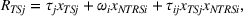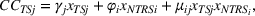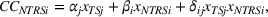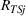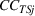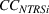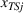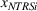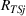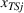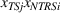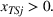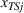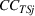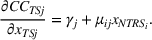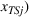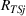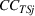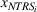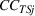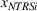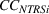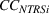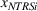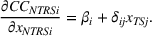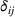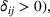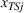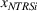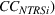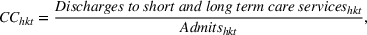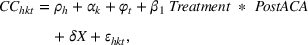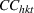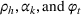Abstract
Ensuring continuity of care for patients after an intensive hospitalization episode is a complex dilemma that plagues the US health‐care system. Despite its influence on health outcomes such as mortality and readmissions, it is difficult to construct policy instruments such as report cards or penalties for improving continuity of care due to the fragmented nature of post‐intensive hospitalization care. However, policy instruments that target other related health outcomes can also benefit continuity of care. We examine whether a quality regulation that penalized hospitals for excess readmissions has implications for an unregulated aspect, that is, continuity of care through intra‐ailment and spillover effects. Intra ‐ailment effects occur from the effects of penalty regulation on the continuity of care of ailments targeted for regulation. Spillovers occur from the effects of penalty regulation on continuity of care for ailments that were not targeted by the policy but share complementarities with targeted ailments. We conduct difference‐in‐differences analyses using patient‐level data for 2004–2014 from the state of California. Our empirical strategy utilizes the nature of the hospital production function, which is organized by medically related specialties. We construct three cohorts of patients, all of whom belong to specialties that house the ailments targeted for readmission penalties. These include (1) ailments targeted by the penalty policy for readmissions, (2) closely related, non‐targeted ailments, and (3) unrelated ailments. Results reveal evidence of intra‐ailment effects, which manifest as increases in continuity of care of targeted ailments, and spillovers, which manifest as increases in continuity of care of non‐targeted but related ailments. We find that processual mechanisms, such as the source of patient admissions and length of stay, and structural mechanisms, such as system size, accentuate the intra‐ailment effects. Our study provides novel insights into how quality regulation can have intra‐ailment and spillover effects and bespeaks the importance of incorporating these effects in the regulatory benefit‐cost calculus.
INTRODUCTION
To assess the overall effects of any regulation, it is important to determine whether the regulatory policy influences outcomes that are not a target of the regulation (Greenwald & Stiglitz, 1986). We examine the effects of the Hospital Readmissions Reduction Program (HRRP), which is an important policy instrument of the Affordable Care Act (ACA) regulation. The HRRP imposed a penalty on hospitals whose risk‐adjusted readmissions for three ailments (heart attacks, heart failure, and pneumonia) were greater than the national average. Evidence indicates that the HRRP reduced readmissions (Gupta, 2021; Qiu et al., 2022). To achieve the policy goals of fewer readmissions, hospitals are likely to take actions that improve the entire spectrum of care for ailments that were the target of regulation. Thus, regulation for outcome A of targeted ailment K could plausibly influence an unregulated outcome B of ailment K. We label this as an intra‐ailment effect of regulation. Additionally, we posit that the regulation will influence an unregulated outcome B of ailment J, which shares complementarities with ailment K, but J itself is not the target of regulation for either outcome A or B. We label this as a spillover effect of regulation.
We examine these two effects of the HRRP quality penalties on an important determinant of health‐care quality, namely, continuity of care for ailments targeted (intra‐ailment effects) and ailments not targeted (spillover effects) by the regulation. Continuity of care, broadly defined as enabling appropriate care across patient conditions, settings, and time, is an important part of the health‐care delivery continuum that helps patients discharged after intensive hospital‐based care to smoothly transition into their communities (Berwick et al., 2008; Buntin et al., 2009; Liu et al., 2001; Senot, 2019). Continuity of care can refer to relational continuity, where a patient's care is managed by the same provider over the entire care cycle; informational continuity, where the patient's information is transferred over care episodes; continuity of physical location, where the entirety of a patient's care occurs in the same location; management continuity, which refers to how care is integrated across sites and episodes (Haggerty et al., 2003; Senot, 2019); or transitional continuity, which occurs when patients are discharged from intensive care to another type of care (Verhaegh et al., 2014). We focus on transitional continuity, which encompasses “a broad range of time‐limited services designed to ensure health‐care continuity, avoid preventable poor outcomes among at‐risk populations, and promote the safe and timely transfer of patients from one level of care to another or from one type of setting to another” (Naylor et al., 2011, p. 747). Transitional continuity of care can occur in hospital‐based step‐down or recovery units or in other facilities such as skilled nursing facilities (SNFs), inpatient rehabilitation facilities, long‐term care hospitals, and home health agencies.
Research on the antecedents of continuity of care and their effectiveness is nascent. Policy instruments that target continuity of care are challenging to design, owing to the fragmented nature of the industry. However, to assess the comprehensive impact of a regulatory policy such as HRRP, it is important to examine whether the regulation that targets another relatively more discernable quality outcome (readmissions) can improve other patient care outcomes such as continuity of care within the targeted service. Such intra‐ailment effects arise from learning, resource sharing, and innovation (Thornton & Thompson, 2001). Moreover, non‐targeted but related ailments can also benefit from the regulation due to the complementarities between the targeted and non‐targeted ailments. However, managerial actions such as strategic reallocation of resources can limit such spillovers. Further, if resources are taken away from the activities that benefit non‐targeted areas, it can potentially even lead to negative spillovers.
The HRRP quality penalties targeted a few key ailments because comprehensive regulation would be prohibitively complex and cognitively cumbersome, which defeats its ability to provide information for decision making (Arya et al., 2014). The regulatory design that targets an outcome (readmission) of a few ailments facilitates our empirical analysis with respect to the identification of intra‐ailment effects. Further, the logistics of health‐care delivery wherein process flows are organized around medical specialties (Batt et al., 2020) enables the assessment of spillovers. We use patient‐level data for 2004–2014 for the state of California and a difference‐in‐differences design to calibrate intra‐ailment and spillover effects. Each patient record in our sample contains a principal diagnosis, which is the ailment for which the patient was admitted (e.g., heart attack). Additionally, the patient record lists comorbidities, which are other diagnoses that exist but are not the reason for the current hospitalization episode (e.g., kidney disease, encephalitis, etc.). Each of these ailments is classified into specialties that are based on the anatomy (e.g., heart or lungs) of where the ailment originates. For example, the cardiovascular specialty consists of ailments that relate to the heart and blood vessels. We restrict our analysis to only patients whose principal diagnosis is in the two specialties that treat one of the three targeted ailments. These specialties are cardiovascular (which houses heart attack and heart failure) and respiratory (which houses pneumonia).
