Abstract
1. Introduction
Isotretinoin (13-cis-retinoic acid, 13-RA), a vitamin A derivative (retinoid), is commonly prescribed for severe acne that is resistant to other treatments [1]. It has been effective in the clinical control of acne. However, it has a wide range of side effects, such as teratogenesis in gestation [2, 3], arthritis [4], sacroiliitis with severe disability [5], liver damage [6], and psychosocial symptoms such as anxiety, depression, disturbance in satisfaction, and quality of life [7, 8]. Some studies reported that the administration of 13-RA can cause physiological changes that are pathways for the pathology of different psychiatric conditions such as increased oxidative stress [6, 9, 10], headache, seizures, confusion, and cerebral ischemia [11]. Consequently, stress is believed to be the root cause of any central nervous system disorder such as depression. Some experimental approaches have shown hypothalamo-pituitary-adrenal (HPA) axis stimulation in stress-induced disruption of the BBB. The BBB is the neurovascular unit that controls the transition of matter and blood-borne immune cells into the brain parenchyma. Plus, the BBB prevents any peripheral injury to the brain tissue or its material.
Persons who use 13-RA to treat acne can develop mood swings such as cognitive neuropsychological disturbances, stress-induced mood swings, anxiety, amnesia, insomnia, and depression as well as suicide ideation or even suicide itself [12–15]. The pathogenic mechanism is unknown. It is known, however, that 13-RA affects steroid production in rats [16] and that many steroids can cause mood swings in humans [17, 18] and vasodilation [19, 20]. The mechanism behind these changes lies in the ability of isotretinoin to regulate gene expression in the brain [13, 21].
Few studies have also shown a relationship between 13-RA treatment and stress in general and between 13-RA treatment and the brain as a specific organ. This study investigated if 13-RA treatment in rats would affect their brain blood flow. Therefore, a rat model was used to determine if 13-RA administration induces stress. Many studies have used rat models for stress [22, 23] because they have similar characteristics to humans in their expression of emotions [24, 25]. In an experiment on rats, chronic unpredictable mild stress (CUMS) induced depressive-like behaviors [25, 26].
In nuclear medicine, technetium-99m hexamethyl propylene amine (99mTc-HMPAO) is used as a radiotracer for investigating regional cerebral blood flow in humans. It is potentially useful for assessing cerebral blood flow in small animals [27, 28] and in several disease models [29, 30]. It has also been used to label leukocytes for the detection of inflammation [31, 32]. Scientists believe that 99mTc-HMPAO accumulates in the brain through its intracellular conversion from a lipophilic form to a hydrophilic form in the brain parenchyma [33].
2. Materials and Methods
2.1. Materials
The HMPAO pharmaceutical kit (exametazime) was purchased from GE Healthcare (Chalfont St. Giles, UK). The 99m-technetium pertechnetate (99m Tc-pertechnetate, 99mTcO-4) radionuclide was obtained from a molybdenum99-technetium-99 m (99Mo99mTc) generator purchased from Amersham International PLC (Amersham, UK). The following authentic lipids were purchased from Sigma (St. Louis, Mo, USA): fatty acids (palmitic acid (C16:0), pentadecanoic acid (C15:0), adrenic acid (C22:4), docosapentaenoic acid (C22:5), and cervonic acid (docosahexaenoic acid; C22:6)); cholesterol heptadecanoate; heptadecanoyl CoA; tripalmitin; and D-
2.2. Experimental Animals and Ethics Statement
Adult male Wistar rats (
2.3. 99mTc-HMPAO Labeling
The labeling procedure was performed following the manufacturer’s instructions. 2,960 MBq of 99m Tc-pertechnetate in 3 ml saline was added to a freeze-dried exametazime kit to prepare 99mTc-HMPAO. In all cases, the labeled 99mTc-HMPAO was fresh and used within 30 min from the time of its preparation. Its radiochemical purity was verified through thin-layer chromatography according to the manufacturer’s recommendations and ranged from 95% to 99%.
