Abstract
Effects of pregabalin (PGB) on patient-reported health outcomes were assessed in 65 PGB-naive subjects with trigeminal neuralgia refractory to previous analgesic therapy in a prospective, multicentre observational study carried out in primary care. Twelve weeks' monotherapy with PGB (n = 36) or add-on (n = 29), reduced baseline intensity of pain by a mean ± S.D. of −40.0 ± 22.1 mm [−55.4%, effect size (ES) 2.32; P < 0.0001] with 59.4% of responders (pain reduction ≥ 50%), and produced 34.6 ± 29.3 additional days with no/mild pain. Anxiety/depression symptoms decreased by −3.8 ± 3.5 and −4.5 ± 4.2 points (ES 0.95 and 1.02; P < 0.0001), respectively. PGB improved sleep by −17.9 ± 19.6 points (ES 1.18; P < 0.0001) and improved patient functioning (Sheehan Disability Index) by decreasing overall scoring by −8.6 ± 5.9 points (ES 1.59; P < 0.0001). Health state (EQ-5D) increased by 31.6 ± 22.2 mm (ES 1.67; P < 0.0001), with 0.0388 ± 0.0374 gained quality-adjusted life-years. In spite of the small sample size, results support the effectiveness of PGB for the improvement in pain and related health symptoms.
Introduction
Trigeminal neuralgia (TGN) is a form of neuropathic pain (NeP) characterized by the occurrence of abrupt pain, generally one-sided, severe, brief, sharp and recurrent in the distribution area of one or several branches of the fifth cranial nerve (1). It is a relatively infrequent condition, with 70/100 000 lifetime prevalence, an annual incidence of 8/100 000 cases and a slight predominance in women (2). Although not well studied, patients with TGN, as with other forms of NeP, may frequently develop anxiety, depression and sleep disturbance symptoms to some extent as a consequence of pain itself (3). Furthermore, TGN pain significantly affects their state of health, including functioning and well-being, health resources consumption and individual productivity (3).
Pharmacological treatment of TGN is primarily based on the use of anticonvulsant drugs such as carbamazepine (4–8), this drug being the treatment of choice. Also, carbamazepine use is extended to other forms of NeP despite its side-effects profile (6, 8). In primary care settings, gabapentin, oxcarbazepine or tricyclic antidepressant drugs are recommended as alternatives to carbamazepine when the drug is not tolerated (5). However, this is frequently a treatment-resistant condition with suboptimal management where drugs are often used whose efficacy has not been demonstrated in this condition [e.g. non-steroidal anti-inflammatory drugs (NSAIDs)], or in situations where lower than recommended doses of drugs such as carbamazepine or gabapentin are used (3).
Pregabalin (PGB) is an α2-δ binding drug displaying analgesic, anxiolytic and anticonvulsant properties (9). It is structurally related to gabapentin. In randomized, placebo-controlled clinical trials, PGB has demonstrated efficacy for pain relief in patients with diabetic neuropathy and peripheral postherpetic neuralgia, significantly improving affective symptoms, sleep and quality of life (10). Furthermore, the pharmacokinetic profile of PGB allows for easy management and rapid dose escalation to therapeutic dosages (11). However, no randomized clinical trials evaluating the usefulness of PGB for the treatment of TGN have been conducted so far. Recently, results of a prospective open-label study have been published in which the efficacy of 150–600 mg/day PGB doses administered to 53 patients with TGN in a tertiary care centre was assessed during 1 year (12). Treatment with PGB resulted in the alleviation or at least 50% reduction of pain intensity in 25% and 49% of patients, respectively (12). Our study presents clinical practice evidence of the effect of PGB on patient-reported outcomes, including pain, symptoms of anxiety and depression, sleep disturbances, disability and calculation of quality-adjusted life-year (QALY) gain, representing the multidimensional nature of this condition, in a large group of patients with painful TGN treated in primary care settings (PCS).
