Abstract
Hepatocellular carcinoma (HCC) is a notoriously difficult cancer to treat. The recent development of immune checkpoint inhibitors has revolutionised HCC therapy; however, successful response is only observed in a small percentage of patients. Biomarkers typically used to predict treatment response in other tumour types are ineffective in HCC, which arises in an immune-suppressive environment. However, imaging markers that measure changes in tumour infiltrating immune cells may supply information that can be used to determine which patients are responding to therapy posttreatment. We have evaluated [18F]AlF-mNOTA-GZP, a radiolabeled peptide targeting granzyme B, to stratify response to ICIs in a HEPA 1-tumours, a syngeneic model of HCC. Posttherapy, in vivo tumour retention of [18F]AlF-mNOTA-GZP was correlated to changes in tumour volume and tumour-infiltrating immune cells. [18F]AlF-mNOTA-GZP successfully stratified response to immune checkpoint inhibition in the syngeneic HEPA 1-6 model. FACS indicated significant changes in the immune environment including a decrease in immune suppressive CD4+ T regulatory cells and increases in tumour-associated GZB+ NK+ cells, which correlated well with tumour radiopharmaceutical uptake. While the immune response to ICI therapies differs in HCC compared to many other cancers, [18F]AlF-mNOTA-GZP retention is able to stratify response to ICI therapy associated with tumour infiltrating GZB+ NK+ cells in this complex tumour microenvironment.
1. Introduction
Hepatocellular carcinoma (HCC) is the most common primary liver cancer and is typically diagnosed at an advanced state [1]. First-line therapies for advanced HCC include systemic treatments such as the tyrosine kinase inhibitor sorafenib, but these are relatively ineffective and are associated with significant adverse effects [2]. The introduction of immune checkpoint inhibitor (ICI) therapies has been a major development in the treatment of advanced HCC, however, not all patients respond. The causes for this lack of response are poorly understood, biomarkers able to stratify response are an area of significant unmet clinical need, and accurate stratification of treatment response to ICIs is particularly important in HCC where many patients have existing chronic hepatic viral infections. Biomarkers associated with ICI efficacy in other cancer types appear to have little utility in HCC. Microsatellite instability (MSI) is a robust predictor of response to ICIs; however, the percentage of MSI-high cancers among HCC patients is very low [3, 4]. Likewise, tumour mutational burden (TMB) is a widely recognized biomarker of response but TMB high cancers are rare in HCC [5]. PD-L1 expression has also been demonstrated as a predictive biomarker of response in several cancer types; however, the efficacy of ICIs in HCC does not seem to be related to PD-L1 expression [6, 7]. In many cancers, the degree of tumour infiltrating lymphocytes (TILs) has been shown to be an accurate predictive of response to ICIs, and numerous biomarkers have been developed in an effort to quantify immune cell changes in the tumour microenvironment, usually targeting cytotoxic T cells [8–12]. However, the presence of TILs alone may not be enough for accurate stratification of response to ICI therapy in HCC where tumours have been found to harbour enriched populations of exhausted T cells, which show impaired immune surveillance, and a lack of response to ICIs [13]. Biomarkers of immune cell activation and tumouricidal activity may prove more successful [14]. Recently, noninvasive imaging of granzyme B, the serine protease released from active tumouricidal TILs, has been demonstrated to accurately stratify response to immunotherapy in syngeneic models of colon cancer [15–17]. In the current study, we have evaluated whether the granzyme B targeting peptide biomarker, [18F]AlF-mNOTA-GZP, is able to stratify responders to ICI therapy in the HEPA 1-6 syngeneic model of HCC, using flow cytometry to correlate tumour biomarker retention with tumour-associated immune cells.
