Abstract
Antiepileptic drugs are important psychotropic agents that are commonly used to treat psychiatric disorders. The behavioral effects of antiepileptic drugs may differ between epilepsy and psychiatric patient populations. Randomized, double-blind, controlled data on the psychotropic efficacy of antiepileptic drugs are limited mainly to bipolar disorder.
Antiepileptic drugs (AEDs) are psychotropic agents; that is, they act on the mind and can positively or negatively influence behavior. This result is expected, given their mechanisms of action, which are to alter ion channel and neurotransmitter system functions and, thereby, modulate the electrochemical systems that underlie behavior. Behavioral effects (e.g., cognitive and mood) associated with AEDs are complex and can vary dramatically between patients. It is currently not possible to predict which patient with epilepsy will tolerate an AED and which one will develop adverse, as opposed to positive, psychotropic effects.
AEDs and Behavioral Effects
Studies involving either adverse or beneficial effects of AEDs in one patient population (e.g., epileptic, psychiatric, or other clinical groups, such as patients with migraine and diabetic neuropathy) cannot be assumed to apply to another patient population. A spectrum of behavioral effects from these medications can be experienced by patients with or without epilepsy. In epilepsy patients, suppression of seizures or interictal epileptiform activity may be accompanied by positive or negative behavioral effects. In general, sedating AEDs, such as valproic acid and carbamazepine, possess anxiolytic, antimanic, and sleep-promoting benefits but may cause fatigue, impaired attention, and mood depression. Activating AEDs, such as felbamate and lamotrigine, may possess antidepressant and attention-enhancing efficacy but may cause anxiety, insomnia, and agitation. These generalizations of the effects of AEDs are limited by the wide variability of clinical responses.
Even among epilepsy patients, striking differences may be seen in the effects of an AED, based on both individual reactions and other clinical factors. For example, in comparison to patients with new-onset epilepsy, refractory epilepsy patients may be resistant to adverse neurobehavioral effects when administered a new or additional AED. Studies of inpatients with medically refractory epilepsy who were administered felbamate showed that rapid escalation of the dose to 3,600 mg/day was often well tolerated (1). However, with outpatient populations, slower titrations more commonly elicited side effects, such as gastrointestinal, headache, insomnia, restlessness, and agitation (2).
Epilepsy patients, in general, may be more susceptible to the adverse behavioral effects of AEDs than are other populations, possibly resulting from structural or functional changes that increase their risk of psychiatric disorders. For example, affective and psychotic symptoms exhibited with levetiracetam administration are significantly more common among patients with epilepsy than among patients with cognitive or anxiety disorders (3). For other medications, such as ethosuximide, vigabatrin, and topiramate, a forced normalization like process may contribute to behavioral changes, such as psychosis (4–6). A hypothesis relating to this phenomenon posits that epileptiform discharges may mimic electroconvulsive therapy in a focal area, and discharge suppression may lead to psychopathology. However, multiple other mechanisms are likely.
Off-label Use of AEDs
The vast majority of data regarding psychotropic effects associated with AEDs show negative changes, such as anxiety, irritability, agitation, depression, and psychosis, possibly as an artifact of study designs focused on screening for adverse effects. Many of the reports regarding positive psychotropic effects associated with AEDs are based on small samples, often using retrospective, nonrandomized, or nonblinded data. As a result, a bias exists toward focusing on adverse effects that are epiphenomena of antiepileptic treatment, whereas the need for proactive assessment of positive effects of AEDs has not been met.
