Abstract
In cerebral blood flow studies, migraine aura is characterized by a posterior cortical hypoperfusion. In contrast, only rare and mild changes in brain perfusion have been demonstrated in migraine without aura, suggesting two different haemodynamic patterns in migraine with and without aura. Our aim was to study hypoperfusion with positron emission tomography (PET) as early as possible during spontaneous migraine without aura attacks. We used H2 15O PET to investigate seven patients (six female, one male) with migraine without aura (International Classification of Headache Diseases-II code 1.1) in three situations: during the headache phase, after headache relief following sumatriptan injection, and during an attack-free interval. Statistical analysis was performed with SPM2. Within 4 h after the attack onset, significant relative bilateral posterior cortical hypoperfusion was found and persisted after headache relief following sumatriptan injection. A posterior cortical hypoperfusion demonstrated in migraine without aura could suggest a common pathogenesis in migraine with and without aura. The significance of relative posterior hypoperfusion in migraine without aura is discussed according to the current knowledge of migraine pathogenesis.
Introduction
Most of the cerebral blood flow (CBF) studies in migraine have introduced the concept of a pattern different in migraine with aura (MA) and migraine without aura (MoA).
Migraine aura is characterized by a focal reduction of regional CBF in the posterior part of one hemisphere that usually, but not always, corresponds to the topography and timing of the reported symptoms (1–5). The hypothesis of cortical spreading depression (CSD) during migraine aura was suggested by Milner in 1958 (6). Recently, blood oxygen level-dependent (BOLD) functional magnetic resonance imaging (fMRI) has shown cerebrovascular changes in the cortex of migraineurs while experiencing a visual aura that closely resemble CSD (7). In this study, a clear temporal and spatial correlation was established between the visual symptoms of the aura and the BOLD signal changes.
In MoA, no blood flow changes were noticed in several studies (8–10). However, Woods and colleagues have reported a bilateral decrease in regional CBF (rCBF) spreading forward from visual associative cortex to parietal and occipitotemporal areas in a patient from the start of a spontaneous attack of MoA (11).
Since the initial positron emission tomography (PET) study performed by Weiller and colleagues (12), further studies have shown activation in brainstem structures during MA and MoA (13–16). Then brainstem nuclei may participate in migraine pathogenesis, probably in a dysfunctional mode of the anti-nociceptive network and cerebrovascular control.
In a previous publication using PET, we have reported hypothalamic and brainstem activation in spontaneous migraine attacks recorded as soon as possible after onset. The aim of the present study using the same design and the same patients was to focus on cerebral relative hypoperfusion.
Methods
We used H2 15O PET to study seven patients (six female, one male, mean age 38.1 years) during a spontaneous migraine attack. None of the patients had had prophylactic treatment. The study was approved by the local ethics committee. Informed consent was obtained from all patients, who were studied within 6 h after the onset of migraine symptoms. To avoid visual and auditory stimulation, subjects wore opaque goggles while being scanned in a darkened, quiet room. Each patient had six rCBF measurements under three conditions (two scans for each condition): during migraine headache, after headache relief following 6 mg sumatriptan subcutaneous injection, and during an attack-free interval (15–60 days later). PET scans were done with an EXACT HR+ scanning system (CTI/Siemens, Saint-Denis, France). Statistical Parametric Mapping 2 (SPM 2; Wellcome Department of Cognitive Neurology, London, UK) was used for data analysis. Images were realigned with the first image as the reference, normalized into the space defined by the atlas of Talairach and Tournoux, and smoothed with a Gaussian filter (8 mm at full width at half maximum). Statistical parametric maps were generated using an analysis of covariance model after normalization for global CBF changes (17). So with this method, rCBF measurements are adjusted value. The analysis included data from all seven patients. There were three right-sided, three left-sided and one bilateral headache. Analysis was performed both with and without taking lateralization into account. The first analysis discounted the side of the headache. In the second analysis, the PET scans from patients with left hemicranias were reflected through the sagittal plane for a ‘flipped’ analysis (incorporating right hemicranias, flipped left hemicranias and bilateral headaches). We chose a corrected threshold of P < 0.01 (false discovery rate) and a cluster extent of > 200 voxels for statistical analysis (see Table 1 and Fig. 1). Two comparisons were performed: comparison 1, rCBF during headache-free interval vs. rCBF during migraine headache; and comparison 2, rCBF during headache-free interval vs. rCBF after headache relief. Second order analysis (paired t-test, with a threshold of P < 0.01 uncorrected) was used to compare the ‘unflipped’ and the ‘flipped’ group and the rCBF variation between comparison 1 and comparison 2. To evaluate the degree of hypoperfusion between the headache-free condition and the migraine attack conditions, we compared the adjusted rCBF in each of those two conditions given by SPM at the most significant statistical peak and converted the modifications in percentage.
