Abstract
We describe two patients with thunderclap headaches due to reversible cerebral vasoconstriction syndrome (RCVS). The first patient illustrates multilobar intracerebral haemorrhages as an under-appreciated feature of RCVS, and the second illustrates recurrent thunderclap headache (presumed recurrent RCVS) after a long interval of 4 years. These cases demonstrate the spectrum of presentation of RCVS, a clinically under-recognized condition.
Keywords
Introduction
Reversible cerebral vasoconstriction syndrome (RCVS) is an under-recognized cause of thunderclap headache. Recognizing it is important in its correct management and in identifying possible precipitants. We present two cases highlighting features of this condition. We also illustrate how complications may occur with initial misdiagnosis, and the late recurrence of thunderclap headache (presumed, but not proven, RCVS) in one case.
Patient 1
A 59-year-old woman presented thunderclap headache, described by the patient as if ‘hit by a bus’ graded 10 out of 10 on a notional analogue severity scale, vomiting and a distortion of vision on the left-hand side from headache onset. Five days earlier, she had had a similar episode that started with vomiting, immediately preceding a thunderclap headache, which partially recovered. There was no prior history of migraine or other thunderclap headache, and she was otherwise well. There was no history of vasoconstrictive drug exposure or use of cannabinoids.
On examination, she was normotensive. Her neurological and general examination was normal.
Cranial computed tomography (CT) on presentation (day 5 from first thunderclap headache and within 7 h of second episode) was normal. Cerebrospinal fluid (CSF) examination performed 22 h after second thunderclap, was normal (white blood cells < 4/mm3, red blood cells 9/mm3, glucose 3.2 mmol/l, protein 0.39 g/l) and negative for xanthochromia on spectophotometry. Day 6 after the first episode, magnetic resonance imaging (MRI), including magnetic resonance venography (MRV), showed a left occipito-parietal haemorrhage and right occipito-parietal oedema (Fig. 1). Apparent diffusion coefficient and diffusion-weighted imaging sequences were not