The dramatic increase in the use of AEDs as therapy for psychiatric disorders outpaces evidence of their efficacy. Although behavioral effects as a side effect have been studied, large, randomized, controlled trials that directly assess behavioral effects associated with AED administration are scarce. However, studies do provide evidence that carbamazepine, valproate, and lamotrigine act both as antimanic agents and mood stabilizers in bipolar disorder (7). The increased off-label use of AEDs, other than these three, for psychiatric disorders is potentially dangerous. The data supporting the use of AEDs for psychiatric disorders often is based on case reports, small open-label series, or controlled studies that are limited by sample size and statistical power or by methods biased toward positive findings. For instance, presenting topiramate as a treatment for major depression or anxiety that also helps with weight loss is misleading. Data solidly show that topiramate helps patients lose weight, but no data demonstrate a beneficial effect with affective disorders. No AED has been shown to be efficacious for the treatment of depression, generalized anxiety disorder, panic disorder, schizophrenia, or most other psychiatric disorders (8).
Frequently Used AEDs
The following are commonly administered AEDs and brief descriptions of their potential psychotropic effects.
Barbiturates
The barbiturates are rarely prescribed as psychotropic agents, although Rickels (9) and Shaffer (10) found that they possess anxiolytic, sedative hypnotic, and moodstabilizing properties in some patients. Barbiturates can impair cognition and motivation; depress mental and physical energy as well as mood; and cause hyperactivity, irritability, aggressive behavior, and impotence. Brental (11) showed that barbiturates are most likely to cause depression and suicidal ideation in patients with a family or personal history of affective disorder. With depressed, irritable, or aggressive epilepsy patients taking long-term barbiturate therapy, gradual conversion to a nonbarbiturate AED may avoid the need for prescribing a psychotropic medication.
Carbamazepine
Although carbamazepine is structurally similar to the tricyclic antidepressant imipramine, it does not have antidepressant effects. DeLeon (7) demonstrated that CBZ effectively treats mania and stabilizes mood in bipolar disorder. Uncontrolled studies support a role for CBZ in treating impulse-control disorders, such as borderline personality disorder with aggression and episodic dyscontrol syndrome. Stein (12), Uhde (13), and Lima (14) showed that CBZ is not effective in treating panic disorder or cocaine dependence. CBZ may cause behavioral problems. Friedman (15) showed that 10% of patients with mental retardation, who were treated with CBZ for mood disorders, developed adverse behavioral reactions. CBZ-induced behavioral disorders usually occur in patients with existing behavioral difficulties.
Ethosuximide
Ethosuximide, used to treat absence seizures, may cause confusion, sleep disturbances, aggressive behavior, depression, and, rarely, psychosis. Forced normalization with ethosuximide may cause behavioral abnormalities (4).
Felbamate
Felbamate has stimulant properties, which can cause anxiety, irritability, or insomnia (16).
Gabapentin
Pande (17) showed that social phobia and other forms of anxiety are effectively treated with gabapentin. In two double-blind studies, Pande (17) and Frye (18) revealed that gabapentin was ineffective for bipolar or unipolar depressive disorders, although Schaffer et al. (10) and McElroy (19) demonstrated that gabapentin improved mania and the depressive phase of bipolar disorder in open-label studies. Ryback (20) found that preliminary studies suggest a beneficial effect of gabapentin on behavioral dyscontrol, agitation in senile dementia (21), and self-injurious behaviors in neurologic syndromes (22). In uncontrolled studies with epilepsy patients, gabapentin improved the sense of well-being and mood dysfunction, independent of seizure reduction (23). Notably, this effect just reached statistical significance (p = 0.04) in one of three depression scales. The other two depression inventories had p values >0.57, whereas the anxiety scale had a p value >0.9. This study found no reduction in seizure frequency in the GBP-treated group (mean, 1,615 mg/day), a difference from double-blind placebo-controlled trials that showed efficacy of GBP at this dosage. GBP has minimal detrimental cognitive side effects with epilepsy patients (24,25) but occasionally causes irritability and agitation. This problem most often occurs in children with developmental disabilities but may affect children with normal intelligence and sensorimotor function as well as adults (26,27).
