Abstract
Status migrainosus is a condition characterized by a migraine attack causing disability, with or without aura, lasting for > 72 h. The pathophysiological mechanisms underlying this complication of migraine remain a matter of debate. We describe a migraine without aura patient who presented two episodes of status migrainosus associated with recurrent and reversible brain magnetic resonance imaging abnormalities. These abnormalities, confirmed also by positron emission tomography, suggest that status migrainosus can be associated with a condition of vasogenic cerebral oedema.
Introduction
Status migrainosus (SM) is a complication of migraine characterized by a headache attack (with or without aura) lasting > 72 h despite treatment (1). SM is generally considered a rare condition. However, a recent prospective study has shown that approximately 20% of migraine patients, during their attacks, experience pain for > 72 h (2). The pathophysiological mechanisms underlying SM have been rarely investigated. Emotional stress, medication overuse, depression and anxiety and discontinuation of prophylactic medications have been considered risk factors for SM (3). Altered circadian secretion of melatonin has been shown in patients with SM, suggesting a role for the pineal gland in the pathophysiology of migraine attack (4). Angiographic examination in a 26-year-old woman with SM demonstrated the presence of multiple segmental stenoses and dilations of cerebral arteries and suggested the presence of an inflammatory process (5). At present, however, the exact mechanisms involved in SM are unknown.
We report the case of a migraine without aura patient hospitalized during two episodes of SM. Repeated magnetic resonance imaging (MRI) examinations revealed recurrent and reversible brain abnormalities, suggestive of vasogenic brain oedema, localized in the occipital and temporal lobes. Lateralized brain abnormalities were confirmed both by electroencephalography (EEG) and by positron emission tomography (PET). This case report shows evidence of altered blood–brain barrier (BBB) during SM.
Case report
A 18-year-old woman was admitted as inpatient to our Neurological Department for an acute headache attack lasting from 5 days. Family history was positive for migraine without aura. From the age of 10–14 years the patient had suffered from partial seizures with complex semiology, treated with sodium valproate. At the time, brain MRI was normal. No recurrence of epilepsy occurred after stopping the therapy. At age 15 years, the patient had observed the onset of sporadic headache attacks, all fulfilling the International Classification of Headache Disorders, 2nd edn criteria (1) for migraine without aura, generally related to menses. The headache attacks were responsive to paracetamol and were not debilitating.
In January 2006 the patient developed an acute migraine without aura attack not responding to common analgesic. Twenty-four hours after headache onset the patient experienced intermittent visual disturbances, in the form of metamorphopsia, macropsia, micropsia, scintillating scotoma and negative scotoma, which were present when the patient presented to the emergency room of our hospital. None of the described symptoms was suggestive of migraine aura. In order to formulate an exhaustive diagnosis, the patient was admitted to our Neurological Department. General and neurological examinations were normal. Her blood pressure was 120/80, her apical pulse was 68/min and she was afebrile.
At admission, cranial computed tomography (CT) was performed, with normal results. Two days after admission, cranial T2-weighted (T2W) MRI and fluid-attenuated inversion recovery (FLAIR) images demonstrated hyperintense signal changes with swelling in the supratentorial white matter and adjacent cortex in the right temporal and occipital lobes (Fig. 1A). Diffusion-weighted imaging demonstrated slightly increased diffusion in these regions. The apparent diffusion coefficient was elevated in the territories supplied by the branches of the right posterior cerebral artery, suggesting the presence of vasogenic oedema. There was no enhancement of the lesions after administration of gadolinium. PET with 18F-fluorodeoxyglucose showed a reduction in the fixation of the tracer in the right temporal-occipital cerebral cortex, a finding suggestive of focal hypometabolism probably related to vasogenic oedema (Fig. 2). Visual field examination was normal. EEG showed evidence of right temporal-occipital focal delta activity.
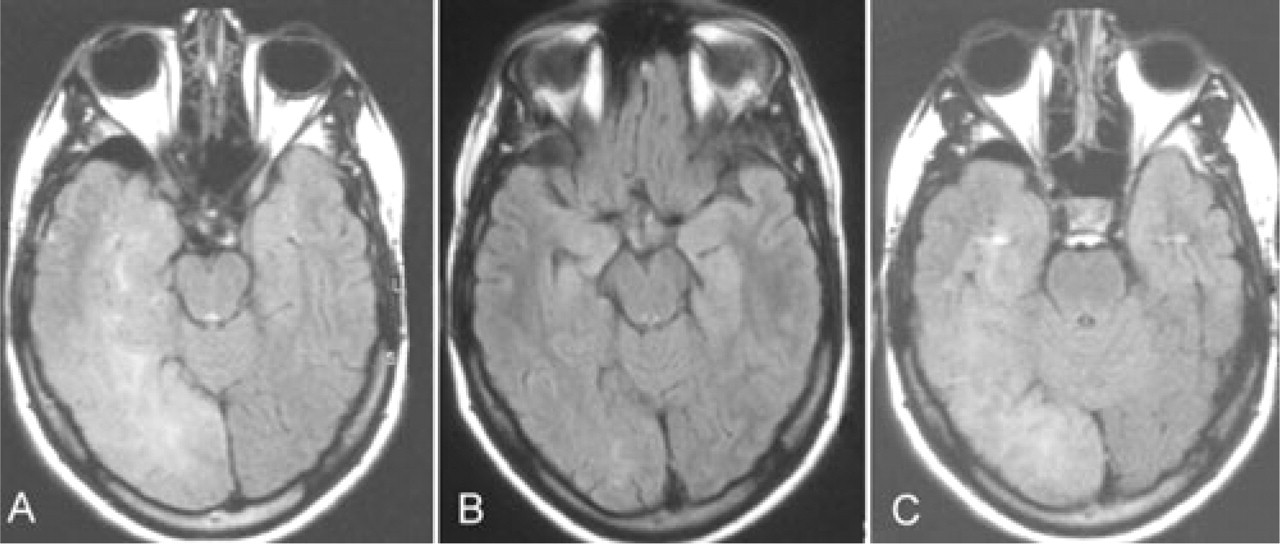
(A) Fluid-attenuated inversion recovery (FLAIR) image demonstrating an area of bright signal intensity over right temporal-occipital cortex (date of examination 8 January 2006). (B) Resolution of the previously identified lesions on FLAIR imaging (date of examination 29 June 2006). (C) The new magnetic resonance imaging scan shows the reappearance of abnormal FLAIR signal (date of examination 8 June 2007).