performed to confirm infarction. There were clinical concerns with regard to possible cortical vein thrombosis as a differential, and she was anticoagulated with intravenous heparin and subsequently warfarin. MRI of the brain was repeated on day 14 due to persistent symptoms of headache and vomiting, showing left frontal and bilateral occipito-parietal haemorrhages and a right subdural haematoma (Fig. 2). MRV was repeated and considered normal. Review of initial MRV was now considered normal. Anticoagulation was stopped and headaches resolved after 22 days from first symptoms.
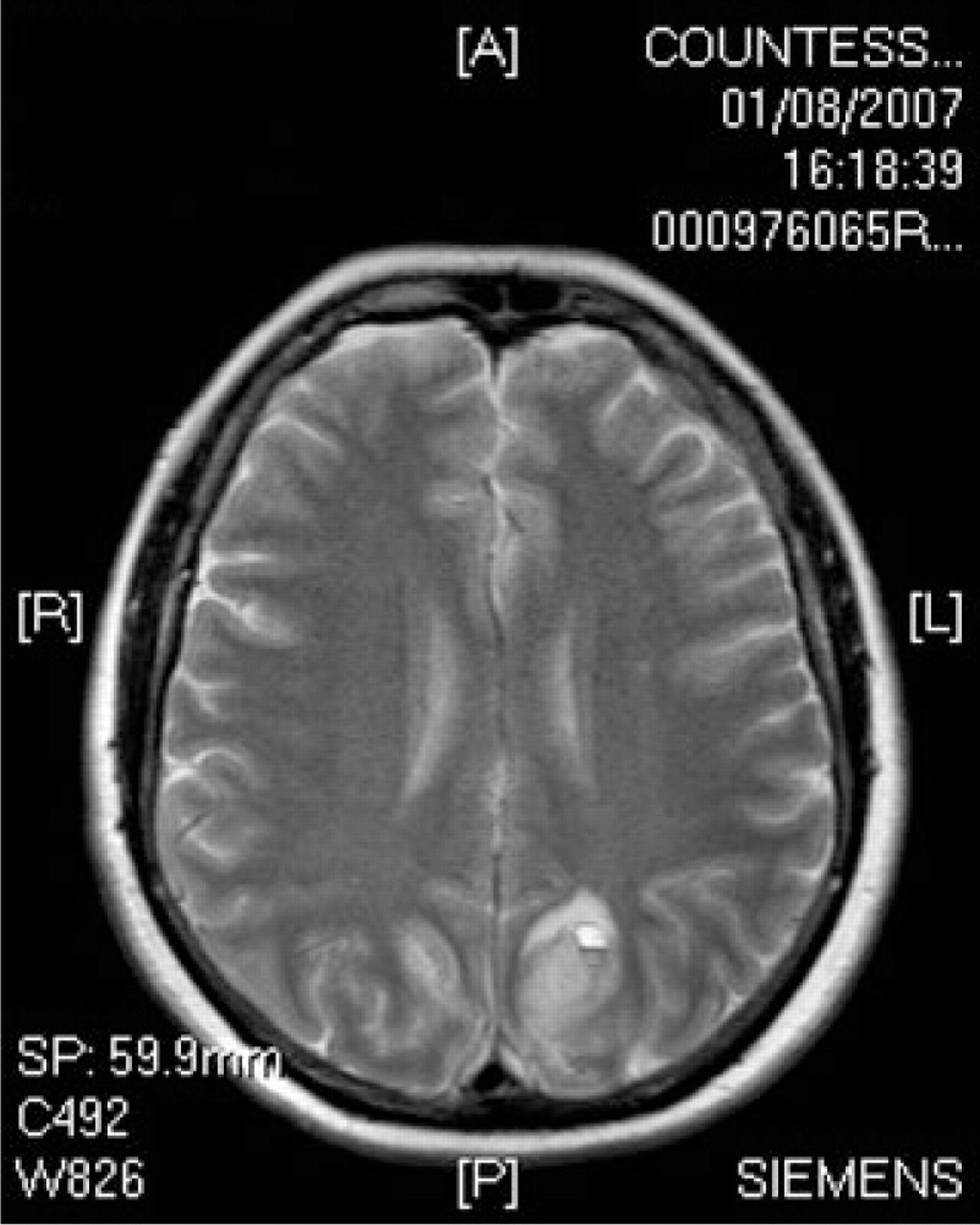
T2 axial magnetic resonance image demonstrating left occipito-parietal haemorrhage and right occipito-parietal oedema.

T2 axial magnetic resonance image demonstrating left frontal haemorrhage, bilateral occipito-parietal haemorrhage and a small right subdural haematoma.
Magnetic resonance angiography (MRA) on day 21 showed multiple areas of segmental narrowing and beading (Fig. 3). The multilobar intracranial haemorrhages were unchanged from day 14. Repeat MRA on day 29 showed significant improvement with spontaneous reversal of vasospasm, and correlated with clinical improvement. MRA at 2 months was normal. There was no residual neurological deficit.

Magnetic resonance angiogram (MIP) demonstrating arterial beading.
Patient 2
A 33-year-old woman with a life-long history of migraine without aura presented with her first episode of thunderclap headache (reaching immediate peak intensity graded 10 out of 10 on a notional analogue scale), nausea, vomiting, photosensitivity, left leg monoparesis and dysphasia. She was an occasional user of cannabis, and a small quantity of cannabis was used in the week preceding her symptoms.
On examination, she was normotensive and had a normal cardiorespiratory examination. Neurological examination showed left leg monoparesis with pyramidal distribution with no reflex changes and mild dysphasia.
Cranial CT (day 1) and MRI (day 3) were normal. Lumbar puncture was attempted unsuccessfully and complicated by the development of a lumbar epidural haematoma, confirmed on MRI. CT angiography on day 8 was non-diagnostic due to patient movement.
