Abstract
It has been a big challenge to distinguish synchronous multiple primary lung cancer (sMPLC) from primary lung cancer with intrapulmonary metastases (IPM). We aimed to assess the clinical application of dynamic 18F-FDG PET/CT in patients with multiple lung cancer nodules. We enrolled patients with multiple pulmonary nodules who had undergone dynamic 18F-FDG PET/CT and divided them into sMPLC and IPM groups based on comprehensive features. The SUVmax, fitted
1. Introduction
Lung cancer is among the most common cancers and the leading cause of cancer-related death worldwide [1]. Given the advanced diagnostic and surveillance methods, as well as the increasing aging population, there has been a recent increase in the incidence of synchronous multiple primary lung cancer (sMPLC). It is crucial to discriminate sMPLC from primary lung cancer with intrapulmonary metastases (IPM) due to their varying clinical staging, treatment strategies, management, and prognosis. Patients with sMPLC are staged separately and generally treated with curative surgical resection because of early stage and favorable prognosis while those with IPM in advanced stages receive chemotherapy or radiotherapy with palliative intent [2].
Differential diagnosis between sMPLC and advanced lung cancer is traditionally based on conventional histopathologic features. These include location, morphology, histologic type, time interval, lymphatic invasion, metastases, and clinical manifestation, as described by the Martini and Melamed criteria published in 1975 [3], which was subsequently modified by the American College of Chest Physicians (ACCP) [2]. However, this traditional diagnostic process often involves an overlap in a significant proportion of cases, especially for tumors with a similar morphology or histotype, which impedes the determination of whether they share the same clone origin. Moreover, some patients cannot undergo preoperative histological examination or relative surgery. Further, some tumors cannot be sampled and tested pathologically if the patient has a poor physical condition, including limited cardiopulmonary reserves.
Novel molecular and genomic analyses, including biomarker assessment (driver gene mutations) [4], array comparative genomic hybridization [5], shallow whole-genome sequencing [6], and TP53 mutation analysis [7], can define the between-lesion correlation. These molecular biological techniques allow the detection and analysis of specific molecular markers or mutation sites to determine the heterogenicity of two cancer foci. However, given the substantial misclassification rate, which results from limited sensitivity, stabilization, repeatability, and economic benefits, techniques that could be able to precisely define sMPLC or IPM have not been widely applied in clinic [8].
Preoperative differentiation of primary tumors from metastases is more clinically pivotal than postoperative differentiation. Moreover, preoperative imaging examination plays a significant role. However, there is limited information regarding the imaging and metabolic characteristics of multiple lung cancer nodules [8]. Pretreatment chest high-resolution computed tomography (HRCT) is a conventional and cost-effective means of preliminary diagnosis. Since distinguishing IPM from MPLC is empirically based on the morphological pulmonary nodule features, it is difficult to obtain a definitive diagnosis of a primary tumor or metastasis solely based on CT characteristics. Moreover, according to the ACCP clinical practice guidelines, 18F-fluorodeoxyglucose positron emission tomography/computed tomography (18F-FDG PET/CT) plays a significant role in guiding clinical decisions and is a recommended standard workflow for patients with potentially curable lung cancer [2]. 18F-FDG PET/CT has been extensively used in patients with lung cancer for diagnosis [9], staging [10], prediction (i.e., monitoring therapy response) [11], and prognosis [12]. These mentioned applications indicate that 18F-FDG PET could be used to identify early-stage sMPLC, which involves multiple pulmonary sites. Several heterogeneous methods could be used to determine 18F-FDG uptake. Dynamic PET acquisition, immediately starting from radiotracer injection, measures drug activity change over time. Dynamic PET including multiple frames usually requires long image acquisition time (for 18F-FDG, generally lasting for 60 minutes) and only allows the assessment of one FOV. Generally speaking, the radioactivity concentration of radiotracer in blood and tissue will be changing most rapidly early after injection; therefore, most dynamic PET acquisition has finer time frames at early time points and wider time frames at later time points [13]. On the other hand, static PET acquisition is a common clinical PET acquisition mode. After radiotracer injection for a period of time, when the physiological metabolism or binding in vivo is stable, the multiple-FOV and single frame static emission scan is acquired. For 18F-FDG, static emission scan lasting for 10-15 minutes is usually started at 45-60 minutes postinjection. In routine clinical practice, the standardized uptake value (SUV) based on static PET scanning, which is a simple semiquantitative index that reflects the metabolic activity of tumor lesions, is widely applied in PET imaging evaluation. SUV is associated with several tumor characteristics, including histopathological subtypes [14]; tumor proliferation [14], differentiation [15], and aggressiveness [16]; and tumor stage, recurrence, and survival [17, 18]. However, the SUV outcome is affected by several factors, including the image acquisition time [19], acquisition mode [20], reconstruction mode [21], serum glucose and insulin levels [22, 23], and positive contrast agent [24]. Theoretically, Patlak graphic analysis based on dynamic PET scanning is the most classical fully quantitative measure of glucose metabolism that involves irreversible trapping. It calculates the 18F-FDG net influx rate constant (i.e., uptake rate constant,
Since sMPLC with a separate clonal origin often indicates early-stage and less than two-year interval tumorigenesis, we hypothesized that there would be the same SUV or
This study is aimed at assessing the differential diagnostic ability of dynamic 18F-FDG PET scan in patients with lung cancer involving multiple pulmonary sites. This could be able to provide a novel method for sMPLC diagnosis in clinic.
