. Control samples were prepared without treating cells with IL-1β or in the absence of F(ab′)2-DIG. Cell imaging experiment in a 96-well plate format was accomplished by plating 2 × 104 cells/well and treating the cells with IL-1β, anti-E-selectin-DIG and anti-DIG antibody-peroxidase/D-DOTA(Gd) as described above. MR signal intensity was quantified as described above and compared to aqueous Gd standard solutions.
Results
Enzyme-Mediated Oxidation and Relaxivity Changes
The synthesized paramagnetic substrate hydroxytyraminyl-glycylmethylDOTA [D-DOTA(Gd)] (Figure 1A) was purified and tested as an electron donor. Time curves of peroxidase-mediated catalysis in the presence of hydrogen peroxide showed a rapid increase in relaxation rate (1/T1) with an apparent pseudo-first-order constant of k1 = 0.04 s−1. Conversely, relaxation time measurements showed a rapid concentration-dependent increase in R1 relaxivity following enzyme addition (from 3.75 to 11.50 (mM s)−1 at 0.47 T, 40°C). Control reactions without enzyme or peroxide did not yield any measurable R1 changes. To test whether the observed R1 increase could be a result of the dissociation of gadolinium cation and oxidized D-DOTA, we treated the reaction mixture with Chelex-100 and compared it to the control with no resin added. No differences in R1 could be observed, indicating that this was not the case.
To determine whether the peroxidase-mediated R1 increase was due to polymerization as hypothesized (Figure 1B), we performed size-exclusion HPLC at different time points (10–60 min) during the reaction and compared elution profiles to control substrates (Figure 2A). Indeed, higher-molecular-weight oligomers were formed having a mean hydrodynamic radius of a 7-kDa molecule. The elution profile did not change when initial substrate concentration was varied from 10 to 100 μM. Mass spectrometry of reaction products showed various oligomers with polymerization degrees ranging from 2 to 12 (Figure 2B). Nuclear MR dispersion measurements performed using the oligomerized D-DOTA(Gd) suggested a fourfold increase in rotational reorientation time (τr) of chelated gadolinium following the treatment of substrate with peroxidase (J. Chen, A. Bogdanov, R. Weissleder, in preparation).
Magnetic Resonance Imaging
The first set of MRI experiment was designed to visualize enzyme-mediated conversion of the paramagnetic substrate. A 96-well plate containing different dilutions of the substrate in the presence or in the absence of peroxidase was used. The MR signal intensity increase was clearly visible using T1-weighted spin-echo sequences (Figure 2C). Overall, a 60% enhancement of the MR signal was measured using gadolinium concentrations between 50 and 200 μM (Figure 2D).
To determine the sensitivity of the amplification method, we also varied the concentration of peroxidase in reaction mixtures (fixed concentration of 100 μM D-DOTA(Gd); Figure 3A). Peroxidase was clearly detectable at concentrations of 50 ng/ml (e.g., 10 ng of peroxidase in the volume of 200 μl, i.e.,1 pM; Figure 3B). At concentrations of peroxidase exceeding 500 ng/ml (100 ng peroxidase/well), all the substrate was converted during the reaction time (1 hr), resulting in the saturation of signal intensity (Figure 3B).
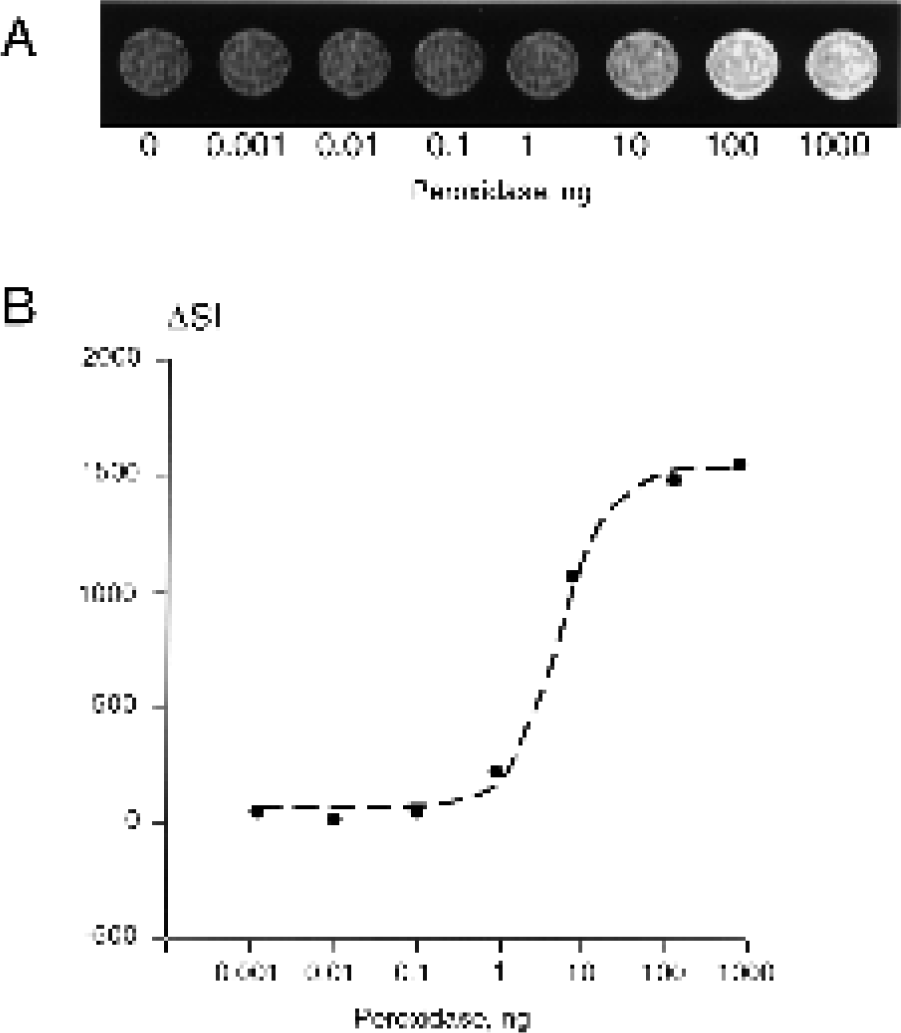
Detection of peroxidase using MRamp. (A) MR image of 96-well plate fragment containing D-DOTA (Gd) in the presence or the absence of the enzyme; (B) the corresponding signal intensity increase over the control with no peroxidase present (ΔSI). Reaction mixture contained 0.1 mM substrate, 3.5 mM H2O2 in a volume of 0.2 ml. Imaging was performed at 1.5 T using spin-echo sequence as described in Materials and Methods.
