Abstract
Nitric oxide (NO) has been implicated in migraine attacks, but the role of NO in migraine remains unclear. We here hypothesize that increased NO in the headache-free period is associated with migraine. One hundred and thirty probands participated in this study. Various parameters of the NO pathway, such as nitrate, nitrite, arginine, citrulline, nitrosylated proteins, asymmetric dimethylarginine, symmetrical dimethylarginine, expression of endothelial nitric oxide synthase (eNOS) and inducible nitric oxide synthase and two polymorphisms of eNOS were investigated. We found significant increased nitrate and decreased nitrite levels in migraineurs in the headache-free period. Nitrate and nitrite levels showed a significant inverse correlation. Logistic regression revealed an odds ratio of 3.6 for migraine. Other parameters of the NO pathway were neither altered in migraineurs nor correlated with nitrate. We show here that migraine patients suffer under sustained increased nitrosative stress in the headache-free period, which is associated with a 3.6-fold higher risk for migraine.
Keywords
Introduction
Nitric oxide (NO) is synthesized from the amino acid arginine by the enzyme family of nitric oxide synthases (NOS) which consists of three isoforms, including endothelial NOS (eNOS), inducible NOS (iNOS) and neuronal NOS (nNOS). NO is a reactive labile molecule and rapidly oxidized to its stable end-products nitrate and nitrite. NO has been implicated in the pathophysiology of migraine on the basis of a wide variety of approaches, including several clinical studies showing increased NO levels during migraine attacks (1–3). Therefore it is hypothesized that NO plays a critical and causative role in the pathophysiology of migraine by triggering migraine attacks, as currently reviewed in detail by Olesen (4). However, the pathophysiological mechanisms of NO in migraine remain unclear. The role of NO in the headache-free period, especially, is in part controversial. The role of the NO pathway in the headache-free period is of particular interest, when hypothesizing that the liability to migraine attacks is triggered by NO. Further, two functional polymorphisms in the eNOS gene are hypothesized to be involved in migraine (5–7). A point mutation of guanine to thymine at nucleotide 894 in exon 7 of the eNOS gene (dbSNP number rs1799983) and a substitution in the promoter region at nucleotide -786 (dbSNP number rs1800779) have been associated with reduced basal NO production. The impact of these polymorphisms on migraine is controversial (8–10). A further parameter involved in the NO pathway is the NOS inhibitor asymmetric dimethylarginine (ADMA) (11). To our knowledge, the role of ADMA in migraine has not been investigated to date. In general, the impact of aura symptoms on the NO pathway is also not yet clear. While some studies suggest an influence of aura symptoms on the NO pathway during migraine attacks, others have found no such association (4).
Taking all data together, there is substantial evidence for a causative role of NO in the pathophysiology of migraine. However, the exact mechanisms as well as the impact of the NO pathway in migraine are not yet clear. The role of NO in the headache-free period remains especially elusive. We hypothesize that beyond triggering migraine attacks, NO is already increased in the headache-free period and associated with migraine. Therefore, the aim of the present study was a comprehensive detailed examination of the NO pathway in the headache-free period to prove the hypothesis that increased NO in the headache-free period is associated with migraine. This was achieved by investigating various parameters of the NO pathway in migraineurs with and without aura. In detail, we investigated the amino acids of the NO pathway arginine and citrulline and the stable end-products of NO, nitrite, nitrate and nitrosylated proteins. We also investigated the NOS inhibitor ADMA and the expression of eNOS and iNOS as well as two genetic polymorphisms of eNOS.
Methods
Patients
Baseline characteristics of migraine patients and healthy controls

Results are expressed as mean ±
MoA, migraineurs without aura; MA, migraineurs with aura; eNOS, endothelial nitric oxide synthase; iNOS, inducible nitric oxide synthase; ADMA, asymmetric dimethylarginine; SDMA, symmetrical dimethylarginine.
Blood collection
Blood collection was performed after written informed consent was given. All probands were free of common infectious diseases and were not under medication. The median time period between the last attack and blood collection was 8 days (mean 19 ± 26 days). Blood was obtained after an overnight fast by venous puncture, immediately centrifuged at ambient temperature, and serum was stored at −80 °C until analysis. Blood collection for gene expression analysis was performed with the PAXgene System (PreAnalytiX, Hombrechtikon, Switzerland), as recommended by the manufacturer. Whole blood samples were collected and stored at −80 °C for single nucleotide polymorphism analysis.