2. Materials and Methods
2.1. Patients
In our center, firstly, all first-visit patients with related symptoms underwent preliminary screening using chest HRCT. Then, other examinations mainly including serologic examination, tumor-associated antigen, brain magnetic resonance imaging, and cervical and abdominal CT scan further clarified the general condition and clinical TNM classification. Fifty-three patients with multiple pulmonary nodules regardless of extrapulmonary metastases received dynamic 18F-FDG PET scans and had not received any medical treatment before this. After careful preoperative evaluation based on above examination, twenty-seven suitable patients (stage I, stage II, and partial stage IIIA) without medical contraindications received radical surgical resection. Partial inoperable patients (stage IV and partial stage IIIA) were followed by CT-guided percutaneous lung puncture biopsy, bronchoscopic biopsy, or superficial lymph node biopsy mainly based on tumor location. What calls for special attention was that two cases whose

Diagram of case inclusion and exclusion.
The study was approved by the institutional review board of the Fifth Affiliated Hospital of Sun Yat-sen University (IRB protocol number ZDWY.FZYX.002). All the included patients provided signed informed consent. The clinical trial registration number is NCT03679936. Baseline clinical characteristics, including sex, age, height, weight, smoking history, and tumor characteristics, were obtained from electronic medical records with permission.
2.2. Dynamic PET Data Acquisition and Reconstruction
Dynamic PET/CT scan was performed using 112-ring digital light guide PET/CT (uMI780, United Imaging, China). The patients were fasted for at least 6 h before scanning. The patient was restricted to movement in the scanner to avoid motion artifacts and conducive to subsequent accurate fusion of PET and CT. The scan covered the region between the thoracic inlet and the lower liver margin. Each PET/CT scan began with a transmission CT scan for 5 seconds that was used for attenuation correction. Next, an 18F-FDG bolus (range 143–327 MBq) was intravenously injected and a dynamic PET scan was acquired immediately as follows. Dynamic data were collected for 60 min comprising 48 frames with the following dimensions:
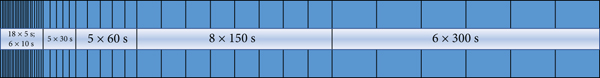
Dynamic acquisition mode and Patlak model. (a) Dynamic data were collected for 60 min comprising 48 frames:

2.3. PET Data Analysis
Two experienced nuclear medicine physicians analyzed the dynamic 18F-FDG PET/CT images using Carimas 2.10 software (Turku PET center, Finland). Reconstructed PET images were analyzed as follows: (1) definition of volumes of interest (VOIs); (2) obtaining the time-activity curve (TAC) of the left ventricle (i.e., plasma input function) and lung cancer nodules; (3)
First, three-dimensional VOIs were manually drawn over the left ventricle (arterial blood pool) and tumors with a landmark using the Carimas 2.10 software. VOIs were visually localized using CT images. Moreover, the maximum diameters (
Second, by projecting the VOIs onto the complete dynamic dataset, the Carimas analysis software automatically output relative TAC data of the VOIs. The left ventricle TAC was used for plasma input function, also known as image-derived input function (IDIF).
Third, the TAC of the left ventricle and tumors were used to fit standard Patlak modeling to assess FDG tissue kinetics via the least square regression method using Matlab 2018b (MathWorks Inc., Natick, MA, USA) [25, 26]. Patlak analysis was performed using data obtained between 20 and 60 postinjection minutes (i.e., between frames 38 and 48). The Patlak model for 18F-FDG metabolism in lung cancer (Figure 2(b)) has been previously described in detail [30].