In another series of experiments, we determined whether MRamp is useful in detecting model ligands in an ELISA-like assay. A serially diluted F(ab′)2 fragment of monoclonal antibody (concentration range 0.1–1000 ng), covalently labeled with digoxigenin, was adsorbed on the surface of a 96-well plate and incubated with antidigoxigenin-peroxidase conjugate followed by 100 μM D-DOTA(Gd) (Figure 4A). MRI was performed in a single plate and in stacked plates assays. The antibody fragment was detectable at 1–80 ng with 5–8 ng F(ab′)2-digoxigenin fragments being detectable at the lower threshold (Figure 4B, C). The sensitivity of the MRamp assay was comparable to that of a standard colorimetric ELISA performed under identical conditions, the only difference was that a chromogenic substrate was used (Figure 4C).

(A) Set-up of MRI experiment using 96-well microtiter plate format; (B) MR image of 96-well plate central section containing D-DOTA(Gd) in the presence or the absence of adsorbed digoxigenin-labeled F(ab′)2 detected using antidigoxigenin-peroxidase conjugate; (C) MR ELISA signal change (circles) and standard ELISA signal (triangles) as a function of adsorbed amount of F(ab′)2. Reaction composition and imaging set up are described in Materials and Methods.
The latter experiment strongly suggested that it should be feasible to image specific antigen expressions on the cell surface by MRI. To test this, we treated human endothelial cells (HUVEC) with interleukin-1β (IL-1β) to induce expression of E-selectin on the cell surface [31]. Overexpression was confirmed by using fluorescently labeled anti-E-selectin F(ab′)2 (Figure 5A, B). MRI clearly showed high signal intensity in E-selectin expressing cells that had been treated with IL-1β followed by digoxigenin-labeled antibody and antidigoxigenin-peroxidase conjugate (Figure 5C). In control experiments, nontreated cells or IL-1β-treated cells that were not incubated with anti-E-selectin antibody showed significantly lower MR signal over the background (Figure 5C, D). The observed changes in MR signal intensity were statistically significant in both HUVEC precipitates (Figure 5C) and supernatants in 96-well plates with substrate-attached cells (Figure 5D; p #x003C; 05) (Figure 5D). There were no appreciable MR signal intensity change in control cells that were not treated with IL-1β (Figure 5C) or controls that were treated with IL-1β but were not incubated with DIG-labeled anti-E-selectin F(ab′)2 (Figure 5C, D). MR signal could be determined in as few cells as 3.5 × 104 cells/well and using 200 ng F(ab′)2 fragment (Figure 5D) at 1.5 T using a clinical MRI system during scan times of #x003C; 2 min.

Detection of E-selectin expression on human endothelial cells. (A) Fluorescent microscopy of human endothelial cells treated with IL-1β; (B) control, nontreated cells. (C) MRI of HUVECs at 1.5 T: (1) standard solution of 50 μM GdCl3; (2) HUVEC cells treated with IL-1β, anti-E-selectin-DIG antibody and anti-DIG peroxidase conjugate followed by 100 μM substrate and 3.5 mM H2O2; (3) control with no IL-1β stimulation; (4) control IL-1β stimulation with no anti-E-selectin-DIG antibody. (D) Detection of E-selectin on HUVEC grown and imaged in 96-well plates (simultaneous imaging of two stacked plates). The dependence of MR signal intensity on the concentration of digoxigenin-labeled anti-E-selectin F (ab′)2 is shown. Data is presented as m ± SD (n = 4). Imaging performed as described in Materials and Methods.