Patients and methods
Study design
The results represent a secondary analysis of a multicentre, observational, prospective 12-week study, with the objective of assessing the cost of treatment of refractory NeP [due to painful diabetes neuropathy (DPN), postherpetic neuralgia (PHN) or TGN] under real-life conditions in PCS: the LIDO study (13). The study was conducted between September 2005 and April 2006, and 391 primary care physicians, representative of all Spanish territories, participated. The study was non-interventional, and the analgesic treatment prescribed was decided in accordance with the clinical judgment of the physician responsible for the patient's management. Doctors could substitute patients' previous treatments with one or several other drugs, or add a new drug to the background therapy, but this new therapeutic schedule should remain up to the end of the 12-week study. In accordance with Spanish recommendations, the study was approved by the Ethics Committee of Clinical Research from the Hospital de la Princesa (Madrid, Spain) and was conducted in agreement with the principles contained in the Declaration of Helsinki for human studies.
The objective of this post hoc analysis was to assess the effect of two PGB treatment patterns, add-on and monotherapy, in a subgroup of subjects with TGN.
Study population
The original study included patients of ≥ 18 years old, of both genders and with an established diagnosis of NeP secondary to DPN, PHN or TGN made by a specialist. TGN diagnosis was usually done in pain or neurology clinics. Study subjects had to be refractory to previous analgesic therapy and have suffered chronic NeP for ≥ 6 months. The term refractory was defined as the absence of pain reduction after treatment with at least one course of an analgesic drug in monotherapy. In addition, study requirements included a score ≥ 4 (range 0–10) in the NeP Douleur Neuropathique 4 questions (DN4) questionnaire (14–16) (see below), cultural and educational levels sufficient to complete a written health questionnaire in Spanish, and to obtain patients' informed consent. This secondary analysis included those patients who fulfilled the previously mentioned selection criteria, had a diagnosis of TGN, and had not received PGB treatment prior to study initiation.
The primary end-point of the LIDO study was to determine the use and calculate the cost of healthcare resources devoted to the treatment of chronic NeP during a 12-week follow-up period under common medical practice conditions, in PCS. No sample size was predetermined for the secondary analysis presented in this study.
Clinical assessments and measurement instruments
In this multicentre, observational, prospective 12-week study, patients were assessed twice, at baseline and the final study visits (after 12 weeks). During the baseline visit, the Spanish version of the NeP DN4 diagnostic questionnaire was completed by clinicians (14). Selection criteria were verified and sociodemographic and disease and treatment duration data were collected, in addition to data related to the use of healthcare and non-healthcare resources during the 12 weeks prior to study inclusion, in accordance with patients' medical records. At baseline and the end of the 12-week follow-up, patients completed the Short Form McGill Pain Questionnaire (SF-MPQ), the Sheehan Disability Inventory (SDI), the Medical Outcomes Study Sleep Scale (MOS-Sleep), the Hospital Anxiety and Depression Scale (HADS) and the EQ-5D questionnaire. Furthermore, patients completed a diary in which they recorded weekly pain intensity on a visual analogue scale (SF-MPQ VAS) and patient's health status (EQ-5D VAS). We used the Spanish validated versions of all patients' reported outcomes instruments used in this study.
The NeP DN4 diagnostic questionnaire has demonstrated 83% sensitivity and 90% specificity, consists of 10 items describing different pain characteristics and allows distinction between NeP and non-NeP conditions. A score of at least 4 of 10 possible points is suggestive of NeP (14–16).
The primary component of the SF-MPQ consists on 15 descriptors (11 sensory and four affective) to be assessed on an intensity scale ranging from 0 = no pain to 3 = severe pain (17). Three pain scores are obtained by adding the different intensities: sensory, affective and overall. The second part of the SF-MPQ consists of a 100-mm VAS assessing the patient's pain intensity during the previous week. The third part is designed to evaluate pain intensity at the time of assessment through an ordinal six-point scale, ranging from 0 = no pain to 5 = unbearable pain. The SDI is a validated measure that assesses functional impairment of patients in three domains: work, social life and family life/home responsibilities (18). Each domain is evaluated on an 11-point scale, ranging from 0 = no impairment to 10 = extremely impaired. The instrument contains two additional items assessing perceived stress and perceived social support. The SDI provides three scores: a disability score consisting of the sum of the three first item scores and ranging from 0 to 30, the fourth item or perceived stress score, with a range of 0–10, and the fifth item or social support score in which values are recorded, inverse to the other four.
