Abstract

Chronic daily headache is a difficult problem to treat for many adolescents, with a natural tendency for parents to look into ‘alternative’ treatments such as acupuncture, herbal remedies, biofeedback and relaxation techniques (1). Data on food trigger avoidance are controversial and not universally recommended even as an adjunctive treatment (2–4). Obesity may be a factor in frequent migraines (5) and therefore weight loss through exercise and diets may be theoretically beneficial (6).
The modified Atkins diet (MAD) is a high-fat, very low carbohydrate diet used in the treatment of children and adults with intractable epilepsy (7). This diet creates a ketotic state similar to the traditional ketogenic diet, but is started as an out-patient without a fast, or calorie, fluid or protein restriction (7). Weight loss can occur as well. Similar to anticonvulsant drugs, there has been recent interest in the potential benefits of ketogenic diets for the treatment of neurological conditions other than epilepsy, including Alzheimer disease, amyotrophic lateral sclerosis, brain tumours and autism (8). In 2006, a single case report was published regarding a 43-year-old woman with daily migraines who completely responded to a ketosis-inducing diet containing several high-protein and low-carbohydrate shakes per day (9).
Case series
A prospective, open-label study design was employed. Subjects included adolescents aged 12–19 years with chronic daily headaches (minimum of 15 headaches per week) for at least 3 months' duration (10). All subjects had failed at least two pharmacological preventative treatments prior to enrolment. Exclusion criteria included prior use of the Atkins diet for > 1 week, pregnancy, significant heart or kidney disease, hypercholesterolaemia, body mass index < 18 kg/m2, recent changes in preventative medications within the past week, and use of abortive migraine treatments (e.g. triptans or non-steroidal anti-inflammatory drugs) for > 11 days in the past month. This study was approved by the Johns Hopkins Institutional Review Board andwas listed at htttp://www.clinicaltrials.gov as NCT00181064.
At the first study visit, all adolescents were examined by a neurologist (E. H. K.) and laboratory values, baseline paediatric migraine disability scale (PedMIDAS) score (11), 3-day food record, and headache frequency were obtained. Approximately 30 min in the clinic was then spent with both the neurologist (E. H. K.) and a registered ketogenic dietitian (Z. T.) discussing food choices, with written dietary information provided. On the MAD, children were restricted to 15 g/day of carbohydrates. There were no specifications on which carbohydrates could be eaten. High-fat foods (e.g. heavy whipping cream, oils, butter and mayonnaise) were encouraged and neither calories nor fluids were restricted. A daily carbohydrate-free multivitamin and calcium supplement (any over-the-counter supplement) was recommended. Patients were told not to give additional supplementation with riboflavin (2) beyond this daily multivitamin.
Patients were instructed to check urine ketones twice weekly using over-the-counter reagent strips and measure body weight weekly. Subjects were seen again in clinic at 6 and 12 weeks, with a repeat 3-day food record, review of a headache calendar, and neurological examination at each visit. Carbohydrates could be increased to 20 g/day after 6 weeks if desired, but no medication changes were allowed. Laboratory studies and PedMIDAS were repeated after 12 weeks, which marked the study conclusion.