2. Materials and Methods
2.1. [18F]AlF-mNOTA-GZP Radiochemistry
The synthesis of NOTA–β-Ala-Gly-Gly-Ile-Glu-Phe-Asp-CHO (mNOTA-GZP) and characterisation details have been described previously [16]. The radiosynthesis of [18F]AlF-mNOTA-GZP has also been described previously [16]. No-carrier-added aqueous [18F]fluoride ion was produced via the [18O(p,n)18F] nuclear reaction (GE PETtrace 860 cyclotron). Quality control analytical radio-HPLC was performed on a UFLC Shimazdu HPLC system equipped with dual-wavelength UV detector and a NaI/PMT-radiodetector (Flow-Ram, LabLogic). Radioactivity measurements were made with a CRC-55tPET dose calibrator (Capintec, USA). [18F]AlF-mNOTA-GZP was formulated as a colourless solution of 10% ethanol in saline (
2.2. Animal Procedures
Animal procedures adhered to Institutional Animal Care and Use Committee Singapore regulations (IACUC No. 181399). C57/BL6 mice were acquired from In Vivos (Singapore) at 6-8 weeks of age, housed at standard room temperature with food and water provided ad libitum. HEPA 1-6 cells were purchased from ATCC, cultured in RPMI supplemented with 10% FBS, 100 U/mL penicillin, and 100 μg/mL streptomycin at 37°C in a humidified atmosphere at 5% CO2. HEPA 1-6 cells were mixed 1 : 1 with Matrigel (Sigma) and injected subcutaneously into the right shoulder with a final concentration of 2X10 [6] per mouse. Tumour volumes were quantified using Vernier by callipers every 3 days from day 6 after tumour implantation. Tumour volumes were calculated as previously described using the modified ellipsoid formula (
Immune checkpoint inhibitor antibodies were purchased from Bio-X Cell (rat IgG2a isotype control (α-trinitrophenol mAb), rat IgG2a anti-mouse PD-1 (αPD1 mAb RMP1-14), and mouse IgG2b anti-mouse CTLA-4 (αCTLA4 mAb 9D9)) and dosed by intraperitoneal (i.p.) injection on days 6, 9, and 12 after tumour implantation (control IgG at 5 mg/kg, αPD1 monotherapy at 10 mg/kg, αCTLA4 monotherapy at 5 mg/kg or combined αPD1, and αCTLA4 therapy at 10 mg/kg and 5 mg/kg, respectively).
Response to therapy was determined using tumour growth inhibition (%TGI) on day 19 as previously described using the formula
2.3. PET-CT Imaging
Animals were imaged 12 days after tumour implantation as previously described using a Siemens Inveon PET-CT [15]. PET acquisitions were performed under isoflurane anaesthesia and acquired from 60–80 minutes postinjection (p.i.); CT was used for anatomical coregistration. Animal physiology was recorded using the Biovet physiological monitoring system. Calibrated images were reconstructed and analysed using FIJI and Amide software (version 10.3 Sourceforge). Volumes of interest (VOI) delineated by CT imaging were used to determine tissue uptake. Data are expressed as % of the injected dose per gram (%ID/g) of tumour tissue in the VOI.
2.4. Flow Cytometry
Tumours were removed post imaging and processed for flow cytometry as previously described [15]. Briefly, a single-cell suspension was generated from the tumour tissue and viable cells stained with the following antibodies for flow cytometry assessment on a BD FACSymphony; CD45 (clone 30-F11 BV570; Biolegend), CD3 (clone 500A2 BUV563; BD Biosciences), CD4 (clone RM4-5 BV650; BD Biosciences), CD8 (clone 53-6.7 BV510; BD Biosciences), CD25 (clone PC61 BUV395; BD Biosciences), F4/80 (clone BM8 biotin; Biolegend), CD206 (clone C068C2 PE-Cy7; Biolegend), Ly6C (clone HK1.4 BV605; Biolegend), NKp46 (clone 29A1.4 BUV737; BD Biosciences), CD11b (clone M1/70 APC-Cy7; Biolegend), I-A/I-E (clone M5/114.15.2 BV785; Biolegend), Ly6G (clone 1A8 BV480; BD Biosciences), FoxP3 (clone 150D AlexaFluor647; Biolegend), Fixable Live/Dead Blue (Invitrogen), Streptavidin BUV805 (BD Biosciences), PD-L1 (clone MIH5 BV421; BD Biosciences), CD107a (clone 1D4B FITC; Biolegend), CD170 (clone E50-2440 PECF594; BD Biosciences), Perforin (clone S16009A PE; Biolegend), CD11c (clone N418 BV711, Biolegend), and granzyme B (clone QA16A02 AlexaFluor700; Biolegend). Data was recompensated and analysed using FlowJo V10.5 software (FlowJo LLC).