The MOS-Sleep Scale is a self-assessed questionnaire that examines key aspects of sleep (19). It consists of 12 items comprising six subscales or domains: Sleep Disturbances, Snoring, Shortness of Breath or Headache upon Awakening, Sleep Adequacy, Somnolence and Sleep Quantity. In addition, the MOS-Sleep Scale provides a summary index (Sleep Problems Index) obtained from nine of the scale item scores; the higher the score, the worse the sleep, with the exception of dimensions related to the amount of sleep and adequacy of sleep, which are scored in the opposite direction. This scale has shown appropriate psychometric properties in NeP patients (20). The HADS is a self-assessed instrument also, consisting of 14 items, seven of which explore depression symptoms and the other seven anxiety symptoms (21). Each item score ranges from 0 to 3, where 0 represents the absence of the symptom and 3 the highest severity or frequency of the symptom. By adding the seven items of each subscale, two scores are obtained ranging from 0 to 21, for depression and anxiety (HADS-D and HADS-A), respectively.
The EQ-5D is designed to assess a patient's perceived health status (22). This is a five-item generic measure of health status assessing the level of abnormality of five dimensions: movement, self-care, day-life activities, pain/discomfort and anxiety/depression. A utility index with a −0.6 to 1.0 range may be calculated from these five item scores, with higher scores representing better health status. The instrument also includes a VAS (EQ-5D VAS) ranging from 0 = the worst imaginable health status to 100 = the best imaginable health status.
Statistical analysis
For statistical analyses, patients where divided into two groups, depending on the treatment initiated at the baseline visit, in accordance with clinical judgment: patients to whom PGB in monotherapy was prescribed as a substitute of the previous therapy (PGB monotherapy group); and patients to whom PGB was added to the previous therapeutic schedule (PGB add-on group). The subjects included in the PGB monotherapy were those receiving solely PGB as the unique analgesic therapy.
Patients' baseline characteristics were described by means and
In order to evaluate the relevance or clinical significance of changes in different measures, the obtained effect size (ES) was calculated using the difference of means, before and after treatment, of a particular measure, divided by the combined
Results
Patients disposition
The LIDO study originally included 1845 patients, 65 of whom fulfilled the selection criteria for this post hoc analysis. Of these 65 patients, 36 (55%) received PGB in monotherapy and 29 (45%) received PGB in combination with other drugs (add-on group). Only one patient, of the monotherapy group, did not complete the 12-week follow-up.
Baseline demographic and clinical characteristics