Laboratory procedures
Serum nitrate and nitrite were determined with the NO Quantitation Kit from Active Motif (Rixensart, Belgium), as recommended by the manufacturer. Serum arginine and citrulline levels were determined by triple quadrupole mass spectrometry (Applied Biosystems API 2000 LC/MS/MS system; Vienna, Austria) with the EZ:Faast Amino Acid Kit from Phenomenex (Aschaffenburg, Germany) as previously described (13). Serum nitrosylated proteins were determined with the NWLSS Nitrotyrosine ELISA from Northwest Life Science Specialties (Vancouver, Canada). Serum ADMA as well as symmetrical dimethylarginine (SDMA) levels were measured by high-performance liquid chromatography and precolumn derivatization with o-phthaldialdehyde as described previously (14). Genotypes were analysed using TaqMan™ fluorogenic 5′-exonuclease assays. Primer and probe sets were designed and manufactured using Applied Biosystems ‘Assay-by-Design’ custom service (Applied Biosystems). End-point fluorescence was measured in a POLARstar plate reader (BMG Labtech, Durham, NC, USA). Fluorescence data were exported into Excel format and analysed as scatter plot. Expression analysis of eNOS and iNOS from whole blood cells was performed with the PAXgene Blood RNA Kit from PreAnalytiX. Isolated RNA was transcribed into a cDNA library with the cDNA Archive Kit from Applied Biosystems. Quantitative real-time polymerase chain reaction was performed with the Taqman Universal Master Mix and gene expression assays on demand for eNOS, iNOS and glyceraldehyde 3-phosphate dehydrogenase as a house-keeping gene for normalization on a AB7900 (Applied Biosystems). Calculation of eNOS and iNOS expression was performed by using the delta crossing point method (15).
Statistical analysis
Data are presented as means ± standard deviations. Continuous variables were compared using Students
Results
Baseline characteristics
Analysis of the NO pathway during the headache-free period in migraineurs revealed significant alterations compared with healthy controls (Table 1). Migraine patients showed significant increased nitrate (
Correlations of the NO pathway
To determine possible interactions of the NO pathway, we performed linear regression analysis between nitrate, nitrite and NOX with parameters of the NO pathway. We found a highly significant negative correlation between nitrate and nitrite ( Correlation between nitrate and nitrite in healthy controls (○), migraineurs without aura (×) and migraineurs with aura (▵). 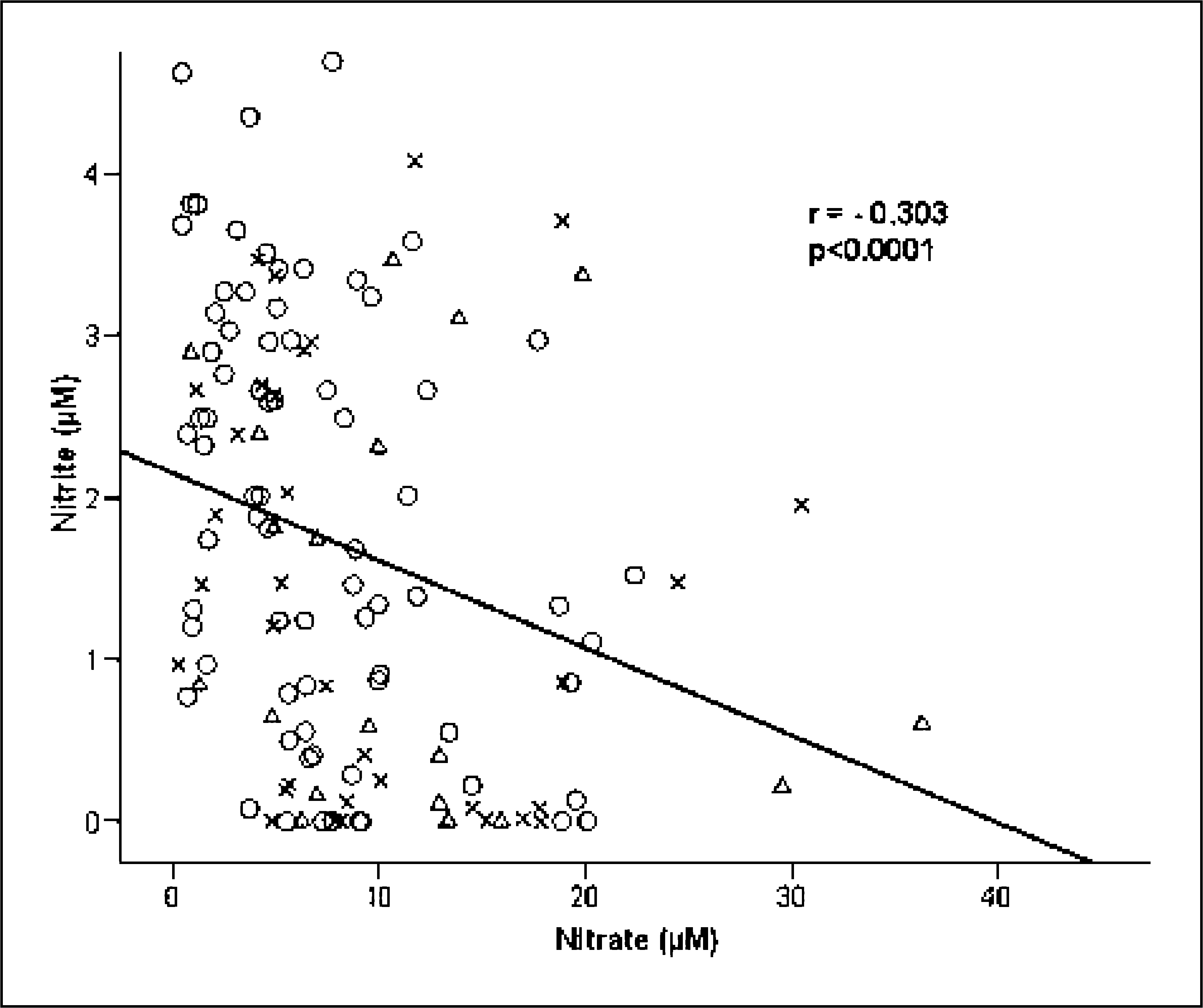
Plasma nitrate level and risk of migraine
Logistic regression analysis of plasma nitrate and odds of migraine

*
Model 1 is unadjusted. Model 2 is adjusted for age and gender.
Association of endothelial nitric oxide synthase polymorphisms
Genotype distribution and allele frequencies of endothelial nitric oxide synthase rs1799983 and rs1800779 among clinical groups

All comparisons were not significant.
MoA, migraineurs without aura; MA, migraineurs with aura.
Comparison of endothelial nitric oxide synthase (eNOS) polymorphisms with parameters of the nitric oxide pathway in healthy controls and migraineurs

All comparisons were not significant (
Discussion