Significant decreases in blood flow during spontaneous migraine attack before and after sumatriptan compared with the headache free-interval
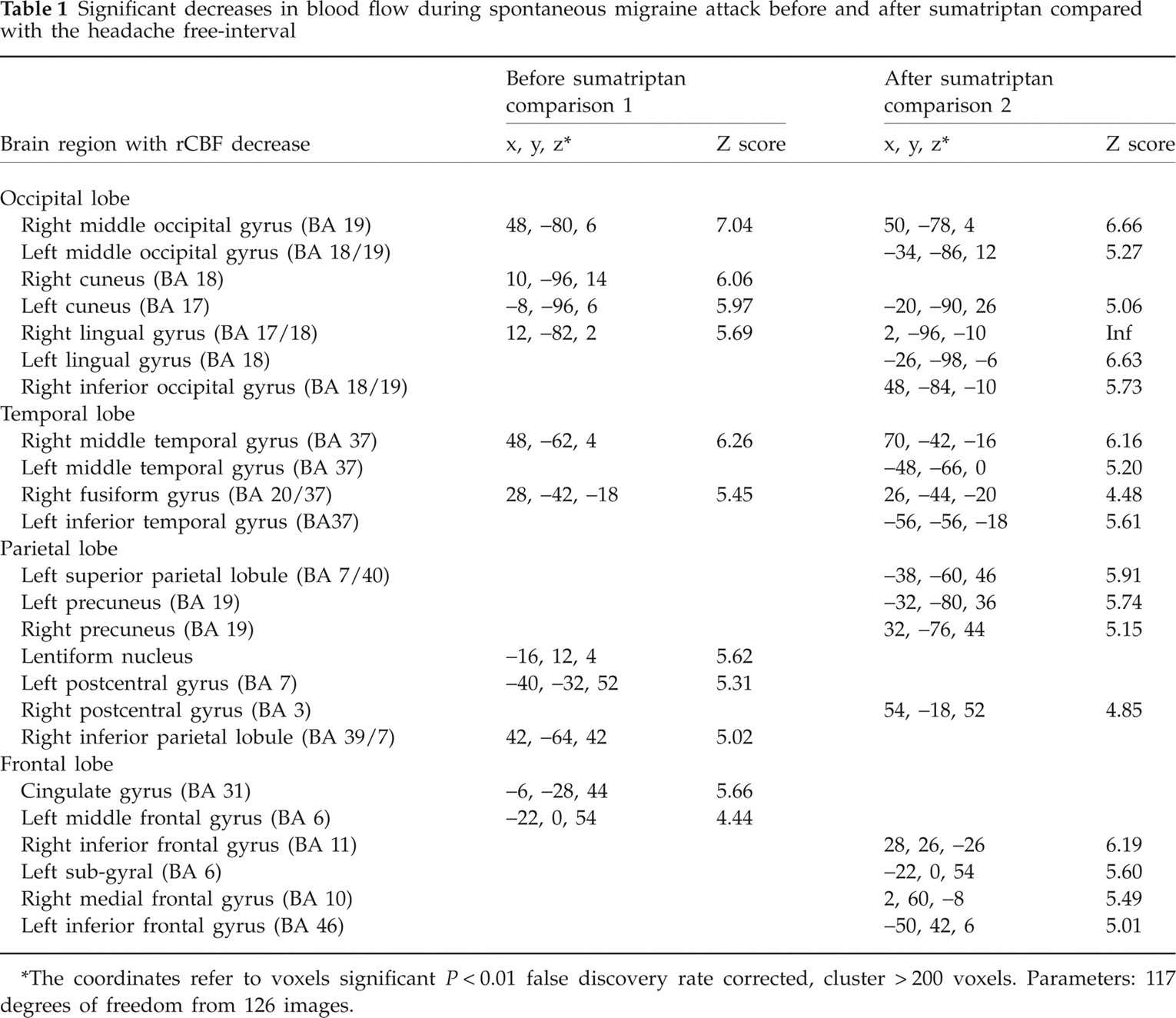
The coordinates refer to voxels significant P < 0.01 false discovery rate corrected, cluster > 200 voxels. Parameters: 117 degrees of freedom from 126 images.