The mean age of patients included in this sample was about 60 years, with a predominance of female patients; approximately one-third were actively working (Table 1). The mean time from diagnosis of TGN was > 2 years, most patients were receiving polytherapy before entering the study, and the most frequently used drugs were NSAIDs, especially in the PGB add-on group (76%), whereas antiepileptic agents (39%) and paracetamol (31%) were more frequently used in the monotherapy group (Table 1).
Demographic and clinical characteristics
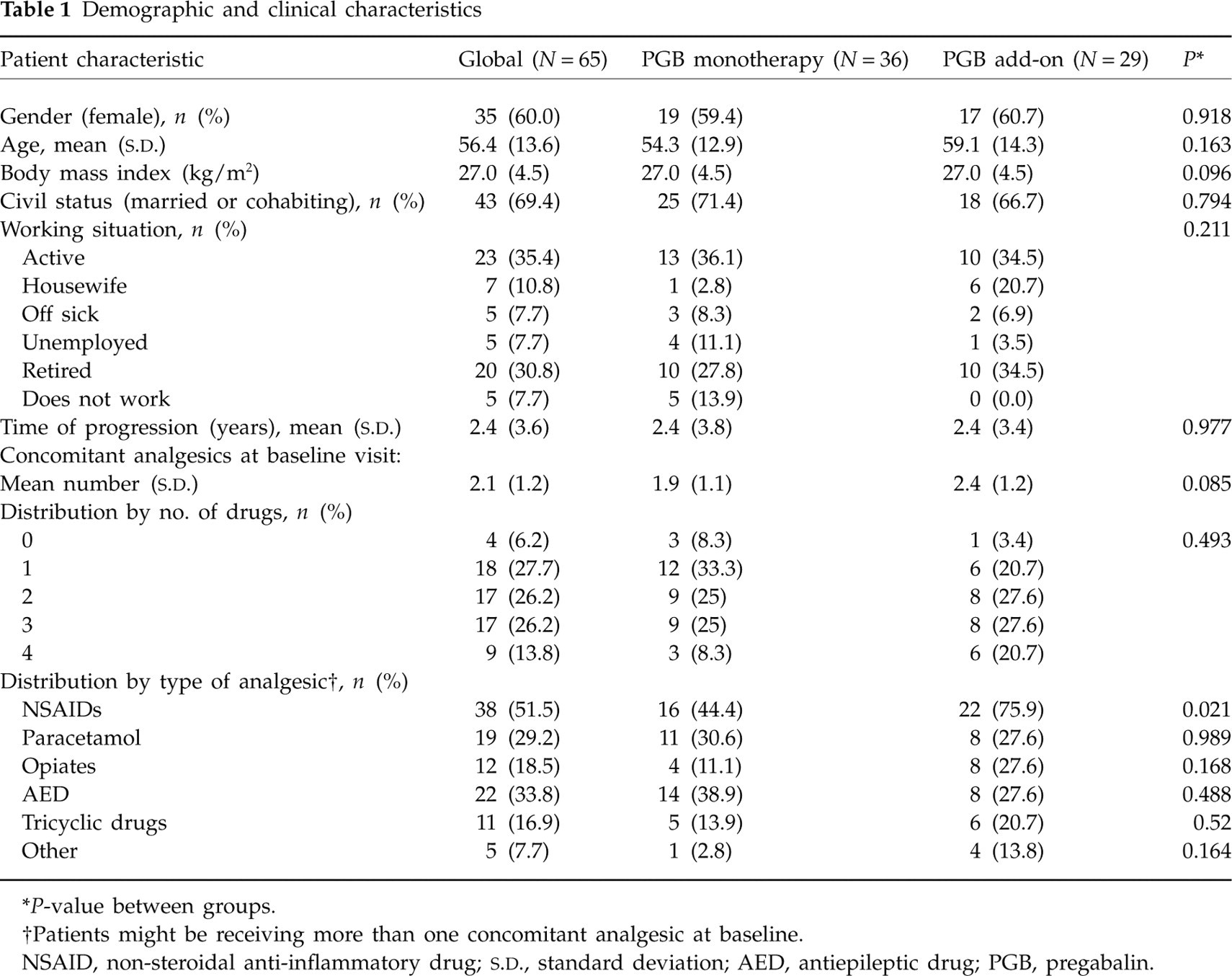
∗ P-value between groups.
†Patients might be receiving more than one concomitant analgesic at baseline.
NSAID, non-steroidal anti-inflammatory drug;
With regard to baseline clinical characteristics (Table 2), patients showed moderate to severe pain intensity, and important effects on their quality of life assessed by the EQ-5D. Sleep and psychic domains were affected also, but at a lower level than pain and quality of life. With the exception of a significantly higher DN4 questionnaire score in the add-on group, no differences were found between the PGB add-on and monotherapy groups in any of the health outcomes evaluated at baseline.
Patient-reported outcomes at baseline visit

∗ P-value between groups.
†Sum of scores of the three disability items.
‡Patients responding ‘without problems in the item’.
Drug treatment
Patients in the PGB add-on group received additionally NSAIDs (34%), paracetamol (20%) and opiates (11%) among other analgesic drugs in lower proportion. The mean final dose of PGB in monotherapy was 196 ± 105 mg/day, and higher, 234 ± 107 mg/day, in the add-on group.
Patients' reported outcomes
Pain reduction
Treatment with PGB was related to a significant and clinically relevant reduction (ES ≥ 0.8) in pain according to all pain scores of the SF-MPQ (Table 3). At study completion, 60% of patients showed a reduction of pain intensity of ≥ 50% from baseline. The reduction in the SF-MPQ sensory pain or pain intensity in the previous week was significantly lower in patients who received PGB add-on. The mean change of pain in the previous week (Table 3) was significantly more pronounced in the patient group receiving PGB monotherapy compared with the add-on therapy group (−45.1 ± 20.9 mm vs. −33.7 ± 22.2 mm, respectively, P = 0.038), almost doubling the effect of PGB add-on. Although the mean cumulative number of days with no or mild pain was greater in the PGB monotherapy group, however, the difference did not attain statistical significance. Differences were also found in the onset of pain reduction; whereas in the PGB monotherapy group clinically significant reductions were observed beginning in week 2, the patient group receiving PGB add-on therapy showed a clinically relevant pain reduction beginning in week 6 (Fig. 1, ES not shown).