To empirically operationalize intra‐ailment effects, we investigate whether HRRP quality penalties are associated with increases in continuity of care for patients whose principal diagnosis is one of the three ailments that were targeted for regulation, namely, heart attack, heart failure, and pneumonia. We label this cohort of patients as targeted services (TS). We focus on continuity of care for TS, relative to a control cohort consisting of patients whose principal diagnosis is in the same specialty as the TS, but all their comorbidities lie in different medical specialties. This control group, labeled as unrelated services (URS) consists of patients who were admitted for an ailment in the same specialty as the TS but have all their comorbidities in different medical specialties. Our design controls for scientific, technological, and other trends that are ailment‐specific and thus shared between TS and URS for their principal diagnosis. To empirically operationalize spillovers, we use a sample of non‐targeted patients whose principal diagnosis is in the same specialty as the TS, and at least one of their comorbidities also lies in the same medical specialty. Such patients will have greater scope for spillovers, relative to URS. We label this cohort of patients as non‐targeted but related services (NTRS).
Intra‐ailment effects arise from knowledge and resource sharing due to which efforts invested to improve one outcome of a service (readmissions) permeate through the system of care and benefit another outcome (continuity of care) within the targeted service. We explore routines as a mechanism driving intra‐ailment effects. Routines produce “repetitive, recognizable patterns of interdependent action carried out by multiple participants” (Feldman & Pentland, 2003, p. 103). Routines emerge from work patterns responses to stimuli and enable the retention of codified knowledge (Zollo & Winter, 2002). Work patterns can also incorporate scope for learning, adaptation, and improvisation in response to external changes (Nembhard & Tucker, 2016). We posit that processual and structural routines facilitate intra‐ailment effects. Processual routines emerge from the sequence of interdependent events that contribute to task execution, while structural routines emerge from organizational design and enable or constrain individual and collective actions and interactions (Felin et al., 2012).
We examine two processual routines—the channel through which the hospital admission occurred and the length of time for which care occurred in the hospital before the patient is discharged. In terms of the channel through which admissions occurred, we examine emergency department (ED) admits. EDs are characterized by severe time pressures, and process‐based routines can enable quick responses (Batista et al., 2016). EDs also face temporal uncertainty with respect to how events will unfold, which requires speedy adaptation (Geiger et al., 2021). Although the discharge to continuity of care occurs outside of the ED, after the patient has been stabilized and moved to a hospital room, discharge planning begins at the time the patient is first admitted (Albert, 2016). ED routines have also been linked with better quality of care throughout the care process (Jarvis, 2016). We develop an argumentation that a larger proportion of ED admits in TS would enable better routines in the entire care continuum. Thus, we expect that increases in continuity of care for TS (relative to URS) will be greater when a relatively higher proportion of TS patients are ED admits.
Our second processual driver is the length of stay (LOS) in the hospital. After a patient is admitted, stabilized, and transferred for inpatient care, additional care processes occur over the length of the patient's stay until the patient is discharged to home or to another care facility. Each additional day of stay in the hospital reduces the chances of readmission but also reduces the capacity available for new patients. Once patients recover from the most acute phase of their illness, discharges to continuity of care facilities can free up hospital bed capacity for critically ill patients (Grabowski & Maddox, 2020). However, a lower LOS carries an associated requirement to manage the care processes, adapt routines, and modify care intensity to reduce readmission risk. Therefore, we expect continuity of care increases in TS (relative to URS) to be higher when TS patients have relatively lower LOS.
We examine hospital system size as a structural factor. A larger multi‐hospital system could face rigidities in the diffusion of knowledge, updating of learning repositories, and implementation of new routines (Edmondson et al., 2001). We expect larger increases in continuity of care from intra‐ailment effects to occur in hospitals that belong to smaller systems. However, larger hospital systems can also achieve intra‐ailment effects by investing in continuity of care facilities. Finally, we show that the greater the extent of intra‐ailment effects, the greater the spillovers to the NTRS.
We make several contributions to the accounting, operations, and health‐care literature. First, we contribute to accounting literature that examines the effects of regulatory policies that incentivize desirable behaviors or penalize undesirable behaviors (Leuz & Wysocki, 2016). Research emphasizes the need to identify spillover effects of regulation to obtain a more complete assessment of the welfare effects. We contribute by showing that incentivizing quality performance for a few select services can have externalities beyond those targeted by policy. Thus, like T. Lu et al. (2021), we demonstrate a broader impact of regulation that extends beyond the effects on the targeted outcomes of targeted services. Second, by constructing within‐specialty treatment and control groups, our methodology reduces the exposure to a common problem that plagues studies about the effects of regulation, namely, that treated firms may differ systematically from untreated firms. Researchers can mistakenly interpret results that occur from differences in baseline conditions between the two groups. Evidence from parallel trends using a short pre‐regulatory time series does not mitigate the potential for baseline differences because changes between the treatment and control groups (e.g., technology or customer attitudes) can occur in a lumpy manner. Third, while the accounting literature has focused on the impact of financial reporting regulation, the operations literature has examined policy mechanisms to improve quality, particularly in health care (Andritsos & Tang, 2014; L. X. Lu & Lu, 2018). Our study suggests that targeted regulation can impact quality in other parts of the health‐care value chain. Finally, our study provides two additional factors—namely, intra‐ailment effects and spillovers—that must be considered before making a conclusion about the effectiveness of a health‐care regulatory policy such as the HRRP (Arifoğlu et al., 2021). Continuity of care reduces the likelihood of readmissions and improves health outcomes (Werner et al., 2019), and we discuss this link with empirical support. We contribute by showing how policy instruments such as quality penalties for readmission can have beneficial effects on continuity of care, which is an important determinant of overall patient health status.
The next section contains our hypotheses. Section 3 describes the data, measures, and the methodology, with results in Section 4. Section 5 discusses the study's implications and its limitations.
LITERATURE REVIEW AND HYPOTHESES DEVELOPMENT
Quality regulation with or without financial penalties is an important health‐care policy tool. States such as New York and Pennsylvania experimented with various forms of mandatory quality disclosures aimed at reducing losses to patients and insurance companies from providers’ information advantage (Dranove & White, 1987). In 2005, the Centers for Medicare and Medicaid Services (CMS) designed a free website, Hospital Compare, which provided outcome quality (mortality) and process quality (protocols followed at admission and during hospital stay) information about patients discharged from US hospitals. These disclosure requirements were initially restricted to three highly prevalent ailments, namely, heart attack, heart failure, and pneumonia. Research finds that the disclosure of mortality was associated with an improvement in mortality performance for disclosed as well as related ailments (Deore et al., 2023; Totten et al., 2012).
