Abstract
Despite the advent of new antiepileptic drugs (AEDs) over the past 15 years, the treatment of uncontrolled partial-onset seizures remains a dilemma for clinicians. The most recent AEDs offer new mechanisms of action and more favorable safety profiles than the first generation of AEDs. Lacosamide (LCM) is the latest AED awaiting approval by the FDA for adjunctive use in partial-onset seizures. It differs from all other approved AEDs in that it has two novel mechanisms of action and favorable pharmacokinetic and safety profiles. The purposes of this article are to present the significant parameters for its use in clinical practice, by summarizing the preliminary results of phase II and III clinical trials, and to compare its efficacy data with other second-generation AEDs.
Lacosamide (LCM) (SPM 927, formerly harkoseride), the R-enantiomer of 2-acetamido-N-benzyl-3-methoxypropionamide, is a chemical compound with anticonvulsant and antinociceptive properties. In November 2007, a new drug application was filed with the FDA for use of LCM as adjunctive therapy in the treatment of partial-onset seizures in adults with epilepsy. The application includes three formulations: tablets, syrup, and an intravenous injection. LCM was approved in Europe on September 3, 2008 as adjunctive therapy in the treatment of partial-onset seizures, with or without secondary generalization, for patients with epilepsy 16 years or older (1).
The purposes of this article are to present the significant parameters for its use in clinical practice and to summarize the results of phase II and III clinical trials. A Medline search was conducted using the term “lacosamide,” “SPM 927,” and “harkoseride.” Cross-reference was also conducted from various articles. Bibliography and abstracts were provided by the manufacturer. Three major phase II/III clinical trials were found, but only one has been published in full. In addition, a summary of clinical trial results (with a subject number over approximately 100), comparing lacosamide with other commonly used second-generation AEDs for adjunctive therapy in adults with refractory partial seizures, is presented.
Pharmacology
LCM is a member of a series of functionalized amino acids that have been screened for anticonvulsant properties (2–4). It causes a general decrease in neuronal discharge frequency and synaptic excitability in laboratory testing. Since these actions are not mediated at major excitatory (AMPA/NMDA) or inhibitory (GABAA) postsynaptic receptors (5), the mechanism of action of LCM was initially unknown. Recently, two different probable mechanisms of action have been discovered. First, LCM selectively enhances slow inactivation of sodium channels. All sodium channels undergo both slow and fast inactivation. Classical sodium channel modulators selectively enhance fast inactivation. Since slow inactivation of sodium channels is an endogenous mechanism by which neurons reduce ectopic hyper-activity, this modulation represents an effective mechanism to selectively reduce pathophysiological hyperactivity, while leaving physiological activity intact (6,7). Second, LCM binds to the collapsin response mediator protein-2 (CRMP-2) and modulates mCRMP2 function in vitro. It is unclear whether the effect on this protein is stimulatory or inhibitory, but as a consequence, LCM appears to attenuate the effects of neurotrophic factors on axon outgrowth (8).
LCM has shown a unique profile when studied across a range of animal models of epilepsy, demonstrating an anticonvulsant effect similar to many of the newer antiepileptic medications. It is effective in the maximal electroshock model (9). At both the median effective dose for maximal electroshock model protection and the median toxic dose for rotarod impairment, LCM elevates the seizure threshold in the pentylenetetrazol seizure test (9,10). LCM also decreases self-sustaining status epilepticus in rats (10), prevents spontaneous generalized tonic–clonic seizures in the cobalt/homocysteine model of self-sustaining status epilepticus (9), inhibits NMDA-induced seizures in mice (9), and is able to completely block 4-aminopyridine-induced seizures in the visual cortex in vitro (11). LCM is unique, however, as shown in a diverse set of investigations. First, using the 6-Hz model of psychomotor seizures, which is considered a model for treatment-resistant epilepsy (12), LCM has a high potency to reduce seizures (9,13,14). Second, while the ability to protect genetically bred mice against audiogenic seizures is a feature of many antiepileptic drugs (AEDs), LCM is particularly effective in decreasing these sound-induced seizures in Fringe mice (9). Finally, LCM is conspicuously inactive against clonic seizures induced by subcutaneous administration of the chemoconvulsants pentylenetetrazol, bicuculline, and picrotoxin (9).