Fourth, the last frame (at 55-60 min postinjection) of the dynamic scans was used for static analysis to obtain the SUV. The SUVmax of each pulmonary malignant lesion on PET/CT was extracted from the dynamic data.
2.4. Calculation of Indicators:
and
Based on SUVmax measured from 18F-FDG PET, the fitted
2.5. Histopathologic Analysis and Genetic Mutation Analysis
All the punctured tissue and resected specimens were fixed, dehydrated, embedded, sectioned, and stained for microscopic examination. Two experienced pathologists recorded the histopathologic diagnoses and features, including the histotype; lymphatic, nerve, and pleural invasion; extranodal extension; and regional lymph node metastasis.
Gene sequencing of some patients (18 out of 19 in sMPLC group; 20 out of 26 in IPM group) was carried out by the Beijing Genomics Institute. Gene sequencing projects include (1) Oseq TM-T tumor individualized diagnosis and treatment gene detection: 508 gene coding regions and partial intron regions closely related to solid tumors were detected; (2) Oseq TM-ctDNA noninvasive tumor individualized diagnosis and treatment gene detection: 508 gene coding regions and partial intron regions closely related to solid tumors were detected; (3) Oseq TM-T lung cancer individualized diagnosis and treatment gene detection: 20 specific gene mutations (i.e., ALK, EGFR, KRAS, BRAF, ERBB2, RET, MET, ROS1, NRAS, HRAS, DDR2, PIK3CA, AKT1, FBXW7, MAP2K1, FGFR3, NTRK1, KIT, PTEN, and TP53) associated with individualized drug use in lung cancer were detected.
2.6. Statistical Analysis
Between-group differences in baseline characteristics were assessed using an unpaired two-tailed Student’s
3. Results
3.1. Patient and Tumor Characteristics and Gene Detection Results
There were no differences in the baseline characteristics between the sMPLC and IPM groups. Table 1 summarizes the gender distribution, age, height, body weight, FDG injection dose, and smoking history. IPM diagnoses were mainly based on radiologic and clinical patterns although the primary tumor was pathologically confirmed. sMPLC diagnoses were primarily based on histopathologic features. In both the sMPLC and IPM groups, the most commonly diagnosed tumor was adenocarcinoma with a majority being unilateral (68% and 71%, respectively). Table 1 presents the tumor characteristics. Supplementary Tables S1 and S2, respectively, show the detailed gene mutation detection information of the sMPLC and IPM groups, mainly including sample type, tumor mutation burden, microsatellite instability, and mutations detected. EGFR mutations were the most common mutations whether in the sMPLC group or in the IPM group. Particularly, in the sMPLC group, almost two primary tumors were tested for mutation detection because of high resection rate and diagnosis requirement; however, only primary tumors were tested for mutation detection in the IPM group because almost only one lesion was resected or biopsied (Supplementary Tables S1 and S2).
Patient and tumor characteristics.
sMPLC: synchronous multiple primary lung cancer; IPM: intrapulmonary metastases; SCC: squamous cell carcinoma; AC: adenocarcinoma; SD: standard deviation of the mean.
3.2. sMPLC vs. IPM: Absolute Differences of
There was no significant difference in
Statistical results of the sMPLC and IPM groups.
Data are shown as median (minimum–maximum). sMPLC: synchronous multiple primary lung cancer; IPM: intrapulmonary metastases;
Individual results in the sMPLC group.
aSpecifically, tumor 1 and tumor 2 in patient no. 16 have the same pathological subtype, but the different gene mutation. sMPLC: synchronous multiple primary lung cancer;
Individual results in the IPM group.