2.4. Experiment Protocol
The rats were randomly divided into three groups: the control group (C), which was given 0 mg of 13-RA per kg of body weight; the low-dose group (L), which was given 0.3 mg/kg of 13-RA; and the high-dose group (H), which was given 0.5 mg/kg of 13-RA. The dose of 13-RA was dissolved in 2 ml DMSO and suspended in 2 ml propylene glycol. Each rat received the dose subcutaneously for seven consecutive days, after which their brain blood flow was immediately and dynamically studied using 99mTc-HMPAO imaging. Control animals received subcutaneous injections of 0.5 ml saline solution (pH 8.6). Ten minutes prior to imaging, each rat was anesthetized through intraperitoneal injection of 25 mg/kg intraval sodium. Then, an intravenous line was inserted into the tail vein of each rat, after which each rat was transferred to and fixed on a fixed board against the table of the gamma camera. The rat was positioned so that its head was localized at the center of the field of view. Then, it was injected with 125 MBq of 99mTc-HMPAO intravenously, followed by a saline push during the brain image acquisition. The acquisition lasted 60 min and was captured in images with the gamma camera for the entire experiment.
2.5. 99mTc-HMPAO Brain Flow Gamma Camera Imaging
To study the brain blood flow, the injected rats were imaged. Each scan was performed using a single-head gamma (
2.6. Image Processing
The brain scans and frame data were collected and combined in one frame. Then, the region of interest (ROI) around the brain (target, T), the whole-body region (WB), and the background soft tissues of the rat (BG) was delimited. The net 99mTc-HMPAO uptake of the brain was calculated as the net target count,
2.7. 99mTc-HMPAO Uptake
After imaging, the rats were euthanized by cervical dislocation to collect their blood, heart, lung, brain, kidneys, liver, gut, bone, adrenals, and testes for further study. The rats from each group were resected, and the respective organs were counted in kilobecquerels (kBq) using a dose calibrator. The counts were recorded and expressed as the
2.8. Brain Blood Flow Quantification
In three experiments, for each group (C, L, and H), the cerebral blood flow was quantified using the method done by Hale et al. [34] with minor modifications using colored microspheres. In brief, the 12
2.9. Histological Studies and Brain Damage Scoring
Brain light microscopy and histology were performed on a total of three rats from each group. After each animal was sacrificed, 4 ml of blood was collected. The animal’s body was perfused with normal saline accompanied by a cold fixative (4% paraformaldehyde and 0.1% glutaraldehyde in 0.1-M PO4 buffer, pH 7.3). After perfusion, 5 mm tissue samples of the brain were collected, placed in the fixative for 24 h, and washed in 0.1 M PO4 buffer for 72 h. Then, the samples were dried in graded ethanol and cleared in xylene using an automatic processor, after which they were embedded in paraffin wax. Thin, 5
2.10. Modified Unpredictable Mild Stress Model
The unpredictable mild stress model was followed, as described by [36–38], with timing modifications. The remaining animals assigned to the stress group were exposed to one dose of 0.4 mg/kg of 13-RA subcutaneously for seven consecutive days compared to C group, which was given 0 mg of 13-RA per kg of body weight. The control animals were manipulated in a separate room at the same start and end times of the restraint session. All animal groups were regularly provided food and water. After seven days, the animals were not given water a day before the conduct of the sucrose preference test (SPT), as described in [39, 40]. The rats were kept in their assigned cages with free access to two bottles, one of which contained 300 ml of sucrose solution (1%,
2.11. Adrenocorticotropic Hormone (ACTH) and Lipid Adrenal Measurement for Stress
In this study, we used two groups: the control group, which, as mentioned, received 0 mg/kg 13-RA, and the ACTH group, which was given 0.4 mg/kg 13-RA subcutaneously for seven consecutive days. Three milliliters of adrenal homogenates was mixed with a solution that contained 850 mM sucrose (5 mg wet weight/ml), centrifuged at 3,600 rpm (1,452 g), then assayed and analyzed for fatty acids according to the validation protocol of fatty acid quantification, the extraction ratio, hydrolysis, and the methylation efficacy, which was originally established by Ingalls et al. [41], and used in this study with the volume modifications [42, 43].