While Hospital Compare used disclosures as a policy tool, the HRRP (under the ACA) provided financial incentives to improve health‐care quality. Under the HRRP, CMS imposed financial penalties of up to 3% for hospitals with poor quality performance, defined as excess 30‐day readmission rates for three ailments (heart attack, heart failure, and pneumonia). This measure was constructed with a predictive regression model that used patient mix, location, size, and other hospital‐level factors. Extant research indicates that HRRP quality penalties reduced readmission rates among Medicare beneficiaries for the targeted conditions (Gupta, 2021; Zuckerman et al., 2016). To achieve the policy goal of fewer readmissions, hospitals had incentives to improve the entire spectrum of a patient's health plan, including post‐discharge planning (e.g., evaluating post‐hospitalization care options, arranging for transfer to another care facility; James, 2013). Thus, the benefits of HRRP can influence continuity of care, which is a longstanding concern and a key challenge in health‐care delivery (Holland & Harris, 2007; Reid et al., 2002).
Continuity of care
Transitioning patients to care facilities is inevitable because keeping patients in an intensive care unit (ICU) or critical care unit (CCU)‐type care at a hospital when their health needs could be addressed in less resource‐intense settings is an inefficient use of hospital capacity. With aging population and more incidences of chronic and infectious diseases, the demands on hospital capacity resources have increased (Prince et al., 2015). The ability of hospitals to manage health‐care delivery by coordinating with transitional care facilities and ensuring continuity of care presents an opportunity to improve health‐care value, defined as patient health outcomes achieved per dollar spent (Porter, 2010). Drawing on prior evidence (Werner et al., 2019), we conclude that an increase in the discharge of patients to post‐intensive facilities, rather than home, improves continuity of care quality. Continuity of care has been shown to improve health‐care outcomes such as readmissions (Queenan et al., 2019). An integrative review of 20 articles by Stamp et al. (2014) indicates that continuity of care can not only decrease readmissions but also can increase the quality of life and reduce the overall cost of care for patients with heart failure. Another review of 133 studies by Sezgin et al. (2020) concludes that continuity of care can reduce readmissions and improve quality of life, measured by the patient's ability to perform activities of daily living.
Therefore, continuity of care through timely transitional care can achieve the triple aim of better health, higher quality care, and lower costs. This requires following a care plan that is accessible to all authorized care providers to provide patients with an experience that is coherent, connected, and consistent with their health‐care needs (Haggerty et al., 2003). However, the siloed process of health‐care delivery has been a deterrent in providing continuity of care to patients. Care providers engage with patients at defined “touchpoints” that often limit their ability to see the system‐wide view of patient care.
To improve continuity of care, some health‐care organizations have reengineered their processes and established services platforms to coordinate care (Meyer et al., 2007). These platforms help in care coordination by managing the interface between patients, health‐care providers, and technology. Specific individuals, referred to as “case managers,” actively manage patients’ interactions with health‐care delivery systems. A set of routines supported by an integrated care plan across multiple settings is needed to identify and determine the needs of patients, plan and coordinate the care, assess progress, and evaluate the outcomes. Executive initiative supported by compatible information systems and a common sense of purpose across different departments and providers help leading health‐care organizations to establish these routines (Meyer et al., 2007).
Continuity of care is facilitated by various mechanisms. Effective execution of various activities involved in continuity of care calls for management of interdependencies among tasks by creating coordinating mechanisms (Gittell, 2002). These coordination mechanisms can range from routines (Winter & Nelson, 1982) in the form of clinical pathways, establishing boundary spanners (Davenport & Nohria, 1994; Lawrence & Lorsch, 1967) such as case managers and nurses, setting team meetings to facilitate interactions among participants (Lawrence & Lorsch, 1967; Tushman & Nadler, 1978), and relational coordination (Gittell, 2002; Van de Ven et al., 1976). These coordination mechanisms require process innovations. Research has noted the role of organizational leadership, multi‐specialty group practice, aligned incentives, information technology and guidelines, accountability for performance, a physician–management partnership, and organizational culture related to the degree to which coordination mechanisms are established (Gardner et al., 2014). Additionally, the contextual environment such as government regulations also influences the innovation process (Thakur et al., 2012). Yet there is a paucity of research on how the contextual environment influences continuity of care in hospitals. Our study attempts to fill this lacuna and examines whether regulation aimed at reducing readmissions can influence continuity of care outcomes.
Hypotheses development
When a patient is readmitted for an ailment for which they were previously hospitalized, it gives rise to a variety of costs and increases the risks for adverse outcomes such as mortality (Jencks et al., 2009). Evidence indicates that the passage of HRRP is associated with a reduction in readmissions (Gupta, 2021; Qiu et al., 2022; Wasfy et al., 2017), and lower readmission rates are associated with reductions in mortality rates for targeted ailments (Dharmarajan et al., 2017). When readmissions are penalized, hospitals have incentives to reduce readmission rates for targeted ailments. HRRP also encourages quality strategies that focus on the entire care cycle (Albritton et al., 2018). These include improved case management, discharge planning, information sharing, and so on. Studies have shown discharge planning such as the use of post‐acute care facilities to influence readmissions (Li et al., 2020; Sezgin et al., 2020). Research has also found reductions in readmissions for non‐targeted ailments after the HRRP regulation, consistent with the notion that hospitals implemented quality improvement strategies across the board (Demiralp et al., 2018).
Strategies undertaken to reduce readmissions can also influence unregulated outcomes of care of the targeted ailments. Such intra‐ailment effects occur when efforts toward a regulated outcome permeate through the system of care and produce benefits to another unregulated outcome for the same ailment. We study such intra‐ailment effects between the regulated outcome (readmissions) and an unregulated outcome ( i.e., continuity of care) for targeted services (TS, i.e., heart attack, heart failure, and pneumonia). Strategies undertaken to reduce readmissions for a targeted ailment can also influence continuty of care for non‐targeted but related services (NTRS) through spillover effects.
Consider the following production functions for hospital readmission management and continuity of care:
We start with
A positive γj
indicates that increasing effort ( Quality penalty regulation for excess readmissions of a targeted service is associated with an increase in continuity of care for the targeted service.
Quality penalties for readmissions could yield positive effects for patients in non‐targeted but related services as per Equation (III). The nature and organization of the health‐care delivery system generate scope for such spillovers. Medical training, treatment, and recovery are administered through bundles of specialties, which relate to an anatomical system (such as cardiology, neurology, injuries, etc.). Hospitals are usually organized by clinical specialties, and each specialty contains unique care pathways (Best et al., 2015). It is common to have heart attack patients (TS) and angina patients (NTRS) receiving treatment in the same room, with the same equipment, and the same personnel. For example, if a patient is admitted with chest pain, a common test is to measure Troponin I and B Natriuretic Pep (BNP) levels. BNP is elevated in both heart attack and angina. If the patient is not having a heart attack (based on Troponin I levels), but the BNP level is elevated, the diagnosis is that the patient is having an angina attack. Thus, the test for heart attacks has spillover effects to the diagnosis and treatment of angina.