In the present study we investigated the role of the NO pathway in patients with MA and MoA in the headache-free period as we hypothesized that migraineurs are characterized by increased NO levels irrespective of attacks. We determined nitrate, nitrite and nitrosylated proteins as the stable end-products of NO and, further, the metabolites of the NO pathway arginine and citrulline, the expression of eNOS and iNOS as well as two eNOS polymorphisms. Furthermore, we investigated the NOS inhibitor ADMA. Our results clearly show that migraineurs exhibit increased serum nitrate and decreased serum nitrite levels in the headache-free period. The increase of nitrate and accompanying decrease of nitrite is strengthened by the finding that nitrate and nitrite showed a significant inverse correlation. Nitrate is built from NO by interaction with free radical oxygen species, especially via peroxynitrite. Nitrite is built from NO by interaction with free nitrogen species, especially via nitrogen trioxide. Therefore it is clear that the combination of increased nitrate levels and decreased nitrite levels can be explained only by additional oxidative stress in migraineurs. Our conclusion is in line with several studies indicating that oxidative stress is involved in the pathophysiology of migraine (16,17). Interestingly, a study by Tuncel et al. has shown that oxidative stress in migraineurs is more pronounced in patients with MA, as we found higher NO levels in patients with aura (18). Furthermore, we also found a significant increase in the total amount of serum NOX in migraineurs. The amount of nitrosylated proteins was also increased in migraineurs, although, because of the extremely high spread of this parameter, the difference from controls was not significant. We next investigated if the observed NO levels in the headache-free period were correlated with the time interval from the patients' last migraine attack. We found no correlation between the NO pathway and patients' last migraine attack or the annual frequency of attacks. These results indicate that migraineurs suffer under sustained nitrosative stress in the headache-free period. We further analysed the possible effects of NO on symptoms of aura, and found significantly increased nitrosative stress in patients with aura. In the MoA group NO was increased, but not statistically significantly so. The impact of aura symptoms on the pathophysiology of migraine is controversial (19). Whereas some studies found clinical and pathophysiological differences between MA and MoA patients, others did not (4). As currently outlined by Purdy, the latest research suggests that migraine with and without aura may share the same underlying pathogenic mechanisms (20). To determine the impact of NO on migraine we performed logistic regression, revealing an OR of 3.6 for migraine, when comparing the lowest with the highest quartile of nitrate. As we found a significant OR only in the highest quartile, one may speculate that the impact of increased NO on migraine does not follow a linear relation and only high levels of NO are associated with migraine. Taking all data together, we show here that increased nitrosative stress in the headache-free period is a risk factor for migraine.