2.12. Study of Plasma Free Fatty Acids via Gas Chromatography/Mass Spectrometry (GC/MS)
Rat lipid samples at a volume of 0.25 ml were used to measure the number of unesterified (free) fatty acids. Each sample was vortexed vigorously for 1 min and then centrifuged at 3,000 rpm (1,615 g) for 10 min at room temperature using an MSE Mistral 2000 centrifuge (with a
2.13. Data Presentation and Statistical Analysis
All data, unless otherwise stated, were expressed as the
3. Results
3.1. Analysis of the Brain Radioactivity Concentration
The results showed that the brain radioactivity concentration after the intravenous administration of 99mTc-HMPAO was steady 30–60 min postinjection. The uptake was determined using the ROI around the T to WB ratio. The next T count, NTC, showed increased uptake (to double) in a dose-dependent manner. Figure 1 shows the images of the 99mTc-HMPAO (brain) male rats. The composite 0–60 min image and the ROIs were used as C, L, and H groups. The unpaired multiple comparison tests showed highly significant differences between C and L and H (

99mTc-HMPAO (brain) male rat imaging. Composite 0–60 min images and regions of interest (ROIs) used as (a) receiving 0 mg/kg 13-RA as the control (C), (b) receiving 0.3 mg/kg 13-RA as the low-dose group (L), and (c) receiving 0.5 mg/kg 13-RA as the high-dose group (H). Brain ROI: BG: background and WB: whole body (


3.2. 99mTc-HMPAO Uptake
Table 1 shows the resected organs. The uptakes were arranged in descending order from the highest to the lowest uptakes, which were in the brain, kidney, heart, liver, gut, adrenal, lung, testes, blood, and bone. The ANOVA showed
99mTc-HMPAO uptake for control (C), low-dose 13-RA-injected rats (L), and high-dose 13-RA-injected rats (H).
Activity in resected organs, normalized and corrected by background counts and whole-body radioactivity. The values are expressed as percentages.
3.3. Brain Blood Flow Quantification
The results of the microsphere method showed the evidence that the composite images at 0-60 min postinjection of 99mTc-HMPAO reflected cerebral blood flow (CBF). There was a strong correlation (

Quantification of cerebral blood flow using the microsphere method. C: control group receiving 0 mg/kg 13-RA; L: low-dose group receiving 0.3 mg/kg 13-RA; H: high-dose group receiving 0.5 mg/kg 13-RA.
3.4. Histological Changes in the Brain
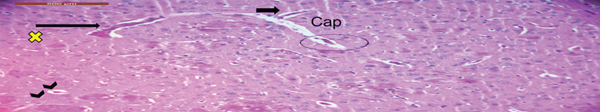
Figure 3 shows the histological changes of the brain light microscopy of each of the C, L, and H groups using the HE and cresyl violet stain. Figure 3(a) shows a normal rat brain with a uniform and normal arrangement and structure of cerebral capillaries (capillary structure, Cap) and blood vessels with a normal configuration. However, with the use of the 13-RA dose, the L group rats showed increased glycolipids, which signified congestion of the cerebral blood vessels (small arrow) and focal hemorrhage (long arrows). The hippocampal neurons were reduced to ghost cells, with no nuclear staining, a barely visible cell outline, and softening and hemorrhage (chevron arrows). The H group showed necrosis of neurons (small stars), neuronophagia (small circles), and neuronal edema (small crescent shape). In addition, subarachnoid hemorrhage was seen.

Histological findings of brain tissues stained with hematoxylin-eosin (HE) in rats using light microscopy. (a) The normal (control group) rat brain showed a uniform and normal arrangement and structure of cerebral capillaries (capillary structure, Cap) and blood vessels with normal configuration. (b) The L group (low13-RA dose) showed an increase in glycolipids (yellow x sign), congestion of cerebral blood vessels (short wide small arrow), and focal hemorrhage (long arrow). The hippocampal neurons were reduced to ghost cells, with no nuclear staining and a barely visible cell outline plus softening and hemorrhage (chevron arrows). (c) The H group (high dose) showed necrosis of neurons (small stars), neuronophagia (small circles), and neuronal edema (small crescent shape). Subarachnoid hemorrhage was also seen.

3.5. Modified Unpredictable Mild Stress Model
According to the results of the stress model, the stress group showed a highly significant unpaired multiple comparison test result with a
3.6. GC/MS
Figure 4 shows the GC/MS results for the free fatty acids in both C and the stress group, indicated by the ACTH adrenal homogenates. There were highly significant differences among the experiment groups in both the unpaired

GC/MS results of free fatty acids extracted from the control and adrenocorticotropic hormone (ACTH) adrenals. Values are expressed as
4. Discussion
The main objective of this study was to investigate the relationship between stress and 13-RA treatment. It was hypothesized that 13-RA treatment affects brain blood flow, thus playing a role in the development of depression. The study was conducted by imaging brain perfusion in rat models treated with 13-RA.
Our results showed that 13-RA increases blood flow, perfusion, and uptake in rat brain in a dose-dependent manner. Our study presents evidence for the experimental design that composite images at 0–60 min reflected the brain or cerebral blood flow (CBF). The well-known microsphere method absolutely quantified CBF (ml/min/g tissue) [45–47] and correlated strongly with the 99mTc-HMPAO radioactivity images and results. Even, the results of the 99mTc-HMPAO brain uptake in the L group increased to twice that in the control, and the brain uptake in the H group was fourfold that in the control.