Clinical Trials on Partial-Onset Epilepsy
One phase II study and two phase III studies (reported only in abstract formats) of LCM for the treatment of partial epilepsy have been completed. All three studies included between 400 and 500 subjects with uncontrolled partial-onset seizures; subjects were required to have had at least four partial-onset seizures per 28-day period, within an 8-week baseline phase, and no seizure-free period longer than 21 days. LCM was studied as add-on therapy to current AEDs, with or without a vagus nerve stimulator. The maintenance phase in all three trials was 12 weeks. The methodological differences between these three studies are listed in Table 1.
Summary of Phase II/III LCM Clinical Studies for Refractory Partial-Onset Epilepsy
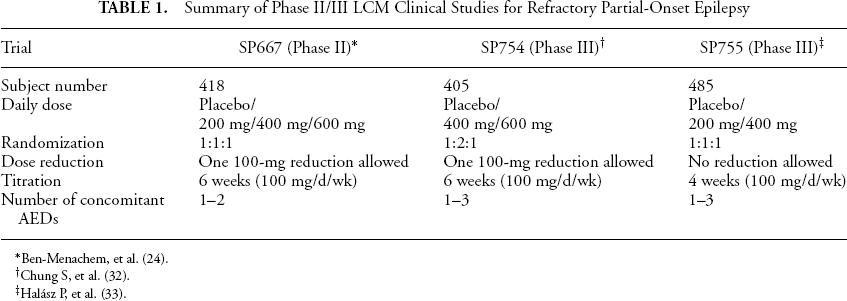
Ben-Menachem, et al. (24).
Chung S, et al. (32).
Halász P, et al. (33).
The two primary outcome measures for these three studies were: 1) 50% responder rate and 2) median percentage seizure reduction over the maintenance phase, as compared to the previous 28-day baseline. All three studies showed that LCM provided statistically significant benefit in these two outcome measures at 400 mg/day. Two of the studies (SP667 and SP754) that tested the 600-mg/day dose also found statistically significant benefits, but not significantly greater than the 400-mg/day dose. The 200-mg/day dose showed statistically significant benefit in one of two studies that tested this dose (SP755); however, the positive benefit was only in regard to median percentage seizure reduction and not the 50% responder rate (see Figures 1 and 2) (15,16,17). Of interest in the SP667 trial, the proportion of patients experiencing a 75% reduction in seizure frequency was also significantly better among the 400-mg (22.4%, p = 0.002) and 600-mg (16.2%, p = 0.03) groups, as compared with placebo (6.3%) (24).
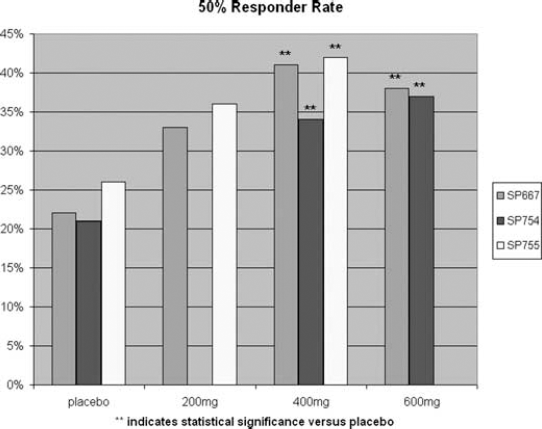
Summary of LCM phase II/III studies efficacy outcome: 50% responder rate.
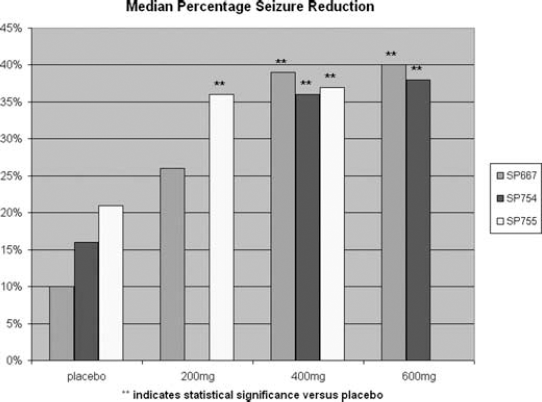
Summary of LCM phase II/III studies efficacy outcome: median percentage seizure reduction.