Lamotrigine
Lamotrigine effectively treats refractory bipolar disorder (28–30). During a 1-year follow-up, McElroy (31) found that bipolar patients experienced sustained improvement in depressive symptoms. The proportion of patients achieving remission by week 4 of the study was 81%, and episodes of mania/hypomania occurred less frequently than in the prior year. In a placebo-controlled trial of bipolar patients with recent mania or hypomania, lithium was superior to placebo at prolonging the time to a manic, hypomanic, or mixed episode, whereas LTG was superior to placebo at prolonging the time to a depressive episode (32). For patients with bipolar disorder I, substitution of LTG for other psychotropic medications was associated with improved cognitive function (33). Unlike some other psychotropic agents used to treat bipolar disorder, LTG does not destabilize mood or cause sexually adverse effects, weight gain, or withdrawal symptoms (32). In preliminary studies, LTG has demonstrated beneficial effects on unipolar depression (18), borderline personality disorder (34), and schizoaffective disorder (35). LTG may cause mild stimulation and insomnia, which can be managed by shifting most of the dose to the morning or early afternoon.
In patients with epilepsy, LTG has a favorable cognitive and behavioral profile (36). Patients with developmental disabilities and epilepsy may experience both positive and negative psychotropic effects (37,38). Positive effects include diminished irritability, hyperactivity, and perseveration, as well as improved energy and social function. Negative effects include irritability, hyperactivity, and stereotypic or aggressive behavior. Serum LTG levels do not predict a psychotropic response.
Levetiracetam
No positive psychotropic effects are established for LEV, although it is chemically related to the putative nootropic drug piracetam. Approximately 5% to 10% of adults and 12% to 25% of children taking LEV may exhibit irritability, anxiety, depression, and other behavioral disorders. These problems may occur more often in patients with developmental delays (39). Among 118 patients with epilepsy and learning disabilities who were treated with LEV, 15 (12.7%) had behavioral symptoms: 9 (7.6%), aggressive behavior; 2 (1.7%), affective disorder; 2 (1.7%), emotion lability; and 2 (1.7%), other personality changes (39).
Phenytoin
Once promoted as an antidepressant (40), PHT now is rarely used as a psychotropic agent, although a controlled study found efficacy for mania (41). PHT has a cognitive and behavioral profile similar to that of CBZ (42). Some patients experience sedation, psychomotor slowing, mild cognitive impairment, and depression. A chronic, cumulative encephalopathy may occur after long-term exposure to high doses, perhaps resulting partly from cerebellar atrophy. An acute encephalopathy and seizures may develop with toxicity (blood PHT levels, >35 μg/mL).
Tiagabine
In patients with epilepsy, tiagabine may improve mood or cause irritability, emotional lability, and dysphoria (43,44). Preliminary studies indicate that TGB is not effective in bipolar disorder (45).
Topiramate
TPM may improve mania and stabilize mood in bipolar disorder (3). TPM may help to treat binge-eating disorder (46) and to reduce aggression and other behavioral disorders in intellectually impaired adults (47). An open-label study supports its use for posttraumatic stress disorder (48) and social phobia (49). The weight loss associated with TPM can benefit many patients, especially those treated with drugs promoting weight gain (e.g., antipsychotic drugs, valproate, GBP, and selective serotonin reuptake inhibitors).
Cognitive and behavioral problems associated with TPM use are a significant concern with epilepsy patients. However, both cognitive and behavioral problems are less frequent and severe when the starting dose is low and is increased slowly (50). Cognitive disorders include impaired attention, word-finding, verbal fluency, memory, and psychomotor slowing. Behavioral changes include depression, irritability, and, rarely, psychosis (51). Among 103 patients in whom behavioral disorders developed while they were being administered TPM, 46 were affective, 22 were aggressive, 16 were psychotic, 11 were anxious, and 8 had personality changes (46). In a study of 470 refractory epilepsy patients, behavioral side effects were the most common cause for discontinuation (70%), followed by mental slowing (27.6%), dysphasia (16.0%), and mood problems (e.g., agitation, 11.9%) (51). In a randomized, controlled trial of TPM, GBP, and LTG in healthy young adults, only TPM-treated subjects showed significant declines in attention and word fluency, as well as increases on an anger–hostility scale (24). These changes were observed with acute doses and at 2- and 4-week visits. In patients with Lennox–Gastaut syndrome, adverse events included somnolence, mood problems, nervousness, personality disorder, and language problems (52).