2.5. Statistical Analysis
All data sets were assessed for normal distribution and analysed using GraphPad Prism 8.0.0. A Kruskal-Wallis 1-way ANOVA with a Dunn’s posttest was used for analysis (
3. Results
3.1. [18F]AlF-NOTA-GZP Is Associated with Positive Therapy Response
HEPA 1-6 liver tumour bearing mice were treated with control antibody or αPD1 monotherapy, αCTLA4 monotherapy or a combination of αPD1, and αCTLA4 and tumour volumes measured over the course of the study. Assessment of the tumour growth curves demonstrated normal distribution (Shapiro-Wilk p0.0588), and tumour volumes varied depending on the treatment regime and individual response (tumour volumes are depicted in Figures 1(a) and 1(b) and Supplementary Table S1, response rates in Supplementary Table S2). %TGI and tumour retention of [18F]AlF-NOTA-GZP are well correlated for all data (Pearson
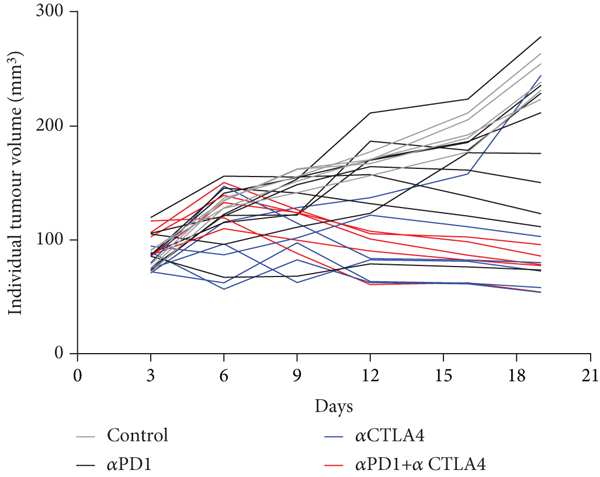
Graph displaying tumour volumes in response to administration of ICI therapy. Mice (


Tumour retention of [18F]AlF-NOTA-GZP was heterogeneous across the different treatment arms (Figure 2(a)), and the control group and TNRs displayed low tumour retention whereas tumour retention in the TR groups was significantly higher; αPD1 (

(a) Maximum intensity projection images depicting [18F]AlF-NOTA-GZP tumour retention. Yellow dotted areas indicate the HEPA 1-6 tumour border. Mice were injected with [18F]AlF-NOTA-GZP (~10 MBq intravenously) and static images acquired from 60-80 mins postinjection. (b) Graph showing differences in [18F]AlF-NOTA-GZP tumour retention from each treatment arm. [18F]AlF-NOTA-GZP tumour retention was significantly increased in αPD-1, αCTLA4 and combined


Table showing HEPA 1-6 tumour retention of [18F]AlF-NOTA-GZP after completion of ICI treatment. Data are shown as
3.2. [18F]AlF-NOTA-GZP Tumour Retention Is Dependent on Granzyme B Expression on Tumour-Infiltrating NK Cells
We analysed the changes in HEPA 1-6 tumour uptake of different infiltrating immune cells in response to therapy, and t-SNE and Rphenoptype clustering were used to determine immunophenotypic changes across the different treatment arms comparing each TR group against the TNR group (Figure 3). Many immunophenotypic changes were detected between the responsive tumours and TNRs (Supplementary Table S4). Significant changes were attributed to CD3+ and CD4+ T cells and GZB+ NK+ cells (Figure 3 and Table 2). Overall, the strongest correlation was observed between [18F]AlF-NOTA-GZP and GZB+ NK+ cells (Supplementary Figure 1, Pearson

Multicolour flow cytometry analysis of HEPA 1-6 tumour-associated immune cells after treatment. Percentages of (a) CD3+ T cells relative to CD45+ cells, (b) CD4+ TILS relative to total CD3+ T cells, (c) CD4+ Treg cells relative to total CD4+ cells, and (d) GZB+ NK+ cells relative to total NK+ cells. Data are shown as individual values with
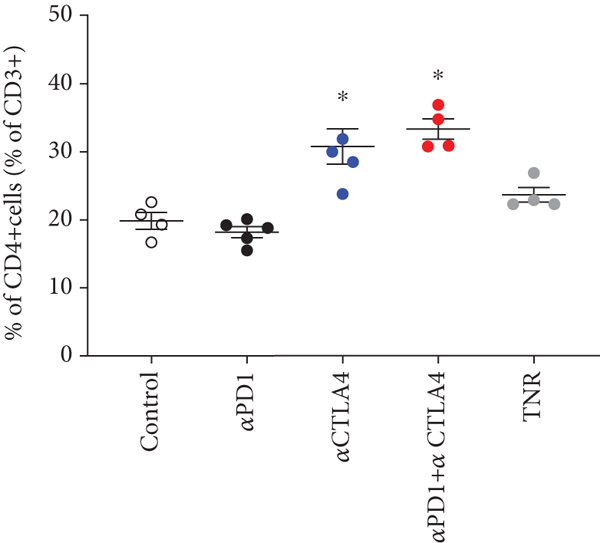

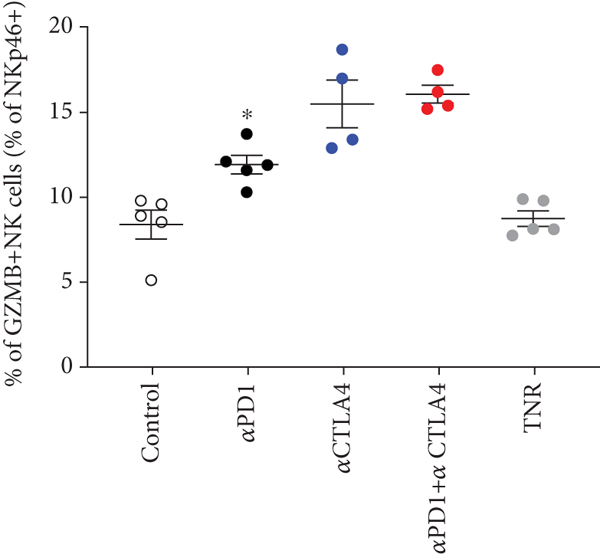
Table showing HEPA 1-6 tumour infiltrating immune cells after completion of ICI treatment. Data are shown as
4. Discussion
The liver is a tolerogenic organ, able to regulate immune responses to a constant stream of antigens from the gut [18], this immune-suppressive environment, however, is detrimental for tumorigenesis. Chronic inflammation caused by viral infections or long-term damage (alcoholic cirrhosis, steatohepatitis, or nonalcoholic fatty liver disease) drives the development of HCC [19]. Chronic inflammation also leads to key players in the immune system becoming suppressed, resulting in incomplete activation of T cells and immune exhaustion [20, 21]. Despite such a chronically immunosuppressed environment, ICI therapy has shown great promise in HCC with recent clinical trials showing the benefit of anti-PD1 or anti-CTLA4 treatment alone or in combination with antiangiogenic therapy (CHECKMATE 40, KEYNOTE 240, and KEYNOTE 