Comparison of LCM to other Commonly Used Second-Generation AEDs
The placebo response rates for the three LCM clinical trials (10–25%) are consistently higher (>15%) than those observed in historical placebo-controlled trials of AEDs but appear to be comparable to placebo-controlled trials of AEDs in current clinical development. It is thought that this higher placebo-response rate in recent trials may be due to a number of factors, including differences in clinical trial methodology, baseline number of seizures, the number of concomitant or previously used AEDs, concomitant use of enzyme-inducing or enzyme-inhibiting medications, treatment and titration intervals, or the subjects’ nationality. A greater number of concomitant AEDs may be an explanation for the higher placebo-response rate to LCM compared with most other second-generation AEDs, but it does not hold in comparison to pregabalin (see Table 2). Another possible explanation might be the recent increase in the percentage of study subjects recruited for antiepileptic clinical trials outside of the United States and Europe, where placebo-response rates may be higher, although this theory is difficult to investigate since such information is not published.
Placebo Response Rates and Concomitant Therapy in Pivotal Trials of Lacosamide, and Other Second-Generation Antiepileptic Drugs in Medication-Refractory Partial Epilepsy
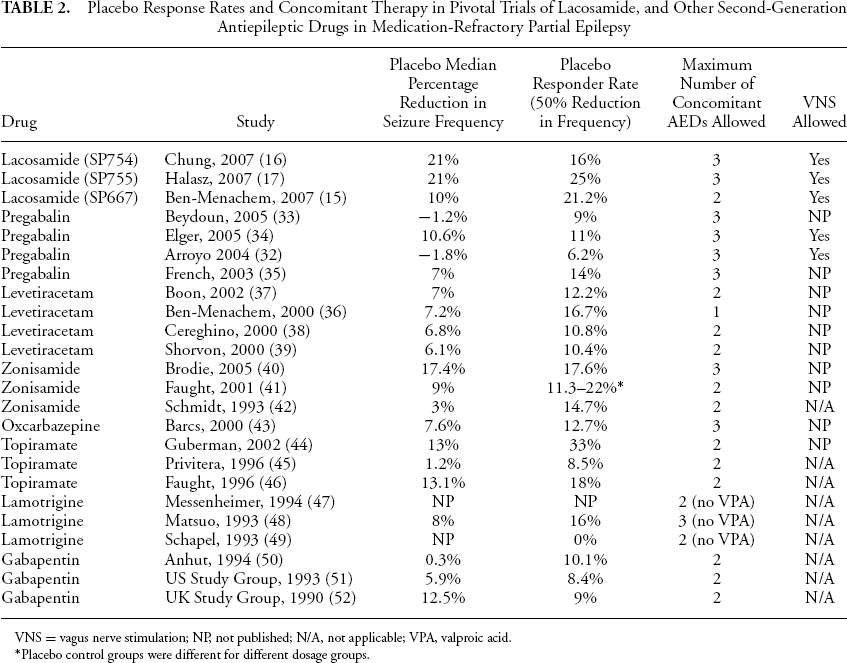
VNS = vagus nerve stimulation; NP, not published; N/A, not applicable; VPA, valproic acid.
Placebo control groups were different for different dosage groups.
The response over the placebo rate of LCM is comparable to that of levetiracetam, zonisamide, oxcarbazepine, lamotrigine, and gabapentin, but clearly lower than that of pregabalin and topiramate. These results are summarized in Table 3A and 3B. This distinction is particularly evident when pregabalin and topiramate were tested in higher dose ranges than those considered to be tolerable to the majority of patients. Oxcarbazepine also produced a high response over placebo effect when it was tested at a generally intolerable dose of 2,400 mg/day. However, certain doses of pregabalin and topiramate (e.g., pregabalin 300 mg/day and topiramate 200 mg/day), which generally were found to be tolerable in the clinical trials, produced a median seizure frequency response over placebo measures that were greater than 40% in at least one study—a response considerably higher than that of LCM. Whether a lower placebo response rate would have improved the performance of LCM is uncertain. It is important to note that any comparison of efficacy data between AED clinical trials is difficult because of the many methodological differences between trials, including flexible-dosing versus fixed-dosing regimens, differing rates of titration (or lack of titration), variable numbers of concomitant AEDs, exclusion of certain concomitant AEDs, and cross-over versus noncross-over studies designs. A meta-analysis using relative response over the placebo rates, adjusted for various study designs, would be more appropriate and is beyond the scope of this article.
