Abstract
Cluster headache (CH) is a debilitating neurovascular condition characterized by severe unilateral periorbital head pain. Deep brain stimulation of the posterior hypothalamus has shown potential in alleviating CH in its most severe, chronic form. During surgical implantation of stimulating macroelectrodes for cluster head pain, one of our patients suffered a CH attack. During the attack local field potentials displayed a significant increase in power of approximately 20 Hz. To the authors' knowledge, this is the first recorded account of neuronal activity observed during a cluster attack. Our results both support and extend the current literature, which has long implicated hypothalamic activation as key to CH generation, predominantly through indirect haemodynamic neuroimaging techniques. Our findings reveal a potential locus in CH neurogenesis and a potential rationale for efficacious stimulator titration.
Keywords
Introduction
The group of trigeminal autonomic cephalalgias (TACs) comprise some of the most severe forms of primary headache known to man (1). As the name suggests, their pathophysiology is considered distinct from that of other primary headaches such as migraine. TACs are characterized by severe short-lived unilateral periorbital head pain, often observed with craniofacial autonomic symptoms (2). Specific syndromes include cluster headache (CH), paroxysmal hemicranias and short-lasting unilateral neuralgiform headache attacks with conjunctival injection and tearing (SUNCT).
CH is a particularly severe form of TAC, characterized by spontaneous unilateral periorbital head pain occurring in cycles that appear to be dependent both on the circadian rhythm and seasonal variations (3). The pathophysiology of CH, as with the more general class of TACs, remains relatively unknown. However, recent neuroimaging studies are finally beginning to elucidate the central mechanisms involved, providing a rationale for the development of new therapeutic strategies (4).
Current evidence points towards the posterior hypothalamic region as a ‘cluster headache generator’ (1, 3–6). This has led to surgical targeting of the region using stereotactic deep brain stimulation (DBS) for the relief of intractable CH. High-frequency stimulation in drug-resistant patient groups has proved highly successful in preventing the headaches (4, 7–9). Although implicated in the pathogenesis of CH attacks, the precise nature of hypothalamic involvement remains unclear. While positron emission tomography (1, 6) and functional magnetic resonance imaging (MRI) (10, 11) provide good evidence for increased regional activation, such approaches are incapable of revealing the rich temporal dynamics that underlie this activity. DBS, along with offering therapeutic benefit to patients, provides a unique opportunity to observe local field activity within the vicinity of the target nucleus. While recording intra-operative local field potentials (LFPs), one patient suffered a CH attack. This is, to the authors' knowledge, the first recorded instance of hypothalamic LFP activity captured during an attack. Analysis of the data reveals a characteristic structure dissimilar to that obtained in a CH patient in a headache-free state. These observations provide evidence for a specific neural rhythm activated at the onset of cluster attack that may prove useful in efficacious stimulator titration.
Methods
Patients underwent stereotactic implantation of DBS macroelectrodes with leads externalized for a period of 7 days to assess the efficacy of the procedure. LFPs were obtained from two patients during the surgical procedure and postoperatively with informed consent and under local ethics committee approval. DBS consisted of quadripolar stimulating macroelectrodes (Medtronics© 3387 electrodes; Medtronics, Minneapolis, MN, USA) inserted via a stereotactic surgical procedure. Macroelectrode implantation was performed in accordance with the published coordinates of (12, 13), specifically, 6 mm posterior, 2 mm lateral and 8 mm inferior to the mid-commissural point, ipsilateral to pain. This target has been described as providing ‘remarkable’ efficacy in previous clinical trials (13). Targeting was achieved through fused computed tomography/MRI images using Radionics Image Fusion™ and Stereoplan™.
A synopsis of patient history including drug regime and titrated stimulation parameters is provided in Table 1. Both patients were diagnosed with intractable chronic CH (International Headache Society code 3.1.2). Patient 1 responded well to stimulation about the mid-contact pair, with patient 2 gleaning greatest benefit from superior contacts. Both patients demonstrated early intolerance to high-voltage stimulation, reporting light-headedness and diplopia approaching 2 V. During the 7-day externalization period, there were no recurrences of CH attacks. Following surgery, patients were removed from any medication relating to their CH. Long-term patient response was more variable. Patient 1 has obtained almost complete headache relief, reporting only infrequent attacks (total seven sumatriptan injections in the 11 months following surgery). Patient 2 has decreased attack frequency from daily to weekly with massively reduced severity (infrequent sumatriptan injections).
Stimulation settings display voltage, frequency, pulse width and active contact

Stimulation was continuous and bipolar.
CH, cluster headache.