Positron emission tomography (10 January 2006) shows reduction in the fixation of the tracer in the right temporal-occipital cerebral cortex.
A laboratory evaluation, including routine cerebrospinal fluid studies, neoplastic markers, antithrombin 3, antinuclear antibody, plasminogen, protein C, protein S, prothrombin time, activated partial thromboplastin time and serum lactic and pyruvic acid, was normal. Microbiological or viral involvement, with emphasis on herpes simplex virus, varicella zoster virus, Epstein–Barr virus and cytomegalovirus encephalitis, were excluded. Cardiological evaluation, including a transoesophageal echocardiogram, was normal. Mitochondrial DNA, screened for mutations associated with mitochondrial encephalopathy with lactic acidosis and stroke-like episodes (MELAS) syndrome, was normal. Exons 3, 4, 5, 8 and 11 of the NOTCH3 gene were sequenced and no mutation was found.
Headache spontaneously subsided a week after admission and the patient was discharged. MRI examination, a month after the first examination, showed reduced alteration of the signal in the right temporal-occipital site. The control EEG had also clearly improved, showing a slow θ rhythm over the posterior right hemisphere. Six months after admission, a new MRI examination was performed (Fig. 1B) and showed almost complete resolution of the previously described area of T2 hyperintensity in the right occipital and temporal lobe. EEG showed the disappearance of the focal slowing previously present over the right hemisphere. In the following months, the patient presented irregular headache episodes, all treated with symptomatic drugs.
In June 2007, while feeling well, the patient again complained of the same symptoms presented in January 2006. The objective neurological examination showed normal results. Brain MRI showed a large area of altered signal, characterized by hyperintensity both in T2W and FLAIR, with the involvement of the temporal and occipital lobes (Fig. 1C). The signal alteration involved the white matter and, to a minor extent, the cortex. Cranial CT with perfusion study documented increased blood flow in the right temporal-occipital site. The computed visual field examination was normal. A symptomatic therapy was established (triptans, non-steroidal anti-inflammatory drugs), as well as a prophylactic one (topiramate, 100 mg/day). Cranial MRI performed 5 months later demonstrated complete resolution of the pathological signal alterations. In the following 6 months, the patient complained only of sporadic attacks of migraine without aura.
Discussion
The patient described had two rather stereotypical periods of SM.
Extensive evaluation failed to reveal other potential causes, such as migrainous infarction, cerebral autosomal dominant arteriopathy with subcortical infarcts and leukoencephalopathy, MELAS, central nervous system vasculitis, tumorous conditions or inflammatory disease. Repeated brain MRI examinations showed the presence, during both periods of SM, of an area of altered signal, lateralized in the right occipital and also right temporal lobes, suggestive of the presence of vasogenic oedema. To the best of our knowledge, this is the first case report showing reversible brain MRI abnormalities in patients with SM.
Brain MRI abnormalities reported in our patient during SM show some similarities with the neuroimaging findings of patients with both familial and sporadic hemiplegic migraine (6, 7), in migraine with aura and basilar migraine patients (8–10) and in a patient with migralepsy (association of migraine and epilepsy) (11). In these cases, increased vasogenic leakage has been suspected as the primary pathogenic mechanism of disease-associated reversible MRI abnormalities.
Cerebral vasogenic oedema is, classically, defined as fluid originating from blood vessels and accumulating around neuronal and glial cells. The amount of oedema is greatest in the white matter (increased water and sodium in the extracellular spaces, decreased potassium); but the same changes may take place in grey matter. The astrocytes become swollen. The disruption of the cerebral capillary provides the underlying mechanism for vasogenic oedema. Cerebral vasogenic oedema is seen in response to trauma, tumours, focal inflammation and late stages of cerebral ischaemia. The mechanisms leading to cerebral vasogenic oedema in patients with hemiplegic migraine, basilar migraine, migralepsy and SM are, at present, unknown.
Recently, the possible involvement of aquaporins, a family of osmotically driven bidirectional water channel proteins essential for the maintenance of BBE integrity, has been suggested in migraine (12). Up-regulation of aquaporins (mainly aquaporin 4) at the BBE may be an interesting pathophysiological explanation of increased vasogenic oedema observed in our patient with SM.
In conclusion, we report the case of a migraine without aura patient who experienced two episodes of SM. In both episodes, brain MRI showed the presence of reversible alterations in the occipital and temporal cortex suggestive of vasogenic brain oedema. Additional studies are needed in order to elucidate the pathophysiological mechanisms of transient MRI abnormalities in SM.