Spillovers are more likely among related ailments. We draw on Holmstrom and Milgrom's (1991) framework to posit that when two ailments (e.g., heart attack and angina) share complementarities because they belong to the same specialty (e.g., cardiology), then increasing inputs toward improving an outcome (readmissions, R) of one ailment (heart attack, TSj) will not only improve a related outcome of the same ailment (
Complementarities between TSj
and NTRSi
imply Quality penalty regulation for excess readmission of a targeted service is associated with increases to continuity of care in non‐targeted but related services.
DATA AND RESEARCH DESIGN
Data
To examine the impact of quality penalties on continuity of care, we extract patient‐level information from California's Office of Statewide Health Planning and Development (OSHPD) Patient Discharge Data (PDD) for the years 2004–2014. The database contains yearly observations for about 3–4 million patients including the principal diagnosis and up to 24 comorbidities (at the International Classification of Diseases ‐ ICD code level), type of insurer, discharge destination, procedures, LOS, and hospital‐related mortality (i.e., whether the patient died in hospital). Data documentations are available at
Our interest is to examine if continuty of care increases occur after quality penalties were announced for heart attack, heart failure, and pneumonia in 2010 under HRRP. The PDD database provides information about disposition (disp) defined as “the consequent arrangement or event ending a patient's stay in the hospital.” We measure continuity of care as the proportion of patients discharged to the following type of short and long‐term care facilities: acute care (“step down”) within the admitting hospital, other (non‐acute) care within the admitting hospital, skilled nursing/intermediate care (SN/IC) within the admitting hospital, acute care (“step down”) at another hospital, other care (not SN/IC) at another hospital, SN/IC at another facility, and residential care facility. We define continuty of care by patient cohort at a given hospital as
Patient cohorts
Increases in continuity of care after quality penalties can occur not from the quality regulation but from other factors that influence quality, such as technology, scientific advancements, and capacity investments. To identify effects of quality regulation on CC, we use treatment and control cohorts from the same medical specialty. Technological advancements and patient care processes follow similar treatment pathways within a medical specialty. Thus, to control for specialty‐specific trends, we restrict our analysis only to patients who are treated for a principal diagnosis that is in the same medical specialty as an ailment belonging to TS. These include cardiovascular and respiratory diseases. For example, codes in the range ICD 390–459 refer to a broader medical specialty focusing on the treatment of “diseases of the cardiovascular system,” which includes TS ailments such as heart attack and heart failure, as well as other diseases such as ischemic heart disease, aortic aneurysm and dissection, arrythmia, etc. See Appendix A for a list of ailments in each medical specialty.
Within each of the two medical specialties that were the target of readmission penalties, we construct three cohorts—one treatment cohort to calibrate the intra‐ailment effects of readmission quality penalties on continuity of care quality, one treatment cohort to assess spillover effects of penalties, and one control cohort. The first cohort contains only patients whose principal diagnosis is subject to the readmission quality penalties. These patients would be the targets of quality efforts that were inspired by the regulation. The second cohort contains patients whose principal diagnosis and at least one of their comorbidities are in the same medical specialty as TS (but is not the TS itself). These patients benefit from spillovers from quality improvements due to their complementarities with the TS in both the principal diagnoses and comorbidities. The third cohort, that is, the control cohort, contains patients whose principal diagnosis is in the same medical specialty as TS, but none of their comorbidities are in the same specialty as the TS. These patients do not have comorbidities that benefit from spillovers with the treatment group that was subject to quality penalties. Thus, while both the second and third cohorts can benefit from positive externalities in their principal diagnosis, they differ in the potential for externalities in their comorbidities. We exclude patients whose principal diagnosis was “stroke” since this ailment was subject to additional regulations in 2004 and 2006. Thus, our control group stays consistent over time and includes only ailments that did not face additional regulations. We construct the three patient cohorts as follows.
Treatment group 1–targeted services or TS: This cohort comprises patients whose principal diagnosis is an ailment that is targeted by the readmission penalties. To construct this cohort, we use the technical specification issued by the Agency for Healthcare Research and Quality (AHRQ) to identify patients subject to one of the three principal diagnoses (heart attack, heart failure, and pneumonia). The AHRQ defines ailments by the ICD‐9 codes that are used for reporting and reimbursement. For example, “heart attack” is targeted for readmission penalties and belongs to the ICD‐9 code range “410.” A patient with a principal diagnosis of heart attack therefore belongs to this cohort.
Treatment group 2–non‐targeted but related services or NTRS: We define NTRS as patients whose principal diagnosis is a non‐targeted ailment within the same medical specialty as TS, and whose comorbidities also include at least one ailment from the same specialty as TS. For example, a patient with a principal diagnosis of “aortic aneurysm and dissection” (ICD 441) and a comorbidity “deep vein thrombosis” (ICD 453) would be classified as NTRS because both the principal diagnosis and comorbidity are from within the cardiovascular specialty. This cohort is not subject to readmission penalties for either the principal diagnosis or comorbidity. However, this cohort should exhibit improvements in quality after the penalty regulation was introduced due to the existence of complementarities with the TS in their principal diagnosis as well as comorbidities.
Control group (unrelated services or URS): To allow us to reliably estimate the intra‐ailment and spillover effects of the regulation for the two treatment groups, we require a control group that is subject to the same trends in quality arising from scientific or technological advancements affecting care outcomes of ailments in TS but is systematically less likely to exhibit complementariness with TS. We construct this cohort from patients with principal diagnosis from the same specialty as the TS (as is the case with NTRS) but all comorbidities from different specialties. An example is a patient with a principal diagnosis from the cardiovascular specialty, such as aortic aneurysm and dissection (ICD 441) but with all comorbidities from outside the cardiovascular specialty (e.g., with comorbidity encephalitis myelitis, i.e., brain inflammation [ICD 323] from the specialty of diseases of the central nervous system). This cohort of patients shares complementarities with TS because of their principal diagnosis; however, no complementarities exist between their comorbidities and the TS. The different medical specialty comorbidities (e.g., central nervous system ailments) do not share a common pathophysiology with ailments in the targeted specialties (cardiovascular and respiratory ailments). The non‐related comorbidities of a patient in URS are usually treated by the appropriate specialist (e.g., an infectious diseases expert in the case of a comorbidity of an infection such as meningitis, a neurologist in the case of a comorbidity of a central nervous system disorder such as Parkinson's disease). For URS patients, decisions related to the aspects of care such as LOS and discharge destination are taken collaboratively by multiple care givers across medical specialties. The URS control group is also subject to spillovers in their principal diagnosis; hence, our estimates are conservative.
