Abstract
There is growing evidence that alterations in the insulin and glucose metabolism may be involved in the pathogenesis of migraine. Nitric oxide (NO) stress has been associated with migraine. However, the role of NO on the insulin and glucose metabolism in migraineurs has remained elusive to date. The aim of the present study was to investigate the insulin and glucose metabolism in migraineurs and to determine possible interactions with the NO pathway. One hundred and twenty non-obese probands participated in this study, including 48 migraineurs and 72 healthy volunteers. Various parameters of the NO pathway, glucose metabolism as well as body measurement parameters were determined. We found a highly significantly increased insulin and Homeostasis Model Assessment (HOMA)-index in migraine patients, whereas fasting glucose was decreased. Logistic regression revealed an odds ratio of 5.67 for migraine, when comparing the lowest with the highest quartile of HOMA. Multivariate analysis showed that HOMA, waist-to-length ratio and nitrite as parameters of NO stress were highly significantly correlated. We show here that hyperinsulinaemia is associated with migraine and, furthermore, is correlated with increased NO stress. These findings represent a new pathophysiological mechanism that may be of clinical relevance.
Keywords
Introduction
There is growing evidence that insulin and glucose metabolism is altered in migraine patients and may play a pathophysiological role. Current studies by Rainero et al. (1) and Cavestro et al. (2) show that insulin sensitivity is impaired in migraine patients, which suggests a role for insulin resistance in the comorbidity of migraine and vascular diseases. Impaired insulin sensitivity in migraine patients is of particular interest, as insulin resistance has emerged as a risk factor for coronary artery disease, hypertension and stroke (3–5). Insulin also influences brain metabolism and cerebral blood flow (6,7). However, the underlying pathophysiological mechanisms have remained elusive to date. Nitric oxide (NO) has been implicated in migraine attacks in a variety of studies showing increased NO levels during migraine attacks (8–11). We have shown that migraine patients suffer under NO stress in the interictal phase (12). Increased NO stress in the headache-free period is associated with a 3.6-fold higher risk for migraine. NO, synthesized from the amino acid arginine by the enzyme family of NO synthases (NOS) plays a pivotal role in various pathophysiological pathways, including inflammation, the vascular system and the central nervous system (9). NO is a potent anti-atherogenic molecule, which acts as a vasodilatant, inhibits platelet aggregation, smooth muscle cell proliferation and expression of adhesion molecules (13–15). We here hypothesize that increased NO stress in migraineurs is associated with an altered insulin and glucose metabolism. The aim of the present study was to investigate the insulin and glucose metabolism in migraineurs and to determine possible interactions with the NO pathway. This was achieved by investigating various parameters of glucose metabolism and the NO pathway in non-obese migraineurs with (MA) and without aura (MoA). In detail, we investigated fasting glucose and insulin levels, body measurement parameters, blood pressure (BP), the amino acids of the NO pathway arginine and citrulline and the stable end products of NO nitrite, nitrate and nitrosylated proteins. We also investigated the biological NOS inhibitor asymmetric dimethylarginine (ADMA) and the expression of endothelial NOS (eNOS) and inducible NOS (iNOS).
Patients and methods
Patients
Baseline characteristics of migraine patients and healthy controls

*P < 0.05; **P < 0.01; ***P < 0.001 compared with controls.
MoA, migraineurs without aura; MA, migraineurs with aura; HOMA, Homeostasis Model Assessment; BMI, body mass index; BP, blood pressure; NOX, total amount of nitrate and nitrite; eNOS, endothelial nitric oxide synthase; iNOS, inducible nitric oxide synthase; ADMA, asymmetric dimethylarginine; SDMA, symmetrical dimethylarginine.
Blood collection
Blood collection was performed after written informed consent had been given. All probands were free of common infectious diseases and were not under medication. The median time period between the last attack and blood collection was 8 days (mean 19 ± 26 days). The shortest time range from patient's last attack to blood collection was about 53 h. The mean annual frequency of migraine attacks was 28.41 ± 27.92. Blood was obtained after an overnight fast by venous puncture and immediately centrifuged at ambient temperature; serum was stored at −80°C until analysis. Blood collection for gene expression analysis was performed with the PAXgene System (PreAnalytiX, Hombrechtikon, Switzerland), as recommended by the manufacturer.