Mean change and magnitude (effect size) of the different pain attributes measured using the SF-MPQ Questionnaire

∗ P-value between groups.
† P < 0.0001 vs. baseline.
‡Values expressed as means (standard deviation) [effect size].
§Responders: patients with a reduction of ≥ 50% of the pain intensity baseline value assessed using the SF-MPQ VAS.
VAS, visual analogue scale; PGB, pregabalin; PPI, present pain intensity; SF-MPQ, Short Form McGill Pain Questionnaire.
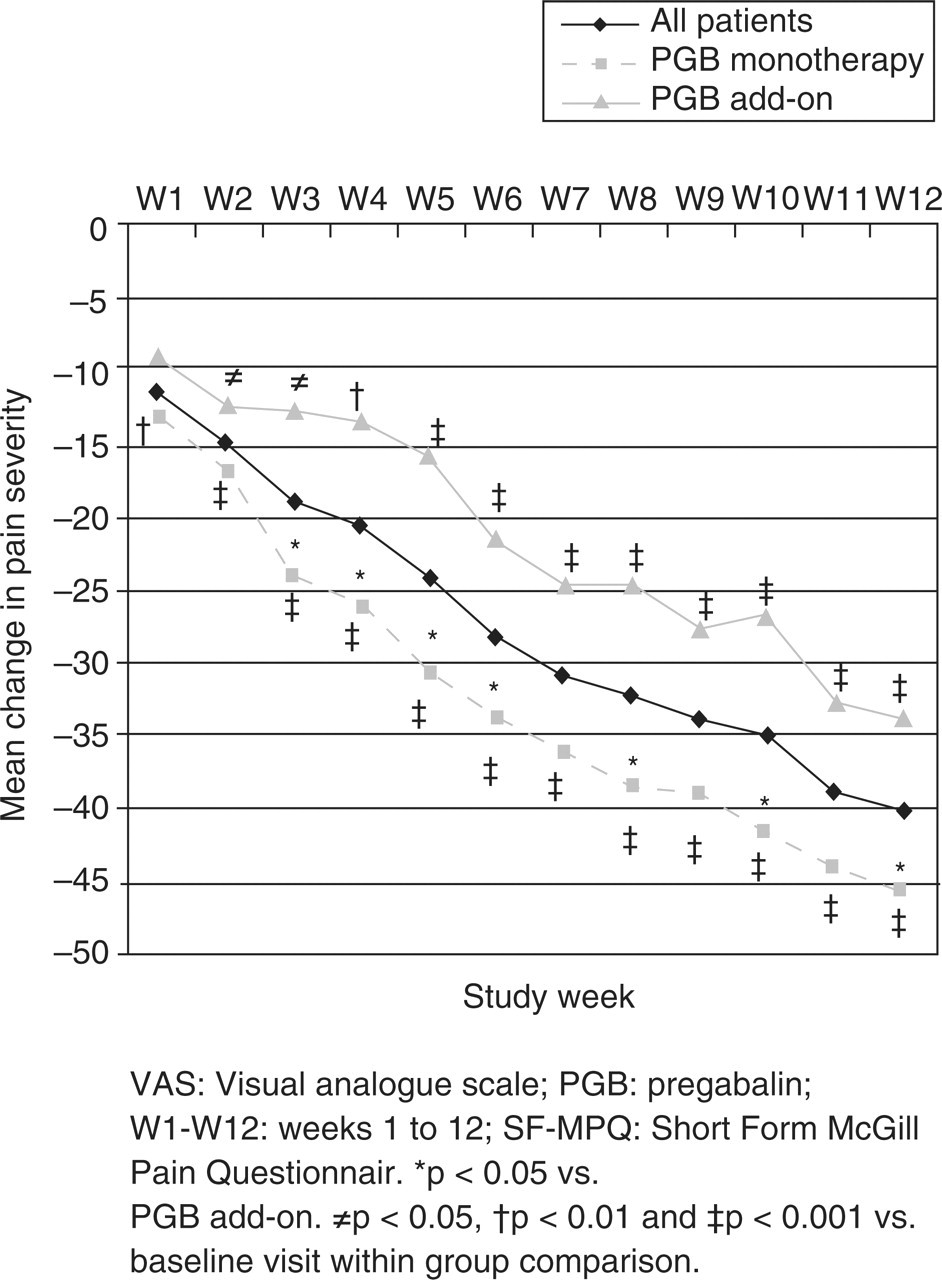
Weekly mean change in pain intensity according to the Short Form McGill Pain Questionnaire visual analogue scale.