LFPs were obtained from the two externalized patients intra-operatively during macroelectrode implantation. Field potentials produced by a population of cells indirectly reflect changes in synchronous neuronal potentials (14). LFPs were recorded using a bipolar configuration from the four adjacent contacts [0(inferior)-3(superior)] of the stimulating macroelectrode (i.e. 0–1, 1–2, 2–3), with a common obtained via disposable Ag/AgCl electromyographic electrodes placed over the back of the neck at approximately the C4 level. A bipolar configuration provides powerful common-mode-rejection of far field activity and volume currents (15).
During surgery patient 1 reported suffering an episode of CH, complete with agitation, autonomic symptoms and head pain comparable to their regular attacks. Attack onset followed immediately from electrode placement. We therefore cannot discount the possibility that the attack may have been triggered by the neural shock of electrode implantation. The attack persisted throughout a 5-min recording period. The cephalgia also gave rise to a mild panic attack, an effect occasionally observed during hypothalamic DBS implantation (16). No further attacks were reported while externalized. Postoperative imaging revealed successful stereotactic targeting of the posterior hypothalamus (Fig. 1).

T1-weighted 1.5T preoperative magnetic resonance imaging of patient 1 overlaid with postoperative CT highlighting electrode placement within the left hemisphere. Enlargement shown in bottom-right quadrant. Presented is the tip (inferior aspect) of the electrode as it begins to ascend through the targeting trajectory.
LFP signals were first amplified (× 10 000) using CED 1902 amplifiers (Cambridge Electronic Design, Cambridge, UK), filtered online (separate 0.5-Hz high-pass and 1000-Hz low-pass second-order Butterworth Infinite Impulse Response filters), then digitized at a sample rate of 2.5 kHz using a CED Micro1401. Data were recorded for offline analysis using CED Spike2 software and analysed using proprietary software developed using Matlab (Mathworks Inc., Natik, MA, USA). Statistical analysis was performed in
To assess statistical significance, independent-samples t-tests were performed between conditions over log-transformed spectra within the frequency bands of interest (e.g. 16–22 Hz). Log-transform spectra are known to provide a good approximation to normality (18). Trends may be identified between patients by applying log-ratio measures to the estimated spectra. Log-ratios provide a quantitative linear assessment of spectral differences between conditions. CIs were determined through jackknife numerical procedures (see (20) for further details).
Spectrograms were constructed via a Hermite function analysis (21). Hermite functions may be considered a natural extension of Thomson's multitaper approach to the time–frequency plane and, as such, benefit from superior leakage, bias and variance properties to their sliding window Short-Time Fourier Transform counterpart (21). Hermite functions were constructed using a 95% energy concentration cut-off (in analogy with (22)) under a unitless time–frequency area A = 6, resulting in K = 2 taper functions. Confidence limits were placed under an effective number of degrees-of-freedom argument (see (22) for details).
Results