Valproate
VPA effectively treats mania and stabilizes mood in patients with bipolar disorder (53,54). Compared with lithium, VPA was superior in providing a longer duration of successful prophylaxis and less deterioration in depressive symptoms, but suicide attempt and death rates were higher with VPA (55). It reduces the severity of acute alcohol withdrawal and reduces benzodiazepines needs (56). VPA may improve mood in patients with epilepsy, developmental disabilities, and schizoaffective disorder (57,58) as well as effectively treat panic disorder (58) and borderline personality disorder (60). Irritability, agitation, aggression, self-injurious behavior, and mood problems in patients with CNS disorders, such as head trauma, seizures, or dementia, may respond well to VPA therapy (58–60). VPA causes sedation and infrequently may cause cognitive impairment, irritability, depression, hyperactivity, and aggressive behavior.
Vigabatrin
Vigabatrin is an irreversible inhibitor of GABA transaminase, which increases GABA levels in the CNS. Vigabatrin has no established positive psychotropic effects and may cause depression, psychosis, and exacerbate hyperactivity (61,62).
Zonisamide
Preliminary studies suggest that zonisamide may help treat mania in patients with bipolar and schizoaffective disorders (63,64). However, ZNS may induce irritability, emotional lability, and rarely, mania, or psychosis.
Pharmacologic properties of the frequently used AEDs are summarized in Table 1.
Potential Psychotropic Effects of Antiepileptic Drugs
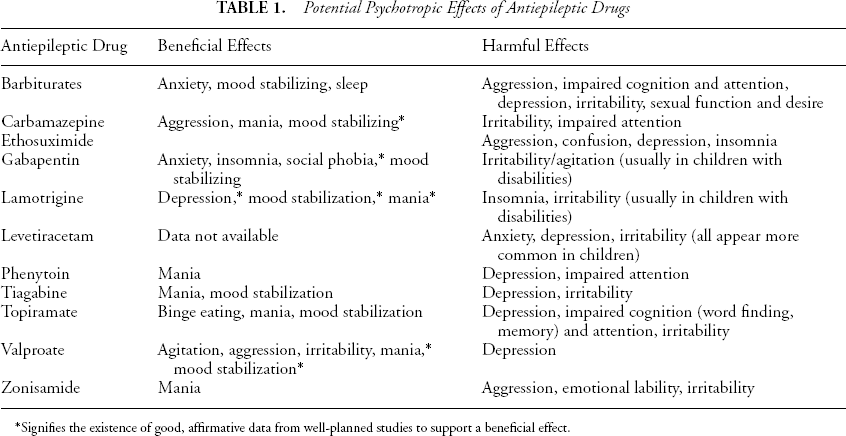
Signifies the existence of good, affirmative data from well-planned studies to support a beneficial effect.
Conclusion
AEDs are psychotropic agents with positive, negative, or no effect in diverse cognitive and behavioral domains. More randomized, controlled, and adequately powered studies are needed to assess their efficacy in treating psychiatric disease. Until such trials are performed, off-label use of AEDs for psychiatric conditions should be judicious. Psychotropic effects in the epilepsy population are variable and unpredictable. Unfortunately, a history of psychiatric illness may be a risk factor for negative psychotropic effects. Thus, beneficial psychotropic effects of AEDs, paradoxically, may be less relevant to epilepsy populations. Use of sedating doses and combinations of AEDs that can impair cognitive and behavioral function should be avoided. Finally, we must be wary of secondary effects of certain AEDs. For example, enzyme-inducing AEDs, such as CBZ, PHT, phenobarbital, and primidone, can increase sex hormone–binding globulin, reduce free (bioactive) testosterone, and thereby reduce libido and impair sexual function. Although sexual interest and function are not considered psychotropic effects, perhaps they ought to be.