Hypoperfusion during migraine without aura attacks in seven patients. Statistical parametric maps showing significant regional cerebral blood flow (rCBF) decreases during migraine without aura attacks. Blue plots correspond to rCBF decrease [P < 0.01 false discovery rate corrected, cluster > 200 voxels] before and after sumatriptan compared with the headache-free interval. Posterior bilateral hypoperfusion can be seen.
Results
Mean time from attack onset to PET scan was 3 h 8 min (range 2 h 15 min to 3 h 50 min). The post-pain relief PET scan was done within 6 h of headache onset for all patients (from 4 h 20 min to 6 h, mean time 5 h 9 min).
During MoA attacks, a relative hypoperfusion was found bilaterally in the occipital cortex and the posterior temporal and parietal cortex compared with the headache-free interval (Table 1 and Fig. 1). The adjusted rCBF decrease was 10.34%. This relative hypoperfusion was significant when the patients are considered as a group as well as individuals (in five out of seven patients). After pain relief by sumatriptan, 4–6 h after onset, the relative hypoperfusion persisted (Table 1 and Fig. 1). The adjusted rCBF decrease was 12.32% after sumatriptan. After flipping the images of left-sided headaches, so that six of seven migraines were on the right side, the relative posterior hypoperfusion remained bilateral before and after sumatriptan. Comparison between ‘flipped’ and ‘unflipped’ data revealed no significant differences.
Second-order analysis demonstrated that hypoperfusion after sumatriptan compared with hypoperfusion before sumatriptan was even more important in the occipital region and appeared in the frontal lobe.
Discussion
When compared with the headache-free period, a decrease in posterior cortical rCBF was found during the headache phase. How can these modifications be interpreted?
H2 15O PET is a method of sequential measurements of rCBF permitting, for example, the attribution of specific functions to brain areas in experimental conditions as specific cognitive or behavioural tasks. Indeed, it has been demonstrated that increases in neuronal activity lead to an increase in rCBF, and decreases in neuronal activity lead to a decrease in rCBF (18). According to previous studies using xenon blood flow techniques or single photon emission computed tomography, demonstrating cortical hypoperfusion (1–5) in MA, we chose to discuss our results in term of relative cortical hypoperfusion.
Is this relative posterior hypoperfusion specific to migraine or is it related to a non-specific pain process?
Similar posterior hypoperfusion has been found in a few pain studies (19–22). However, posterior hypoperfusion in relationship to pain and not specific to migraine seems unlikely, since this hypoperfusion persisted after total pain relief by sumatriptan in our study and could appear before headache (3). Thus far, this hypoperfusion would be specific to migraine because it has been demonstrated only during migraine attacks and not during trigemino-autonomic cephalalgias or experimental headache (23–25).
If posterior cortical hypoperfusion has been demonstrated extensively in MA, only few studies have found similar hypoperfusion during MoA (11, 26–28). Considering that posterior hypoperfusion is present at the beginning of MoA as well as MA, two hypotheses could be advanced to explain this hypoperfusion: (i) hypoperfusion is a consequence of CSD; (ii) hypoperfusion is a primary neurovascular event.
1. Hypoperfusion is a consequence of a CSD
In 1958, Milner hypothesized that the migraine visual aura was associated with CSD, but until recently no study had clearly corroborated this hypothesis (6). Hadjikhani et al. (7) have used BOLD fMRI to map the progression of cerebrovascular changes in three subjects during spontaneous migraine aura and measured the retinotopic nature of the BOLD signal changes. This study revealed multiple neurovascular events in the occipital cortex that closely resemble CSD. Moreover, a clear temporal correlation was established between the initial features of the aura percept and the initial increase in the mean BOLD signal, reflecting cortical hyperaemia. The subsequent decrease in BOLD was correlated with the scotoma that followed the scintillations. More direct evidence that CSD underlies visual aura has been obtained with magnetoencephalography (MEG). MEG studies have revealed that migraine visual aura is associated with shifts in direct current neuromagnetic field potentials similar to those seen during CSD (29, 30).
Recent research in animals has shown that CSD could induce posterior hypoperfusion and would be able to activate the trigeminal meningeal afferents consistent with the development of headache (31). However, this is much debated (32).