Laboratory procedures
Serum nitrate and nitrite were determined with the NO Quantitation Kit (Active Motif, Rixensart, Belgium), as recommended by the manufacturer. Serum arginine and citrulline levels were determined by triple quadrupole mass spectrometry (Applied Biosystems API 2000 LC/MS/MS system; Vienna, Austria) with the EZ:Faast Amino Acid Kit from Phenomenex (Aschaffenburg, Germany) as previously described (17). Serum nitrosylated proteins were determined with the NWLSS Nitrotyrosine Enzyme-linked Immunosorbent Assay from Northwest Life Science Specialities (Vancouver, Canada). Serum ADMA as well as symmetrical dimethylarginine (SDMA) levels were measured by high-performance liquid chromatography and precolumn derivatization with o-phthaldialdehyde as described previously (18). Expression analysis of eNOS and iNOS from whole blood cells was performed with the PAXgene Blood RNA Kit from PreAnalytiX. Isolated RNA was transcribed into a cDNA library with the cDNA Archive Kit from Applied Biosystems. Quantitative real-time polymerase chain reaction was performed with the Taqman Universal Master Mix and gene expression assays on demand for eNOS, iNOS and glyceraldehyde 3-phosphate dehydrogenase as a housekeeping gene for normalization on a AB7900 (Applied Biosystems). Calculation of eNOS and iNOS expression was performed by using the delta crossing point method (19). Fasting glucose and insulin were determined routinely on Roche automated analysers (Roche Diagnostics, Mannheim, Germany). Homeostasis Model Assessment (HOMA) index was calculated as follows: [fasting glucose (mmol/l) × fasting insulin (UE/ml)]/22.5.
Statistical analysis
Data are presented as means ± S.D. Continuous variables were compared using Student's t-test for independent samples or Mann–Whitney U-test depending on the distribution of data. Differences in categorical variables were compared using χ2 test. Correlations between variables were determined by linear regression analysis according to Pearson. P-values < 0.05 were considered statistically significant. Subsequent multiple testing by linear stepwise regression analysis was used to determine the significance of test variables. The association between HOMA index and migraine was analysed using logistic regression to generate odds ratios (ORs) and 95% confidence intervals (CI). HOMA levels were initially examined in quartiles, with the lowest quartile defined as the reference group. Analyses were performed using
Results
Baseline characteristics of study probands are given in Table 1. The study groups did not significantly differ in age, BP, BMI, waist-to-hip or waist-to-height ratio. Analysis of the NO pathway showed significant alterations in migraineurs. Migraine patients showed significant increased nitrate (P < 0.01) and significant decreased nitrite (P < 0.05) levels compared with controls. Subdividing of the migraine study group into MA and MoA revealed that MoA showed increased nitrate levels and decreased nitrite levels similar to MA, although the differences did not reach statistical significance in this subgroup. Analysis of NOX, as the total amount of nitrate and nitrite, showed a significantly higher level in migraineurs (P < 0.01), which was significant in the MA group but not in the MoA group. Determination of nitrosylated proteins revealed an increase in migraineurs, both in MA and MoA, which did not reach significance. Amino acid analysis of arginine and citrulline showed no significant differences in any group. Expression analysis of eNOS and iNOS also showed no significant differences between the study groups. Analysis of the NOS inhibitor ADMA and its biological inactive stereo isomer SDMA revealed no differences in any group.
Analysis of the glucose metabolism revealed significant alterations between migraineurs and healthy controls. Migraineurs showed highly significant increased insulin levels (P < 0.001), which were significant in the MA (P < 0.001) and MoA (P < 0.05) subgroups. Glucose levels were decreased in migraineurs (P < 0.05), which was not statistically significant when subdividing into MA and MoA. Calculated HOMA indices were significantly increased (P < 0.01) in migraineurs compared with healthy controls. Subdividing into MA and MoA showed that the increase in HOMA indices remained highly significant in the MA group (P < 0.001) but not in the MoA group.
Quartiles of Homeostasis Model Assessment (HOMA) indices and odds of migraine

P = 0.003 Model is conditioned on HOMA indices quartiles.