Other patient-reported health outcomes
PGB therapy was associated with significant and clinically relevant improvements, as shown by large ES changes (≥ 0.8), in depression, anxiety and sleep disturbance related symptoms, but globally no significant differences between treatment groups were observed (Tables 4 and 5). However, improvements in anxiety and depressive symptoms were greater in the PGB monotherapy group, and a trend to significance in anxiety symptoms was observed compared with the PGB add-on group (Table 4). Regarding sleep, significant improvements were observed for the nine-item Sleep Problems Index and the six MOS domains (Table 5); however, changes were only clinically relevant (ES ≥ 0.8) for the Sleep Problems Index and in two dimensions: sleep disturbance and quality of sleep (suggest capitalizing domains). The only difference between groups was the item ‘shortness of breath awakening’; patients in the PGB add-on group indicated a significant reduction vs. baseline in this item score, whereas the reduction respective to baseline was not significant in the PGB monotherapy group.
Mean change and magnitude (effect size) of patient-reported outcomes measures at the end of the study

∗ P-value between groups.
† P < 0.0001 vs. baseline.
‡ P < 0.001 vs. baseline.
§ P < 0.01 vs. baseline.
¶ P < 0.05 vs. baseline.
∗∗Values expressed as means (standard deviation) [effect size].
††Sum of scores of the three disability items.
‡‡Patients responding ‘without problems’ in the item.
VAS, visual analogue scale; HADS, Hospital Anxiety Depression Scale; MOS, Medical Outcomes Study; NS, non-significant; PGB, pregabalin; SDI, Sheehan Disability Inventory; QALY, quality-adjusted life-year.
Mean change and magnitude (effect size) of the different dimensions of sleep measured using the MOS Sleep Questionnaire according to treatment group

∗ P-value between groups.
† P < 0.0001 vs. baseline.
‡ P < 0.001 vs. baseline.
§ P < 0.01 vs. baseline.
¶ P < 0.05 vs. baseline.
Values expressed as means (standard deviation) [effect size].
MOS, Medical Outcomes Study; NS, non-significant; PGB, pregabalin.
Clinically relevant (ES ≥ 0.8) and significant improvements of disability and quality of life were observed as well (Table 4). Overall, no significant differences were observed between patients receiving monotherapy and add-on therapy with PGB in improvement of health status (EQ-VAS). However, health status was significantly improved beginning in week 4 in the monotherapy group, and beginning in week 8 in the add-on group, with significant differences observed between the two treatment groups in the first treatment weeks (Fig. 2, ES not shown). Furthermore, significant differences favourable to PGB monotherapy were observed in three of the five heath status dimensions: day-life activities, pain/discomfort and anxiety/depression. A significantly greater QALY gained was observed in the PGB monotherapy group.

Weekly mean change in health state according to the EQ-5D visual analogue scale.
Discussion
The results of this study suggest that PGB, both in monotherapy and in combination with other drugs, is an effective drug for the short-term treatment of TGN in common clinical practice. In our study, treatment with PGB reduced pain and related comorbid symptoms, with clinically relevant improvements of disability and quality of life as well. As regards pain, these results are consistent with a previous non-controlled study of PGB for the treatment of TGN (12). In that study, after 8 weeks' treatment, a positive response to pain was observed in 39 of the 53 included patients (74%), with pain alleviation (13/53, 25%) or reduction of pain intensity observed in at least 50% of patients (26/53, 49%) (12). We noted a reduction in pain in 59% of patients on the SF-MPQ VAS, and in 19 of the 65 patients analysed (31%) pain had disappeared at the end of this 12-week study, as measured with the EQ-5D VAS.