Efficacy Data in Pivotal Trials of Lacosamide and Other Commonly Used Second-Generation Antiepileptic Drugs
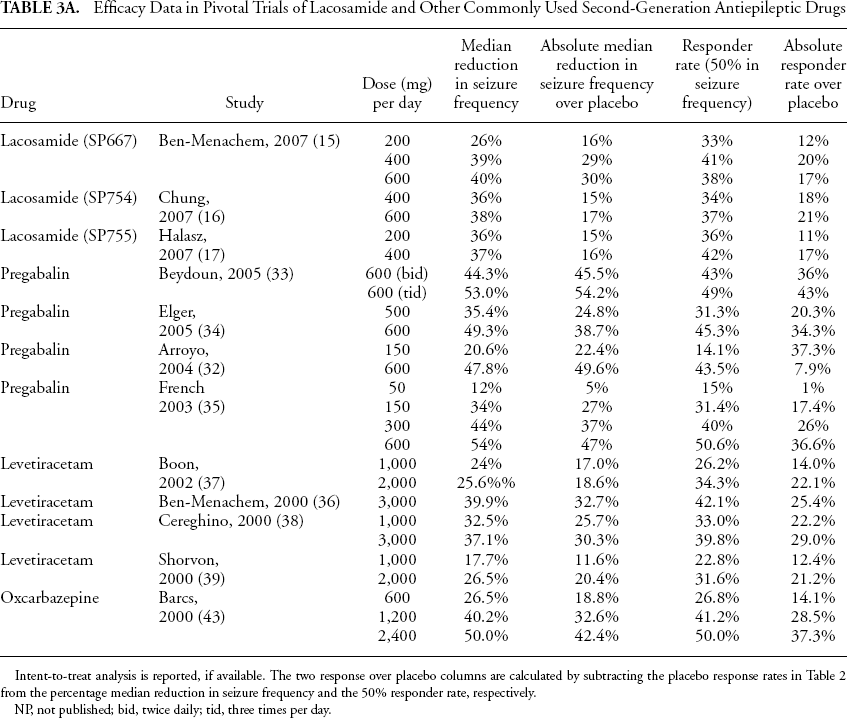
Intent-to-treat analysis is reported, if available. The two response over placebo columns are calculated by subtracting the placebo response rates in Table 2 from the percentage median reduction in seizure frequency and the 50% responder rate, respectively.
NP, not published; bid, twice daily; tid, three times per day.
Efficacy Data in Pivotal Trials of Lacosamide and Other Commonly Used Second-Generation Antiepileptic Drugs
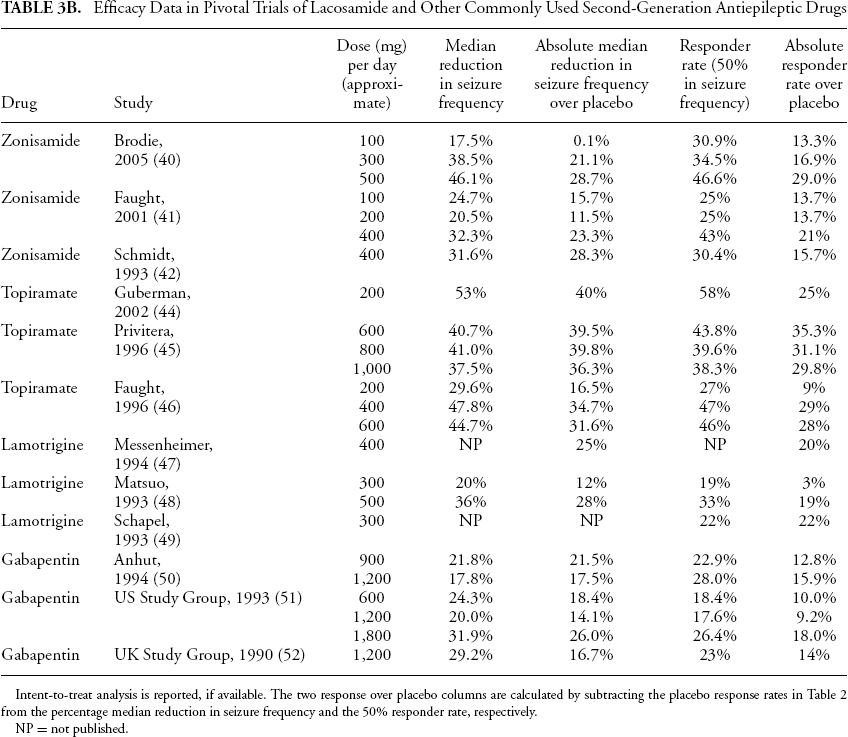
Intent-to-treat analysis is reported, if available. The two response over placebo columns are calculated by subtracting the placebo response rates in Table 2 from the percentage median reduction in seizure frequency and the 50% responder rate, respectively.