These results posit that 99mTc-HMPAO uptake reflects the CBF, which is related to the whole brain or cerebellum CBF. This agrees with many studies that illustrate changes in regional CBF are reflected in parallel to the changes in the 99mTc-HMPAO retention and uptake [48–53], as well as cerebral metabolism.
There were also observations relative to brain histology in the C, L, and H groups. The histological changes of the brain light microscopy of each of the C, L, and H groups with the use of the 13-RA dose showed increased glycolipids, signifying the congestion of the cerebral blood vessels, focal hemorrhage, and soft neurons in the L group. Similar changes were observed in the H group such as neuronal edema and necrosis, neuronophagia, and subarachnoid hemorrhage.
These results therefore proved that 13-RA caused the inhibition of neurogenesis. This has been hypothesized to have a role in depression in a number of studies [54–56]. Besides, this agrees with many studies that reported prominent histopathological changes in the brain following 13-RA and in a dosed manner [57, 58]. The results insinuate that the mechanism behind 13-RA induced depression could be from the ability of the drug to inhibit neurogenesis. This study therefore agrees with several studies that showed histological changes of reductions in the density and size of neurons (neuronal atrophy) and dysfunctional neuronal circuits in the hippocampus and prefrontal cortex [59, 60] in major depression. Different alterations of the regional distribution of 99mTc-HMPAO retention have been reported in depressed patients [61]. Many clinical studies showed that 13-RA causes stress manifested by somatic intracranial hypertension, cerebral infarction, cerebral or thrombotic hemorrhage [62, 63], encephalopathy [64], brain blood vessel inflammation [65], and neurodegenerative disorders [66].
The correlation between brain perfusion and stress is also evident in the experimental results. The findings of this study show that 13-RA caused depression-like behavior in rats following the modified unpredictable mild stress model. This also agrees with many studies, which proved a strong relationship between 13-RA treatment and depression-like behavior [25]. In general, the relationship between stress and depression is well established [67, 68]. It was reported that the pathophysiology of the appearance of depressive symptoms after 13-RA administration in the rat mild stress model is linked to the dysfunctional GABAergic inhibitory system. This system is able to suppress the hippocampal cell division and hippocampal-dependent learning [61]. In addition, this was confirmed impairing the explicit memory and learning and at doses likely to produce serum levels within the range typically used to treat acne in humans [62] in some studies. These studies report that the retention in the brain is proportional to the CBF and is related to both the local hemodynamics status and the cellular content of a reduced glutathione, which reflects the amount of cellular injury or stress [68], the oxidative stress [69, 70], and the psychosocial stress. One study reported a strong association between psychological stress and CBF damage. Induced stress in mice produced cerebrovascular microbleeds in scattered locations, vascular pathology, and blood-brain barrier (BBB) breakdown. Similarly, some studies reported that 13-RA can pass the BBB and confirmed visualizing plasma immunoglobulins and erythrocytes within the parenchyma and perivascular spaces of mouse brains and high gene expression profiles during and after vascular disruptions due to stress [71].
Another major finding from our study is that the gradual increment vasodilation of blood flow to rat brain after 13-RA administration occurred in a dose-dependent manner. This was proved y analyzing stress. In physiology, stress causes the body to act in two directions: the first direction is the nervous system, such as the perception at the level of the brain, and the second direction is the endocrine system. One study presented a link between stress and neurovascular coupling, a physiological process that drives changes in blood flow to specific brain regions. The hemodynamic response function showed an increased signal in several brain regions, including in the temporal and prefrontal cortex, indicating changes in blood flow regulation in response to stress as well as the cortisol secretion and the adrenocorticotropic hormone (ACTH) response [72]. The ACTH is a hormone made by the pituitary gland, a small gland located at the bottom of the brain. ACTH controls the appearance of another hormone known as cortisol. Cortisol is formed by the adrenal glands, two small glands based above the kidneys. Cortisol provides an essential lead to respond to stress and regulate the use of food and energy or body metabolism.
In this study, we proved that ACTH prompts the adrenal cortex to release corticoids (glucocorticoids and mineralocorticoids) for energy by converting glycogen to glucose (glycogenolysis) and by breaking down fat into fatty acids and glycerol (lipolysis). Just like steroids, glucocorticoids and mineralocorticoids are the driving forces and the trigger for many neuropsychiatric symptoms, effects, and disorders, such as depression, quirk, mania, raving, hallucination, agitation, and even a sevenfold increased suicide or attempt average after the release of steroids [73, 74]. In our study, the mechanism of brain perfusion changes is due to the release of steroids. This is in agreement with one study associated between the presence of glucocorticoids and changes in brain volume and white matter microstructure [75]. Another study confirmed a strong association between the presence of glucocorticoids and the local vasodilation of spinal cord [76]. ACTH could also have caused the mobilization of free fatty acids and the vasodilation of the brain in our study.