IPM: intrapulmonary metastases;
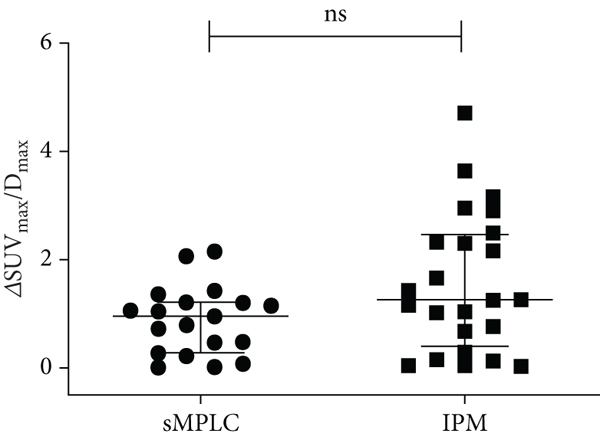
sMPLC vs. IPM: absolute differences of





Representative sMPLC and IPM examples. (a–f) sMPLC examples. (a–c) FDG-PET of a patient (patient #1 in Table 3) with two synchronous primary lung tumors. Dynamic FDG PET/CT revealed an acinar adenocarcinoma in the right upper lobe (b) (white arrow,











3.3. Receiver Operating Characteristic (ROC) Curve Analysis
ROC curve analysis was used to determine the diagnostic accuracy of
3.4. Unilateral sMPLC vs. Bilateral sMPLC
The lesion location has been reported to contribute to the over survival (OS) of MPLC [31, 32]; therefore, we compared the results between unilateral and bilateral sMPLC. The bilateral sMPLC group had a slightly higher
3.5. Unilateral IPM vs. Bilateral IPM
According to the 8th Edition Lung Cancer Stage Classification, unilateral and bilateral IPM could have different T and M categories, which indicates different stages and OS [33]. Therefore, we compared differences in the indicators between unilateral and bilateral IPM. The bilateral IPM group had a slightly higher
3.6. IPM Subgroup Comparisons Based on Primary Tumor Size (≤3 cm vs. 3-5 cm vs. >5 cm)
Size plays a critical role in defining the T category as indicated by the proposed size cut-points of the 8th edition Lung Cancer Stage Classification [34]. We compared differences in the indicators among three IPM subgroups (≤3 cm vs. 3-5 cm vs. >5 cm). Nevertheless, there was no significant among-group difference in
3.7. Diagnostic Results Based on CT Characteristics
Table 5 summarizes the diagnostic results of at least two experienced radiologists based on CT characteristics in the sMPLC group and the IPM group. Based on CT characteristics, 26 out of 43 cases were diagnosed correctly. Based on the optimal cut-off value of
Diagnostic results based on CT characteristics.
“Primary” represents that tumors were considered as primary tumors. “Metastasis” represents that tumors were considered as metastatic tumors. “Unsure” represents that no definite diagnosis could be made based on CT characteristics.
4. Discussion
To our knowledge, this was the first study on the application of dynamic 18F-FDG PET to discriminate between tumors with common (i.e., IPM) and separate (i.e., sMPLC) lineages. Previous PET studies have predominantly focused on static imaging. Our findings indicated that dynamic 18F-FDG PET could be able to provide more detailed parameters for sMPLC identification.
The Martini and Melamed criteria are currently the most widely accepted; however, there remains no standard and uniform clinical guidelines for MPLC. There is no consensus among major lung cancer research institutes regarding MPLC classification. It is difficult to discriminate sMPLC from IPM without a lung, lymph node biopsy, or surgical procedure, which is important since the two conditions have significantly different therapeutic regimens and prognosis. Previous studies have reported that the OS of patients with MPLC was significantly better than those with metastatic tumors [31, 35]. Patients with MPLC are generally treated with curative surgical treatment while those in advanced stages receive chemotherapy or radiotherapy with palliative intent [2].
In routine clinical settings, preoperative differentiation of primary tumors from metastases is more critical than postoperative differentiation. Moreover, preoperative imaging examination plays a crucial role. It is recommended that patients with multiple pulmonary nodules undergo PET scans for careful systemic assessment based on the ACCP guidelines [2]. There is limited information regarding the imaging and metabolic characteristics of multiple lung cancer nodules [8]. Moreover, there have been few studies on the value of preoperative imaging for distinguishing MPLC from IPM in patients with multiple lung cancers [36].