A formal catheter cerebral angiogram on day 10 showed widespread large and medium-sized cerebral arterial vasoconstriction and vasodilation (Fig. 4).

(a) Selective left carotid formal angiogram demonstrating multifocal anterior and middle cerebral arterial segmental vasoconstriction and vasodilation. (b) Selective vertebral formal angiogram showing segmental vasoconstriction and vasodilation of posterior circulation vessels including basilar and posterior cerebral arteries.
A full vasculitic screen was negative, including paraproteins, cryoglobulin, angiotensin-converting enzyme, antineutrophil cytoplasmic antibodies, dsDNA, extractable nuclear antigens and hepatitis serology. Her inflammatory markers were normal, but subsequently rose in response to the development of her epidural collection. Urine toxicology showed the presence of cannabinoids.
She was treated with nimodipine on arrival to the neurosciences centre (day 8) as per our subarachnoid haemorrhage protocol, which was stopped on day 29. On day 23, with the benefit of the angiogram results, she was started on 3 days of intravenous methylprednisolone (1 g/day) followed by a reducing course of oral prednisolone.
Repeat catheter angiography on day 27 was normal. She completely recovered clinically and was headache free by day 29.
Four years later she represented to her local hospital with a similar thunderclap headache with nausea and vomiting without neurological deficit. Cranial CT was normal. Lumbar puncture was refused by the patient. She settled with symptomatic treatment. The admitting medical team did not request early neurological input. There was no enquiry about potential vasoconstrictive drug exposure including cannabinoids and the patient has subsequently been uncontactable. Test for recurrence of RCVS were not performed during her admission.
Discussion
RCVS is characterized by reversible multifocal narrowing of the cerebral arteries heralded by sudden (thunderclap) severe headaches with or without associated neurological deficits (1–3). It has been described under various names, including Call–Fleming syndrome (1), benign angiopathy of the nervous system (4) and postpartum angiopathy (5, 6). It is an under-recognized disorder and may be underdiagnosed, as a recent prospective series reported 67 patients recruited in 3 years in a single institution (3). Another prospective series of 56 patients with recurrent thunderclap headaches reported 22 patients (39%) had RCVS out of 4200 patients seen in a headache clinic (7).
RCVS could be spontaneous or secondary to a number of factors, including postpartum and vasoactive substances (2, 3).
The use of cannabis in patient 2 may have been a trigger, although the patient was an occasional user and claimed to have consumed a small amount 1 week preceding her symptoms. Cannabis has been highlighted as one of the three most common vasoactive substances, alongside selective serotonin reuptake inhibitors (SSRI) and nasal decongestants (3). Interestingly, patient 2 had a history of overdoses with SSRI (up to 200 mg of fluoxetine), which did not cause thunderclap headaches. At the time of presentation with both episodes, she was no longer taking SSRI. Patient 2 therefore may have had secondary RCVS from cannabis use.
It is debatable whether RCVS in patient 1 was spontaneous or secondary RCVS. She started with vomiting immediately preceding her thunderclap headache. Vomiting is not a well-known precipitant of RCVS, but as physical exertion has been described as a trigger (7), it may be argued that vomiting was a trigger. She was on no other medications or vasoactive substances prior to onset of symptoms.
Patient 2 developed an epidural haematoma as a complication of failed lumbar puncture following presentation with thunderclap headache. Dural puncture causing intracranial hypotension has been described as a trigger for RCVS (8–10). However, our patient presented with thunderclap headache with neurological deficit prior to her lumbar puncture. We therefore surmise that her epidural haematoma is unlikely to have caused her RCVS. Furthermore, she did not give symptoms of a low pressure/posturally related headache post lumbar puncture.