A study by Yilmaz et al. has shown a slight but not significant increase of NO in isolated platelets of migraineurs in the headache-free period (17). Taffi et al. found significant increased peroxynitrite, a potent oxidant in platelets of patients with MoA during headache-free periods (16). In contrast, Shimomura et al. showed that NO activity in platelets was decreased in the headache-free period in patients with MA and MoA (3). In contrast, Shukla et al. found no changes in polymorphonuclear leucocyte, platelet and plasma nitrite levels in patients with migraine (21). In summary, the reasons for these different findings may be methodological, at least in part. Although it has been shown that platelets play an important role in the pathophysiology of migraine and are proposed as a promising tool for studying NO variations in migraine (22,23), other sources of NO may also play an essential role. Furthermore, our analysis of metabolites of the NO pathway, such as arginine, citrulline or the expression of eNOS and iNOS, shows no significant alterations in migraineurs compared with healthy controls. Our results indicate that the observed nitrosative stress in migraineurs is rather caused by increased enzyme activity of NOS than by a dysregulation of the NO pathway. Our findings are in line with those of D'Andrea et al., who showed that the plasma levels of arginine were in the same range in migraineurs and controls, although platelet arginine levels were significantly increased in patients with MA (1). To our knowledge, parameters of the NO pathway such as citrulline and the expression of eNOS and iNOS in migraine have not been investigated to date. We next examined the role of the NOS inhibitor ADMA in migraine, to our knowledge also not investigated in migraineurs to date. Proteolysis of methylated nuclear proteins containing arginine results in the formation of ADMA as well as of SDMA, which is the biologically inactive stereo isomer of ADMA (11). ADMA is cleared metabolically by the action of dimethylarginine dimethylaminohydrolase (DDAH). It is suggested that the activity of DDAH is reduced by risk factors such as hypercholesterolaemia, hypertension, diabetes and hyperhomocysteinaemia, leading to accumulation of ADMA (24). Elevated serum levels of ADMA have been found to be associated with a wide variety of pathophysiological mechanisms, including atherogenesis, impaired renal function, hypercholesterolaemia, hypertriglyceridaemia, insulin resistance, diabetes mellitus, hyperhomocysteinaemia and hypertension (25–29). We did not find any differences in ADMA or SDMA levels in migraineurs compared with healthy controls. We also found no correlations between ADMA and parameters of the NO pathway. We therefore conclude that ADMA is not involved in the pathophysiology of migraine.
Finally, we also investigated two polymorphisms in the eNOS gene hypothesized to play a role in migraine, as Borroni et al. found in a pilot study a threefold increased risk for MA compared with MoA (8). It has been shown that both polymorphisms are associated with reduced basal NO production in regard to hyperhomocysteinaemia, predisposition to acute coronary syndromes and hypertension (5–7). We did not find any significant association of the eNOS polymorphisms rs1799983 or rs1800779 in patients with MoA or MA. Our findings are in the line with a current study by Toriello et al., who also found no association with migraine in a much larger cohort than ours (10). Furthermore, we found no correlation between these polymorphisms and variations in the serum levels of NO metabolites or the expression of eNOS in migraine patients and in healthy controls. Taken together, our results provide no evidence for an involvement of these two eNOS polymorphisms in migraine.
In conclusion, we have shown here that migraine patients suffer under sustained increased nitrosative stress in the headache-free period. Increased NO is associated with a 3.6-fold higher risk for migraine. Our results suggest that NO would represent a suitable therapeutic target for a preventive migraine therapy.
Footnotes
Competing interests
None to declare.
Acknowledgements
We gratefully acknowledge the work of Mrs Sabine Pailer, Mrs Martina Waltenstorfer and Mr Firtz Wabenegger. The study was supported by a grant of the Styrian Government.