Linear regression analysis of Homeostasis Model Assessment indices and the nitric oxide pathway

R, Pearson correlation coefficient; P, univariate
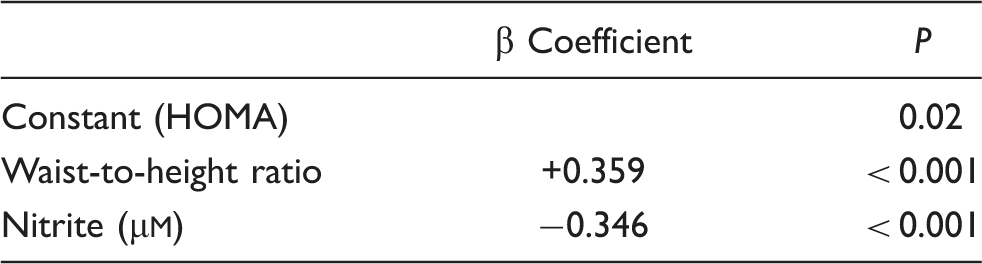
Multiple stepwise linear regression analysis to evaluate correlations of Homeostasis Model Assessment (HOMA)
Discussion
In the present study, we investigated the insulin and glucose metabolism in non-obese migraineurs in the headache-free period and its associations with the NO pathway.
Analysis of the NO pathway in non-obese migraineurs revealed significant increased nitrate and decreased nitrite levels. The increase of nitrate and accompanying decrease of nitrite is explained by interactions of NO with free radical oxygen species (ROS). Nitrate is built from NO by interaction with ROS, especially peroxynitrite. Nitrite is built from NO by interaction with free nitrogen species, especially via nitrogen trioxide. The amount of nitrosylated proteins was also increased in migraineurs, but because of the extremely high spreading of this parameter, the difference from controls was not significant. Further analysis of metabolites of the NO pathway, like arginine, citrulline, the expression of eNOS and iNOS or ADMA and SDMA levels showed no significant alterations in migraineurs compared with healthy controls. Taken together, our results show that migraineurs suffer under NO stress in the headache-free period. The results of the present study are in line with previous studies where NO has been implicated in migraine attacks (8,11,20–22). We currently showed that migraineurs suffer under increased NO stress during the headache-free period, which is associated with a 3.6-fold higher risk for migraine and may have a causative role in the pathophysiology of migraine (12).
We next investigated the insulin and glucose metabolism in migraineurs. We found highly significantly increased fasting insulin in migraineurs compared with healthy controls. Subdividing the study group into MA and MoA revealed that the increased insulin levels were significant both in the MA and MoA group. We next calculated HOMA indices as a combined parameter of fasting glucose and insulin levels. HOMA indices were significantly increased in migraineurs. The increased HOMA index of 1.80 ± 1.56 in migraineurs indicated a mild insulin resistance status. To determine the impact of elevated HOMA indices on migraine we performed logistic regression, revealing an OR of 5.67 for migraine, when comparing the lowest with the highest quartile of HOMA. Taking all data together, we show here that non-obese migraine patients suffer from hyperinsulinaemia. Our data suggest further that the increased HOMA index is a risk factor for migraine.
Hyperinsulinaemia and insulin resistance have been associated with various pathophysiological disorders, such as atherosclerosis, stroke, hypertension and obesity (4,5,23,24). Impaired insulin sensitivity in migraine patients has already been suggested by two previous studies (1,2). Our results are in line with these studies and further support the hypothesis of Bic et al. (25) that insulin resistance may be an underlying pathophysiological factor in the development of migraine.
To determine the underlying pathophysiological mechanisms, we investigated interactions of insulin metabolism with the NO pathway. Linear regression analysis between HOMA and parameters of the NO pathway revealed highly significant correlations between HOMA and the stable end-products of NO, nitrate and nitrite. We further found a correlation between HOMA and the amino acid citrulline. Subsequent multiple modelling revealed that waist-to-height ratio and nitrite turned out as the strongest correlation parameters of HOMA. Lack of correlations between HOMA and patients' annual frequency of attacks as well as the time point of patients' last attack suggests that increased HOMA indices in migraineurs are continuous, sustained and independent of migraine attacks. Taken together, beyond the known interaction partners of insulin and glucose metabolism like age, BMI and BP, we identified correlations between insulin metabolism and the NO pathway.
We have shown here that non-obese migraine patients suffer under hyperinsulinaemia and that hyperinsulinaemia is associated with a 5.67-fold higher risk for migraine. We further found a novel mechanism in the pathophysiology of migraine between hyperinsulinaemia and the NO pathway. These findings may be of clinical relevance, as NO inhibitors are discussed as therapeutics in migraine treatment, both in preventive and acute migraine therapy.
Footnotes
Competing interests
None to declare.
Acknowledgement
We gratefully acknowledge the work of Mrs Martina Waltenstorfer.