Of greatest interest were localized power differences observed between adjacent electrode contacts. These represent focal activity within the vicinity of the contact pairs. Figure 2 displays results from a spectral analysis of patient 1 during their intra-operative CH attack. There is a highly prominent peak in the LFP spectra around 16–22 Hz, localized to central and superficial LFP contacts (1–2 and 2–3, respectively). Thus, at the onset of cluster attack a spatially localized increase in relative LFP power was seen in patient 1. Subsequently, it has become clear that bipolar stimulation between contacts 1–2 and 2–3 were most effective at relieving the patient's CH pain.
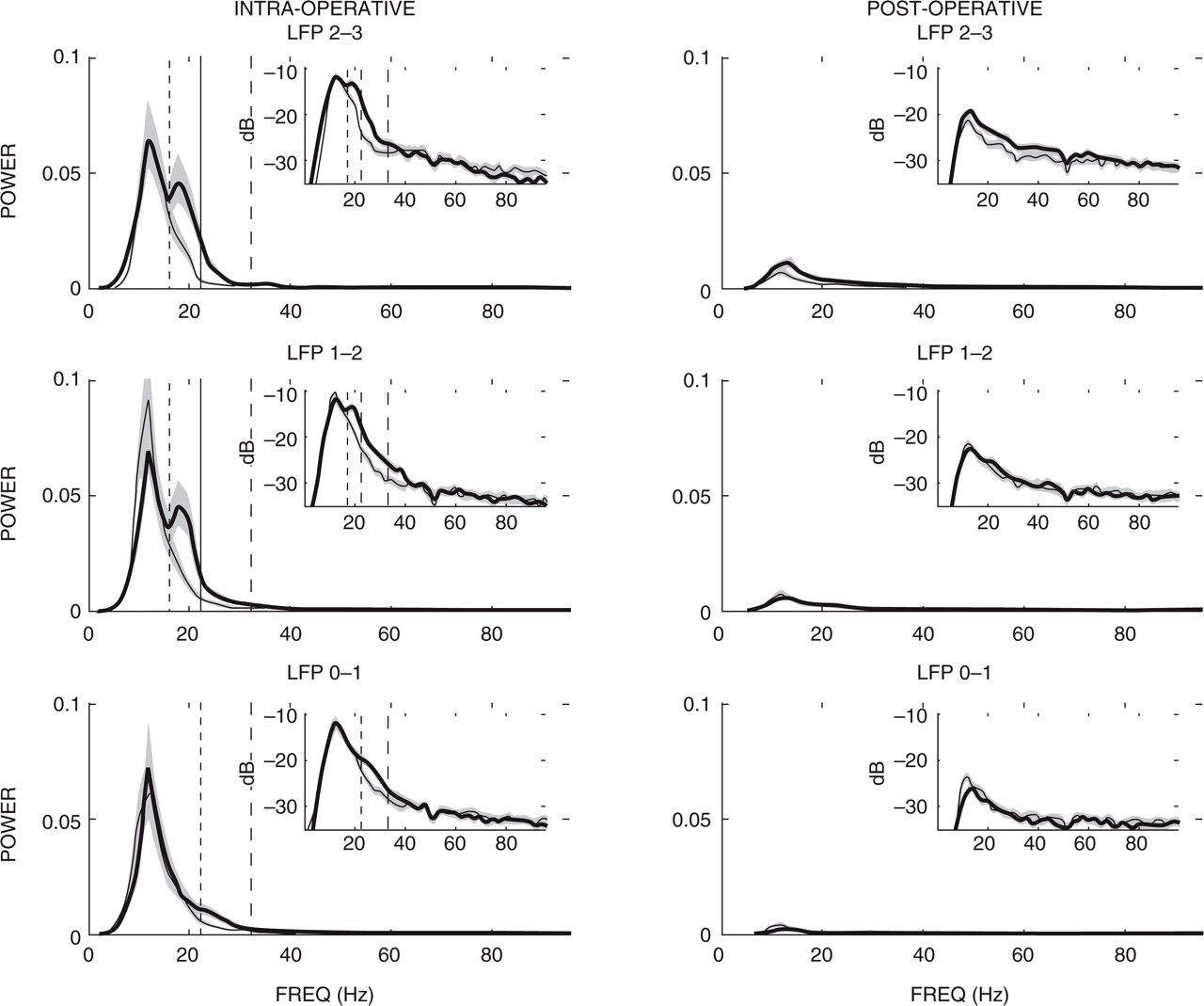
Mean power spectra displayed for intra-operative (left column) and postoperative (right column) local field potential (LFP) recordings. Bold trace denotes patient 1 (who suffered an intra-operative cluster headache attack) with the thin trace patient 2. Shaded regions denote the 95% confidence intervals. Dashed vertical lines highlight the 16–22 Hz and 22–32 Hz frequency bands. Inlay figures depict the same spectra presented on a decibel scale to accentuate global structure. Note that normalization of the power spectra results in a unitless ratio measure, reducing scales to relative differences.
Frequency-localized power within the 16–22 Hz range failed to prove statistically significant under an independent-samples t-test for inferior contacts 0–1 (t 120 = 1.77; NS). Data corresponding to contacts 1–2 (t 120 = 7.62) and 2–3 (t 120 = 7.59), however, proved highly significant at the P < 0.001 level. No other significant differences were observed outside of the 16–40 Hz range. Likewise, there were no obvious deviations in postoperative spectra between patients, a result illustrated in Fig. 2.
Other changes between intra- and postoperative spectra were not spatially localized (i.e. common across contacts). A general sub-30-Hz relative increase in power was observed intra-operatively, peaking around 12 Hz. Since this change is consistent across subjects, it probably reflects a surgical effect. Investigation of this phenomenon would prove useful in further eliciting the mechanism of CH.
There were also non-localized changes in relative power observed between intra- and postoperative spectra that were dissimilar between patients. In this instance a common 22–32-Hz increase may be observed between intra- and postoperative spectra for subject 1, significantly higher than for the same frequency band observed in subject 2. Since this feature is not spatially localized, it probably reflects the result of natural intersubject variability, possibly due to variations in surgical response.
Log-ratio measurements permit the evaluation of trends between patients, depicted for the intra-operative condition in Fig. 3. Figure 3 demonstrates a clear spatial and frequency-localized increase in power observed in patient 1 relative to patient 2. This becomes clear when we examine results from LFPs 2–3 and 1–2, where a highly significant deviation from 0 is observed over a 16–32-Hz band, possibly extending to 40 Hz in LFP 1–2. This range is in agreement with the 16–22-Hz and 22–32-Hz activity previously described (Fig. 2). Likewise, LFP 0–1 demonstrates a significant deviation from 0 only across the 22–32-Hz range. These results support our previous categorization of spatially localized (16–22 Hz) and broader (22–32 Hz) differences between patients. Log-ratio measures are also provided for the postoperative condition, where no specific component deviations from 0 are observed across the LFPs. Thus, the only significant differences that can be observed are obtained intra-operatively during a typical episodic CH attack.