If CSD is a primary event common in MA and MoA, CSD must be predominantly asymptomatic, because only a minority of patients report aura. It seems unlikely that CSD occurring in the occipital cortex, characterized by a very high neuronal density, especially in the primary visual cortex (33), could be clinically silent.
Moreover, if the relationship between the aura symptoms and CSD seems to be clear, the temporal link between posterior hypoperfusion and CSD remains unclear. According to Olesen's study (3), the oligaemia starts before the onset of aura and extends into the headache phase, outlasting the aura symptoms. This discrepancy between haemodynamics and symptoms of aura and headache has also been found in studies of visual triggered attacks using BOLD-fMRI (34). In perfusion-weighted imaging during spontaneous MA, the rCBF decrease persisted up to 2.5 h into the headache phase (8). In a study using PET in spontaneous MoA attacks (27), global hypoperfusion was found within a mean time of 13.3 h from the onset of headache. In our study, the hypoperfusion persisted 6 h after the onset attack. Long-lasting hypoperfusion could not be explained by a single wave of CSD. To produce such a long-lasting event, a succession of waves would be required, all of them asymptomatic, which seems unlikely.
In our study, the hypoperfusion after sumatriptan is even more important in the occipital region and appears in the frontal lobe. CSD could not explain this frontal area of hypoperfusion, since CSD classically does not cross prominent sulci. However, a direct effect of sumatriptan cannot be ruled out.
Combining electrophysiological measurements with optic intrinsic signal imaging, Brennan and collegues (35) have shown, during experimentally induced CSD in mice and rats, vasomotor changes in cortex travelling at significantly greater velocity than the neuronal changes. These results suggest that there is a dissociation of metabolic demand and vascular response and that the cortical surface arteriolar changes associated with CSD appear to have independent mechanisms of propagation (36).
2. Hypoperfusion is a primary neurovascular event
A primary brainstem dysfunction has been proposed as the origin of migraine attacks. Since the initial PET study performed by Weiller et al. (12), further studies have shown activation in brainstem structures during migraine attacks, either spontaneous (13, 16) or triggered (14, 15, 37) and either with (13, 14, 37) or without aura (12–16). Activation has been described in the midbrain that could correspond to the dorsal raphe nucleus, periaqueductal grey, and locus coeruleus (12, 16), red nucleus, substantia nigra (37) and the dorsolateral pons (13, 14, 16). These nuclei are involved in the central control of nociception and extra- and intracerebral vascular control.
Experimental work in animals has shown how brainstem nuclei disturbance could initiate the vascular changes seen in migraine (38). Activation of noradrenergic neurons in the locus coeruleus would be expected to produce bilateral reductions in cortical blood flow (39–41) that would be more prominent in the occipital region (42), the part of the brain affected by hypoperfusion during migraine attacks.
Some arguments from neuroimaging studies in migraineurs are in favour of a primary brainstem dysfunction responsible for posterior hypoperfusion. In the study of Cao et al. (37), brainstem activation appeared before the neuronal suppression in the occipital cortex or the onset of visually triggered symptoms. The persistence of both brainstem activation (12, 16) and posterior hypoperfusion after sumatriptan injection suggests that the hypoperfusion could be in relationship to the brainstem activation and then would persist as long as the brainstem activation does, independently of the migraine symptoms relieved by the action of the triptan on the peripheral trigeminovascular system.
Concerning the absence of visual symptoms during the hypoperfusion phase, a primary vascular event below the ischaemic level (respectively 10% and 12% before and after sumatriptan in our study) seems to us a better explanation than an asymptomatic CSD.
On this hypothesis, the primary event in both types of migraine could be an oligaemia triggered by the activation of brainstem nuclei. Cortical susceptibility to oligaemia could then trigger CSD, producing visual symptoms in MA.
In conclusion, we found in MoA a posterior hypoperfusion similar to the changes found in other studies in MA. If this hypoperfusion is specific to migraine, it favours the same pathophysiology in MA and MoA. However, the significance of this hypoperfusion needs to be specified.
Footnotes
Acknowledgements
The authors are very grateful to the patients for their cooperation. We thank the PET Centre technicians and nurses for their participation and motivation, and the staff of Toulouse Clinical Investigation Centre for patient management. This work was supported by INSERM and a grant from the Délégation Régionale à la Recherche Clinique des Hôpitaux de Toulouse 2001.