Several case reports have reported incidental detection of sMPLC using 18F-FDG PET [37–40]. Contrastingly, there have been few studies indicating that 18F-FDG PET could be able to locate the clonal origin of synchronous multiple tumors [41–43]. Dijkman et al. reported that the relative between-tumor difference in
Similar to our study, Liu et al. only included patients with both cancers located in the lung and reported significant differences in the SUVmax ratio (bigger SUVmax/smaller SUVmax) between sMPLC and IPM. This indicated that the between-tumor SUVmax ratio could differentiate sMPLC from IPM [43]. In our study, we did not find a significant between-group difference in the SUVmax ratio and
Both Dijkman et al. and Liu et al. suggested that the SUV of tumors with a common lineage were more consistent than those with separate lineage [41, 43]. Moreover, they reported that related indicators (e.g., ΔSUV% and SUVmax ratio) were significantly higher in the sMPLC group than in the IPM group. Inconsistent with these previous findings, we found that
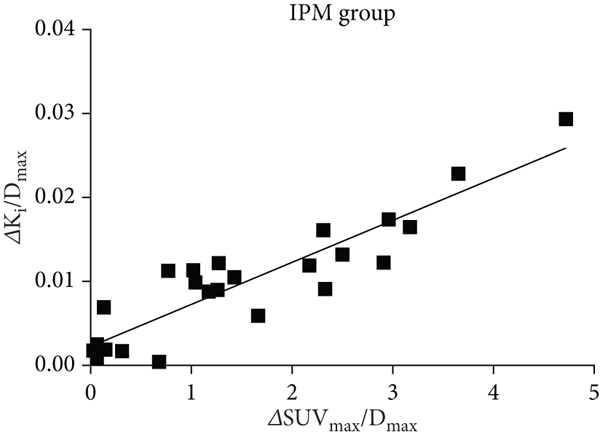
In our study, there was a good correlation between
>According to the 8th Edition Lung Cancer Stage Classification [46], a separate T, N, M category should be designated to each tumor for second primary lung cancer. However, for patients with IPM, tumor nodules located in the same and different lobes of the unilateral lung and in the bilateral lung are staged as T3, T4, and M1a, respectively. As a result, unilateral IPM and bilateral IPM have different T and M categories, which is indicative of different stages. Regarding the site of the separate tumor nodule relative to the primary tumor in clinically staged patients, tumors in the same lobe are associated with a superior OS than those in different ipsilateral lobes. Moreover, the tumors with different contralateral lobes were associated with the worst OS [33]. Consequently, we determined differences in the indicators between unilateral IPM and bilateral IPM. Although there was no significant difference, the bilateral IPM group had a slightly higher
We observed that
This study has several limitations. Firstly, we employed a small sample size (43 cases); therefore, the results are preliminary and exploratory. Second, most of the cases with metastatic disease were diagnosed without histopathological confirmation based on typical clinical and radiological features, although the primary tumor was pathologically confirmed. Third, partial tumors were <1 cm with low SUV, which could have resulted in SUV biases due to the partial volume effect and statistical noise [50]. Fourth, for the optimal cut-off value of
5. Conclusions
Our findings indicated that dynamic 18F-FDG PET/CT could be a useful tool for distinguishing sMPLC from IPM complemented by histopathologic, clinical, and genetic evaluation, especially during preoperative assessment, which is mainly dependent on clinical and imaging characteristics.
Footnotes
Data Availability
The data presented in this study are available on reasonable request from the corresponding author. The data are not publicly available due to privacy or ethical issues.
Ethical Approval
The study was conducted according to the guidelines of the Declaration of Helsinki and approved by the Institutional Review Board of the Fifth Affiliated Hospital of Sun Yat-sen University (IRB protocol number ZDWY.FZYX.002).
Consent
Informed consent was obtained from all subjects involved in the study.
Conflicts of Interest
The authors declare no conflict of interest.
Authors’ Contributions
H.J.J. and Q.D.C. were responsible for conceptualization; W.Z.L. and M.Y. were responsible for methodology; S.Y. and L.B. were responsible for software; X.F.P. and X.J.W. were responsible for formal analysis; X.H.H. and H.C.Z. were responsible for investigation; Q.D.C., Y.W., and Z.L. were responsible for resources; J.Z.X., X.H.H., H.C.Z., and X.J.W. were responsible for data curation; W.Z.L. and M.Y. were responsible for writing—original draft preparation; H.J.J. and Q.D.C. were responsible for writing—review and editing; W.Z.L. and M.Y. were responsible for visualization; H.J.J. was responsible for supervision; H.J.J. and Q.D.C. were responsible for project administration; H.J.J. and H.S. were responsible for funding acquisition. All authors have read and agreed to the published version of the manuscript. Weize Lv and Min Yang are equally contributed to this study as the first author. Hongjun Jin and Qingdong Cao are equally contributed to this study as the corresponding author.
Acknowledgments
The authors would like to thank the patients who participated in the study. Moreover, the authors appreciate Ms. Ping Jiang, Ms. Xiaojuan Jia, Mr. Wenhua Xu, and Mr. Fanwei Zhang from the Department of Nuclear Medicine, the Fifth Affiliated Hospital of Sun Yat-sen University, for their technical supports. Thanks are due to Prof. Chunlei Han for his kind help in model fitting from Turku PET Centre, Finland. This research was funded by the National Key R&D Program of China (2018YFC0910600), the National Natural Science Foundation of China (81871382 and 82150610508), grants from the Guangdong Science and Technology Department to the Guangdong Provincial Key Laboratory of Biomedical Imaging (2018B030322006), and Starting Fund (310103050303-220904094238) from the Fifth Affiliated Hospital, Sun Yat-sen University.