Brain imaging results from this study also illustrate that 13-RA causes vasodilation. This phenomenon suggests a release of catecholamines for a few seconds to accelerate the vasodilatation of arteries throughout the working muscles and the brain. Catecholamines are also involved in stress responses. A unique observation from this study was the response of HPA axis stimulation to stress. The free fatty acids were consumed after 13-RA administration, and the free fatty acids were reduced significantly in the experimental groups in comparison with C. This agrees with the results of several studies that observed that ACTH and pituitary hormones stimulated the reduction of free fatty acid concentration in a dose-dependent manner and increased their consumption by the muscles during activity [58, 77]. The reduction in free fatty acid level was reported to cause a stimulatory effect on ACTH and the cortisol secretion [78, 79]. However, some studies reported that there was no effect of free fatty acids on ACTH and cortisol secretion in normal [80]. Since 13-RA causes mild suppression of pituitary hormone levels [77], the outcome of 13-RA treatment in humans might be directly related to altered steroid levels because of vasodilation in the blood circulation as explained above.
Moreover, this study points out the need to prevent the use of medications or drug mechanisms that may lead to stress. Studies predispose subjects to an increased risk of suicide and depression after reduction in free fatty acids. 13-RA is strongly associated with stress. This agrees with our results. The highest 99mTc-HMPAO uptake counts were ordered from the highest to the lowest organ uptake and were in the brain, kidneys, heart, liver, gut, adrenals, lungs, testes, blood, and bones.
Many studies reported that 13-RA causes disruption in lipids such as dyslipidemia, hypertriglyceridemia [70], and imbalance in endocrine systems [81]. This study proves that free fatty acids are reduced due to depression and stress. Palmitate, a 16-carbon saturated fatty acid, is reduced in the brain after 13-RA administration due to stress formation and depression [82]. The linoleate, oleate, and stearate were reduced; some studies reported that this reduction is due to changes in the phospholipid formation [83]. Similarly, the arachidonate [84] and the docosahexaenoate were reduced and were reported due to depression [85]. The docosatetraenoate free fatty acids were reduced with unknown cause in comparison to the control group. The relationship of ACTH secretion and lipid levels is still unknown. Some studies reported that lipid levels have no effect on the ACTH secretion [86], while other studies reported that plasma lipid profile is affected by the ACTH [87, 88].
Therefore, our 99mTc-HMPAO imaging, behavioral, histological, and fatty acid experiments proved that the CBF is affected and increased by 13-RA. The study presented a rat model to detect the effect of 13-RA in causing stress such as depression using nuclear medicine imaging. Given that there has been an increasing incidence of depression and suicides in patients treated with 13-cis RA, the study has established how the drug affects neuronal signaling and the mechanism of action of neuroactive steroids and hormones to induce the disease. Results illustrate that the mechanism of action may affect and target the gamma-aminobutyric acid (GABA) receptors, which were reported to increase CBF and vasodilation in a dose-dependent manner. Findings that 13-cis RA increases brain perfusion to induce depression could help in the development of new antidepressants.
5. Conclusion
To our knowledge, this is the first study on the effects of 13-RA on rat brain using functional nuclear medicine imaging. Our findings suggest that 13-RA affects brain functioning by increasing blood flow to the brain and consuming free fatty acids, thus providing a possible biological mechanism by which 13-RA treatment could lead to depression and suicidal ideation in a minority of vulnerable acne patients. Studies using female rats and steroid levels to evaluate the effects of 13-RA on brain functioning are ongoing. Further investigation of the cerebral region that is most involved in the accumulation and uptake of 99mTc-HMPAO is needed, as well as which area in the brain is the most sensitive or is more specific for diagnosing the cerebral lobe pathology based on 99mTc-HMPAO. Besides, further analysis software coregistering 99mTc-HMPAO images to an MRI template will be under consideration.
Footnotes
Data Availability
The datasets generated in this study are available from the corresponding author upon reasonable request.
Conflicts of Interest
The authors have no conflict of interest.
Acknowledgments
We would like to acknowledge Kuwait University and King Faisal University for supporting this research using their facilities.