Boxplot diagrams are provided in the first column for log10 spectra corresponding to patient 1 (P1) and patient 2 (P2), intra- and postoperatively. Boxplots were constructed about the peak spectral difference, 20 Hz in this case, given a bandwidth of 1 Hz. The second column displays log-ratio measures of spectra between patients for the intra-operative condition over each of the three bipolar recording configurations. The third column displays similar log-ratio measures for the postoperative condition. Log-ratio 95% confidence limits placed by jackknife numerical procedures. LFP, local field potential.
Finally, spectrograms were constructed using a Hermite function analysis (see Methods) for both patients over intra-operative conditions (see Fig. 4). Spectrograms highlight both the high-power component around 10 Hz common across patients, and a marked increase in 16–22 Hz activity in patient 1, localized to the mid-superior contacts. In both patients the spectra appear tonic, with no modulation in activity observed over the recording period. Indeed, periodic examination of the signal reveals no significant deviation in power content from either patient over the available recording interval.

Spectrogram images corresponding to 40 s of intra-operative data from patient 1 (left column) and patient 2 (right column) local field potential (LFP) recordings. Rows correspond (top-to-bottom) to LFP 2–3, LFP 1–2 and LFP 0–1, respectively. Spectra presented on a decibel scale to elucidate global structure.
Discussion
We present initial follow-up data from two patients undergoing DBS for chronic CH. Our data provide support for targeting the posterior hypothalamus (as (13)) to treat refractory chronic CH. Both patients reported a substantial reduction in headache occurrence with infrequent prophylactic treatment. We also report a distinct 16–22-Hz peak in neural activity observed during an episode of CH, spatially localized in the vicinity of those electrodes that provided the most effective therapeutic stimulation. This observation suggests that isolation of a pathological rhythm in LFPs could guide efficacious stimulator titration.
The CH attack observed in patient 1 occurred outside of their regular cluster pattern. It was therefore probably induced due to the neural shock of surgically implanting the stimulating macroelectrode. This may also contribute to our failure to observe a clear onset of attack, since LFP recordings began post implantation. The ability to induce typical CH through hypothalamic stimulation (a result confirmed by (23)) supports our understanding of the hypothalamus as a fundamental trigger zone in CH attacks.
The presented data represent a preliminary investigation into the precise neural mechanisms underlying hypothalamic involvement in CH. We hypothesize that high-frequency stimulation of the target zone disrupts the synchronous build-up of pathological oscillations that are believed to result in hypothalamic triggering of the trigeminoautonomic reflex. A separate CH attack has been reported in the literature as a direct result of stimulation during DBS surgery (23). In that study, the attack was induced through test stimulation at the target zone (130 Hz), although quickly aborted by high-flow oxygen.
To the authors' knowledge, this is the first published account of LFPs recorded during a cluster attack from the posterior region of the hypothalamus. A series of recent articles, however, have reported microelectrode recordings observed during DBS surgery for chronic CH from a distinct neurosurgical target (23, 24). In (23), single unit activity was observed, displaying a mean discharge rate of 17 Hz (range 13–35 Hz). The observed activity showed no change in response to sensory, motor, autonomic or emotional stimuli. The hypothalamic region targeted in these studies corresponds to a set of revised coordinates published in (25), specifically 2 mm lateral, 3 mm posterior and 5 mm inferior to the mid-commissural point, which are distinct from our own.
Bartsch et al.'s observation of tonic 13–35 Hz activity, peaking at 17 Hz (23), bears striking resemblance to our own observations of raised 16–32-Hz LFP power during a cluster attack. Cordella et al. have also reported spontaneous firing rates in the vicinity of 24 Hz in three TAC patients (24). Neither study reports the low-frequency power observed intra-operatively. This may be due to dissimilarity in surgical procedure, a fundamental difference in the surgical target, or the intrinsic dissimilarities between LFPs and single-unit recordings. LFPs represent aggregate synaptic activity within the vicinity of the DBS macroelectrode, whereas microelectrodes typically represent the action potential firing of isolated neurons.
The identification of a specific neural rhythm during CH attacks provides the first direct support for hypothalamic involvement in CH, long suspected from clinical results and indirect haemodynamic imaging methods (6, 8, 9, 24, 26). The presence of a frequency-localized power modulation isolates a potential trigger mechanism in CH. Identifying the pathogenic origin of this rhythm may provide key insights into the complex mechanisms thought to underlie CH alongside other TACs.