Main estimations
To estimate the impact of the ACA quality penalties on continuity of care, we rely on a research design involving the three patient cohorts—TS, NTRS, and URS. We examine the (1) intra‐ailment effects on the change in continuity of care of TS and (2) spillover effects from TS to continuity of care of NTRS. The URS cohort acts as a common control group. To estimate intra‐ailment (spillover) effects on continuity of care, we compare continuity of care rates between TS (NTRS) and URS cohorts before and after the announcement of HRRP under the ACA regulation. We use the following empirical specification, which provides difference‐in‐differences estimates around the HRRP announcement:
RESULTS
Descriptives
Panel A of Table 1 presents means and standard deviations of continuity of care (CC) across all the patient cohorts (TS, NTRS, and URS) in pre‐ and post‐ACA periods. Table 1 shows that while continuity of care increased in the post‐ACA period for TS and NTRS, no increases were apparent in URS. This provides initial evidence of intra‐ailment and spillover effects of the quality penalty regulation. Panel B of Table 1 presents means and standard deviations of all control variables used. Differences in Risk, Procedures, and Mortality across cohorts necessitate their inclusion in the regression estimation model.
Descriptives.
Abbreviations: NTRS, non‐targeted but related services; TS, targeted services; URS, unrelated services.
Intra‐ailment and spillover effects—Full sample
To examine intra‐ailment effects, we estimate Equation (VI) using the TS cohort as Treatment and URS cohort as the control. The coefficient on Treatment X PostACA (β 1), captures the difference‐in‐differences estimate. Results in column 1 of Table 2 indicate a positive and significant coefficient. Thus, continuty of care for TS patients increased more than the continuty of care for URS patients following the regulation. The magnitude of this effect is 1.78 percentage points, which translates to a 4.6% (0.0178/0.3884) increase in continuty of care of TS relative to URS in the post‐regulation period.
Intra‐ailment and spillover effects of quality penalties—Full sample.
Note: Robust standard errors are in parentheses.
Abbreviations: NTRS, non‐targeted but related services; TS, targeted services; URS, unrelated services.
p < 0.01; **p < 0.05; *p < 0.1.
Next, we examine regulatory spillovers by comparing continuty of care for the NTRS cohort against continuty of care of the URS cohort. We estimate Equation (VI) using the NTRS cohort as Treatment and the URS cohort as the control. The coefficient on Treatment X PostACA (β 1) is an estimate of the magnitude of relative change in the continuty of care of NTRS relative to URS after the announcement of readmission penalties. Positive and significant coefficients as in column 2 of Table 2 indicate positive spillovers. Specifically, continuity of care for patients in the NTRS cohort increased to a greater extent than the continuity of care for patients in the URS cohort in the years following regulation. The magnitude of this effect is 1.38 percentage points, which translates to a 4.4% (0.0138/0.3155) increase in continuty of care of NTRS relative to URS in the post‐quality penalty regulation period. This shows that quality penalties had spillover effects for ailments that were not the target of penalties. Further, the lack of negative spillover effects indicates that hospitals did not reallocate resources away from other non‐targeted services toward the increase in continuty of care of TS.
The analyses assume that trends in continuity of care rates in the absence of quality penalty regulation would have been similar for the treatment and benchmark groups. To validate this assumption, we plot continuity of care rates around the introduction of quality penalties in Figure 1A,B. The HRRP was announced in 2010, which implies that 2010 is the first year in the post‐HRRP period. Thus, we use 2009 as the base year and find evidence of parallel trends between the treatment group (TS and NTRS) and the benchmark group (URS) in the period leading up to the introduction of quality penalties. The continuity of care rates of TS relative to URS (Figure 1A) and NTRS relative to URS (Figure 1B) in the years preceding the quality penalties display no pre‐existing trends. The increase in continuity of care rates of TS and NTRS relative to URS aligns with the introduction of quality penalty regulation, indicating that the observed intra‐ailment and spillover effects can be attributed to the regulation. Online Appendix OS1 in Supporting Information discusses how our empirical design attempts to minimize violations to the stable unit treatment value assumption.
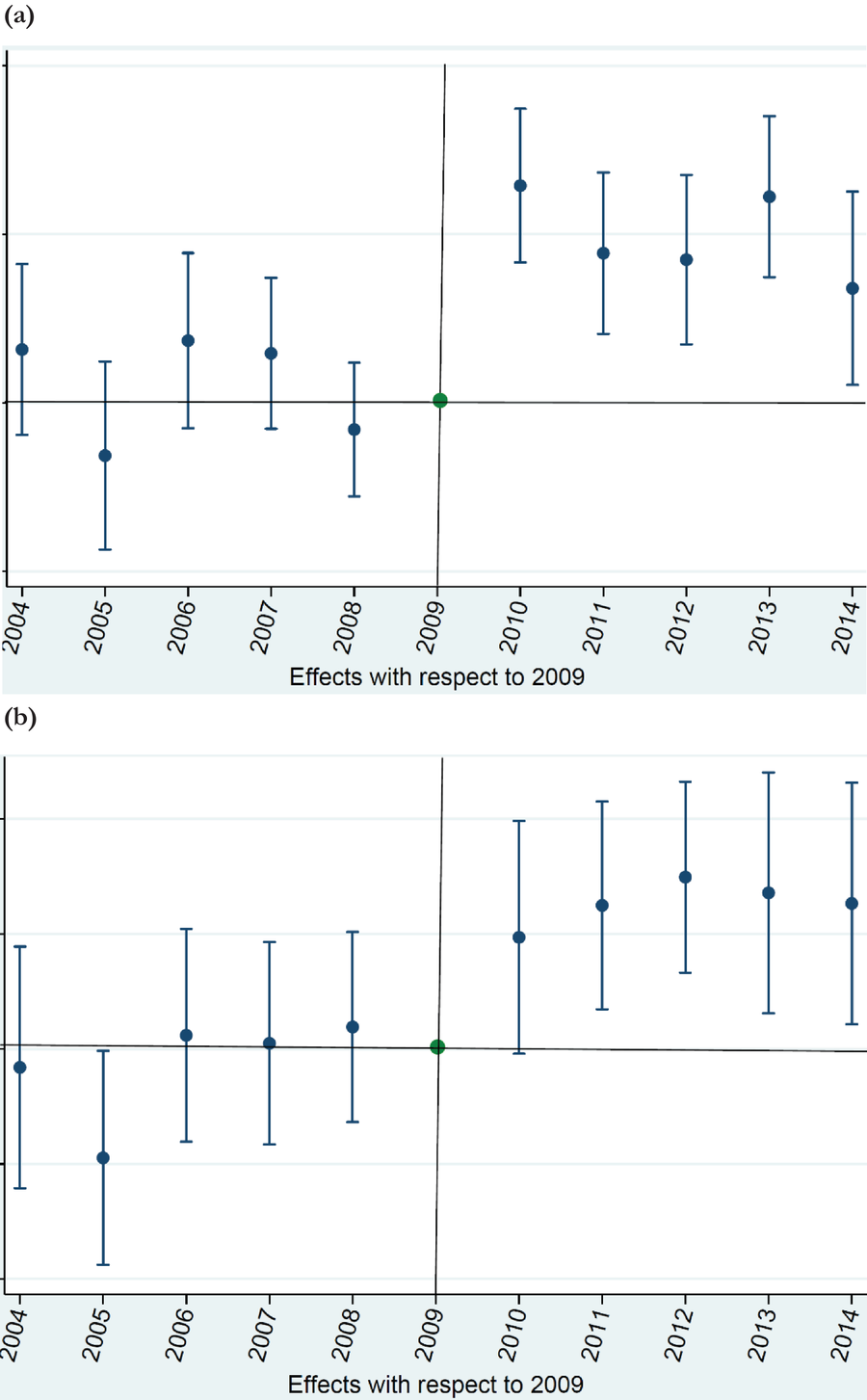
(A) Parallel trends graph—intra‐ailment effect (TS vs. URS). (B) Parallel trends graph—spillover effect (NTRS vs. URS). NTRS, non‐targeted but related services; TS, targeted services; URS, unrelated services.