NP = not published.
Clinical Trials Data on Adverse Events and Long-Term Safety
Based on the three pivotal clinical trials previously discussed, LCM appears to be well tolerated. Most adverse events were mild to moderate and were more commonly observed during the titration phase. The most common adverse events were related to CNS and gastrointestinal side effects, and they appeared to be dose related. The two studies comparing 400 mg/day and 600-mg/day dosing revealed that the emergent adverse events leading to discontinuation of LCM were dizziness, nausea, diplopia, abnormal coordination, ataxia, vomiting, and nystagmus. Of these, dizziness was the most common. LCM appears to have a low incidence of psychiatric side effects with psychosis reported in only 0.3% of patients (18). There is no report of rash, hyponatremia, or cognitive slowing. There is no evidence of abuse potential. Among other clinically relevant side effects seen with other AEDs, LCM does not cause weight gain or weight loss, neither does it change patterns in hematology, blood chemistry, and vital signs (15,17,19). The percentage of patients who withdrew from the studies increased with higher doses, particularly with the 600-mg/day dose (see Figure 3). Although the LCM efficacy data may be slightly better for 600 mg/day than 400 mg/day, when data from all three clinical trials are pooled, the tolerability of LCM is better at 400 mg/day, thus making it appear to be the optimal dose.
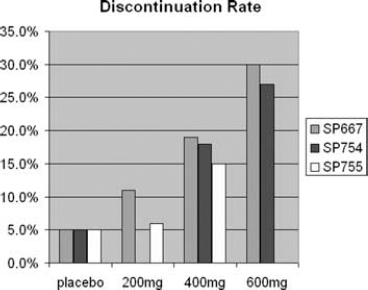
Summary of LCM Phase II/III studies—compliance outcomes.
Long-term safety data are also available for the open-label study SP615 (which is the continuation of SP667). In this study, the drug is well tolerated, and there is evidence for well-maintained seizure reduction by LCM for up to 2.5 years. Similar to the previous findings, long-term LCM treatment was not associated with any pattern of change in basic testing of blood chemistry and hematology or weight measurements. A small increase in median PR interval (5–9 msec) across all subjects (all modal doses) was observed on the EKG (15,19).
Pharmacokinetics
Studies in healthy adult volunteers have shown that oral LCM is rapidly and almost completely absorbed from the gastrointestinal tract, with an oral bioavailability of approximately 100%. LCM is absorbed within 4 hours and food does not affect its bioavailability (20,21). The intravenous formulation of LCM has been studied in short-term replacement therapy and appeared to be safe and well tolerated (22).
LCM undergoes minimal first-pass metabolism and is eliminated by renal excretion (40%) and biotransformation. The primary metabolite from demethylation (CYP2C19) has no activity and is eliminated by the kidney (30% of the dose). LCM has a half-life of 13 hours, displays a first-order pharmacokinetic elimination profile, and is administered twice daily. Pharmacokinetic studies showed a predictable volume of distribution and elimination rate (23), and no clinically significant differences between the pharmacokinetics of LCM in young and elderly subjects were observed. AUC and Cmax are increased by 20% in the elderly, but no dose adjustment is recommended (24). Protein-binding is minimal (less than 15%), and plasma concentrations are consequently linear and proportional for doses up to 800 mg per day and steady state is achieved at 3 days (21). Therapeutic concentrations with LCM have not been established; drug level monitoring is not necessary. Moderate hepatic and renal impairment have both been shown to increase systemic drug exposure up to approximately 40% (18). Titration, therefore, should be carried out with caution in these patient populations.