Multilobar intracranial haemorrhages have been described with RCVS (11, 12). Multilobar intracranial haemorrhages found in a single patient may be unusual, as it was reported in only one of 89 patients in the updated prospective series presented at a recent stroke conference (13), although combinations of cortical subarachnoid haemorrhages and intracranial haemorrhages, as seen in patient 1, were reported in 8/89 of their patients (13). Haemorrhagic complications in RCVS occur earlier (within the first 3–10 days) compared with ischaemic events (approximately 12 days) from headache onset (3).
It could be argued that our patient developed multilobar haemorrhages due to a combination of the reversible arterial vasospasm and anticoagulation, as she first presented with a single-lobe haemorrhage plus infarction. The infarcted right occipito-parietal cortex would have been prone to haemorrhagic transformation when on anticoagulation. Furthermore, vasospasm could have resulted in ischaemia of intracranial vessel walls leading to necrosis, increased vascular permeability, perivascular microhaemorrhage and subsequent vessel rupture when perfusion pressure was restored (6).
Our first patient was initially misdiagnosed as having a cerebral venous thrombosis, which is increasingly recognized as a cause of thunderclap headache (14), with lobar intracranial haemorrhage resulting from venous back-pressure effect. MRV can be misleading, as transverse sinus flow gaps occur in up to 31% of normal patients, and may be mistaken for dural sinus thrombosis (15). The diagnostic accuracy of MRV may be improved with gadolinium-enhanced three-dimensional gradient echo sequences (16) (in comparison with two-dimensional time-of-flight sequences) and breath-holding manoeuvres (17). The gold standard is the combination of MRI (T1, T2, fluid-attenuated inversion recovery and T2∗ sequences) to visualize the thrombosed vessel and MRV to detect the non-visualization of the same vessel (18).
Recurrent thunderclap headaches over a few days to 2 weeks is the clinical hallmark of RCVS (3), as in our first patient with two episodes of thunderclap headache within 5 days of presentation. Less common are recurrent episodes of RCVS. Recurrences have been reported after an interval of up to 36 months, in a patient with RCVS secondary to two normal consecutive pregnancies (5). Our second patient's recurrence of thunderclap headache and/or RCVS after an interval of 4 years is unusual. Unfortunately, MRA was not performed when our second patient represented with thunderclap headache after 4 years, as she presented to a different local hospital and early neurological advice was not requested. Although the patient felt the symptoms were similar to the first episode 4 years previously, MRA was not performed and therefore we cannot definitively conclude that the latter episode was a recurrence of RCVS. Patients with thunderclap headache, normal CT scan and normal CSF analysis should be fully investigated by cranial and cervical imaging, including arteries and veins, if one is to exclude absolutely all vascular causes of secondary thunderclap headache.
Recurrent benign thunderclap headache after a 4-year interval is uncommon. If we compare our second patient with patients with non-subarachnoid haemorrhage thunderclap headache, only 1.5% (one of 71 patients) had recurrence after 4 years (19). In these patients with non-subarachnoid haemorrhage thunderclap headache, identical recurrences tend to occur within 30 days (20). It is not known whether some of these patients actually had RCVS.
It is not known whether patients who developed secondary RCVS from one vasoactive substance should avoid another known vasoactive substance, as recommended by some authors (3). Our second patient, who had a presumed secondary RCVS from cannabis, previously took overdoses of SSRI with alcohol a few times, without precipitating secondary RCVS.
Our two cases highlight reversible cerebral vasoconstriction syndrome as a cause of thunderclap headache and multilobar intracranial haemorrhage. The presentation may cause diagnostic confusion with cerebral venous thrombosis, and MRV may be misleading. Secondary causes of RCVS, especially if precipitated by use of vasoactive substances, should be considered. Recurrence of thunderclap headache after an interval of 4 years may occur and should be investigated for recurrence of RCVS.
Footnotes
Competing interests
None to declare.
Acknowledgements
The authors thank Dr Kumar Das, consultant neuroradiologist, for his help with the radiology images in the preparation of this manuscript.