Appendix B contains robustness checks. These include alternative specification of a difference‐in‐differences analyses, analyses that account for differences in pre‐regulation continuity of care rates, and intra‐ailment and spillover effects of quality penalties estimated separately in the Medicare and non‐Medicare subsamples.
Discharges to home health and SNF could be better indicators of future quality of life of the patient than discharges to hospices or long‐term care facilities. Thus, we perform additional analyses separately for discharges to the following subset—SNF, home health, and residential care facilities. We find results similar to our main estimations (see Table OS2, panel A, in Supporting Information). The analysis also indicates that discharges to SNF, home health care, and residential care increases for all the three targeted ailments (see Table OS2, panel B, in Supporting Information). The increases are highest in heart failure, relative to pneumonia (p‐value based on t‐test of differences < 0.01) and relative to heart attack (p‐value based on t‐test of differences < 0.01). The results reflect the greater need for continuity of care for chronic diseases relative to non‐chronic diseases. While heart failure is classified by the CDC as a chronic ailment (Bernell & Howard, 2016), pneumonia could include both chronic and non‐chronic infections, and heart attacks can occur as an acute manifestation (e.g., sudden metabolic deprivation) or from an underlying chronic condition (e.g., from blocked arteries).
To determine whether readmissions penalties have an impact on CC, we examine the association between continuity of care and reductions to readmissions. We find a negative association as reported in Appendix C, indicating that continuty of care reduces readmission. Thus, regulation that penalized readmissions would likely have motivated increases to continuity of care. Finally, we conducted an analysis of the association between LOS and readmissions for each of the three ailments separately. Our results indicate that there is no significant link between LOS and readmissions (see Table OS3 in Supporting Information). To ensure that hospitals are not differentially treating patients with private insurance versus no insurance, we perform cross‐sectional analyses of intra‐ailment and spillover effects for hospitals with low or high proportions of private insurance and/or no insurance patients. We find no significant differences based on insurance status (see Table OS4 in Supporting Information). Intra‐ailment and spillover effects are present in hospitals with (a) high proportion as well as low proportion of privately insured patients (Table OS4, panel A, in Supporting Information), (b) high proportion as well low proportion of no insurance patients (Table OS4, panel B, in Supporting Information), and (c) mix of high proportion private and low‐proportion of no insurance patients or low proportion of private and high proportion of no insurance patients (Table OS4, panel C, in Supporting Information).
Factors driving intra‐ailment effects
We examine organizational routines as a mechanism for intra‐ailment effects. Routines are “generative systems that produce repetitive, recognizable patterns of interdependent action carried out by multiple participants” (Pentland & Feldman, 2008, p. 236). Hospitals use care protocols that contain the pattern of actions to be taken in different situations. Such routines are central to health‐care organizations and assist in managing quality and cost while also providing a patient‐centric approach to care (Goh et al., 2011). Routines have a flexible component that arises from improvisation as the need for change arises (Feldman & Pentland, 2003). Literature on routines that uses a practice perspective underscores the importance of adjustments to routines based on the context (Birnholtz et al., 2007).
Routines flow from organizational processes and organizational structure (Felin et al., 2012). Processual routines involve application of logic to sequencing interdependent events. Such routines are common in health‐care organizations—for example, process maps tap on the clinical knowledge of providers and staff members. These process (or workflow) maps describe the clinical and administrative process steps that a patient's cycle of care is required to follow. Workflows differ based on whether the patient was admitted through the ED or based on a previously scheduled appointment. Different from processual routines, structural routines emerge from organizational design elements, such as hierarchical levels, size, complexity, and vertical integration. Structures can enable or hamper individual actions or processes where routines are embedded. We examine how processual and structural mechanisms influence spillovers from quality penalties.
Processual mechanisms driving intra‐ailment effects of quality penalties
Process‐based routines in health care can emerge from outside the organization or can develop organically. For example, hospitals are required to follow strict regulatory protocols when a patient is admitted with chest pain for a suspected heart attack. Other routines are developed internally based on benchmarking or other knowledge management techniques (Batista et al., 2016). Process routines manifest in the form of clinical guidelines, professional certifications, best practices, and standardized pathways of care (Sackett & Rosenberg, 1995). Hospitals use explicit mechanisms such as scheduling, planning, and formal as well as informal communications purposely with the aim of improving continuity of care. Hospitals can also have implicit mechanisms that accrue from shared knowledge. We examine two types of processual routines that can encompass both explicit and implicit mechanisms to drive quality improvements. These include service‐level routines originating from the source of the patient admittance into the hospital and related to the patient's LOS in the hospital.
Source of patient admissions
In health care, it is common for work groups to face problems that occur daily in large frequencies. For example, hospitals have a high frequency of patients arriving at the ED with chest pain or other complaints (Edmondson et al., 2001). Promptly guiding an ED patient into appropriate care pathways is critical, given the relationship between time‐to‐treatment and survival in many ailments such as heart attacks (Veinot et al., 2012). Such guidance is necessary because flexing capacity is difficult in EDs due to the high cost of capacity (e.g., diagnostic equipment such as Computed Tomography (CT) scanners), uncertain demand, congestion costs, and space constraints. A vast literature underscores the importance of routines in the ED (McHugh et al., 2012) to manage patient flows using methods such as rapid assessment and early intervention. Routines can enable hospitals to provide care for ED patients within the constraints of resource utilization (Popovich et al., 2012), and additionally ED routines include planning for transitional care (Albert, 2016).
When the proportion of ED patients for a TS at a hospital, that is, the balance of unplanned visits, is greater, it places greater stress on the hospital's treatment of the TS and increases the importance of using routines to manage patient flows. Drawing from research that indicates that ED routines are associated with improved quality of the entire care process (Jarvis, 2016), we expect hospitals with a higher proportion of ED admits in a TS to design quality routines in other areas, including continuity of care.