Discussion
Several MR amplification strategies can be used to increase the atomic relaxivity of paramagnetic lanthanides. This is most commonly achieved by modulating the rotational correlation time (τr). So far, three different ways of decreasing rotational mobility of metal complexes had been considered: (1) dispersed paramagnetic chelated metal ions into high-viscosity compartments; (2) covalent attachment of paramagnetic complexes to macromolecules; and (3) noncovalent binding of chelated complexes to macromolecules (reviewed in Lauffer [27]). In this research, we show that there is a fourth strategy leading to a similar τr increase. This strategy includes condensation (or polymerization) of low relaxivity complexes into higher relaxivity products. We tested this hypothesis by synthesizing a peroxidase substrate using a derivative of DOTA, a commonly used chelate. Modification of DOTA with hydroxytyramine (dopamine) resulted in an electron donor substrate for catalytic hydrogen peroxide reduction. Tyraminelabeled ligands (e.g., biotin) has been successfully used previously for the “amplification” of signal generated by an enzyme-linked conjugate in immunohistochemistry [32],[33]. It had also been suggested that the amplification stage includes the covalent attachment of oxidized substrate to the molecules surrounding peroxidase-labeled ligand and requires the removal of nonreacted or nonbound substrate. As our experiments demonstrate, a typical MRamp assay is similar in design to a standard colorimetric ELISA in that the attachment of the oxidized substrates to any surface is not required (Figure 1B). Moreover, the amplification does not require the presence of proteins for enhancing relaxation effects (e.g., serum proteins) [34],[35].
Analysis of reaction products by size-exclusion HPLC and mass spectrometry confirmed condensation of oxidized substrates. We further demonstrated that oligomerization results in strong relaxation effects directly detectable by MRI (Figure 2C). MR signal intensity of peroxidase-treated substrates was higher than that of aqueous gadolinium solutions at equal concentrations (Figure 2C) because of the higher relaxivity of polymer-bound gadolinium. NMRD spectrometry showed a substantial τr increase, and this appears to be the main reason for the observed relaxation effects. The strong relaxation effects indeed allowed nanogram amounts of peroxidase to be detectable by MRI (Figure 3). These encouraging results prompted us to further develop a “MR-imageable” ELISA-like assay, which could be reproducibly performed in at least two stacked plates (Figure 4A). The sensitivity of such an MR assay was similar to that of standard colorimetric ELISA assays (Figure 4C).
To extrapolate MRamp antigen-detection assay to cell surface expressed proteins, we used a well-characterized model — highly specific expression of E-selectin on the surface of human endothelial cells (HUVEC) stimulated by IL-1β (Figure 5A). This model is relevant to several disease states as it is well established that E-selectin expression on endothelium occurs in inflammation and atherosclerosis [36–38]. A strong signal enhancement in cell precipitates treated with IL-1β followed by E-selectin detection (i.e., sequential treatments with digoxigenin-labeled antibody, antidigoxigenin-peroxidase conjugate, and D-DOTA(Gd)) showed that E-selectin-expressing cells bind at least some of the oligomerized D-DOTA(Gd). This observation may be explained by high local enzymatic activity of antibody-bound peroxidase associated with plasma membrane of E-selectin-positive cells. In alternative experimental setting that involved small numbers of attached endothelial cells in a 96-well culture plate (1-2 × 104 cells/well, Figure 5D), we observed a measurable MR signal intensity increase in supernatants of cells expressing E-selectin. These experiments demonstrated that MRamp detection of cell-surface molecular determinants is potentially achievable with or without the removal of the bulk of the paramagnetic MRamp reaction product. The latter results suggests that the amplification strategy, ultimately, may result in biological amplification in vivo, as the resultant paramagnetic oligomers may have different clearance rates than monomeric precursors. Differential excretion of products of MRamp reaction may potentially result in differential enhancement of blood pool in experimental and control animals. Further in vivo experiments are currently underway to test these projections.
In conclusion, we demonstrated in several model studies the utility of a novel MRamp strategy. We showed that the catalysis-mediated threefold increase in relaxivity translates into a strong proton MR signal enhancement that can be utilized for positive identification of cytokine-regulated cell surface receptor expression. Furthermore, the developed substrates and secondary enzyme-linked antibodies can be universally used when combined with different primary antibodies.
Footnotes
Acknowledgments
The authors are grateful to Drs. Hanns-Joachim Weinmann and Bernt Raduechel (Schering) for providing GlyMetDOTA, tri tBu ester. The authors acknowledge the help of Jeanne-Marie Kiely for providing H18/7antibody fragment, Kay Case for the assistance with endothelial cell culture, and Dr. Michael Gimbrone, Jr. for the useful discussions (Vascular Research Division, Department of Pathology, Brigham and Women's Hospital). We acknowledge the input made by Dr. John Chen who contributed preliminary NMRD data analysis and made useful comments. This work has been supported in part by 1P50CA86355-01 (RW) and 5RO1 CA74424-01 (AB). CB was supported by a grant from Deutsche Forschungsgemeinschaft. AP was supported by the Russian Department of Education Presidential Award.