Our study has extended the results reported by these authors to other important health outcomes. The improvements in anxiety, depression and sleep disturbance symptoms with PGB are very important, as these comorbid conditions frequently seem to be related to TGN (3). A relatively high percentage of patients (25–50%) receive concomitant treatment for their comorbid conditions (3) and, as in other types of neuropathic pain, related symptoms may exacerbate the disease, worsening the severity of clinical features and causing disability (26–29). It should be highlighted that, in our analysis, PGB monotherapy produced significant and clinically relevant improvement in these associated symptoms.
An important aspect of this study is PGB dosing. The treatment of TGN frequently requires the use of high doses of antiepileptic agents (5). In our study, mean doses of PGB (196 mg/day in the monotherapy subgroup, 234 mg/day in the add-on subgroup) were close to the lower limit of doses recommended for seizures (150–600 mg), slightly lower than the dose used by Obermann et al. (270 mg/day) (12), and close to the lowest dose of PGB recommended to treat other types of NeP conditions (150 mg/day) (30), although within its therapeutic range. These data suggest that, although the efficient dose range of PGB for TGN is still unknown, the mean dose used in our study may have been too low (somewhat higher in the PGB add-on group), which is even more notable, as patients had to have failed with a previous analgesic treatment.
A recommended treatment alternative for monotherapy failures in the treatment of TGN is to use a combination of drugs (7). In our study, clinicians chose PGB combination or add-on therapy for almost 50% of patients with TGN. PGB added to patient medications significantly improved pain, related symptoms, disability and quality of life. However, our analyses suggest that the response to pain was less and had a slower onset in the group receiving PGB add-on compared with patients who received PGB monotherapy. Due to the observational nature of this study, it is possible that participating physicians chose the PGB add-on option for more refractory cases. The only differences observed between the PGB monotherapy and the add-on groups in patients' baseline clinical characteristics were the DN4 questionnaire scores and the percentage of patients taking NSAIDs. The DN4 mean score (range = 1–10) was one point higher in the PGB add-on group. Although the DN4 is a questionnaire supporting diagnosis, there is a lack of specific cut-points for interpreting the results; however, a higher score suggests the presence of more complex and painful symptoms in the PGB add-on group. Likewise, a significantly greater percentage of patients in the PGB add-on group were being treated with NSAIDs, which have little to no demonstrated efficacy in the treatment of TGN.
When interpreting this study, some limitations should be noted. First, this was an observational, non-comparative study, and is therefore susceptible to bias, and effectiveness may have been overestimated. Another limitation is the lack of systematic assessment of treatment tolerability, due to the absence of a specific evaluation of the effect of the pharmacological intervention in the original study. However, the greater number of patients who completed the study suggests that, in general, PGB was well tolerated. Furthermore, assessments other than those present in our study could have been included to measure treatment objectives (31), such as the reduction of duration and frequency of TGN crises (5). Finally, this study was of short duration. However, as clinical trials with PGB in this condition are lacking and currently available therapeutic alternatives limited (6, 8), we believe this study provides valuable information on the design and conduct of formal randomized clinical trials with PGB to confirm present findings, particularly when health decision could be made from these data. According with these results, PGB could be used to alleviate pain in patients with TGN, while a definitive or aetiological therapy may be implemented.
Despite the limitations of this analysis, our results suggest that PGB could play a role in the effective management of pain, anxiety, depression and sleep disturbance in patients with TGN treated under common clinical practice conditions in PCS. Taking into account the pharmacokinetic profile and tolerability of PGB, and the efficacy demonstrated in other forms of NeP, both central and peripheral, PGB may be considered a good alternative for the treatment of TGN patients in PCS. Randomized clinical trials are necessary to confirm the efficacy of this drug in this population, the range of dosage to be used and the long-term maintenance of its beneficial effects.
Footnotes
Acknowledgements
The authors thank Fernando Rico-Villademoros, MD, for his assistance in the preparation of the draft of this manuscript, and especially thank all participants in the LIDO study for their contribution to attaining the goals of this research. This study has been funded by an unrestricted grant from Pfizer Spain