To test this, we construct subsamples at the cohort level, using a median spilt based on the proportion of emergency admits to total admits in the TS cohorts. We re‐estimate intra‐ailment effects as in Equation (VI) for the two subsamples, namely, above median proportion of ED admits in TS (labeled “high ED in TS”) and at or below median proportion of ED admits in TS (labeled “low ED in TS”). Results (panel A, Table 3) indicate that, as predicted, positive intra‐ailment effects of quality penalties on continuty of care are significantly higher for high ED in TS cohorts, relative to low ED in TS cohorts.
Variation in intra‐ailment and spillover effects.
Note: Robust standard errors are in parentheses.
Abbreviations: NTRS, non‐targeted but related services; TS, targeted services; URS, unrelated services.
p < 0.01; **p < 0.05; *p < 0.1; hospital, ailment, and year fixed effects (FE) included
Hospitals use a variety of intervention programs aimed at reducing preventable hospital readmissions. A majority of readmissions occur soon after discharge (Harrison et al., 2011). Premature discharge is often a cause for readmissions (Chrusch et al., 2009), but keeping patients longer requires expensive intensive care capacity, reducing its availability for newly admitted critically ill patients. Discharge to continuity of care facilities, once the patients recover from the most acute phase of their illness, is beneficial to capacity management because it frees up intensive‐care hospital beds (Grabowski & Maddox, 2020). Hospital LOS is, thus, a strategic decision and involves capacity planning, case management, and design of appropriate continuity of care.
We predict that in response to readmission penalties, TS cohorts that have low LOS make greater increases to their continuty of care to reduce the risk of readmissions. We construct cohort‐level subsamples based on a median spilt based on LOS in TS. We re‐estimate intra‐ailment effects as in Equation (VI) for the two subsamples—TS cohorts with above median LOS (labeled “high LOS in TS”) and TS cohorts with at or below median LOS (labeled “low LOS in TS”). Results in panel A of Table 3 indicate that low LOS in TS cohorts show greater post‐regulatory increases in CC, relative to High LOS in TS cohorts.
Structural mechanisms driving intra‐ailment effects of quality penalties
A variety of structural factors can influence hospital capabilities including organizational structure, size, and resources (Felin et al., 2012). We examine two hospital‐level structural factors, namely, size and resources invested in continuity of care. These factors influence knowledge development, coordination, and integration, with implications for continuity of care routines.
Hospital system size
Hospitals often are members of multi‐hospital systems. System membership provides benefits such as reputational enhancement, demand pooling, bargaining power, and enhancing efficiency through resource sharing (Melnick & Keeler, 2007). The size of the multi‐hospital system can impact the speed at which knowledge is shared, diffused, and replicated. When quality penalties disrupt existing work routines, organizations must undergo a learning process, agree on modified routines, and make organizational adaptation to implement the new routines successfully (Edmondson et al., 2001). Larger systems could be handicapped by inertia and entrenched routines that inhibit adaptation (Hannan & Freeman, 1989). Employees of multi‐hospital systems could lose their clinical and managerial autonomy, thwarting their ability to effect changes. We posit that modifications to continuity of care routines are easier to implement in smaller hospital systems.
To test this prediction, we obtain system size data from the Healthcare Information and Management Systems Society (HIMSS) database and spilt our sample into two hospital‐level subsamples based on system size relative to median in the year immediately preceding quality penalty regulation. System size, which refers to the number of individual hospitals that belong to the system, range from 1 to 37, with a median of 2. We estimate Equation (VI) for the subsamples of hospitals that belong to relatively smaller systems (labeled “small system”) and hospitals that belong to larger systems (labeled “large system”). Results in panel A of Table 3 indicate that intra‐ailment effects of quality penalties on continuty of care are marginally greater in small systems than large systems.
Continuity of care increases require investments in capacity. We posit that larger hospital systems can implement and modify routines and achieve quality increases, if such systems have investments in continuity of care capacity. Using financial and utilization data from HCAI, we examine if increases in continuty of care in TS (relative to URS) are greater in larger systems that have pre‐existing investments in continuity of care services for acute care (step down), other care, and SN/IC. To calculate this, we use “Full Data Sets” as provided by OSHPD's financial and utilization datasets and examined the “Financial and Utilization data” page. An example of a variable used to capture whether a hospital had investments in continuty of care is “Beds_Staffed (Avg)_Skilled Nursing Care.” Results in panel A of Table 3 indicate greater intra‐ailment effects (i.e., post‐regulation increases in continuty of care of TS relative to URS) in the subsample of large system hospitals that had made investments in continuity of care facilities prior to the introduction of quality penalties, relative to large system hospitals without such investments.
Complementarities as a source of spillovers
H2 identifies complementarities between TS and NTRS as the driver of spillovers. Evidence of higher continuty of care increases in NTRS in instances where the targeted ailments (TS) have higher continuty of care increases would provide evidence for spillovers from complementarities, which accrue at the specialty level. Accordingly, we spilt our sample at the specialty level, into two subsamples based on the CC increases in TS (relative to URS) in 2010 relative to 2009. For each specialty, we calculate the difference in continuity of care between TS and URS for 2009 and 2010. We then rank the improvements in TS (relative to URS) in 2010 versus 2009. For example, if continuity of care for pneumonia (respiratory specialty URS) was 0.20 (0.15) in 2009 and was 0.23 (0.16) in 2010, the difference in continuity of care between pneumonia (TS) and the corresponding URS for 2009 and 2010 would be 0.05 and 0.07, respectively. This specialty would have shown an increase of 0.02 in continuty of care of TS (relative to URS) in 2010 relative to 2009.
Results based on estimating Equation (VI) using NTRS as the treatment group and URS as the control group in panel B of Table 3 indicate greater spillovers for specialties where TS showed relatively high increases in continuty of care relative to URS in 2010 relative to 2009. Spillover effects are not significant in the subsample with relatively low continuty of care increases in TS. This indicates that the spillover effects observed in NTRS (relative to URS) post‐quality penalty regulation occurred due to complementarities between the TS and NTRS cohorts.
Resource reallocation and gaming
Hospitals may redirect resources from other patients to treatment of TS patients, which would increase the continuity of care quality of TS patients but worsen outcomes for other patients. Our research design that compares changes in targeted and non‐targeted services within the same medical specialty is robust to such concerns because knowledge and infrastructure reside at the medical‐specialty level (e.g., physicians are trained in cardiology, and the same physician treats patients with heart attack and patients with angina). Similarly, technologies and processes adopted to treat TS are used to treat other ailments in the same medical specialty. Using URS as a control group allows us to address this concern because it controls for specialty‐level changes in resource allocations. Reallocation of resources within a specialty (e.g., from URS or NTRS to TS) would be infeasible because it would entail that hospital resources be deliberately withheld to a URS or NTRS patient.