LCM displays a favorable interaction profile with currently prescribed AEDs and other commonly used medications. A clinical pharmacokinetic study with healthy subjects demonstrated that concomitant administration of LCM with the commonly prescribed AEDs, such as valproic acid and carbamazepine, does not significantly affect its rate and its extent of absorption or that of the AED (25); however, this was a short-term study that did not take into account carbamazepine-induced liver enzyme induction. LCM had no relevant influence on plasma concentrations of concomitant AEDs (including gabapentin, topiramate, lamotrigine, carbamazepine, levetiracetam, phenytoin, phenobarbital, oxcarbazepine, valproic acid) for patients with partial seizures, with or without secondary generalization (15,26). In population pharmacokinetic studies, the blood level of LCM is decreased by approximately 15–20% by enzyme-inducing antiepileptic medications (18) No food–drug or drug–drug interactions with other marketed agents (i.e., digoxin, metformin, omeprazole, and oral contraceptives containing ethinylestradiol and levonorgestrel) that could be affected by the CYP2C19 pathway or protein binding at therapeutic levels have been identified (25).
Safety
LCM has a dose-dependent, behavior-depressant effect, which is manifested by sedation and reduced locomotor and coordination activities in animals (2). These adverse events were observed in clinical trials when study subjects were exposed to high doses.
Two studies have shown that LCM solution for infusion is safe and well tolerated when given as an intravenous infusion over the time periods of 10, 15, 30, and 60 minutes in doses ranging from 200 mg to 600 mg per day (27,28). Few adverse events were reported during these intravenous studies, and the safety profile for LCM injection was comparable to oral LCM tablets, based on analyses of adverse events, EKGs, vital signs, and laboratory parameters (22,27,28).
LCM does not exert a clinically relevant prolonging effect on QTc or QRS intervals, and it is unlikely that LCM could influence the conduction and ventricular repolarization (29). A study of QTc interval in healthy subjects administered 400 mg and 600 mg per day of LCM did not show any QTc prolongation (30). A small increase in median PR interval (5–9 msec) across all subjects (all modal doses) has been observed on the EKG in recent clinical trials (15,19); however, the clinical significance of this finding is still unknown and does not appear to be clinically relevant for noncardiac patients.
Based on animal studies to date (mostly in rats and dogs), LCM has no effect on the autonomic nervous system and does not have clinically significant adverse effects on the gastrointestinal, renal, or respiratory systems (2). LCM has no effect on fertility and reproductive function of male or female rats and shows no teratogenic properties in rats or rabbits; however, developmental toxicity was observed at a maternally toxic dose (2). No age-specific toxicity was observed in a study with juvenile rats except for a slight delay in overall development at the highest dose. Overall, LCM showed a favorable profile in preclinical reproductive, developmental, and juvenile toxicity studies (30).
Conclusion
LCM is a new AED with two novel mechanisms of action that may be suitable for use with uncontrolled partial-onset epilepsy. Its pharmacokinetic profile is favorable, and the drug does not appear to have potentially severe adverse effects. Most of its side effect profile is dose-dependent and related to the CNS. The potential for drug interactions with other AEDs and currently prescribed medications is very low. Overall, there is a minimal dosing and clinical monitoring requirement with LCM.
In phase II and III clinical trials, LCM has been shown to be an effective add-on therapy, compared with placebo. A 400-mg daily dose displays the best risk/benefit ratio, as 200 mg daily was not found to be consistently better than placebo in clinical trials thus far. A maximum of 600 mg daily can be efficacious for some patients, but it may well be at the expense of more CNS side effects.
Disclosures
Dr. Halford: UCB, Inc. Consultant and Speaker Bureau on levetiracetam. Principal investigator in two clinical trials with LCM (SP754 and SP756). Dr. Lapointe: UCB, Inc. Speaker Bureau on levetiracetam. Co-Study Coordinator in two clinical trials with LCM (SP754 and SP756).