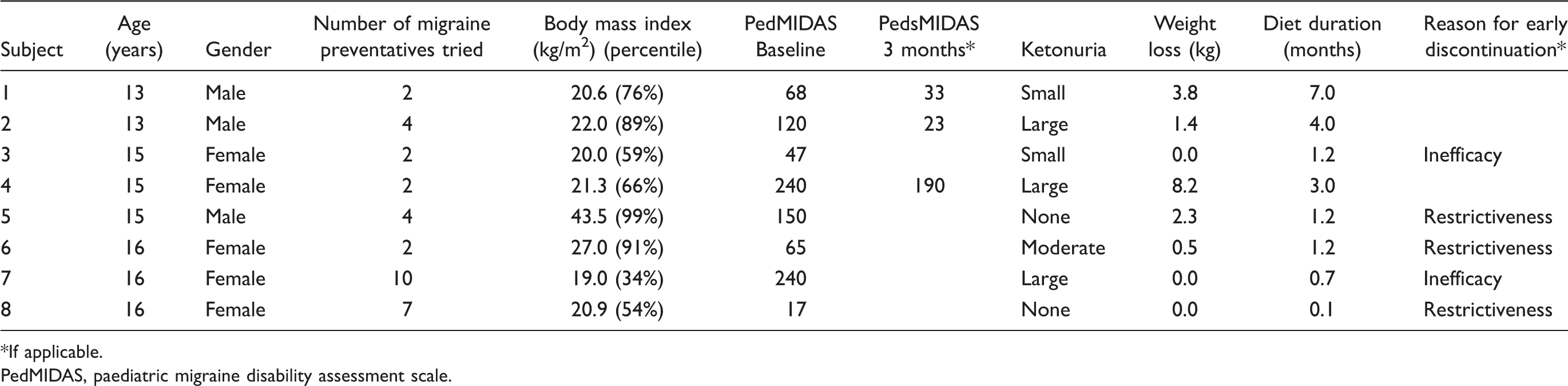
Demographics and results of modified Atkins dietary intervention in the eight subjects
If applicable.
PedMIDAS, paediatric migraine disability assessment scale.
Three subjects (38%) completed the 3-month study, with the other five discontinuing prior to the 6-week visit because of lack of efficacy (3) and restrictiveness (2). No adolescent had any reduction in headache frequency, with all right still having daily headaches when they concluded participation. Two subjects, both male and the youngest in this cohort (subjects 1 and 2), had improvements in their headache severity with a > 50% decrease in PedMIDAS scores. Both chose to remain on the diet after 3 months. Subject 2 is currently headache free, with reported resolution of his headaches 2 months after eventual diet discontinuation because of restrictiveness.
Large urinary ketosis was achieved in six (75%) subjects, with no clear correlation to either reduction in headache severity or diet duration. Weight loss occurred in five (63%), was highest in those who completed the study (subjects 1, 2 and 4), but also did not correlate with the reduction in PedMIDAS score. For the three subjects with follow-up laboratory studies at 3 months, liver and kidney functions were normal but there were slight elevations of total cholesterol (215, 215 and 217 mg/dl). Other adverse effects included temporary fatigue in two subjects (6 and 8).
Discussion
To our knowledge, this study represents the first published prospective study of a dietary treatment for migraine in 16 years (12). Unfortunately, not only were results unimpressive, but in this particular population recruitment was so poor as to lead to early study conclusion. Even the three subjects with a reported improvement in headache severity and quality of life continued to have daily migraines and require pharmacological therapy. There were insufficient study completers to analyse statistically for any potential beneficial aspects of those who responded.
There are probably several reasons for the failure of this study to demonstrate efficacy. First, dietary change is difficult for normal teenagers; adults treated in the past with this dietary therapy have had similar issues with poor compliance (13). Younger children, dealing with intractable epilepsy and who have strongly motivated parents, are probably likely to maintain this diet longer. Second, we chose a difficult neurological disorder, with an often poor prognosis for improvement (14). Children with less frequent, common migraines (e.g. weekly or fewer) may have responded better, as were the subjects of previous studies of oligoantigenic diets (12,15,16). Finally, most of these adolescents were normal weight at baseline, which may have prevented both a motivating secondary outcome and potentially beneficial mechanism from occurring.
In summary, there does not appear to be any clear benefit for the MAD in the treatment of adolescents with chronic daily headache. In addition, had improvement even been noted in this pilot study, this diet was perceived as too restrictive and most adolescents refused to try. Future studies could evaluate a younger, less intractable population using a less restrictive diet if desired. These results demonstrate the difficulties inherent in implementing dietary treatments for migraine.
Footnotes
Competing interests
E. H. K. is a consultant to Atkins Nutritionals, Inc.