In supplemental analysis (untabulated), we investigate hospital charges, which is an aggregation of the prices for every service received by a patient and thus reflect total resources consumed by a patient. We examine whether charges for TS increased after HRRP regulation relative to those of the remaining patient cohorts (NTRS and URS) and do not find significant results. Assuming that reallocations of resources would eventually be reflected in higher charges, this analysis rules out the possibility that hospitals redirected resources within a specialty to treat TS. Untabulated analyses also indicate that continuity of care in URS cohorts did not decline. Thus, hospitals did not appear to divert resources away from other non‐targeted services.
DISCUSSION
We examine intra‐ailment and spillover effects from regulation that introduced quality penalties for an adverse health‐care outcome—namely, 30‐day readmissions for three medical ailments (heart attack, heart failure, and pneumonia). Our results show that quality penalties for excess readmissions yielded increases in another type of health‐care quality, that is, continuity of care. Our empirical analysis indicates that continuity of care improved for all ailments targeted for penalties under the HRRP. Additionally, continuity of care improved for non‐targeted but related ailments. Further, increases in continuity of care accrue not just to targeted patients (Medicare) but spill over to non‐targeted patients (non‐Medicare).
Our study indicates an important role for regulatory spillovers with consideration to where these spillovers occur. Intra‐ailment effects of quality penalties for readmissions benefit continuity of care for the targeted ailments, and spillover to continuity of care increases for related but non‐targeted ailments. Moreover, continuity of care did not diminish for unrelated ailments, which assuages concerns that hospitals responded strategically redirecting resources. There are important theoretical and practical implications that we discuss next. We conclude this section by pointing to the limitations and directions for future research.
Theoretical implications
Our findings reveal that quality regulation aimed at penalizing hospitals for excess readmissions has implications for continuity of care through intra‐ailment and spillover effects. Evidence of intra‐ailment effects highlights how incentive structures can instigate changes to quality routines in health‐care organizations. Changes required for attaining the regulatory objectives result in the establishment and embedding of new routines for patient care. This presents a new perspective on the institutionalizing process resulting from an external stimulus and the associated organizational learning. Intra‐ailment effects on metrics not specifically targeted have implications for the theory of incentive design.
Results pertaining to spillovers provide insights into the interplay between routines that operate at different levels. A distinguishing feature of the health‐care context is that medical specialties are organized around organs and systems of the body, which results in complementarities associated with medical training, asset allocation, and routines. These complementarities pave the way for knowledge spillovers across related services as defined by these medical specialties. Our study shows that regulation‐induced change in routines for a service can spillover to related services within a firm. By shedding light on these spillovers, we contribute to complementarity theory and demonstrate its application within the health‐care value chain.
The linkage between knowledge spillovers and complementarities contributes to the ongoing debate on the microfoundations of routines, which calls for an understanding of the differences in the behavior and performance of firms by unpacking routines and capabilities in terms of their underlying primary components (Felin et al., 2012). Extant research distinguishes microfoundations in terms of individuals, processes and interactions, and structure. Health‐care professionals, especially physicians and surgeons, undergo rigorous training based on the medical specialty. This training results in the development of specialized knowledge and distinct routines that form the basis for spillovers between related services due to complementarities. We distinguish between processual and structural routines to capture the sources of heterogeneity in routines.
Practical implications
The findings of this study offer directions for considering routines when developing regulatory policies in the health‐care context. Health‐care organizations are structured around medical specialties, and the services within these specialties share strong complementarities owing to common knowledge‐base, processes, and physical assets. Since it is not practically possible to devise all‐encompassing regulations, information about complementarities can help in developing policies that consider potential spillovers due to the underlying complementary processes and assets. Additionally, our findings also shed light on how policymakers can consider spillovers due to these complementarities and the underlying routines to positively influence performance measures that are often difficult to regulate. Post‐acute care facilities are scattered, and it is not easy to create policy instrument/regulations to directly improve the quality of transitional continuity of care. We show that current policies that focus on readmissions, bundled care, and accountable care organization can address continuity of care‐related quality measures.
For health‐care organizations, our study has implications for considering processual and structural routines in quality improvement efforts. Resource allocation decisions can take these routines and complementarities into account. One key criticism of regulatory policies is that they motivate organizations to redirect resources from other activities to respond to the requirement. We show that these regulations can trigger a different sequence of actions whereby organizations develop routines that can aid in not only improving the outcome required by the regulation but also of other related tasks. This finding motivates careful considerations of routines at individual, processual, and structural levels to not only fulfill the requirements of a regulation but to gain operational and strategic benefits. Based on the results of this study, large health‐care organizations should invest in acute care facilities to develop necessary routines for improving transitional continuity of care.
Our study has implications beyond healthcare. Manufacturing, merchandise, and service industries can experience quality spillovers from regulatory changes owing to complementarities in production functions. For example, the department of transportation requires the disclosure of on‐time performance for domestic flights (Forbes et al., 2015). Process reengineering to improve ground turnaround times for domestic flights motivated by such focused disclosures can also benefit international flights. Our study speaks to outcomes from other types of regulations, such as environmental regulations. Bui and Kapon (2012) find that environmental disclosures reduce facility‐level toxic releases for substances that face formal regulation and spill over to substances that do not face formal regulation. Ignoring spillovers in such settings could lead to under‐estimation of the effectiveness of regulation on outcomes.
Limitations and directions for future research
There are some limitations of this study that provide directions for future research. This study focuses on a specific policy instrument of the ACA regulation. It is important that future research examines a broader set of benefits and costs of regulatory programs, including spillovers into related products and services to facilitate meaningful policy decisions. In our study, we consider processual and structural routines but could not capture individual‐level routines due to data limitations. By examining routines emanating at the three levels—individual, processes and interactions, and structure—future studies can help with a fuller understanding of microfoundations of routines in the health‐care setting. The results from the analysis of mechanisms in our study are consistent with routines, but we do not examine the creation, development, operation, maintenance, and change of specific routines. Further research is needed to understand how microfoundations contribute in this regard and the temporal sequence in which these microfoundations operate. Field‐based research would be helpful in identifying the types of routines in a granular manner. Further work is also required about organizational mechanisms and strategies for achieving changes to quality‐based routines. In our investigation of discharges to post‐acute care facilities, we only consider heterogeneity among patients as it relates to their clinical diagnoses. Future studies can examine if the spillovers identified in this study vary across ethnic classes and socioeconomic strata that patients belong to. Post‐acute care facilities have been reported to serve as “pop‐off valve” for hospitals to manage their capacity (Grabowski & Maddox, 2020). Such capacity management tactics, if followed by several hospitals, can constrain capacities of post‐acute care facilities, thereby impacting their quality of care. Future studies can examine the effect of regulations in transferring patients to post‐acute care facilities considering the relative availability of capacity in these facilities.
Footnotes
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.