224) [1, 3, 19, 22–26]. Overall survival remains the primary endpoint in HCC clinical trials; however, imaging-based assessment allows for estimations of objective response rates (ORR) used to identify strong early signals of efficacy [2]. However, assessment of treatment response in HCC is complex, the lack of tumour shrinkage with some effective therapies and the coexistence of cancer and cirrhosis in HCC patients makes conventional RECIST-based imaging assessment unreliable [27, 28]. Imaging biomarkers that provide information about changes in the tumoural microenvironment after treatment may provide reliable markers of objective response. Previous studies have shown that GZB-targeting peptides can stratify ICI therapeutic response in syngeneic models of colon cancer [16, 17]; however, this has not been demonstrated in a syngeneic HCC before. HCC tumour retention of [18F]AlF-mNOTA-GZP was significantly higher in treatment responsive tumours compared to TNRs (Table 1, Figure 2(b), Supplementary Figure 2); however, the immune mechanisms involved were very different to other syngeneic cancers. In previous studies, syngeneic colon cancers responding to anti-PD1, CTLA4, or combination therapy displayed increases in CD8+ and GZB+ CD8+ cells and decreases in F4/80 myeloid cells [15, 16]. Treatment responsive HCCs, however, responded via different immune pathways, with no significant changes observed in CD8+ or myeloid cell populations but instead a significant increase in tumour infiltrating GZB+ NK+ cells and a concomitant decrease in CD4+ T-regulatory cells. NK cells make up a significant part of the intrahepatic lymphocyte population and are positively correlated with HCC patients’ survival and prognosis [29, 30]. NK cells are involved in the innate and adaptive immune response through cross talk with dendritic cells and T cells and are rapidly gaining popularity as a target for the development of new ICIs [31, 32]. The presence of the CD4+ T-reg cells (CD4+/FOXP3+) has been linked with poor prognosis in HCC and a reduced response to ICI therapy [33, 34] so decreases in tumour infiltration of CD4+ Treg cells may ease the immunosuppressive environment allowing immune recognition.
Recently, new granzyme B targeting tracers have been developed whose tumour retention is dependent on granzyme B proteolytic activity as opposed to the current aldehyde tracer which quantifies the amount of biochemically active GZMB molecules at the tumour [35]. One of the documented shortcomings of the aldehyde granzyme B tracers is low tumour retention, which may be masked in tumours originating in tissues with higher nonspecific retention including liver and kidneys. The new granzyme B tracer, dependent on proteolytic activity, shows higher tumour retention, but due to its considerable size is cleared much more slowly from blood, leading to higher background. It will be interesting to see how these different radiopharmaceuticals can be used to provide complementary information to stratify therapy response in future.
Overall, [18F]AlF-mNOTA-GZP demonstrates the ability to accurately stratify ICI response in HCCs, showing that granzyme B radiopharmaceuticals can be used across multiple tumour types, including those arising in immune suppressive environments. While the immune response to ICI therapies differs in HCC compared to many other cancers, granzyme B may still be a potential biomarker for immunotherapy efficacy in liver cancer providing useful information to help improve patient management.
Footnotes
Data Availability
All data is available on request to the corresponding author.
Conflicts of Interest
The authors declare no conflict of interest.
Acknowledgments
This work was supported by Singapore’s Health and Biomedical Sciences (HBMS) Industry Alignment Fund Pre-Positioning (IAF-PP) grant H18/01/a0/018, administered by the Agency for Science, Technology and Research (A
